Abstract
We report on the toxicity of ZnO nanoparticles (NPs) to gram-negative and gram-positive bacterial systems, Escherichia coli (E. coli) and Staphylococcus aureus (S. aureus), and primary human immune cells. ZnO NP (~13 nm) showed complete inhibition of E. coli growth at concentrations ⩾ 3.4 mM, whereas growth of S. aureus was completely inhibited for ⩾1 mM. Parallel experiments using flow cytometry based assays clearly demonstrated that growth inhibitory properties of ZnO NP were accompanied by a corresponding loss of cell viability. Identical ZnO NP had minimal effects on primary human T cell viability at concentrations toxic to both gram-negative and gram-positive bacteria. Collectively, these experiments demonstrate selectivity in the toxic nature of ZnO NP to different bacterial systems and human T lymphocytes. Developing selective toxicity to biological systems and controlling it by NP design could lead to biomedical and antibacterial applications.
Nanoparticles (NPs) with sizes in the 1 – 100 nm range, comparable to the size range of biological structures1 are attractive materials for the manipulation, sensing, and detection of biological systems.2–4 When reduced to the nanoscale realm, many benign materials develop toxicity.5–8 The increased presence of NP in the environment suggests that a fundamental understanding of their mode and range of toxicity is needed. If NP displays selective toxicity to biological systems and this property can be controlled by their design, it could lead to biomedical/antibacterial applications. On the other hand, developing methods to prepare NP with reduced toxicity will be useful for applications such as cell imaging,2 drug delivery,3 and cancer therapy.4
Recent studies have shown that NP of some materials, including metal oxides, can induce cell death in eukaryotic cells5,6 and growth inhibition in prokaryotic cells7,8 due to cytotoxicity. Most studies reporting on metal oxide NP toxicity have focused on mammalian cells, primarily on transformed cancer cell lines; however, their effects on bacterial systems have not been investigated except in a few recent studies.7,8 Brayner et al.7 and Thill et al.8 investigated the toxicity of 11 nm ZnO and 7 nm CeO2 NPs on E. coli and reported significant growth inhibition at concentrations >3 mM. Although these few investigations on prokaryotic systems7,8 have shown growth inhibition, they did not determine if this response was due to bacterial cell death or simply prevention of growth. The purpose of this study is to determine ZnO NP toxicity—are these NPs toxic to both gram-positive and gram-negative bacteria as well as primary human immune cells? Is there a difference in toxicity levels between any of these cell types? No such studies have been conducted on ZnO NP to date.
Zinc oxide NP was synthesized in diethylene glycol (DEG) medium by forced hydrolysis of zinc acetate at 160 °C (Ref. 9) and was thoroughly characterized using x-ray diffraction (XRD), spectrophotometry, and transmission electron microscopy (TEM). NP-induced toxicity tests on bacteria were conducted in liquid and solid Luria-Bertani (LB) media containing different concentrations of ZnO NP in the 0–10 mM range. Bacterial cultures were grown in NP-free LB media overnight at 37 °C with continuous shaking at ~200 rpm. To assess toxicity thresholds of ZnO NPs to E. coli and S. aureus, cultures were plated on LB containing ZnO NP, incubated overnight at 37 °C, and colony forming units (CFUs) enumerated. DEG had no effect on bacterial growth and colony formation (data not shown). Time dependent NP toxicity tests were performed as follows. Equal densities of overnight S. aureus and E. coli cultures (based on OD600 nm values) were used to inoculate LB broth with and without NP. Broth cultures were incubated with shaking as above, sampled repeatedly, and viable cell densities were measured via CFU enumeration by plating on NP-free LB media. Additional experiments using bacteria and primary human T lymphocytes evaluated the effects of ZnO NP resuspended in phosphate buffered saline (PBS). No treatment-difference was observed between DEG suspended and PBS suspended ZnO NP (data not shown).
NP-induced toxicity tests on human T lymphocytes were performed as follows. First, peripheral blood mononuclear cells were obtained by Ficoll-Hypaque (Histopaque-1077, Sigma, St. Louis, MO) gradient centrifugation using heparinized blood samples from healthy volunteers.10 This cell mixture was washed three times with Hank’s buffer (Sigma) and incubated at 1 × 106 cells/ml in Roswell Park Memorial Institute (RPMI)-1640 (Sigma) containing 10% fetal bovine serum. CD4+ T cells were subsequently isolated using negative immunomagnetic selection per manufacturer’s instructions using a cocktail of antibodies against CD45RO, CD8, CD19, CD14, CD16, CD56, CD8, and glycophorin A (StemCell Technologies, Vancouver, B.C.) with collection of unlabeled T cells (typically >96% CD4+ and >93% viable). Purified CD4+ T cells were cultured in RPMI/10% fetol calf serum at 1 × 106 cells/ml in 96-well microtiter plates and treated with various concentrations of commercially available (Aldrich) bulk ZnO powder or laboratory synthesized ZnO NP resuspended in PBS. After 24 h of culture, cells were dually stained with propidium iodide (PI) (BD Biosciences) to monitor loss of membrane integrity and a fluorescein isothiocyanate-labeled anti-CD4 antibody (Beckman Coulter) to stain T cells.10 10 000 events gated on CD4 positive cells were analyzed for changes in PI staining to allow quantification of cell death. NP was excluded from analysis based on absence of fluorescence.
