Abstract
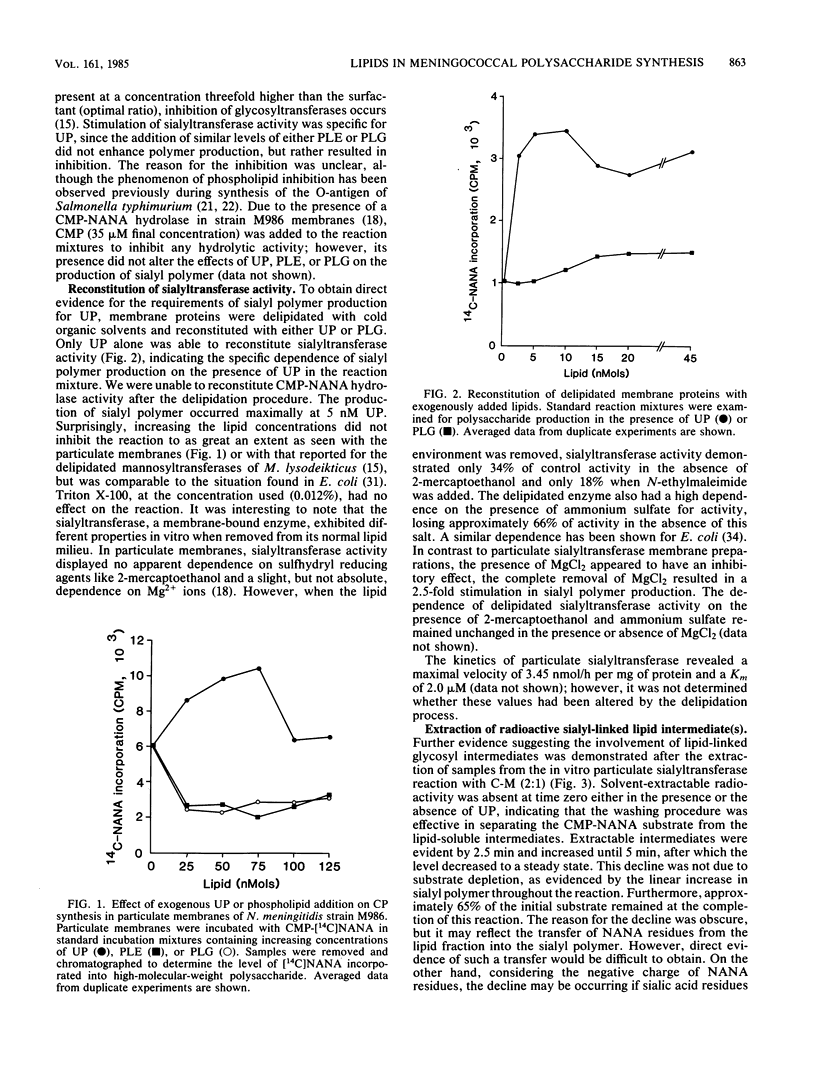
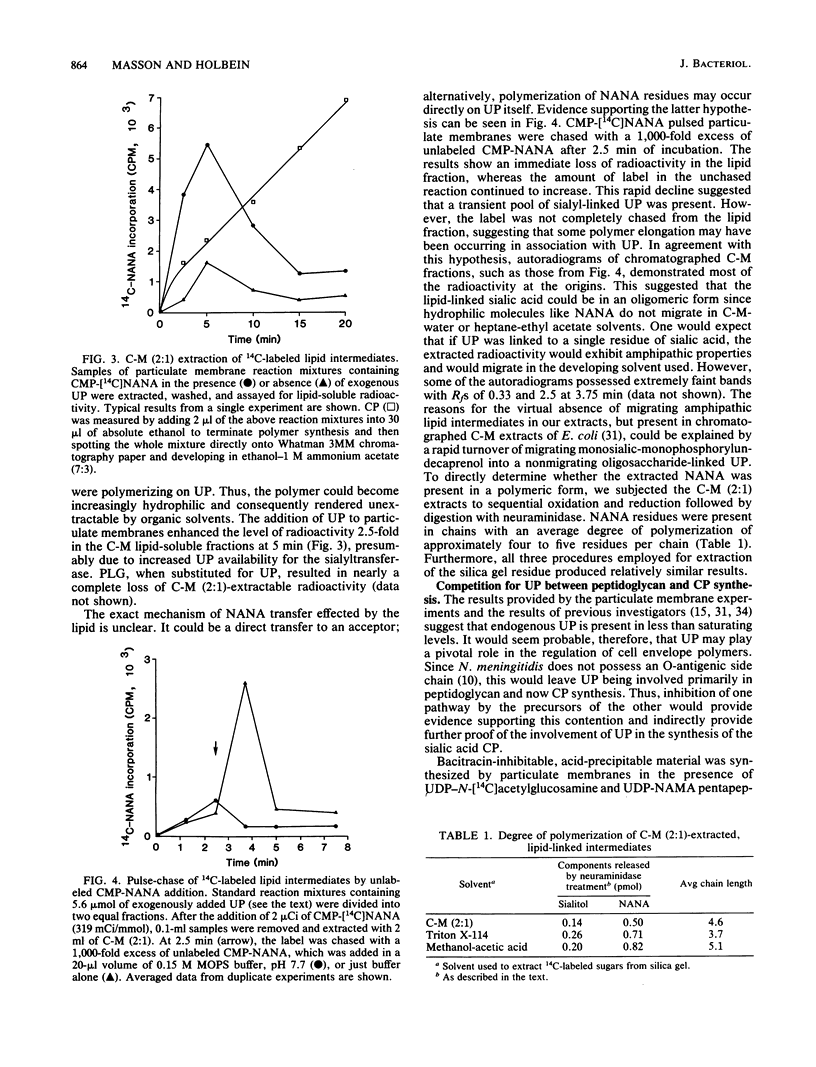
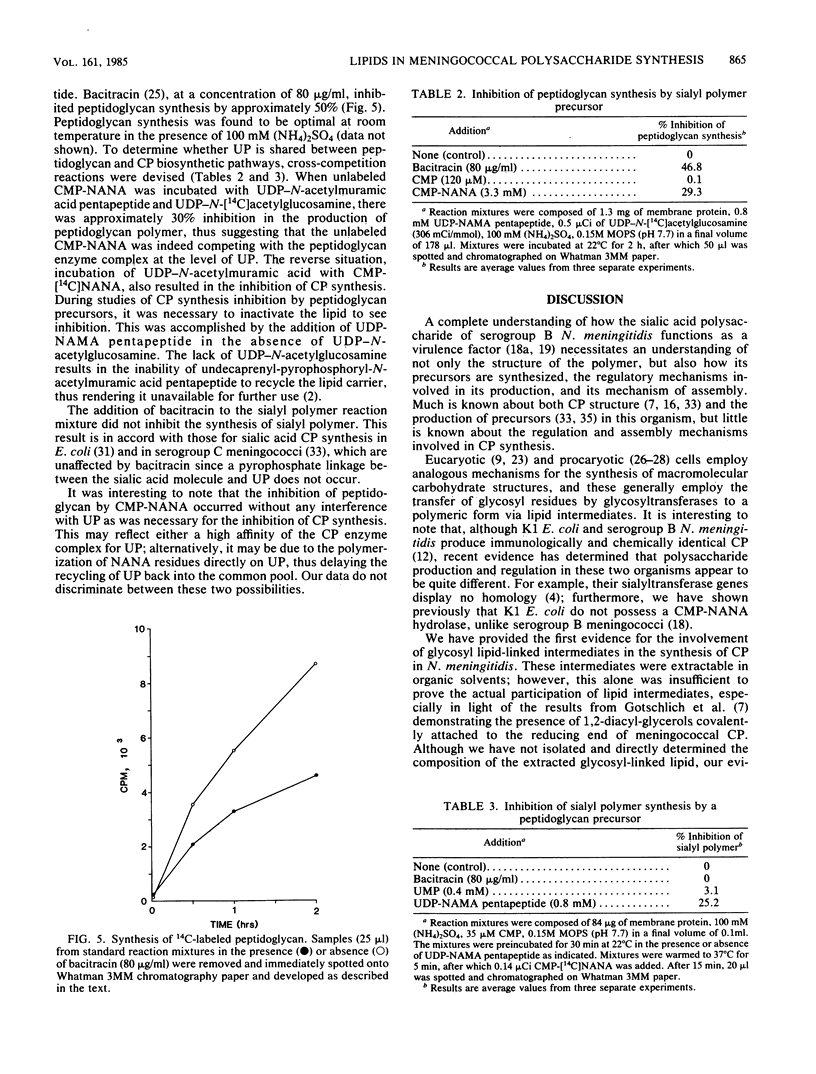
Neisseria meningitidis serogroup B strain M986 was examined for the involvement of lipid intermediate(s) participating in the biosynthesis of the sialic acid capsular polysaccharide. The addition of exogenous undecaprenyl phosphate, phosphatidylethanolamine, or phosphatidylglycerol to particulate membranes, in the presence of cytidine 5'-monophosphosialic acid, resulted in the stimulation of sialyltransferase activity specifically by undecaprenyl phosphate. Sialyltransferase activity, after delipidation of particulate membrane proteins, was specifically reconstituted by undecaprenyl phosphate. After the addition of 14C-labeled cytidine 5'-monophosphosialic acid to particulate membranes, the level of labeled lipid intermediate(s), extracted by chloroform-methanol (2:1), increased up to a maximum level between 3.75 and 5.0 min, which subsequently decreased to a lower steady-state level. Pulse-chase experiments revealed a transient, solvent-extractable, lipid-linked component. The extracted N-acetylneuraminic acid was in polymeric form. Sequential oxidation and reduction of the extracted radioactivity followed by neuraminidase treatment revealed an average degree of polymerization of four or five N-acetylneuraminic acid residues. Bacitracin-sensitive peptidoglycan was synthesized in vitro by particulate membranes. Cross-competition experiments between peptidoglycan and capsular polysaccharide synthesis by preincubation of precursors of one pathway during synthesis of the other revealed a competitive effect for a common component. This component was believed to be a common pool of undecaprenyl phosphate. A model for the production and regulation of the capsular polysaccharide is proposed.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Anderson J. S., Matsuhashi M., Haskin M. A., Strominger J. L. Biosythesis of the peptidoglycan of bacterial cell walls. II. Phospholipid carriers in the reaction sequence. J Biol Chem. 1967 Jul 10;242(13):3180–3190. [PubMed] [Google Scholar]
