Abstract
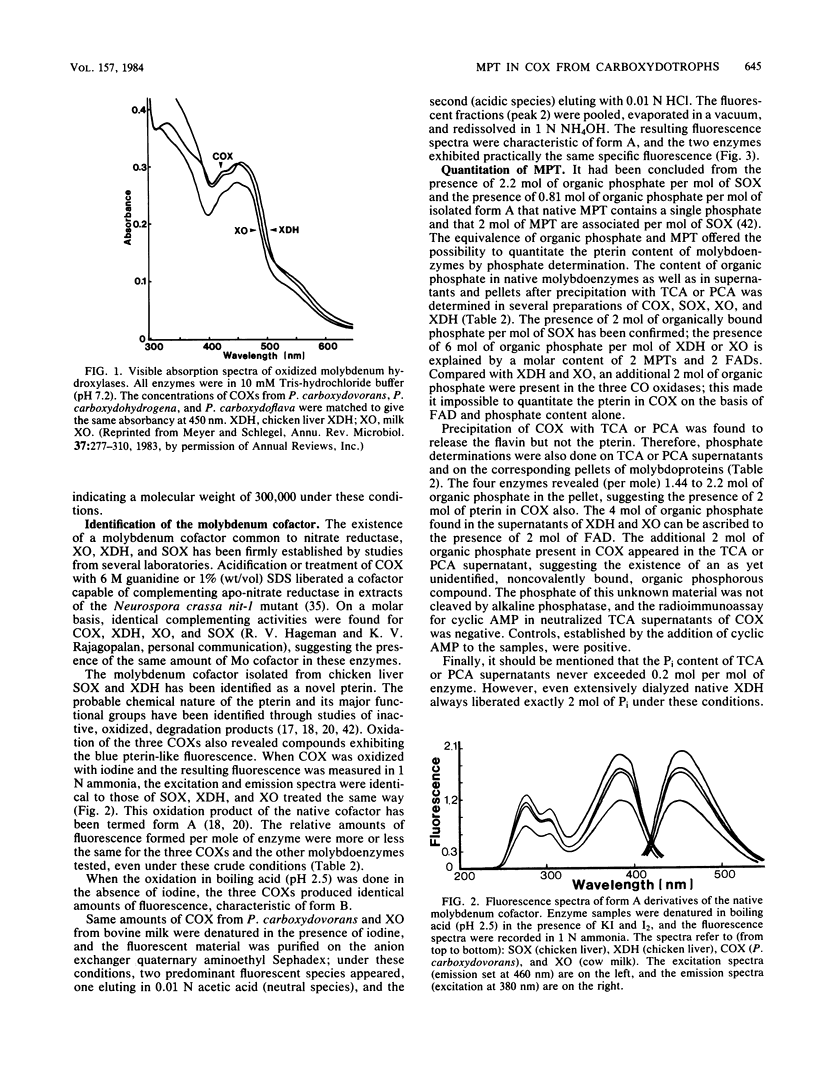
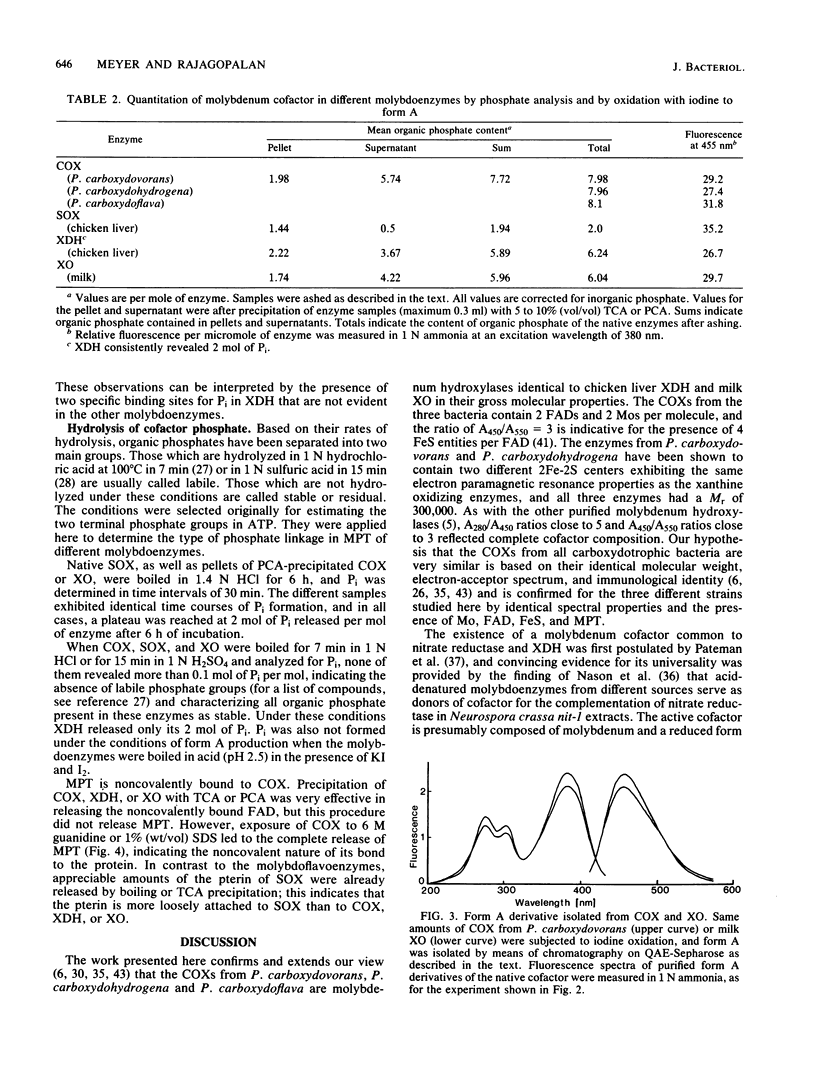
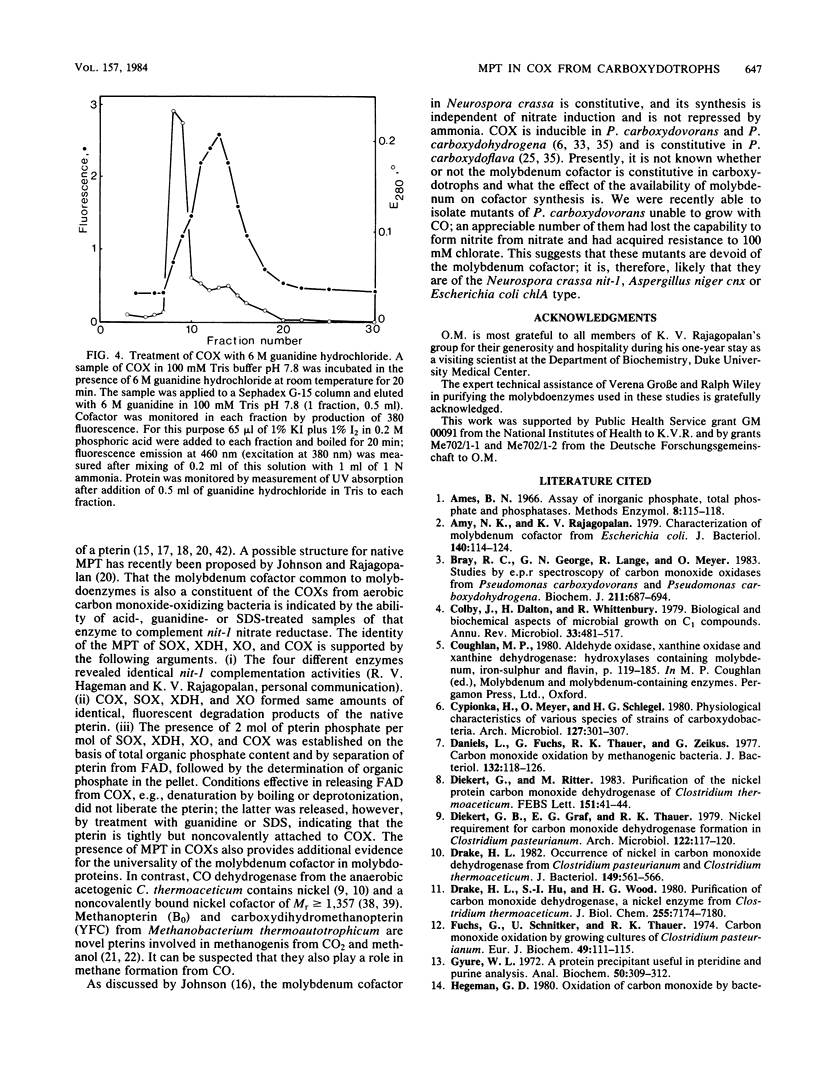
The carbon monoxide oxidases (COXs) purified from the carboxydotrophic bacteria Pseudomonas carboxydohydrogena and Pseudomonas carboxydoflava were found to be molybdenum hydroxylases, identical in cofactor composition and spectral properties to the recently characterized enzyme from Pseudomonas carboxydovorans (O. Meyer, J. Biol. Chem. 257:1333-1341, 1982). All three enzymes exhibited a cofactor composition of two flavin adenine dinucleotides, two molybdenums, eight irons and eight labile sulfides per dimeric molecule, typical for molybdenum-containing iron-sulfur flavoproteins. The millimolar extinction coefficient of the COXs at 450 nm was 72 (per two flavin adenine dinucleotides), a value similar to that of milk xanthine oxidase and chicken liver xanthine dehydrogenase at 450 nm. That molybdopterin, the novel prosthetic group of the molybdenum cofactor of a variety of molybdoenzymes (J. Johnson and K. V. Rajagopalan, Proc. Natl. Acad. Sci. U.S.A. 79:6856-6860, 1982) is also a constituent of COXs from carboxydotrophic bacteria is indicated by the formation of identical fluorescent cofactor derivatives, by complementation of the nitrate reductase activity in extracts of Neurospora crassa nit-l, and by the presence of organic phosphate additional to flavin adenine dinucleotides. Molybdopterin is tightly but noncovalently bound to the protein. COX, sulfite oxidase, xanthine oxidase, and xanthine dehydrogenase each contains 2 mol of molybdopterin per mol of enzyme. The presence of a trichloroacetic acid-releasable, so-far-unidentified, phosphorous-containing moiety in COX is suggested by the results of phosphate analysis.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Amy N. K., Rajagopalan K. V. Characterization of molybdenum cofactor from Escherichia coli. J Bacteriol. 