Abstract
Blastocyst implantation is a critical stage in the establishment of pregnancy. Leukemia inhibitory factor (LIF) is essential for mouse blastocyst implantation and also plays a role in human pregnancy. We examined the effect of a potent LIF antagonist (LA) on mouse implantation. In mice, LIF expression peaks on day 3.5 of pregnancy (D3.5) (D0.5 = day of mating plug detection) in the uterine glandular epithelium. LA (7 mg/kg per day) administered from D2.5 to D4.5 via four hourly i.p. injections plus continuous administration via miniosmotic pump resulted in complete implantation failure. To improve its pharmacokinetic properties, we conjugated LA to polyethylene glycol (PEG), achieving a significant increase in serum levels. PEGylated LA (PEGLA) (37.5 mg/kg per day) administered via three i.p. injections between D2.5 and D3.5 also resulted in complete implantation failure. PEGLA immunolocalized to the uterine luminal epithelium at the time of blastocyst implantation. Both LA and PEGLA reduced phosphorylation of the downstream signaling molecule STAT3 in luminal epithelial cells on D3.5. The effects of PEGLA were found to be endometrial, with no embryo-lethal effects observed. These data demonstrate that administration of a PEGylated LIF antagonist is an effective method of targeting LIF signaling in the endometrium and a promising novel approach in the development of nonhormonal contraceptives for women.
Keywords: embryo implantation, leukemia inhibitory factor, mouse
Blastocyst implantation into the endometrium is a critical step in the establishment of pregnancy. Synchronized endometrial receptivity and blastocyst competence are achieved via a network of endocrine, paracrine, and autocrine factors, including cytokines (1). However, the molecular mechanisms are still incompletely understood, hampering efforts to develop targeted, nonhormonal methods of contraception, for which there is a recognized worldwide need (2).
Leukemia inhibitory factor (LIF) is one of few molecules known to be indispensable for mouse blastocyst implantation (3). Gene-targeting experiments have shown that LIF-deficient female mice are infertile (4). LIF is a multifunctional glycoprotein that can be induced in most mouse and human tissues (5). In mice, LIF mRNA and protein expression are maximal in the uterine endometrial glands on day 3.5 of pregnancy (D3.5). LIF is secreted into the uterine lumen coincident with blastocyst formation and preceding implantation (6–8). Female LIF-null mice (LIF−/−) have normal blastocysts that attach to the uterine luminal epithelium but do not implant (4, 8). Implantation can be rescued by transferring LIF−/− blastocysts to a wild-type female (4) or by injecting LIF−/− females with recombinant LIF on D3.5 or D4.5 (8). Maternal LIF signaling is therefore essential for mouse blastocyst implantation.
LIF belongs to the IL-6 family of cytokines, which includes IL-11, and signals via a cell surface receptor complex composed of the LIF receptor α-chain (LIFR) and the signal-transducing subunit gp130, activating the JAK/STAT pathway (9). Upon activation, STAT proteins are tyrosine-phosphorylated, homodimerize, and translocate to the nucleus to modulate the expression of target genes. Both LIFR and gp130 are highly expressed on D3.5 in the uterine luminal epithelium, coincident with the onset of uterine receptivity (7, 10). At this time, LIF specifically activates STAT3 in the luminal epithelium, resulting in the translocation of tyrosine-phosphorylated STAT3 (P-STAT3) to the nuclei (11). STAT3 activation has an important role in mouse implantation. Deleting the region of gp130 responsible for STAT activation (gp130ΔSTAT mice) results in complete implantation failure (12). Female mice heterozygous for the LIFR are fertile; however, LIFR−/− offspring show placental defects, are underrepresented during gestation, and do not survive to 4 weeks of age (13).
The importance of LIF in human pregnancy is suggested by its endometrial expression pattern, which mimics that in mice. LIF mRNA is present in human endometrium at higher levels during the secretory vs. proliferative phase of the menstrual cycle and is also detected in first-trimester decidua (14). LIF protein is maximal in the endometrial glands during the secretory phase, when blastocyst implantation is most likely to occur (15, 16), and uterine fluid contains maximal levels of LIF protein during the mid to late secretory phase (17). LIFR and gp130 are expressed in the luminal epithelium at this time, indicating a role for LIF signaling in preparing the human uterus for blastocyst attachment (15). In addition, decreased endometrial LIF production may be a factor in some cases of human infertility (17–19). An effective LIF antagonist could therefore prevent blastocyst implantation in women. As a first step in examining this, we explored the effect of targeting maternal LIF on mouse blastocyst implantation in vivo.
A highly potent LIF antagonist, MH35-BD/Q29A+G124R (hereafter referred to as LA), was previously produced by mutating human LIF at regions that bind human LIFR and gp130 (20). Relative to wild-type human LIF, LA has a >1,000-fold higher affinity for binding to LIFR but does not bind gp130 as required for productive signaling. LA is species cross-reactive and is also a potent antagonist of murine LIFR-induced bioactivity (20). However, the pharmacokinetics of LA would be expected to be similar to that of iodinated LIF, which has a very short initial half-life in vivo (21). We therefore aimed to conjugate LA to PEG, a strategy previously used to extend the serum half-life of cytokines (22). We compared the effect of PEGylated LA (PEGLA) and non-PEGylated LA on blastocyst implantation in vivo.
Results
Continuous Administration of LA Completely Blocks Blastocyst Implantation.