- Anderson R. G., Hussey H., Baddiley J. The mechanism of wall synthesis in bacteria. The organization of enzymes and isoprenoid phosphates in the membrane. Biochem J. 1972 Mar;127(1):11–25. doi: 10.1042/bj1270011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brooks D., Baddiley J. A lipid intermediate in the synthesis of a poly-(N-acetylglucosamine 1-phosphate) from the wall of Staphylococcus lactis N.C.T.C. 2102. Biochem J. 1969 Nov;115(2):307–314. doi: 10.1042/bj1150307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Echarti C., Hirschel B., Boulnois G. J., Varley J. M., Waldvogel F., Timmis K. N. Cloning and analysis of the K1 capsule biosynthesis genes of Escherichia coli: lack of homology with Neisseria meningitidis group B DNA sequences. Infect Immun. 1983 Jul;41(1):54–60. doi: 10.1128/iai.41.1.54-60.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- FOLCH J., LEES M., SLOANE STANLEY G. H. A simple method for the isolation and purification of total lipides from animal tissues. J Biol Chem. 1957 May;226(1):497–509. [PubMed] [Google Scholar]
- Fiedler F., Glaser L. The synthesis of polyribitol phosphate. II. On the mechanism of polyribitol phosphate polymerase. J Biol Chem. 1974 May 10;249(9):2690–2695. [PubMed] [Google Scholar]
- Gotschlich E. C., Fraser B. A., Nishimura O., Robbins J. B., Liu T. Y. Lipid on capsular polysaccharides of gram-negative bacteria. J Biol Chem. 1981 Sep 10;256(17):8915–8921. [PubMed] [Google Scholar]
- Hebeler B. H., Young F. E. Chemical composition and turnover of peptidoglycan in Neisseria gonorrhoeae. J Bacteriol. 1976 Jun;126(3):1180–1185. doi: 10.1128/jb.126.3.1180-1185.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hubbard S. C., Ivatt R. J. Synthesis and processing of asparagine-linked oligosaccharides. Annu Rev Biochem. 1981;50:555–583. doi: 10.1146/annurev.bi.50.070181.003011. [DOI] [PubMed] [Google Scholar]
- Jennings H. J., Bhattacharjee A. K., Kenne L., Kenny C. P., Calver G. The R-type lipopolysaccharides of Neisseria meningitidis. Can J Biochem. 1980 Feb;58(2):128–136. doi: 10.1139/o80-018. [DOI] [PubMed] [Google Scholar]