For flow cytometry viability assays, a two color live/dead BacLight kit (molecular probes, Carlsbad, CA) was used. Bacterial cultures were grown for 3 h and subsequently treated with ZnO NP resuspended in PBS for 15 h. Populations of live and dead bacteria were discriminated based on green (Syto9) and red (PI) fluorescence.
Room temperature XRD and optical spectra [shown in Fig. 1(a)] of powder ZnO NP collected after separating them from DEG medium via centrifugation confirmed the wurtzite ZnO phase. Optical spectra of ZnO NP, shown in Fig. 1(a), displayed band gap energy of 3.33 eV which is close to bulk ZnO. TEM images shown in Fig. 1(b) indicate an average particle size of ~13 nm, in agreement with the crystallite size estimated using XRD linewidth.11 Electron diffraction data also confirmed the ZnO phase [inset of Fig. 1(b)]. Although the average particle size was ~13 nm, a range of particle sizes (7–19 nm) was observed in the TEM images; the distribution of which is presented in Fig. 1(c).
FIG. 1.
(a) Optical absorption spectrum of ZnO NP. The inset shows the XRD pattern of the sample and (b) TEM image of the ZnO NPs. The inset shows a selected area electron diffraction pattern confirming the crystalline ZnO phase and (c) plot showing particle size distribution.
Figure 2(a) shows the number of CFU produced by E. coli and S. aureus after being grown in the presence of ZnO NP overnight. ZnO NP prevented growth of E. coli at concentrations ⩾3.4 mM, whereas concentrations ⩾1 mM prevented growth of S. aureus. Observed toxicity of ZnO NP to E. coli is similar to that reported recently by Brayner et al.7 Our experiments build on this finding by comparing the toxicity of the same NP to two different bacterial systems. Further, the experiments by Brayner et al.,7 and some of those performed here [Fig. 2(a)], were not designed to establish if the bacterial response to ZnO NP was bacteriocidal (e.g., inducing cell death) or bacteriostatic (e.g., preventing cell growth) in nature. The consistent exposure of bacterial cells to ZnO NP in these experiments precludes the ability to determine if the observed absence of CFU’s for ZnO NP concentrations ⩾3.4 and 1 mM for E. coli and S. aureus, respectively, was due to bacterial cell death or prevention of cell growth.
FIG. 2.
(Color online) (a) Means and standard errors of CFU per milliliter of E. coli and S. aureus incubated for 24 h in LB media in the presence of different concentrations of ZnO NP; n=3. X and * indicate measured ZnO NP concentrations at which no CFU counts of E. coli and S. aureus, respectively, were observed; (b) viability of primary human CD4+ T cells isolated from peripheral blood samples treated with various concentrations of ZnO NP or bulk ZnO for 20 hrs; n=3.
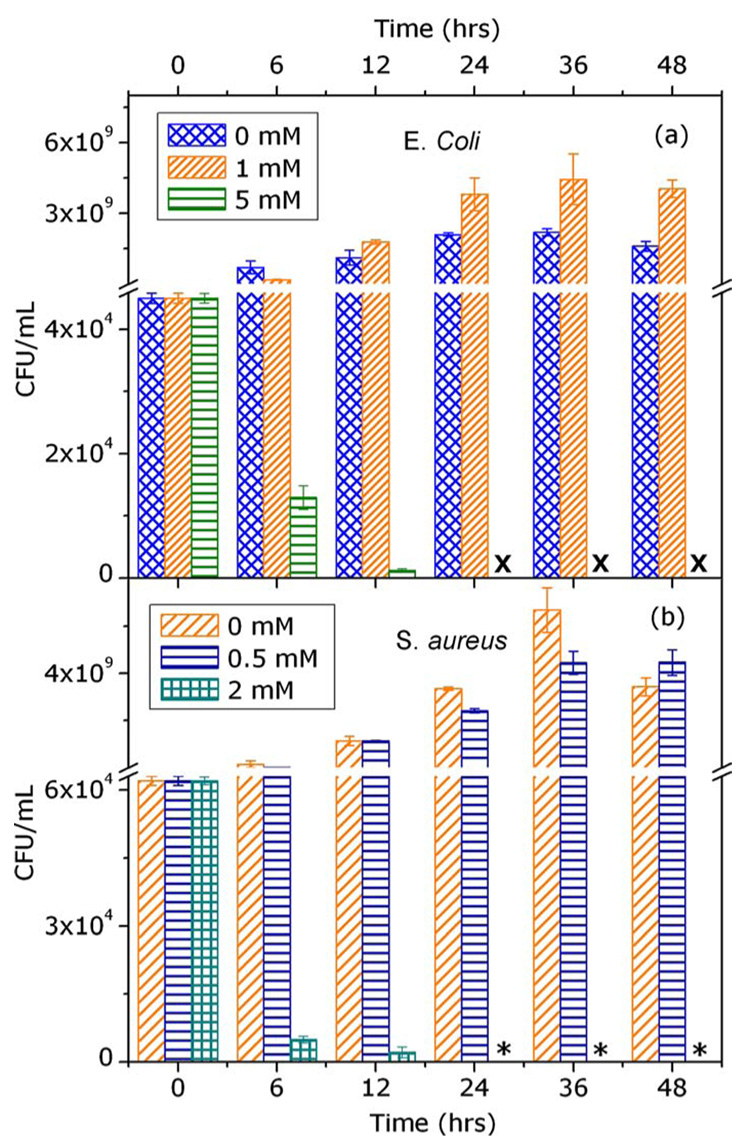