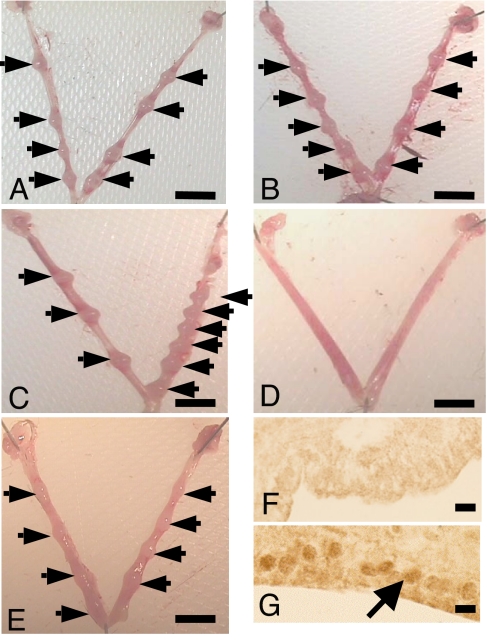
LA or PBS was administered to mated mice to span the time of maximal endometrial LIF expression during the periimplantation period. LA (7 mg/kg per day), or PBS was administered from 2100 hours on D2.5 to 2100 hours on D4.5 via one of four methods: (i) four hourly i.p. injections, (ii) four hourly s.c. injections, (iii) continuous administration via Alzet miniosmotic pump within the peritoneal cavity, or (iv) miniosmotic pump plus four hourly i.p. injections. Numbers of implantation sites and corpora lutea (CL) were counted on D6.5. Whereas administration regimens i–iii had no effect on implantation rates (Fig. 1 A–C), continuous administration of LA by miniosmotic pump plus four hourly i.p. injections completely blocked blastocyst implantation (Table 1 and Fig. 1 D and E).
Fig. 1.
LA blocks blastocyst implantation and reduces STAT3 phosphorylation in uterine luminal epithelium. Representative photomicrographs of uterus collected on D6.5 from mated mice treated with LA (7 mg/kg per day) by i.p. injection (A), s.c. injection (B), Alzet miniosmotic pump (C), i.p. injection plus miniosmotic pump (D) or with PBS by i.p. injection plus miniosmotic pump (control) (E). Arrows indicate implantation sites. (F and G) P-STAT3 immunostaining in mouse uterus on D3.5, 2 h after intrauterine injection of LA (1 mg/kg) (F) or PBS (G). The arrow shows positive nuclear staining in the uterine luminal epithelium. (Scale bars: A–E, 5 mm; F and G, 10 μm.)
Table 1.
Continuous administration of LA blocks blastocyst implantation in mice
| Treatment | n, i.p. plus Alzet | Implantation sites |
Corpora lutea, i.p. plus Alzet | |||
|---|---|---|---|---|---|---|
| i.p. | s.c. | Alzet | i.p. plus Alzet | |||
| LA | 5 | 7.0 ± 1.5 | 8.0 ± 1.7 | 6.0 ± 1.8 | 0.0 ± 0.0* | 7.2 ± 0.8 |
| Control | 4 | 8.0 ± 1.2 | 7.0 ± 1.5 | 7.0 ± 1.3 | 6.0 ± 1.5 | 7.8 ± 0.6 |
LA or control (PBS) was administered to mated mice at a dose of 7 mg/kg per day via one of four regimens as described in Materials and Methods. Numbers of implantation sites and CL were recorded on D6.5. Data represent the mean number of implantation sites or CL per mouse ± SEM.
*, P < 0.05 vs. control.
Administration of LA Reduces STAT3 Phosphorylation in the Uterine Luminal Epithelium.
LIF phosphorylates STAT3 in the luminal epithelium at the time of maximal uterine LIF expression during early pregnancy in mice (11). To determine the effect of LA on STAT3 phosphorylation at the time of implantation, LA (1 mg/kg) or PBS was injected directly into the uterine lumen at 0900 hours on D3.5, and the uterus was examined at time points up to 6 h later. Immunostaining for P-STAT3 was absent in the LA-treated uterus 2 h after administration, whereas P-STAT3 localized to luminal epithelial nuclei in the PBS-treated uterus (Fig. 1 F and G). These data demonstrated that LA was biologically active, targeted the uterine luminal epithelium, and blocked LIF action in the mouse uterus.
Preparation of the PEGylated LIF Antagonist PEGLA.
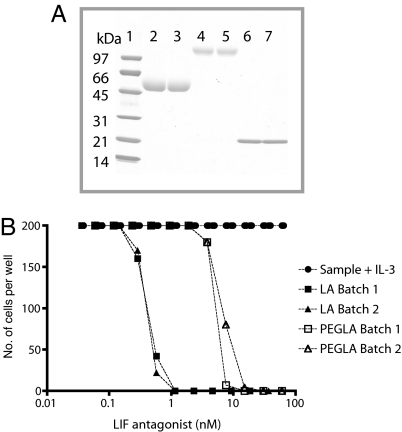
Blastocyst implantation was blocked by continuous administration of LA by miniosmotic pump and i.p. injections; however, this regimen would not be suitable for contraceptive purposes. To increase its serum stability, and thereby reduce the frequency of administration required to block implantation, LA was conjugated to PEG. SDS/PAGE analysis of purified PEGLA showed a broad band migrating with an apparent molecular weight of 100,000–120,000 (Fig. 2A), suggesting that two PEG molecules were attached to each molecule of LA. Preliminary analysis of PEGLA by MALDI-TOF mass spectrometry yielded a broad peak with an average molecular weight of ≈62,000 (data not shown), suggesting that LA was mono-PEGylated. It has been reported previously that, because of the large hydrodynamic volume of PEG, estimates of the molecular weight of PEGylated proteins by SDS/PAGE are significantly higher than those determined by mass spectrometry (23). PEGLA was capable of specifically inhibiting LIF-induced Ba/F3 cell proliferation but showed a 10- to 20-fold reduction in potency compared with the unmodified LA (Fig. 2B). We did not anticipate this to be a problem because LA is a superpotent LIF antagonist with an IC50 of 0.38–0.44 nM compared with 5.5–6.8 nM PEGLA when recombinant human LIF (hLIF) is added at a subsaturating concentration of 12 pM in the in vitro bioassay.
Fig. 2.