1979 Oct;140(1):114–124. doi: 10.1128/jb.140.1.114-124.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bray R. C., George G. N., Lange R., Meyer O. Studies by e.p.r. spectroscopy of carbon monoxide oxidases from Pseudomonas carboxydovorans and Pseudomonas carboxydohydrogena. Biochem J. 1983 Jun 1;211(3):687–694. doi: 10.1042/bj2110687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colby J., Dalton H., Whittenbury R. Biological and biochemical aspects of microbial growth on C1 compounds. Annu Rev Microbiol. 1979;33:481–517. doi: 10.1146/annurev.mi.33.100179.002405. [DOI] [PubMed] [Google Scholar]
- Daniels L., Fuchs G., Thauer R. K., Zeikus J. G. Carbon monoxide oxidation by methanogenic bacteria. J Bacteriol. 1977 Oct;132(1):118–126. doi: 10.1128/jb.132.1.118-126.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diekert G., Ritter M. Purification of the nickel protein carbon monoxide dehydrogenase of Clostridium thermoaceticum. FEBS Lett. 1983 Jan 10;151(1):41–44. doi: 10.1016/0014-5793(83)80338-x. [DOI] [PubMed] [Google Scholar]
- Drake H. L., Hu S. I., Wood H. G. Purification of carbon monoxide dehydrogenase, a nickel enzyme from Clostridium thermocaceticum. J Biol Chem. 1980 Aug 10;255(15):7174–7180. [PubMed] [Google Scholar]
- Drake H. L. Occurrence of nickel in carbon monoxide dehydrogenase from Clostridium pasteurianum and Clostridium thermoaceticum. J Bacteriol. 1982 Feb;149(2):561–566. doi: 10.1128/jb.149.2.561-566.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- ERNSTER L., LINDBERG O. Determination of organic phosphorus compounds by phosphate analysis. Methods Biochem Anal. 1956;3:1–22. doi: 10.1002/9780470110195.ch1. [DOI] [PubMed] [Google Scholar]
- Fuchs G., Schnitker U., Thauer R. K. Carbon monoxide oxidation by growing cultures of Clostridium pasteurianum. Eur J Biochem. 1974 Nov 1;49(1):111–115. doi: 10.1111/j.1432-1033.1974.tb03816.x. [DOI] [PubMed] [Google Scholar]