- Kanegasaki S., Wright A., Warren C. D. Chemically synthesized galactosyl ficaprenyl diphosphate as an intermediate in the biosynthesis of the Salmonella O-antigenic polysaccharide. Eur J Biochem. 1983 Jun 1;133(1):77–81. doi: 10.1111/j.1432-1033.1983.tb07431.x. [DOI] [PubMed] [Google Scholar]
- Kasper D. L., Winkelhake J. L., Zollinger W. D., Brandt B. L., Artenstein M. S. Immunochemical similarity between polysaccharide antigens of Escherichia coli 07: K1(L):NM and group B Neisseria meningitidis. J Immunol. 1973 Jan;110(1):262–268. [PubMed] [Google Scholar]
- Kundig J. D., Aminoff D., Roseman S. The sialic acids. XII. Synthesis of colominic acid by a sialyltransferase from Escherichia coli K-235. J Biol Chem. 1971 Apr 25;246(8):2543–2550. [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Lahav M., Chiu T. H., Lennarz W. J. Studies on the biosynthesis of mannan in Micrococcus lysodeikticus. II. The enzymatic synthesis of mannosyl-l-phosphoryl-undecaprenol. J Biol Chem. 1969 Nov 10;244(21):5890–5898. [PubMed] [Google Scholar]
- Liu T. Y., Gotschlich E. C., Dunne F. T., Jonssen E. K. Studies on the meningococcal polysaccharides. II. Composition and chemical properties of the group B and group C polysaccharide. J Biol Chem. 1971 Aug 10;246(15):4703–4712. [PubMed] [Google Scholar]
- Masson L., Holbein B. E. Influence of nutrient limitation and low pH on serogroup B Neisseria meningitidis capsular polysaccharide levels: correlation with virulence for mice. Infect Immun. 1985 Feb;47(2):465–471. doi: 10.1128/iai.47.2.465-471.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Masson L., Holbein B. E. Physiology of sialic acid capsular polysaccharide synthesis in serogroup B Neisseria meningitidis. J Bacteriol. 1983 May;154(2):728–736. doi: 10.1128/jb.154.2.728-736.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McArthur H. A., Roberts F. M., Hancock I. C., Baddiley J. Lipid intermediates in the biosynthesis of the linkage unit between teichoic acids and peptidoglycan. FEBS Lett. 1978 Feb 15;86(2):193–200. doi: 10.1016/0014-5793(78)80561-4. [DOI] [PubMed] [Google Scholar]
- Osborn M. J. Structure and biosynthesis of the bacterial cell wall. Annu Rev Biochem. 1969;38:501–538. doi: 10.1146/annurev.bi.38.070169.002441. [DOI] [PubMed] [Google Scholar]