Analysis of PEGLA by SDS/PAGE and inhibition of human LIFR/gp130 Ba/F3 cell proliferation by LIF antagonists. Expression, purification, PEGylation, and testing the bioactivity of LIF antagonists were as described in Materials and Methods. (A) Samples (5 μg per lane) were analyzed by SDS/PAGE on a 4–20% gel under reducing conditions and visualized by Coomassie staining. Lane 1, molecular mass markers (in kDa); lanes 2 and 3, PEGylated G-CSF (pegfilgrastim); lanes 4 and 5, PEGLA; lanes 6 and 7, LA. (B) Inhibition of human LIFR/gp130 Ba/F3 cell proliferation by LIF antagonists (cells stimulated with wild-type hLIF, ≈12 pM). Two independent batches of PEGLA and LA were assayed. Molecular weights used to calculate the molarity of PEGLA, LA, and hLIF were 62,000, 23,344, and 19,700, respectively. The maximum possible value for cell number was 200.
PEGLA Blocks Blastocyst Implantation.
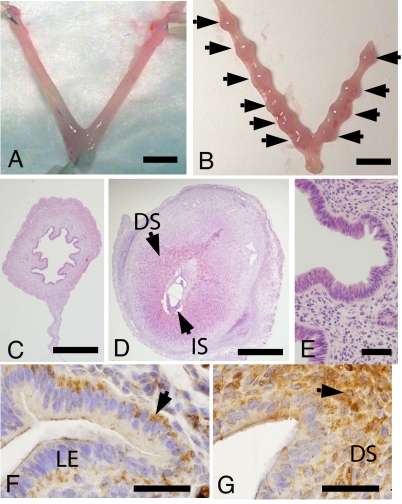
Mated mice were administered PEGLA or control PEGylation reagent (hydrolyzed mPEG2-NHS) by i.p. injection at 1200 hours and 2200 hours on D2.5 and 1000 hours on D3.5, and their uteri were harvested on D6.5. Numbers of implantation sites and CL were counted (Table 2). Implantation was completely blocked in PEGLA-treated mice, which had no visible implantation sites, whereas control-treated mice had the expected numbers of implantation sites (Table 2 and Fig. 3 A and B). There was no difference between groups in the numbers of CL, indicating normal ovulation (Table 2). Histologically, the uteri from the antagonist-treated mice appeared nonpregnant and intact (Fig. 3 C and E), whereas control uteri had a normal postimplantation morphology (Fig. 3D). No side effects were observed in the animals after either treatment. Additionally, i.p. injections of PEGLA into mated mice at 1200 hours and 2200 hours on D1.5 and 1000 hours on D2.5 (24 h earlier than the previous experiment) had no effect on implantation (data not shown).
Table 2.
Three injections of PEGLA block blastocyst implantation in mice
| Treatment | n | Implantation sites | Corpora lutea |
|---|---|---|---|
| PEGLA | 5 | 0.0 ± 0.0* | 5.8 ± 0.7 |
| Control | 4 | 8.8 ± 0.5 | 7.8 ± 0.9 |
PEGLA or control (mPEG2-NHS) was administered to mated mice by i.p. injection (12.5 mg/kg) at 1200 hours and 2200 hours on D2.5 and 1000 hours on D3.5. Numbers of implantation sites and CL were recorded on D6.5. Data represent the mean number of implantation sites or CL per mouse ± SEM.
*, P = 0.0004 vs. control.
Fig. 3.
PEGLA blocks blastocyst implantation and immunolocalizes to uterine luminal epithelium on D3.5. Shown are representative photomicrographs of uteri collected on D6.5 from mated mice treated with 12.5 mg/kg (25 μl) i.p. PEGLA (A) or control (mPEG2-NHS) (B) at 1200 hours and 2200 hours on D2.5 and 1000 hours on D3.5. Arrows indicate implantation sites. (C) H&E-stained cross section of A. (D) H&E-stained cross section of implantation site in B. (E) Higher magnification of C showing normal (intact) epithelial and stromal architecture after PEGLA administration. (F and G) PEG immunostaining in uteri of two different mice on D3.5 after i.p. administration of 12.5 mg/kg (25 μl) PEGLA at 1200 hours and 2200 hours on D2.5 and 1000 hours on D3.5. Arrows indicate positive staining as described in Results. (DS, decidualized stroma; IS, implantation site; LE, luminal epithelium. (Scale bars: A and B, 5 mm; C and D, 0.5 mm; E–G, 50 μm.)
PEGLA Localizes to the Uterine Luminal Epithelium and Stroma.
To assess whether PEGLA was able to reach the required site of action in the uterus (the uterine luminal epithelium), PEGLA or mPEG2-NHS was administered i.p. to mated mice at the same dose that blocked blastocyst implantation. Uteri were collected for PEG immunohistochemistry on D3.5. PEG was detected in the uteri of all antagonist-treated and control-treated mice, with staining primarily localized to the basal surface of luminal epithelial cells and in the cytoplasm of stromal cells (Fig. 3 F and G).
PEGLA Reduces STAT3 Phosphorylation in the Uterine Luminal Epithelium at the Time of Implantation.