CFU counts of E. coli and S. aureus after 0, 6, 12, 24, and 48 h of ZnO NP exposure are shown in Figs. 3(a) and 3(b), respectively. These data demonstrate that exposure to ZnO NP results in cell death (bacteriocidal toxicity) at concentrations of 5 and 1 mM for E. coli and S. aureus, respectively. Further, the number of viable bacterial cells was reduced to below 90% of the initial CFU per milliliter within 12 h for both E. coli and S. aureus, and colonies were completely absent after 24 h of treatment (Fig. 3). E. coli treated with 1 mM ZnO NP showed a slight, albeit, consistent increase in CFU counts compared to control samples. Prior reports12 indicate that E. coli prefers Zn2+ concentrations in growth media of ~1 mM, thus ZnO NP at low concentrations may actually enhance bacterial growth consistent with our findings and those of Brayner et al.7 Zn2+ can stimulate growth of S. aureus also, however, at comparatively much lower (i.e., micromolar) concentrations.13 S. aureus expresses a Zn2+ efflux mechanism during exposure to Zn2+ in the millimolar range,13,14 indicating that millimolar concentrations of Zn2+ represent undesirable and potentially toxic conditions for S. aureus. These differences in the metabolism-dependent processes of E. coli and S. aureus may have a role in the observed difference in toxicity thresholds of these organisms.
FIG. 3.
(Color online) Effect of ZnO NP exposure time on the viability and growth of E. coli and S. aureus. Plots present mean CFU per milliliter (± standard error, n=3) of E. Coli (a) and S. aureus (b) exposed to different concentrations of ZnO NP for 0, 6, 12, 24, and 48 h. X and * indicate measured ZnO concentrations at which no CFU counts of E. coli and S. aureus, respectively, were observed.
To further confirm the bacteriocidal nature of ZnO NP, log phase bacteria were treated with various concentrations of ZnO NP for 15 h and analyzed using flow cytometry. A substantial loss of cell viability/membrane integrity (~30%) was observed in the remaining E. coli following treatment with 5 mM ZnO NP [Fig. 4(a)], whereas a ~59% loss was observed in S. aureus treated with 2 mM ZnO NP [Fig. 4(b)]. These data further indicate that ZnO NP exposure leads to bacterial cell death, mediated in part, by a loss in membrane integrity. Although the percentage of viable cells determined by flow cytometry appears greater than in experiments obtained by independent CFU measures [Fig. 2(a)], these results are generally consistent as the flow cytometry assay provides information relevant to the small population of remaining bacteria that have not yet disintegrated following NP exposure.
FIG. 4.
Relative bacterial viability determined by flow cytometry. Samples of bacteria were prepared, stained, and analyzed as outlined in the text and viability determined by a two-parameter comparison of green and red fluorescence emissions. Bacteria were gated by forward scatter and side scattering light properties and NPs excluded from analysis based on the absence of fluorescence. R3 depicts the region containing dead cells and R4 depicts the region containing live cells. (a) E. coli treated with 5 mM ZnO NPs for 15 h. (b) S. aureus treated with 2 mM ZnO NPs for 15 h.