- Parodi A. J., Leloir L. F. The role of lipid intermediates in the glycosylation of proteins in the eucaryotic cell. Biochim Biophys Acta. 1979 Apr 23;559(1):1–37. doi: 10.1016/0304-4157(79)90006-6. [DOI] [PubMed] [Google Scholar]
- Rohr T. E., Troy F. A. Structure and biosynthesis of surface polymers containing polysialic acid in Escherichia coli. J Biol Chem. 1980 Mar 25;255(6):2332–2342. [PubMed] [Google Scholar]
- Siewert G., Strominger J. L. Bacitracin: an inhibitor of the dephosphorylation of lipid pyrophosphate, an intermediate in the biosynthesis of the peptidoglycan of bacterial cell walls. Proc Natl Acad Sci U S A. 1967 Mar;57(3):767–773. doi: 10.1073/pnas.57.3.767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sutherland I. W. Biosynthesis of microbial exopolysaccharides. Adv Microb Physiol. 1982;23:79–150. doi: 10.1016/s0065-2911(08)60336-7. [DOI] [PubMed] [Google Scholar]
- Tonn S. J., Gander J. E. Biosynthesis of polysaccharides by prokaryotes. Annu Rev Microbiol. 1979;33:169–199. doi: 10.1146/annurev.mi.33.100179.001125. [DOI] [PubMed] [Google Scholar]
- Troy F. A., 2nd The chemistry and biosynthesis of selected bacterial capsular polymers. Annu Rev Microbiol. 1979;33:519–560. doi: 10.1146/annurev.mi.33.100179.002511. [DOI] [PubMed] [Google Scholar]
- Troy F. A., Frerman F. E., Heath E. C. The biosynthesis of capsular polysaccharide in Aerobacter aerogenes. J Biol Chem. 1971 Jan 10;246(1):118–133. [PubMed] [Google Scholar]
- Troy F. A., McCloskey M. A. Role of a membranous sialyltransferase complex in the synthesis of surface polymers containing polysialic acid in Escherichia coli. Temperature-induced alteration in the assembly process. J Biol Chem. 1979 Aug 10;254(15):7377–7387. [PubMed] [Google Scholar]
- Troy F. A., Vijay I. K., Tesche N. Role of undecaprenyl phosphate in synthesis of polymers containing sialic acid in Escherichia coli. J Biol Chem. 1975 Jan 10;250(1):156–163. [PubMed] [Google Scholar]
- Van Lenten L., Ashwell G. Studies on the chemical and enzymatic modification of glycoproteins. A general method for the tritiation of sialic acid-containing glycoproteins. J Biol Chem. 1971 Mar 25;246(6):1889–1894. [PubMed] [Google Scholar]