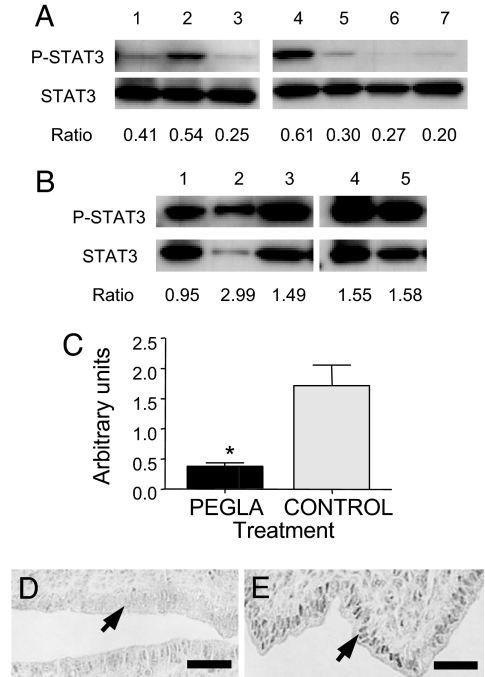
PEGLA or mPEG2-NHS was administered i.p. to mated mice at the same dose and frequency that blocked blastocyst implantation. Uteri were collected on D3.5 for isolation of the luminal epithelium and Western blot analysis. PEGLA treatment caused a consistent and significant reduction in the ratio of P-STAT3 protein to total STAT3 protein in the luminal epithelium compared with control (Fig. 4 A–C). Uterine horns collected at the same time as those used for Western blot were also used for P-STAT3 immunostaining. P-STAT3 immunostaining intensity in the luminal epithelium was reduced in PEGLA-treated mice compared with control (Fig. 4 D and E).
Fig. 4.
PEGLA reduces STAT3 phosphorylation in uterine luminal epithelium on D3.5. Shown are immunoblots for P-STAT3 and total STAT-3 in uterine luminal epithelium isolated on D3.5 from mated mice administered 12.5 mg/kg (25 μl) i.p. PEGLA (A) or control (mPEG2-NHS) (B) at 1200 hours and 2200 hours on D2.5 and 1000 hours on D3.5. Numbers refer to individual animals. (C) Graphical representation of P-STAT3/total STAT3 ratios calculated from densitometry in A and B (mean ± SEM; *, P = 0.0025). (D and E) P-STAT3 immunostaining in uterus on D3.5 after i.p. administration of 12.5 mg/kg (25 μl) PEGLA (D) or control (mPEG2-NHS) (E) at 1200 hours and 2200 hours on D2.5 and 1000 hours on D3.5. The area of uterus shown is the antimesometrial pole, at which the blastocyst will initiate implantation. P-STAT3 immunostaining was specifically reduced in the luminal epithelial nuclei (D and E, arrows) after PEGLA treatment. (Scale bars: 50 μm.)
PEGLA Is Not Lethal to Preimplantation Embryos.
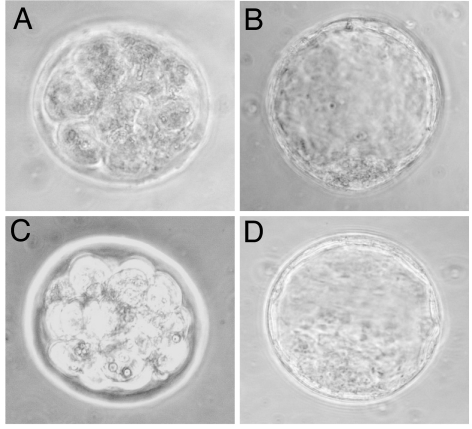
Mice were treated with PEGLA or mPEG2-NHS at the same time and dose used to block implantation, and uteri were flushed at D3.5. The numbers of morulae and blastocysts retrieved were similar between PEGLA-treated and control-treated animals, and there was no difference in the percentage achieving attachment and outgrowth after 72 h of ex vivo culture (Table 3). In addition, the morphology of the morulae and blastocysts at the time of collection did not differ between treatment groups (Fig. 5).
Table 3.
PEGLA is not lethal to preimplantation embryos
| In vivo treatment | n | Morulae (%) | Unhatched blastocysts (%) | Hatched, attached, and outgrowing blastocysts (%) |
|---|---|---|---|---|
| PEGLA | 5 | 16/32 (50) | 5/32 (16) | 11/32 (34) |
| Control | 4 | 14/35 (40) | 7/35 (20) | 14/35 (40) |
Mice were superovulated and mated, and PEGLA or control (mPEG2-NHS) was administered by i.p. injection (12.5 mg/kg) at 1200 hours and 2200 hours on D2.5 and 1000 hours on D3.5. Uteri were flushed, and embryos were collected on D3.5 and scored after 72 h of ex vivo culture.
Fig. 5.
PEGLA was not lethal to preimplantation embryos. Shown are representative photomicrographs of preimplantation embryos collected on D3.5 from mice treated with PEGLA (A and B) or control (mPEG2-NHS) (C and D). Mated mice were administered 12.5 mg/kg (25 μl) PEGLA or control i.p. at 1200 hours and 2200 hours on D2.5 and 1000 hours on D3.5. (A and C) Morulae. (B and D) Blastocysts. (Magnification: ×400.)
Serum Concentration of LA and PEGLA.
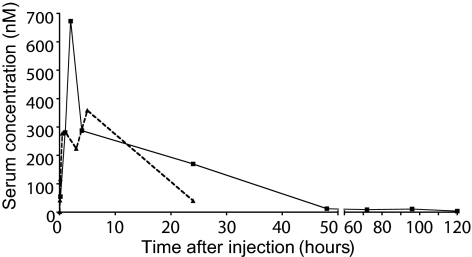
The serum concentration of LA and PEGLA was determined after i.p. injection into nonpregnant mice. Serum concentration of PEGLA peaked at 2 h after injection and was 1.9-fold greater than the peak serum concentration of LA (Fig. 6). The area under the curve for PEGLA was 1.8-fold greater than that of LA. There was no detectable LA, PEGLA, or LIF in serum from untreated mice, or mice injected with the controls, at any of the time points tested (data not shown).
Fig. 6.
LA and PEGLA serum levels. LA (dotted line) or PEGLA (solid line) was injected i.p. into nonpregnant mice (1 mg/kg, n = 2–3 per group), and serum was collected at time points shown for assay by mLIF ELISA. Data are mean values.