Because our results demonstrate a selectivity of ZnO NPs in their toxic nature to prokaryotic systems, additional studies were performed to compare NP toxicity in eukaryotic cells. Human T lymphocytes were evaluated since this cell type is central for protection of the body against pathogens, including bacteria. T cell viability was determined using flow cytometry and PI staining. No effect of micron sized bulk ZnO powder was observed on T cell viability [Fig. 2(b)]. In contrast, ZnO NP treatment caused a significant decrease in viability at concentrations ⩾5 mM (p=0.000) with ~77% of the cells remaining viable at 5 mM and a significant fraction (~43%) remaining viable even at 10 mM ZnO NP [Fig. 2(b)]. These studies indicate that T cells have a larger tolerance range to ZnO NP than bacteria before toxicity is observed [Fig. 2(a)]. In addition, these findings indicate that T cell cytotoxicity is limited to ZnO in the nanoscale size range as no significant effect of bulk ZnO powder was observed.
Collectively, these findings demonstrate the selectivity of metal oxide NPs in their toxic nature to eukaryotic and prokaryotic biological systems. Introduction of ~13 nm ZnO NP kills gram-negative E. coli at concentrations ⩾3.4 mM, whereas growth of gram-positive S. aureus was prevented at much lower concentrations (⩾1 mM). Importantly, human T cells are considerably more resistant to NP toxicity than either E. coli or S. aureus. These findings suggest that ZnO NP may potentially prove useful as nanomedicine based antimicrobial agents at selective therapeutic dosing regimes.
Acknowledgments
This research was supported in part by NSF-Idaho-EPSCoR Program (EPS-0447689), DoE-EPSCoR grant (DE-FG02-04ER46142), NSF grants (DMR-0449639, MRI 0521315), and NIH awards (1R15 AI06277-01A1 and 1R43 AR052955-01).
Contributor Information
K. M. Reddy, Department of Physics, Boise State University, Boise, Idaho 83725
Kevin Feris, Department of Biology, Boise State University, Boise, Idaho 83725.
Jason Bell, Department of Biology, Boise State University, Boise, Idaho 83725.
Denise G. Wingett, Department of Biology, Boise State University, Boise, Idaho 83725
Cory Hanley, Department of Biology, Boise State University, Boise, Idaho 83725.
Alex Punnoose, Department of Physics, Boise State University, Boise, Idaho 83725.
References
- 1.McNeil SE. J. Leukoc Biol. 2005;78:585. doi: 10.1189/jlb.0205074. [DOI] [PubMed] [Google Scholar]
- 2.Jaiswal JK, Simon SM. Trends Cell Biol. 2004;14:497. doi: 10.1016/j.tcb.2004.07.012. [DOI] [PubMed] [Google Scholar]
- 3.Jain TK, Morales MA, Sahoo SK, Leslie‐Pelecky DL, Labhasetwar V. Molecular Pharmaceutics. 2005;2:194. doi: 10.1021/mp0500014. [DOI] [PubMed] [Google Scholar]
- 4.Visaria RK, Griffin RJ, Williams BW, Ebbini ES, Paciotti GF, Song CW, Bischof JC. Mol Cancer Ther. 2006;5:1014. doi: 10.1158/1535-7163.MCT-05-0381. [DOI] [PubMed] [Google Scholar]
- 5.Nel A, Xia T, Madler L, Li N. Science. 2006;311:622. doi: 10.1126/science.1114397. [DOI] [PubMed] [Google Scholar]
- 6.Long TC, Saleh N, Tilton RD, Lowry GV, Veronesi B. Environ. Sci. Technol. 2006;40:4346. doi: 10.1021/es060589n. [DOI] [PubMed] [Google Scholar]
- 7.Brayner R, Ferrari‐Iliou R, Brivois N, Djediat S, Benedetti MF, Fievet F. Nano Lett. 2006;6:866. doi: 10.1021/nl052326h. [DOI] [PubMed] [Google Scholar]
- 8.Thill A, Zeyons O, Spalla O, Chauvat F, Rose J, Auffan M, Flank AM. Environ. Sci. Technol. 2006;40:6151. doi: 10.1021/es060999b. [DOI] [PubMed] [Google Scholar]
- 9.Feldmann C, Jungk HO. Angew. Chem., Int. Ed. 2001;40:359. doi: 10.1002/1521-3773(20010119)40:2<359::AID-ANIE359>3.0.CO;2-B. [DOI] [PubMed] [Google Scholar]
- 10.Coligan JE. Current Protocols in Immunology. Vol. 1 Wiley-Interscience: New York; 1995. Chap. 5. [Google Scholar]
- 11.Punnoose A, Magnone H, Seehra MS, Bonevich J. Phys. Rev. B. 2001;64:174420. [Google Scholar]
- 12.Outten CE, O’Halloran Science. 2001;292:2488. doi: 10.1126/science.1060331. [DOI] [PubMed] [Google Scholar]
- 13.Lindsay JA, Foster SJ. Acta Phys. Univ. Comenianae. 2001;147:1259. [Google Scholar]
- 14.Xiong A, Jayaswal RK. J. Bacteriol. 1998;180:4024. doi: 10.1128/jb.180.16.4024-4029.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]