Abstract
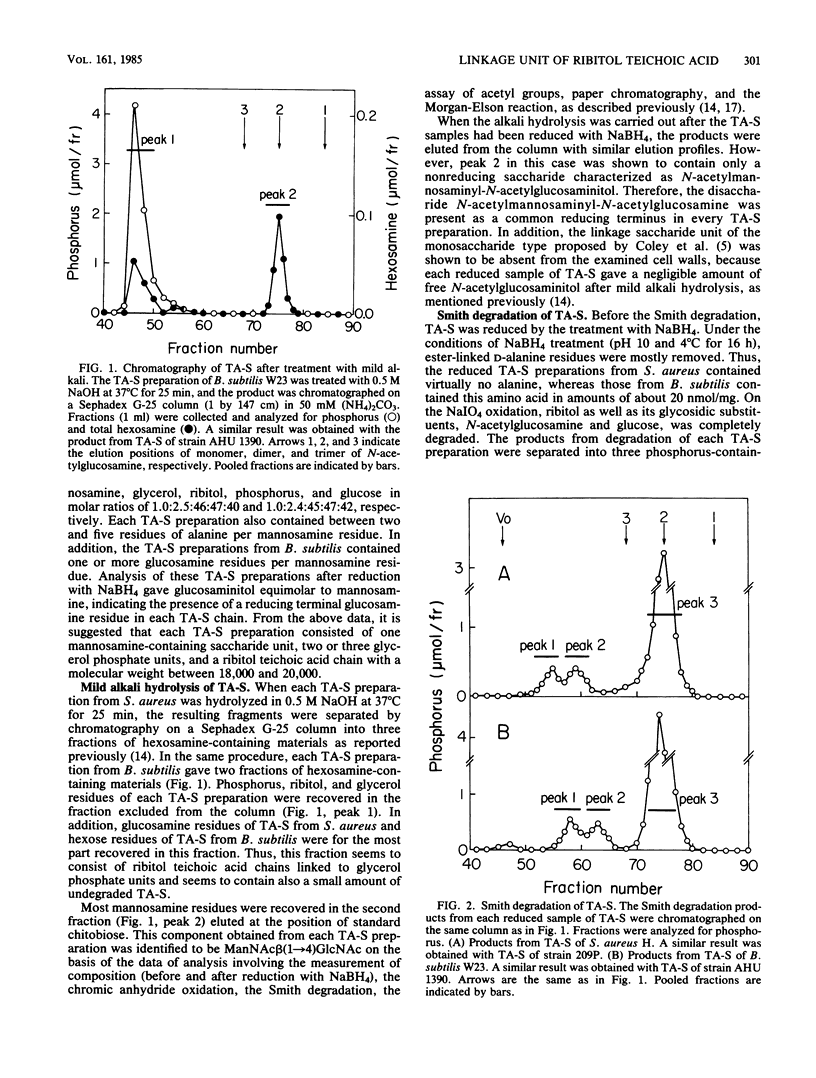
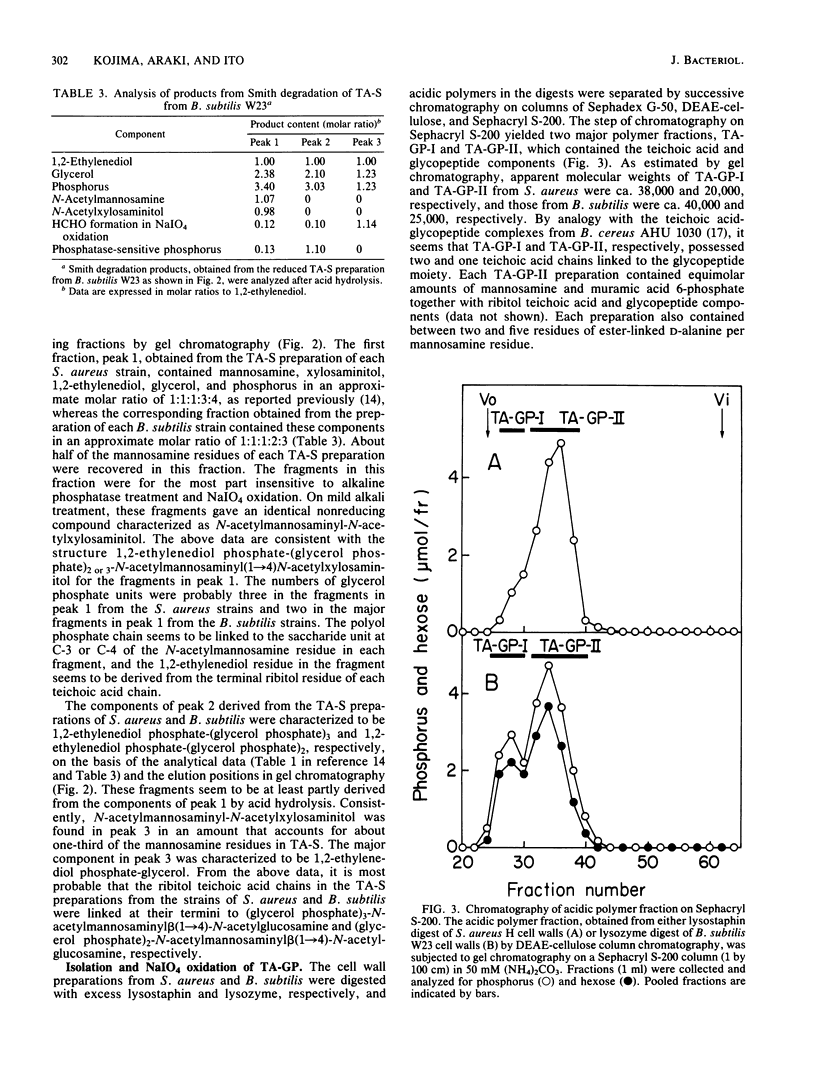
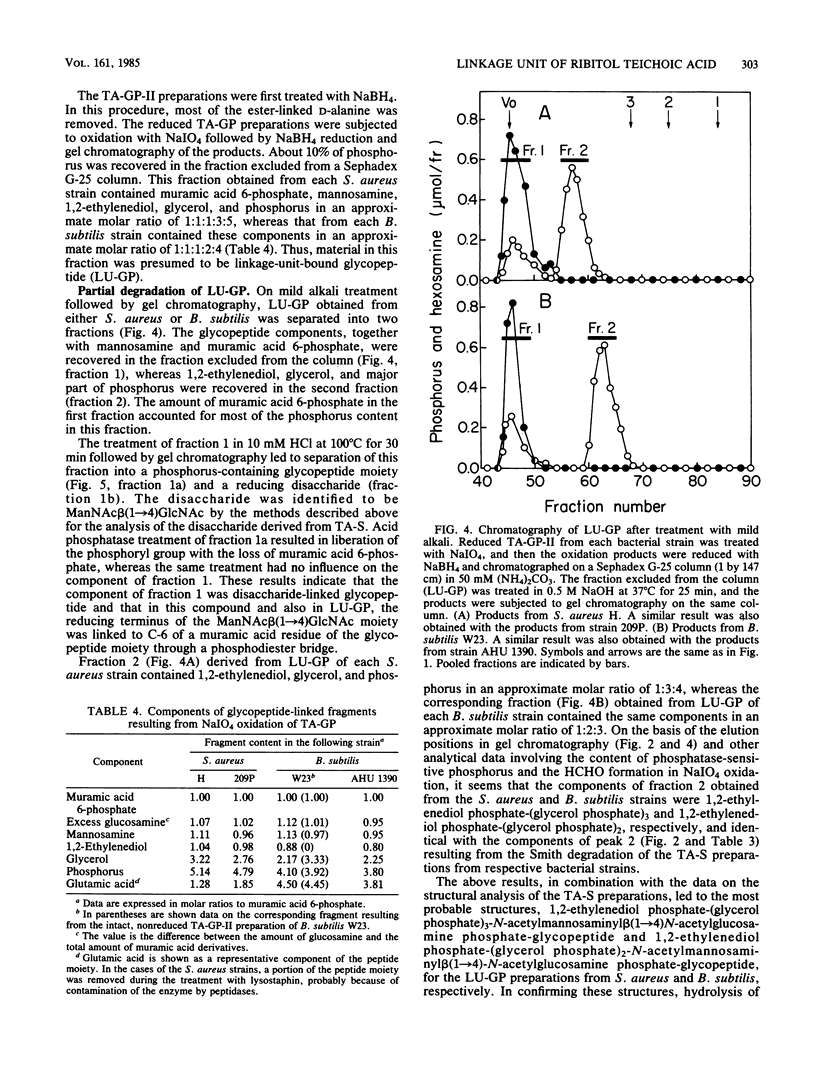
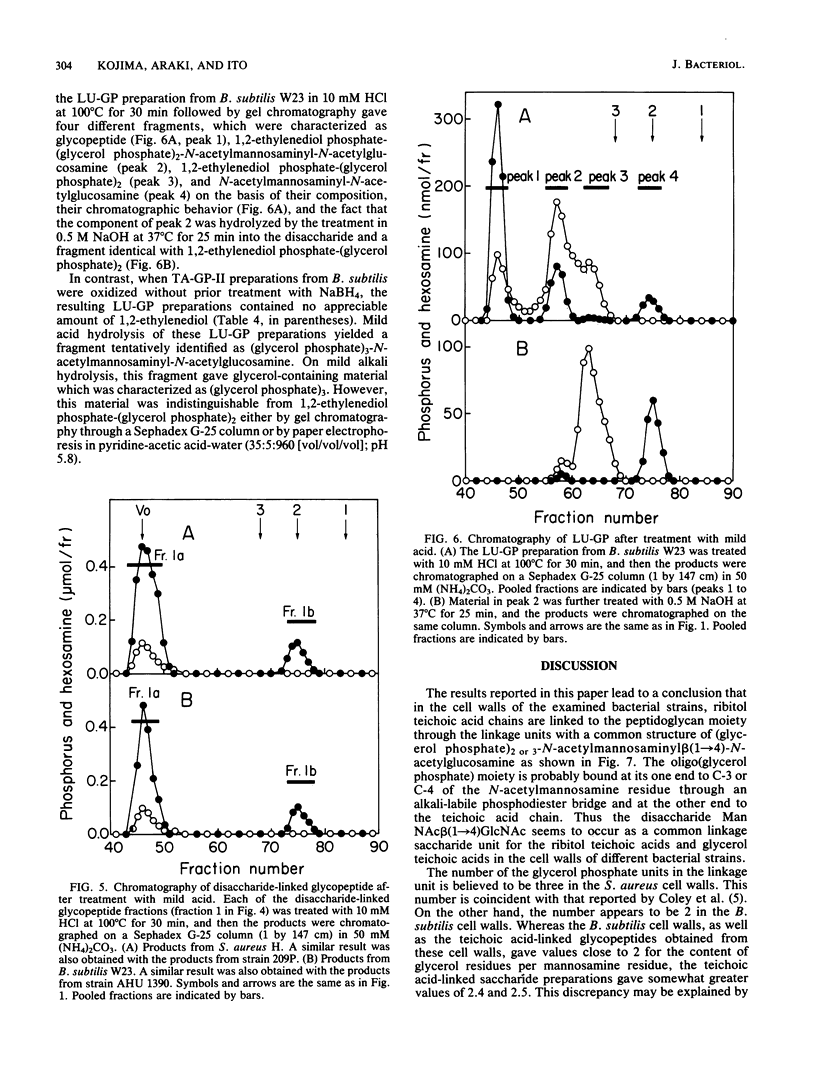
The structure of the linkage regions between ribitol teichoic acids and peptidoglycan in the cell walls of Staphylococcus aureus H and 209P and Bacillus subtilis W23 and AHU 1390 was studied. Teichoic acid-linked saccharide preparations obtained from the cell walls by heating at pH 2.5 contained mannosamine and glycerol in small amounts. On mild alkali treatment, each teichoic acid-linked saccharide preparation was split into a disaccharide identified as N-acetylmannosaminyl beta(1----4)N-acetylglucosamine and the ribitol teichoic acid moiety that contained glycerol residues. The Smith degradation of reduced samples of the teichoic acid-linked saccharide preparations from S. aureus and B. subtilis gave fragments characterized as 1,2-ethylenediol phosphate-(glycerolphosphate)3-N-acetylmannosaminyl beta(1----4)N- -acetylxylosaminitol and 1,2-ethylenediolphosphate-(glycerol phosphate)2-N-acetylmannosaminyl beta(1----4)N-acetylxylosaminitol, respectively. The binding of the disaccharide unit to peptidoglycan was confirmed by the analysis of linkage-unit-bound glycopeptides obtained from NaIO4 oxidation of teichoic acid-glycopeptide complexes. Mild alkali treatment of the linkage-unit-bound glycopeptides yielded disaccharide-linked glycopeptides, which gave the disaccharide and phosphorylated glycopeptides on mild acid treatment. Thus, it is concluded that the ribitol teichoic acid