Discussion
Previous studies have attempted to block LIF signaling in the uterus for contraceptive purposes with limited success. Anti-LIF polyclonal antibodies administered directly into the uterus interfere with blastocyst implantation in the rhesus monkey (24) and in the mouse (25), but neither method completely prevents implantation. Intrauterine injection of a cell-permeable STAT3 peptide inhibitor (26) or a STAT3 decoy oligodeoxynucleotide (27) achieved ≈70% reduction in implantation rates. In contrast, both forms of our highly potent LIF antagonist were 100% effective in blocking implantation and were effective via the systemic i.p. route—a critical step in the development of targeted anti-LIF methods of contraception (28).
In mice, exogenous LIF has an initial half-life of 6–8 min, followed by a more prolonged clearance phase, and shows preferential uptake by organs other than the uterus (21), indicating that targeting the uterus using LA would be difficult to achieve. Indeed, our initial studies showed that, although LA was highly effective in blocking implantation, it required frequent i.p. injections together with continuous administration via miniosmotic pump. The frequency of administration of LA and PEGLA required to block implantation was also likely increased by the presence of circulating soluble LIFR. Soluble LIFR is present at high levels in mouse serum and is elevated during pregnancy (29), which would reduce the efficacy of LIF antagonists that target the receptor. Importantly, although circulating soluble LIFR is present in mice, it has not been detected in women, indicating that LIFR-directed antagonists are likely to have even greater potency in women.
One of the greatest challenges in designing therapeutic agents is to deliver the active compound to the required site of action and extend its serum stability. One option to prolong the biological activity of LA was to PEGylate the molecule (29). PEGylation increases molecular size, reduces renal ultrafiltration, decreases uptake by the liver, and protects the PEGylated molecule from proteolytic cleavage (30–32). Although PEGylation may lower in vivo intrinsic activity, this can be more than compensated for by increased serum stability (33, 34). PEGylated cytokines such as interferons have extended serum half-lives and enhanced clinical efficacy compared with non-PEGylated interferons and are well tolerated in humans (22, 35).
PEGLA was produced and injected i.p. into mice and was detectable in serum for up to 5 days—a significantly extended serum stability compared with that previously shown for exogenous LIF in mice (21) and compared with that of the non-PEGylated LA. PEGLA was biologically active, with enhanced efficacy requiring lower dosing and frequency of injection to prevent implantation compared with LA. Both forms of antagonist reduced LIF action in the uterus in vivo. It is likely that the i.p. injection of both LA and PEGLA acted on the uterus because P-STAT3 was reduced in the uterine luminal epithelium. The timing of PEGLA injection was also critical. Injection of antagonist too early did not abolish implantation, indicating that the antagonist was acting on the uterus at the time LIF is maximally expressed.
A direct effect of PEGLA on the preimplantation embryo is unlikely because we demonstrated no differences between treated and control animals in embryo numbers, morphology, or in vitro viability at the time of implantation. LIFR is present in the preimplantation embryo, but LIFR-null embryos can survive until the perinatal period, suggesting that LIF is not critical for embryo development in mice (13). The data imply that the abolishment of implantation was due to a direct effect on uterine receptivity.
PEGLA shows great potential as a novel contraceptive agent for women. If endometrial LIF is essential for blastocyst implantation in women, as it is in mice, PEGLA could be used on an as-needed basis to prevent implantation. If PEGLA remains in the human circulation for a similar length of time as it does in the mouse, this would be sufficient to cover the implantation window. It remains to test PEGLA in a primate model and then to fully assess its safety, reliability, and reversibility in women.
Materials and Methods
Animals.
Virgin, naturally cycling, 8- to 10-week-old female C57BL/6J mice and male mice of the same strain (Monash University Animal Services, Clayton, Australia) were fed and watered ad libitum and maintained in a 12-h:12-h light:dark cycle. All procedures were approved by the Monash Medical Centre Animal Ethics Committee and complied with the National Health and Medical Research Council Australian Code of Practice for the Care and Use of Animals for Scientific Purposes.
Preparation of the LIF Antagonist LA.
To produce LIF antagonist MH35-BD/Q29A+G124R, the mutant cDNA was subcloned into a modified pGEX-2T vector (GE Healthcare) as a GST fusion protein (20). The GST fusion protein was expressed in BL21 (DE3) Escherichia coli, purified, and cleaved from the fusion partner essentially as described previously (36).
Treatment to Block Blastocyst Implantation Using LA.
Female mice (n = 4–5 per group) were paired with males and checked the following morning for the presence of a vaginal plug (D0.5 = day of mating plug detection). LA or PBS (7 mg/kg per day) was administered from 2100 hours on D2.5 to 2100 hours on D4.5 via one of the following regimens: (i) four hourly i.p. injections, (ii) four hourly s.c. injections, (iii) Alzet miniosmotic pump (Alza) placed into the peritoneal cavity under ketamine/xylazine anesthesia, or (iv) miniosmotic pump plus i.p. injections as in i. Mice were killed on D6.5, and the numbers of implantation sites and CL were recorded. Data were analyzed with Prism 4.0 (GraphPad). Because the numbers of CL were not normally distributed, the nonparametric Mann–Whitney test was used to compare treated and control means. Where the implantation site values for the treated group were 0, a one-sample t test was applied to the control data (theoretical mean = 0).
Preparation of the PEGylated LIF Antagonist PEGLA.
Purified LA protein (in 100 mM sodium phosphate buffer, pH 7.0/0.4 M NaCl) was PEGylated with 40-kDa PEG-NHS (mPEG2-NHS 40 kDa; Nektar Advanced PEGylation) at a protein:reagent molar ratio of 1:3 for 12 h at room temperature. PEGLA was purified by cation-exchange chromatography on a 1-ml HiTrap SP Sepharose column (GE Healthcare), followed by RP-HPLC on a 100-mm × 7.5-mm i.d. Vydac C4 column with a 60-min linear gradient of 0–60% acetonitrile in 0.1% (vol/vol) trifluoroacetic acid. The fractions from RP-HPLC were lyophilized and reconstituted in Milli-Q water. Samples were analyzed by SDS/PAGE using a Novex precast Tris-glycine gel of 4–20% polyacrylamide (Invitrogen) under reducing conditions. After electrophoresis, the gel was stained with 0.1% Coomassie brilliant blue R-250 in 50% methanol and 10% acetic acid and destained in 12% methanol and 7% acetic acid. Control PEGylation reagent was generated by incubating mPEG2-NHS in Milli-Q water for at least 24 h.
