Abstract
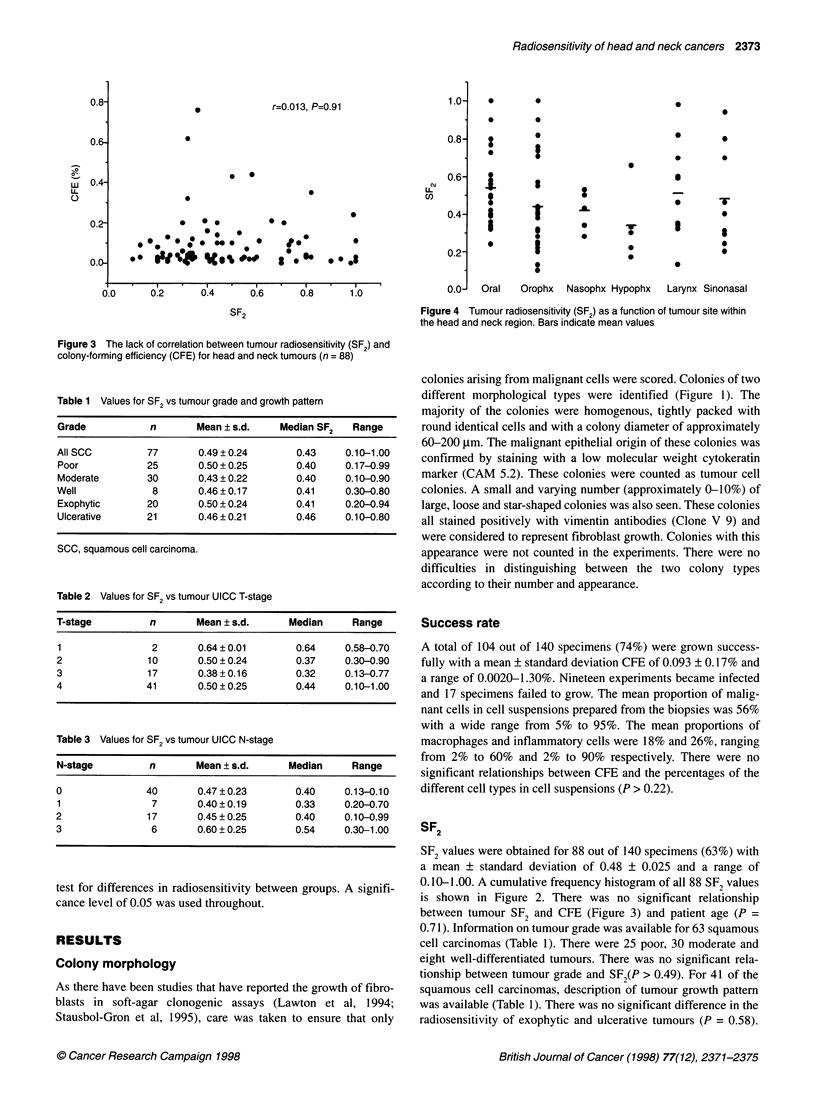
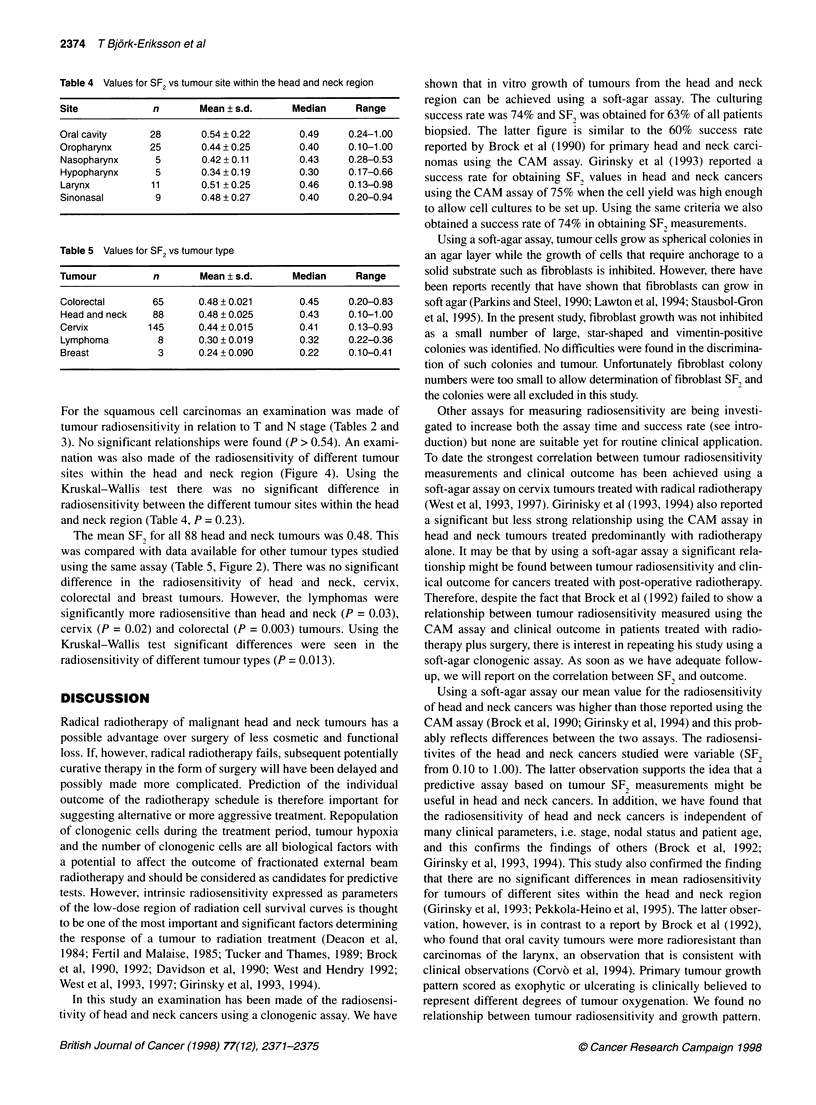
A study was made of the intrinsic radiosensitivity of 140 biopsy and surgical specimens of malignant head and neck tumours of different histologies. Using a soft-agar clonogenic assay, the material was assessed for the ability to grow in culture (colony-forming efficiency; CFE) and inherent tumour radiosensitivity (surviving fraction at 2 Gy, SF2). The success rate for obtaining growth was 74% (104/140) with a mean CFE of 0.093% (median 0.031) and a range of 0.002-1.3%. SF2 was obtained for 88 of 140 specimens, representing a success rate of 63% with a mean SF2 of 0.48 (median 0.43) and a range of 0.10-1.00. There were no significant differences in radiosensitivity between different sites of the head and neck region. There were no significant relationships between SF2 and disease stage, nodal status, tumour grade, patient age, primary tumour growth pattern and CFE. The results were compared with those for other tumour types previously analysed with the same assay. The distribution of the SF2 values for the head and neck tumours was similar to that for 145 cervix carcinomas and there was no significant difference in mean radiosensitivity between the two tumour types. Also, there was no significant difference in radiosensitivity between head and neck tumours and either breast or colorectal cancers. However, a group of eight lymphomas was significantly more radiosensitive. These results confirm the feasibility of carrying out radiosensitivity measurements using a soft-agar clonogenic assay on head and neck tumours. In addition, the work has shown that radiosensitivity is independent of many clinical parameters and that the mean value is similar to that reported for cervix carcinomas.
Full text
PDF

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Brock W. A., Baker F. L., Peters L. J. Radiosensitivity of human head and neck squamous cell carcinomas in primary culture and its potential as a predictive assay of tumor radiocurability. Int J Radiat Biol. 1989 Nov;56(5):751–760. doi: 10.1080/09553008914552001. [DOI] [PubMed] [Google Scholar]
- Brock W. A., Baker F. L., Wike J. L., Sivon S. L., Peters L. J. Cellular radiosensitivity of primary head and neck squamous cell carcinomas and local tumor control. Int J Radiat Oncol Biol Phys. 1990 Jun;18(6):1283–1286. doi: 10.1016/0360-3016(90)90298-x. [DOI] [PubMed] [Google Scholar]
- Coco-Martin J. M., Smeets M. F., Poggensee M., Mooren E., Hofland I., van den Brug M., Ottenheim C., Bartelink H., Begg A. C. Use of fluorescence in situ hybridization to measure chromosome aberrations as a predictor of radiosensitivity in human tumour cells. Int J Radiat Biol. 1994 Sep;66(3):297–307. doi: 10.1080/09553009414551231. [DOI] [PubMed] [Google Scholar]