chains in the cell walls of the strains of S. aureus and B. subtilis are linked to peptidoglycan through linkage units, (glycerol phosphate)3-N-acetylmannosaminyl beta(1----4)N-acetylglucosamine and (glycerol phosphate)2-N-acetylmannosaminyl beta(1----4)N-acetylglucosamine, respectively.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Amano K., Hazama S., Araki Y., Ito E. Isolation and characterization of structural components of Bacillus cereus AHU 1356 cell walls. Eur J Biochem. 1977 May 16;75(2):513–522. doi: 10.1111/j.1432-1033.1977.tb11552.x. [DOI] [PubMed] [Google Scholar]
- Araki Y., Nakatani T., Nakayama K., Ito E. Occurrence of N-nonsubstituted glucosamine residues in peptidoglycan of lysozyme-resistant cell walls from Bacillus cereus. J Biol Chem. 1972 Oct 10;247(19):6312–6322. [PubMed] [Google Scholar]
- BADDILEY J., BUCHANAN J. G., RAJBHANDARY U. L., SANDERSON A. R. Teichoic acid from the walls of Staphylococcus aureus H. Structure of the N-acetylglucosaminyl-ribitol residues. Biochem J. 1962 Mar;82:439–448. doi: 10.1042/bj0820439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chin T., Burger M. M., Glaser L. Synthesis of teichoic acids. VI. The formation of multiple wall polymers in Bacillus subtilis W-23. Arch Biochem Biophys. 1966 Sep 26;116(1):358–367. doi: 10.1016/0003-9861(66)90042-7. [DOI] [PubMed] [Google Scholar]
- Coley J., Tarelli E., Archibald A. R., Baddiley J. The linkage between teichoic acid and peptidoglycan in bacterial cell walls. FEBS Lett. 1978 Apr 1;88(1):1–9. doi: 10.1016/0014-5793(78)80594-8. [DOI] [PubMed] [Google Scholar]