- Gyure W. L. A protein precipitant useful in pteridine and purine analysis. Anal Biochem. 1972 Nov;50(1):309–312. doi: 10.1016/0003-2697(72)90508-8. [DOI] [PubMed] [Google Scholar]
- Ishizuka M., Ushio K., Toraya T., Fukui S. Formation of thieno[3,2-g]pterines from the molybdenum cofactor. Biochem Biophys Res Commun. 1983 Mar 16;111(2):537–543. doi: 10.1016/0006-291x(83)90340-6. [DOI] [PubMed] [Google Scholar]
- Johnson J. L., Hainline B. E., Rajagopalan K. V. Characterization of the molybdenum cofactor of sulfite oxidase, xanthine, oxidase, and nitrate reductase. Identification of a pteridine as a structural component. J Biol Chem. 1980 Mar 10;255(5):1783–1786. [PubMed] [Google Scholar]
- Johnson J. L., Rajagopalan K. V. Purification and properties of sulfite oxidase from human liver. J Clin Invest. 1976 Sep;58(3):543–550. doi: 10.1172/JCI108499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson J. L., Rajagopalan K. V. Structural and metabolic relationship between the molybdenum cofactor and urothione. Proc Natl Acad Sci U S A. 1982 Nov;79(22):6856–6860. doi: 10.1073/pnas.79.22.6856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keltjens J. T., Daniels L., Jannsen H. G., Borm P. J., Vogels G. D. A novel one-carbon carrier (carboxy-5,6,7,8-tetrahydromethanopterin) isolated from Methanobacterium thermoautotrophicum and derived from methanopterin. Eur J Biochem. 1983 Feb 15;130(3):545–552. doi: 10.1111/j.1432-1033.1983.tb07184.x. [DOI] [PubMed] [Google Scholar]
- Kessler D. L., Rajagopalan K. V. Purification and properties of sulfite oxidase from chicken liver. Presence of molybdenum in sulfite oxidase from diverse sources. J Biol Chem. 1972 Oct 25;247(20):6566–6573. [PubMed] [Google Scholar]