- Corvò R., Sanguineti G., Scala M., Garaventa G., Santelli A., Barbieri M., Vitale V. Primary site as predictive factor of local control in advanced head and neck tumors treated by concomitant boost accelerated radiotherapy. Tumori. 1994 Apr 30;80(2):135–138. doi: 10.1177/030089169408000210. [DOI] [PubMed] [Google Scholar]
- Davidson S. E., West C. M., Roberts S. A., Hendry J. H., Hunter R. D. Radiosensitivity testing of primary cervical carcinoma: evaluation of intra- and inter-tumour heterogeneity. Radiother Oncol. 1990 Aug;18(4):349–356. doi: 10.1016/0167-8140(90)90115-d. [DOI] [PubMed] [Google Scholar]
- Deacon J., Peckham M. J., Steel G. G. The radioresponsiveness of human tumours and the initial slope of the cell survival curve. Radiother Oncol. 1984 Dec;2(4):317–323. doi: 10.1016/s0167-8140(84)80074-2. [DOI] [PubMed] [Google Scholar]
- Fertil B., Malaise E. P. Intrinsic radiosensitivity of human cell lines is correlated with radioresponsiveness of human tumors: analysis of 101 published survival curves. Int J Radiat Oncol Biol Phys. 1985 Sep;11(9):1699–1707. doi: 10.1016/0360-3016(85)90223-8. [DOI] [PubMed] [Google Scholar]
- Girinsky T., Bernheim A., Lubin R., Tavakoli-Razavi T., Baker F., Janot F., Wibault P., Cosset J. M., Duvillard P., Duverger A. In vitro parameters and treatment outcome in head and neck cancers treated with surgery and/or radiation: cell characterization and correlations with local control and overall survival. Int J Radiat Oncol Biol Phys. 1994 Nov 15;30(4):789–794. doi: 10.1016/0360-3016(94)90350-6. [DOI] [PubMed] [Google Scholar]
- Girinsky T., Lubin R., Pignon J. P., Chavaudra N., Gazeau J., Dubray B., Cosset J. M., Socie G., Fertil B. Predictive value of in vitro radiosensitivity parameters in head and neck cancers and cervical carcinomas: preliminary correlations with local control and overall survival. Int J Radiat Oncol Biol Phys. 1993 Jan;25(1):3–7. doi: 10.1016/0360-3016(93)90137-k. [DOI] [PubMed] [Google Scholar]
- Lawton P. A., Hodgkiss R. J., Eyden B. P., Joiner M. C. Growth of fibroblasts as a potential confounding factor in soft agar clonogenic assays for tumour cell radiosensitivity. Radiother Oncol. 1994 Sep;32(3):218–225. doi: 10.1016/0167-8140(94)90021-3. [DOI] [PubMed] [Google Scholar]
- Parkins C. S., Steel G. G. Growth and radiosensitivity testing of human tumour cells using the adhesive tumour cell culture system. Br J Cancer. 1990 Dec;62(6):935–941. doi: 10.1038/bjc.1990.413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pekkola-Heino K., Jaakkola M., Kulmala J., Grénman R. Comparison of cellular radiosensitivity between different localizations of head and neck squamous-cell carcinoma. J Cancer Res Clin Oncol. 1995;121(8):452–456. doi: 10.1007/BF01218360. [DOI] [PubMed] [Google Scholar]