- Vann W. F., Liu T. Y., Robbins J. B. Cell-free biosynthesis of the O-acetylated N-acetylneuraminic acid capsular polysaccharide of group C meningococci. J Bacteriol. 1978 Mar;133(3):1300–1306. doi: 10.1128/jb.133.3.1300-1306.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vijay I. K., Troy F. A. Properties of membrane-associated sialyltransferase of Escherichia coli. J Biol Chem. 1975 Jan 10;250(1):164–170. [PubMed] [Google Scholar]
- WARREN L., BLACKLOW R. S. Biosynthesis of N-acetyl-neuraminic acid and cytidine-5'-monophospho-N-acetyl-neuraminic acid in Neisseria meningitidis. Biochem Biophys Res Commun. 1962 Jun 4;7:433–438. doi: 10.1016/0006-291x(62)90330-3. [DOI] [PubMed] [Google Scholar]
- Watkinson R. J., Hussey H., Baddiley J. Shared lipid phosphate carrier in the biosynthesis of teichoic acid and peptidoglycan. Nat New Biol. 1971 Jan 13;229(2):57–59. doi: 10.1038/newbio229057a0. [DOI] [PubMed] [Google Scholar]
- Weiner I. M., Higuchi T., Rothfield L., Saltmarsh-Andrew M., Osborn M. J., Horecker B. L. Biosynthesis of bacterial lipopolysaccharide. V. Lipid-linked intermediates in the biosynthesis of the O-antigen groups of Salmonella typhimurium. Proc Natl Acad Sci U S A. 1965 Jul;54(1):228–235. doi: 10.1073/pnas.54.1.228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams A. G., Wimpenny J. W. Extracellular polysaccharide biosynthesis by Pseudomonas NCIB 11264. Studies on precursor-forming enzymes and factors affecting exopolysaccharide production by washed suspensions. J Gen Microbiol. 1980 Jan;116(1):133–141. doi: 10.1099/00221287-116-1-133. [DOI] [PubMed] [Google Scholar]
- Willoughby E., Highasi Y., Strominger J. L. Enzymatic dephosphorylation of C 55 -isoprenylphosphate. J Biol Chem. 1972 Aug 25;247(16):5113–5115. [PubMed] [Google Scholar]
- Wright A., Dankert M., Fennessey P., Robbins P. W. Characterization of a polyisoprenoid compound functional in O-antigen biosynthesis. Proc Natl Acad Sci U S A. 1967 Jun;57(6):1798–1803. doi: 10.1073/pnas.57.6.1798. [DOI] [PMC free article] [PubMed] [Google Scholar]