- Kim Y. M., Hegeman G. D. Purification and some properties of carbon monoxide dehydrogenase from Pseudomonas carboxydohydrogena. J Bacteriol. 1981 Dec;148(3):904–911. doi: 10.1128/jb.148.3.904-911.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meyer O. Chemical and spectral properties of carbon monoxide: methylene blue oxidoreductase. The molybdenum-containing iron-sulfur flavoprotein from Pseudomonas carboxydovorans. J Biol Chem. 1982 Feb 10;257(3):1333–1341. [PubMed] [Google Scholar]
- Meyer O., Schlegel H. G. Biology of aerobic carbon monoxide-oxidizing bacteria. Annu Rev Microbiol. 1983;37:277–310. doi: 10.1146/annurev.mi.37.100183.001425. [DOI] [PubMed] [Google Scholar]
- Meyer O., Schlegel H. G. Carbon monoxide:methylene blue oxidoreductase from Pseudomonas carboxydovorans. J Bacteriol. 1980 Jan;141(1):74–80. doi: 10.1128/jb.141.1.74-80.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meyer O., Schlegel H. G. Oxidation of carbon monoxide in cell extracts of Pseudomonas carboxydovorans. J Bacteriol. 1979 Feb;137(2):811–817. doi: 10.1128/jb.137.2.811-817.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meyer O., Schlegel H. G. Reisolation of the carbon monoxide utilizing hydrogen bacterium Pseudomonas carboxydovorans (Kistner) comb. nov. Arch Microbiol. 1978 Jul;118(1):35–43. doi: 10.1007/BF00406071. [DOI] [PubMed] [Google Scholar]
- Nason A., Lee K. Y., Pan S. S., Ketchum P. A., Lamberti A., DeVries J. Invitro formation of assimilatory reduced nicotinamide adenine dinucleotide phosphate: nitrate reductase from a Neurospora mutant and a component of molybdenum-enzymes. Proc Natl Acad Sci U S A. 1971 Dec;68(12):3242–3246. doi: 10.1073/pnas.68.12.3242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- PATEMAN J. A., COVE D. J., REVER B. M., ROBERTS D. B. A COMMON CO-FACTOR FOR NITRATE REDUCTASE AND XANTHINE DEHYDROGENASE WHICH ALSO REGULATES THE SYNTHESIS OF NITRATE REDUCTASE. Nature. 1964 Jan 4;201:58–60. doi: 10.1038/201058a0. [DOI] [PubMed] [Google Scholar]