- Ramsay J., Ward R., Bleehen N. M. Radiosensitivity testing of human malignant gliomas. Int J Radiat Oncol Biol Phys. 1992;24(4):675–680. doi: 10.1016/0360-3016(92)90713-r. [DOI] [PubMed] [Google Scholar]
- Rofstad E. K., Wahl A., Brustad T. Radiation sensitivity in vitro of cells isolated from human tumor surgical specimens. Cancer Res. 1987 Jan 1;47(1):106–110. [PubMed] [Google Scholar]
- Schwartz J. L., Mustafi R., Beckett M. A., Weichselbaum R. R. DNA double-strand break rejoining rates, inherent radiation sensitivity and human tumour response to radiotherapy. Br J Cancer. 1996 Jul;74(1):37–42. doi: 10.1038/bjc.1996.312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stausbøl-Grøn B., Nielsen O. S., Møller Bentzen S., Overgaard J. Selective assessment of in vitro radiosensitivity of tumour cells and fibroblasts from single tumour biopsies using immunocytochemical identification of colonies in the soft agar clonogenic assay. Radiother Oncol. 1995 Nov;37(2):87–99. doi: 10.1016/0167-8140(95)98589-d. [DOI] [PubMed] [Google Scholar]
- Tucker S. L., Thames H. D., Jr The effect of patient-to-patient variability on the accuracy of predictive assays of tumor response to radiotherapy: a theoretical evaluation. Int J Radiat Oncol Biol Phys. 1989 Jul;17(1):145–157. doi: 10.1016/0360-3016(89)90382-9. [DOI] [PubMed] [Google Scholar]
- West C. M., Davidson S. E., Pool C., James R. D., Schofield P. F. Lack of a relationship between colony-forming efficiency and surviving fraction at 2 Gy. Radiat Res. 1991 May;126(2):260–263. [PubMed] [Google Scholar]
- West C. M., Davidson S. E., Roberts S. A., Hunter R. D. Intrinsic radiosensitivity and prediction of patient response to radiotherapy for carcinoma of the cervix. Br J Cancer. 1993 Oct;68(4):819–823. doi: 10.1038/bjc.1993.434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- West C. M., Hendry J. H. Intrinsic radiosensitivity as a predictor of patient response to radiotherapy. BJR Suppl. 1992;24:146–152. [PubMed] [Google Scholar]
- West C. M. Invited review: intrinsic radiosensitivity as a predictor of patient response to radiotherapy. Br J Radiol. 1995 Aug;68(812):827–837. doi: 10.1259/0007-1285-68-812-827. [DOI] [PubMed] [Google Scholar]
- Zaffaroni N., Orlandi L., Villa R., Bearzatto A., Rofstad E. K., Silvestrini R. DNA double-strand break repair and radiation response in human tumour primary cultures. Int J Radiat Biol. 1994 Sep;66(3):279–285. doi: 10.1080/09553009414551211. [DOI] [PubMed] [Google Scholar]
- Zölzer F., Alberti W., Pelzer T., Lamberti G., Hülskamp F. H., Streffer C. Changes in S-phase fraction and micronucleus frequency as prognostic factors in radiotherapy of cervical carcinoma. Radiother Oncol. 1995 Aug;36(2):128–132. doi: 10.1016/0167-8140(95)01601-c. [DOI] [PubMed] [Google Scholar]