- HANAHAN D. J., OLLEY J. N. Chemical nature of monophosphoinositides. J Biol Chem. 1958 Apr;231(2):813–828. [PubMed] [Google Scholar]
- Hayashi H., Araki Y., Ito E. Occurrence of glucosamine residues with free amino groups in cell wall peptidoglycan from bacilli as a factor responsible for resistance to lysozyme. J Bacteriol. 1973 Feb;113(2):592–598. doi: 10.1128/jb.113.2.592-598.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heckels J. E., Archibald A. R., Baddiley J. Studies on the linkage between teichoic acid and peptidoglycan in a bacteriophage-resistant mutant of Staphylococcus aureus H. Biochem J. 1975 Sep;149(3):637–647. doi: 10.1042/bj1490637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heptinstall J., Coley J., Ward P. J., Archibald A. R., Baddiley J. The linkage of sugar phosphate polymer to peptidoglycan in walls of Micrococcus sp. 2102. Biochem J. 1978 Feb 1;169(2):329–336. doi: 10.1042/bj1690329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaya S., Yokoyama K., Araki Y., Ito E. N-acetylmannosaminyl(1----4)N-acetylglucosamine, a linkage unit between glycerol teichoic acid and peptidoglycan in cell walls of several Bacillus strains. J Bacteriol. 1984 Jun;158(3):990–996. doi: 10.1128/jb.158.3.990-996.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaya S., Yokoyama K., Araki Y., Ito E. Structural and biosynthetic studies on linkage region between poly(galactosylglycerol phosphate) and peptidoglycan in Bacillus coagulans. Biochem Biophys Res Commun. 1983 Feb 28;111(1):312–318. doi: 10.1016/s0006-291x(83)80153-3. [DOI] [PubMed] [Google Scholar]