Testing the Bioactivity of PEGLA.
Purified LA and PEGLA were tested for their ability to inhibit the action of hLIF in a cell proliferation assay using a Ba/F3 cell line engineered to express hLIFR and gp130. Cells were grown in DME with 10% bovine calf serum, 10% (vol/vol) conditioned medium from WEHI-3B D− as a source of IL-3, 0.8 mg/ml G418, and 20 μg/ml puromycin before assay. Recombinant hLIF was added at a subsaturating concentration (≈12 pM) with various concentrations of either purified LA or PEGLA. After 3 days of incubation, viable cells were counted. A dose–response curve was generated to quantitate the efficacy of the antagonists in inhibiting the activity of wild-type hLIF. As a control for nonspecific inhibition, the antagonists were also tested on the same Ba/F3 cell line stimulated with IL-3 to ascertain that any inhibition was specific for LIF-induced proliferation.
Treatment to Block Blastocyst Implantation Using PEGLA.
Female mice (n = 4–5 per group) were paired with males as above. At 1200 hours and 2200 hours on D2.5 and 1000 hours on D3.5, mice were injected i.p. with 12.5 mg/kg (25 μl) PEGLA or mPEG2-NHS. Mice were killed on D6.5, the numbers of implantation sites and CL recorded, and data were analyzed as above.
Immunostaining.
Five-micrometer sections of formalin-fixed, paraffin-embedded uterus (n = 5 mice per group) were cut onto SuperFrost slides, dewaxed, and rehydrated. Antigen retrieval was performed by microwaving in 10 mM sodium citrate (pH 6.0) for 5 min at 200 W. For PEG immunostaining, endogenous peroxidase activity was quenched with 6% H2O2 (30 min, 25°C), and nonspecific binding was blocked with 10% normal goat serum in TBS (1 h, 25°C). For P-STAT3 immunostaining, 3% H2O2 (10 min, 25°C) and 10% normal swine serum/2% normal mouse serum in TBS (nonimmune block) were used. Primary antibodies were mouse anti-PEG monoclonal IgG1 [E11 (37); a gift from Steve Roffler (Institute of Biomedical Sciences, Academia Sinica, Taiwan); 5 μg/ml in 1% BSA/TBS] or rabbit anti-mouse P-STAT3 [Tyr-705 (Cell Signaling Technology); 90 ng/ml in nonimmune block]. Negative controls were matching concentrations of mouse IgG1 (Dako) or rabbit IgG1 (Sigma). Primary antibody or negative control was applied (16–18 h at 4°C), and sections were washed in TBS/0.1% Tween 20 (TBST). The secondary reagent for PEG immunostaining was the Envision+ System-HRP Labeled Polymer (Dako; 30 min, 25°C), and, for P-STAT3, StreptABComplex-HRP (Dako; 1:200 in nonimmune block; 30 min, 25°C). After washes in TBST and TBS, positive immunostaining was visualized by using DAB (Dako), and sections were counterstained with Harris hematoxylin (Sigma).
Immunoblotting for P-STAT3 and Total STAT3.
Uterine luminal epithelium was isolated as described in ref. 38. Cells were lysed and homogenized in ice-cold lysis buffer [50 mM Trizma Base (Sigma), pH 7.4/150 mM NaCl/2 mM EDTA/2 mM EGTA/25 mM NaF/0.2% Triton X-100 (Sigma)/0.3% Nonidet P-40 (Sigma)] containing Protease Inhibitor Mixture Set III (1:500; Calbiochem). Lysates were centrifuged, and supernatants were assayed for total protein by using the BCA Protein Assay Kit (Pierce). Proteins (30 μg per sample) were resolved on SDS/PAGE gels, transferred to Hybond-P PVDF membranes (GE Healthcare), and blocked in 10% skim milk powder in TBS (1 h, 25°C). Membranes were incubated overnight at 4°C with rabbit anti-mouse P-STAT3 (Tyr-705; Cell Signaling Technology; 1:1,000 in 5% skim milk/TBS). After washes in TBS and TBST, goat anti-rabbit HRP secondary antibody (Dako; 1:1,500 in 5% skim milk/TBS) and the ECL Plus Detection System (GE Healthcare) were applied. Membranes were exposed to autoradiographic film (Hyperfilm ECL; GE Healthcare) for 5 min. Membranes were then washed in stripping buffer (200 mM glycine/0.1% SDS/1% Tween 20) followed by washes in TBS and TBST, methanol rehydration, and a final TBST wash. Membranes were blocked in 10% skim milk/TBS (1 h, 25°C) and incubated with rabbit anti-mouse STAT3 (Cell Signaling Technology; 1:800 in 5% skim milk/TBS, 16–18 h at 4°C). For the detection of total STAT3, membranes were processed as above. Films were scanned and densitometry was performed with GelDoc equipment and Quantity One 1-D Analysis Software (Bio-Rad). Background-corrected density OD/mm2 for P-STAT3/STAT-3 was obtained, and ratios for treated and control animals were compared by using Prism 4.0 (Mann–Whitney test).
Preimplantation Embryo Lethality Assay.