- Ragsdale S. W., Clark J. E., Ljungdahl L. G., Lundie L. L., Drake H. L. Properties of purified carbon monoxide dehydrogenase from Clostridium thermoaceticum, a nickel, iron-sulfur protein. J Biol Chem. 1983 Feb 25;258(4):2364–2369. [PubMed] [Google Scholar]
- Ragsdale S. W., Ljungdahl L. G., DerVartanian D. V. EPR evidence for nickel-substrate interaction in carbon monoxide dehydrogenase from Clostridium thermoaceticum. Biochem Biophys Res Commun. 1982 Sep 30;108(2):658–663. doi: 10.1016/0006-291x(82)90880-4. [DOI] [PubMed] [Google Scholar]
- Rajagopalan K. V., Johnson J. L., Hainline B. E. The pterin of the molybdenum cofactor. Fed Proc. 1982 Jul;41(9):2608–2612. [PubMed] [Google Scholar]
- Thauer R. K., Fuchs G., Käufer B., Schnitker U. Carbon-monoxide oxidation in cell-free extracts of Clostridium pasteurianum. Eur J Biochem. 1974 Jun 15;45(2):343–349. doi: 10.1111/j.1432-1033.1974.tb03559.x. [DOI] [PubMed] [Google Scholar]
- Wakim B. T., Uffen R. L. Membrane association of the carbon monoxide oxidation system in Rhodopseudomonas gelatinosa. J Bacteriol. 1983 Jan;153(1):571–573. doi: 10.1128/jb.153.1.571-573.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Waud W. R., Brady F. O., Wiley R. D., Rajagopalan K. V. A new purification procedure for bovine milk xanthine oxidase: effect of proteolysis on the subunit structure. Arch Biochem Biophys. 1975 Aug;169(2):695–701. doi: 10.1016/0003-9861(75)90214-3. [DOI] [PubMed] [Google Scholar]
- YAGI T. Enzymic oxidation of carbon monoxide. Biochim Biophys Acta. 1958 Oct;30(1):194–195. doi: 10.1016/0006-3002(58)90263-4. [DOI] [PubMed] [Google Scholar]
- YAGI T., TAMIYA N. Enzymic oxidation of carbon monoxide. III. Reversibility. Biochim Biophys Acta. 1962 Dec 17;65:508–509. doi: 10.1016/0006-3002(62)90454-7. [DOI] [PubMed] [Google Scholar]