- Kojima N., Araki Y., Ito E. Structure of linkage region between ribitol teichoic acid and peptidoglycan in cell walls of Staphylococcus aureus H. J Biol Chem. 1983 Aug 10;258(15):9043–9045. [PubMed] [Google Scholar]
- LOWRY O. H., ROBERTS N. R., LEINER K. Y., WU M. L., FARR A. L. The quantitative histochemistry of brain. I. Chemical methods. J Biol Chem. 1954 Mar;207(1):1–17. [PubMed] [Google Scholar]
- REISSIG J. L., STORMINGER J. L., LELOIR L. F. A modified colorimetric method for the estimation of N-acetylamino sugars. J Biol Chem. 1955 Dec;217(2):959–966. [PubMed] [Google Scholar]
- Sasaki Y., Araki Y., Ito E. Structure of teichoic-acid--glycopeptide complexes from cell walls of Bacillus cereus AHU 1030. Eur J Biochem. 1983 Apr 15;132(1):207–213. doi: 10.1111/j.1432-1033.1983.tb07349.x. [DOI] [PubMed] [Google Scholar]
- Schleifer K. H., Stackebrandt E. Molecular systematics of prokaryotes. Annu Rev Microbiol. 1983;37:143–187. doi: 10.1146/annurev.mi.37.100183.001043. [DOI] [PubMed] [Google Scholar]
- Tsuji A., Kinoshita T., Hoshino M. Microdetermination of hexosamines. Chem Pharm Bull (Tokyo) 1969 Jan;17(1):217–218. doi: 10.1248/cpb.17.217. [DOI] [PubMed] [Google Scholar]
- Yoneyama T., Koike Y., Arakawa H., Yokoyama K., Sasaki Y., Kawamura T., Araki Y., Ito E., Takao S. Distribution of mannosamine and mannosaminuronic acid among cell walls of Bacillus species. J Bacteriol. 1982 Jan;149(1):15–21. doi: 10.1128/jb.149.1.15-21.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]