Virgin females (n = 4–5 per group) were superovulated by i.p. injection of 5 units of PMSG followed 48 h later by 5 units of hCG and paired with males. Mice were injected i.p. with 12.5 mg/kg (25 μl) PEGLA or mPEG2-NHS at 1200 hours and 2200 hours on D2.5 and 1000 hours on D3.5. At 1200 hours on D3.5, embryos were collected by flushing uteri with M2 medium (Sigma) containing 1% antibiotic–antimycotic (Invitrogen). Embryos (n = 32 PEGLA-treated, n = 35 mPEG2-NHS-treated) were rinsed and transferred individually to microdrops of M16 medium (Sigma) under embryo-tested light paraffin oil (Sigma) and incubated for 72 h at 37°C and 5% CO2. Embryos were scored at collection and after 72 h, noting stage of development (morula, unhatched blastocyst, or hatched blastocyst). Embryos were considered “attached” if there was no movement with gentle shaking of the culture dish and “outgrowing” if primary giant trophoblast cells were visible around the attachment site.
LA and PEGLA Serum Levels.
Female mice (n = 2–3 per group) were administered 1 mg/kg LA or PEGLA (or control, PBS, or mPEG2-NHS) by i.p. injection and killed at required time points. For LA, these were 1, 3, 5, 10, and 30 min and 1, 3, 5, and 24 h after injection. For PEGLA, time points were 10 min and 1, 2, 4, 24, 48, 72, 96, and 120 h after injection. Blood was collected by cardiac puncture, incubated at 4°C overnight, and serum-separated by centrifugation. Serum was assayed for LA or PEGLA by using a Quantikine Mouse LIF Immunoassay (R & D Systems; catalog no. MLF00) according to the manufacturer's instructions but using standard curves of either LA or PEGLA. Samples were pretreated with 0.625 M acetic acid and neutralized with 2.7 M NaOH/1 M Hepes (resulting in a 2.6-fold dilution) before assay in duplicate. The sensitivity of the assay was 1.94 pg/ml (0.083 pM for LA and 0.031 pM for PEGLA).
Acknowledgments
We thank Yee Lee Tan, Chelsea Stoikos, Tracy Willson, Sandra Mifsud, Ladina Di Rago, Cindy Luo, Andrew Low, and Ellen Menkhorst for excellent technical assistance; Dr. Steve Roffler for donating anti-PEG antibodies; and Dr. Glenn Begley of Amgen for pegfilgrastim. This work was supported by the Consortium for Industrial Collaboration in Contraceptive Research Program of the Contraceptive Research and Development Program, Eastern Virginia Medical School (Subproject CIG-02-82), and the National Health and Medical Research Council of Australia (Grants 388916 and 388901 to E.D. and L.A.S. and Grant 461219 to J.-G.Z., D.M., N.A.N., and L.R.).
Footnotes
Conflict of interest statement: The Walter and Eliza Hall Institute of Medical Research holds a patent for leukemia inhibitory factor.
References
- 1.Sharkey AM, Smith SK. Best Pract Res Clin Obstet Gynaecol. 2003;17:289–307. doi: 10.1016/s1521-6934(02)00130-x. [DOI] [PubMed] [Google Scholar]
- 2.Glasier A, Gulmezoglu AM, Schmid GP, Moreno CG, Van Look PF. Lancet. 2006;368:1595–1607. doi: 10.1016/S0140-6736(06)69478-6. [DOI] [PubMed] [Google Scholar]
- 3.Robb L, Dimitriadis E, Li R, Salamonsen LA. J Reprod Immunol. 2002;57:129–141. doi: 10.1016/s0165-0378(02)00012-8. [DOI] [PubMed] [Google Scholar]
- 4.Stewart CL, Kaspar P, Brunet LJ, Bhatt H, Gadi I, Kontgen F, Abbondanzo SJ. Nature. 1992;359:76–79. doi: 10.1038/359076a0. [DOI] [PubMed] [Google Scholar]
- 5.Metcalf D. Stem Cells. 2003;21:5–14. doi: 10.1634/stemcells.21-1-5. [DOI] [PubMed] [Google Scholar]
- 6.Bhatt H, Brunet LJ, Stewart CL. Proc Natl Acad Sci USA. 1991;88:11408–11412. doi: 10.1073/pnas.88.24.11408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Yang ZM, Le SP, Chen DB, Cota J, Siero V, Yasukawa K, Harper MJ. Mol Reprod Dev. 1995;42:407–414. doi: 10.1002/mrd.1080420406. [DOI] [PubMed] [Google Scholar]
- 8.Chen JR, Cheng JG, Shatzer T, Sewell L, Hernandez L, Stewart CL. Endocrinology. 2000;141:4365–4372. doi: 10.1210/endo.141.12.7855. [DOI] [PubMed] [Google Scholar]
- 9.Kishimoto T, Taga T, Akira S. Cell. 1994;76:253–262. doi: 10.1016/0092-8674(94)90333-6. [DOI] [PubMed] [Google Scholar]
- 10.Song H, Lim H. Reproduction. 2006;131:341–349. doi: 10.1530/rep.1.00956. [DOI] [PubMed] [Google Scholar]
- 11.Cheng JG, Chen JR, Hernandez L, Alvord WG, Stewart CL. Proc Natl Acad Sci USA. 2001;98:8680–8685. doi: 10.1073/pnas.151180898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ernst M, Inglese M, Waring P, Campbell IK, Bao S, Clay FJ, Alexander WS, Wicks IP, Tarlinton DM, Novak U, et al. J Exp Med. 2001;194:189–203. doi: 10.1084/jem.194.2.189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ware CB, Horowitz MC, Renshaw BR, Hunt JS, Liggitt D, Koblar SA, Gliniak BC, McKenna HJ, Papayannopoulou T, Thoma B, et al. Development (Cambridge, UK) 1995;121:1283–1299. doi: 10.1242/dev.121.5.1283. [DOI] [PubMed] [Google Scholar]
- 14.Kojima K, Kanzaki H, Iwai M, Hatayama H, Fujimoto M, Inoue T, Horie K, Nakayama H, Fujita J, Mori T. Biol Reprod. 1994;50:882–887. doi: 10.1095/biolreprod50.4.882. [DOI] [PubMed] [Google Scholar]
- 15.Cullinan EB, Abbondanzo SJ, Anderson PS, Pollard JW, Lessey BA, Stewart CL. Proc Natl Acad Sci USA. 1996;93:3115–3120. doi: 10.1073/pnas.93.7.3115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Vogiagis D, Marsh MM, Fry RC, Salamonsen LA. J Endocrinol. 1996;148:95–102. doi: 10.1677/joe.0.1480095. [DOI] [PubMed] [Google Scholar]
- 17.Laird SM, Tuckerman EM, Dalton CF, Dunphy BC, Li TC, Zhang X. Hum Reprod. 1997;12:569–574. doi: 10.1093/humrep/12.3.569. [DOI] [PubMed] [Google Scholar]
- 18.Giess R, Tanasescu I, Steck T, Sendtner M. Mol Hum Reprod. 1999;5:581–586. doi: 10.1093/molehr/5.6.581. [DOI] [PubMed] [Google Scholar]
- 19.Dimitriadis E, Stoikos C, Stafford-Bell M, Clark I, Paiva P, Kovacs G, Salamonsen LA. J Reprod Immunol. 2006;69:53–64. doi: 10.1016/j.jri.2005.07.004. [DOI] [PubMed] [Google Scholar]
- 20.Fairlie WD, Uboldi AD, McCoubrie JE, Wang CC, Lee EF, Yao S, De Souza DP, Mifsud S, Metcalf D, Nicola NA, et al. J Biol Chem. 2004;279:2125–2134. doi: 10.1074/jbc.M310103200. [DOI] [PubMed] [Google Scholar]
- 21.Hilton DJ, Nicola NA, Waring PM, Metcalf D. J Cell Physiol. 1991;148:430–439. doi: 10.1002/jcp.1041480315. [DOI] [PubMed] [Google Scholar]
- 22.Reddy KR, Wright TL, Pockros PJ, Shiffman M, Everson G, Reindollar R, Fried MW, Purdum PP, III, Jensen D, Smith C, et al. Hepatology. 2001;33:433–438. doi: 10.1053/jhep.2001.21747. [DOI] [PubMed] [Google Scholar]
- 23.Foser S, Schacher A, Weyer KA, Brugger D, Dietel E, Marti S, Schreitmuller T. Protein Expression Purif. 2003;30:78–87. doi: 10.1016/s1046-5928(03)00055-x. [DOI] [PubMed] [Google Scholar]
- 24.Yue ZP, Yang ZM, Wei P, Li SJ, Wang HB, Tan JH, Harper MJ. Biol Reprod. 2000;63:508–512. doi: 10.1095/biolreprod63.2.508. [DOI] [PubMed] [Google Scholar]
- 25.Mitchell MH, Swanson RJ, Oehninger S. Biol Reprod. 2002;67:460–464. doi: 10.1095/biolreprod67.2.460. [DOI] [PubMed] [Google Scholar]
- 26.Catalano RD, Johnson MH, Campbell EA, Charnock-Jones DS, Smith SK, Sharkey AM. Proc Natl Acad Sci USA. 2005;102:8585–8590. doi: 10.1073/pnas.0502343102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Nakamura H, Kimura T, Koyama S, Ogita K, Tsutsui T, Shimoya K, Taniguchi T, Koyama M, Kaneda Y, Murata Y. FEBS Lett. 2006;580:2717–2722. doi: 10.1016/j.febslet.2006.04.029. [DOI] [PubMed] [Google Scholar]
- 28.Gabriel DA, Shea T, Olajida O, Serody JS, Comeau T. Semin Oncol. 2003;30:76–83. doi: 10.1053/j.seminoncol.2003.11.040. [DOI] [PubMed] [Google Scholar]
- 29.Le Bouteiller P. J Gynecol Obstet Biol Reprod. 2004;33:S9–S12. doi: 10.1016/s0368-2315(04)96396-x. [DOI] [PubMed] [Google Scholar]
- 30.Harris JM, Chess RB. Nat Rev. 2003;2:214–221. doi: 10.1038/nrd1033. [DOI] [PubMed] [Google Scholar]
- 31.Roberts MJ, Bentley MD, Harris JM. Adv Drug Deliv Rev. 2002;54:459–476. doi: 10.1016/s0169-409x(02)00022-4. [DOI] [PubMed] [Google Scholar]
- 32.Caliceti P, Veronese FM. Adv Drug Delivery Rev. 2003;55:1261–1277. doi: 10.1016/s0169-409x(03)00108-x. [DOI] [PubMed] [Google Scholar]
- 33.Harris JM, Martin NE, Modi M. Clin Pharmacokinet. 2001;40:539–551. doi: 10.2165/00003088-200140070-00005. [DOI] [PubMed] [Google Scholar]
- 34.Veronese FM. Biomaterials. 2001;22:405–417. doi: 10.1016/s0142-9612(00)00193-9. [DOI] [PubMed] [Google Scholar]
- 35.Lindsay KL, Trepo C, Heintges T, Shiffman ML, Gordon SC, Hoefs JC, Schiff ER, Goodman ZD, Laughlin M, Yao R, et al. Hepatology. 2001;34:395–403. doi: 10.1053/jhep.2001.26371. [DOI] [PubMed] [Google Scholar]
- 36.Gearing DP, Nicola NA, Metcalf D, Foote S, Willson TA, Gough NM, Williams RL. Bio/Technology. 1989;7:1157–1161. [Google Scholar]
- 37.Cheng TL, Cheng CM, Chen BM, Tsao DA, Chuang KH, Hsiao SW, Lin YH, Roffler SR. Bioconjugate Chem. 2005;16:1225–1231. doi: 10.1021/bc050133f. [DOI] [PubMed] [Google Scholar]
- 38.Sidhu SS, Kimber SJ. Biol Reprod. 1999;60:147–157. doi: 10.1095/biolreprod60.1.147. [DOI] [PubMed] [Google Scholar]