Abstract
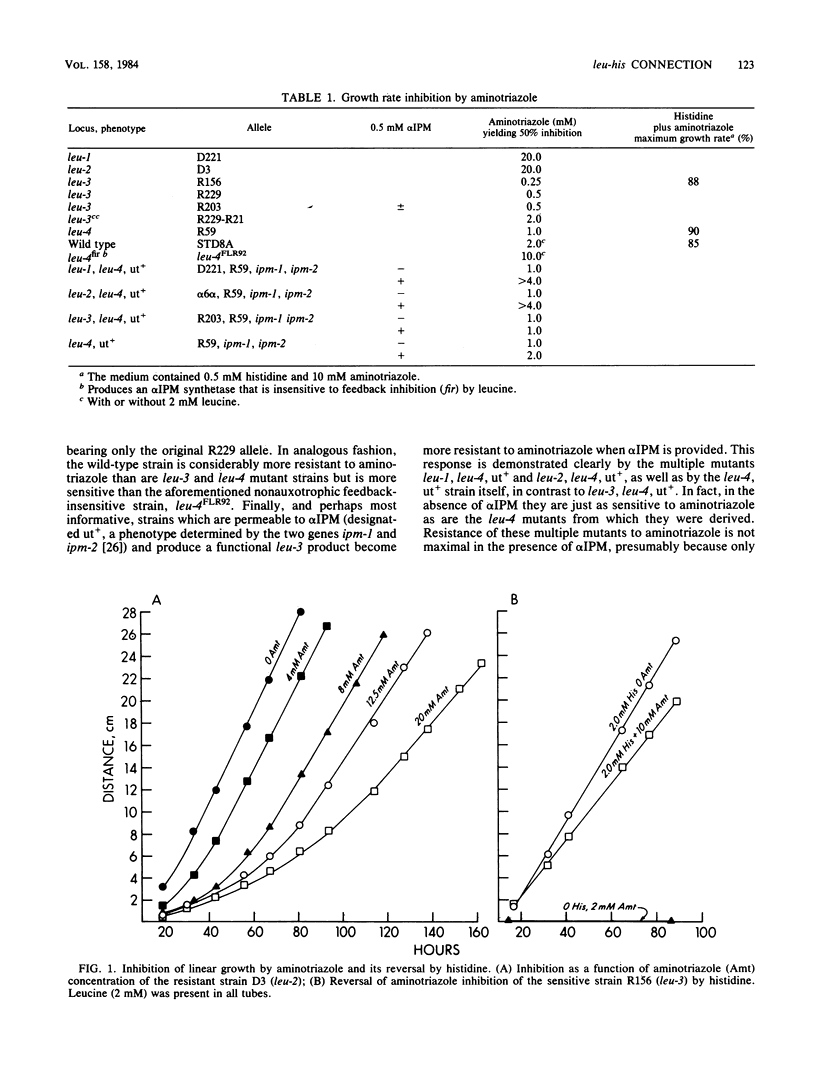
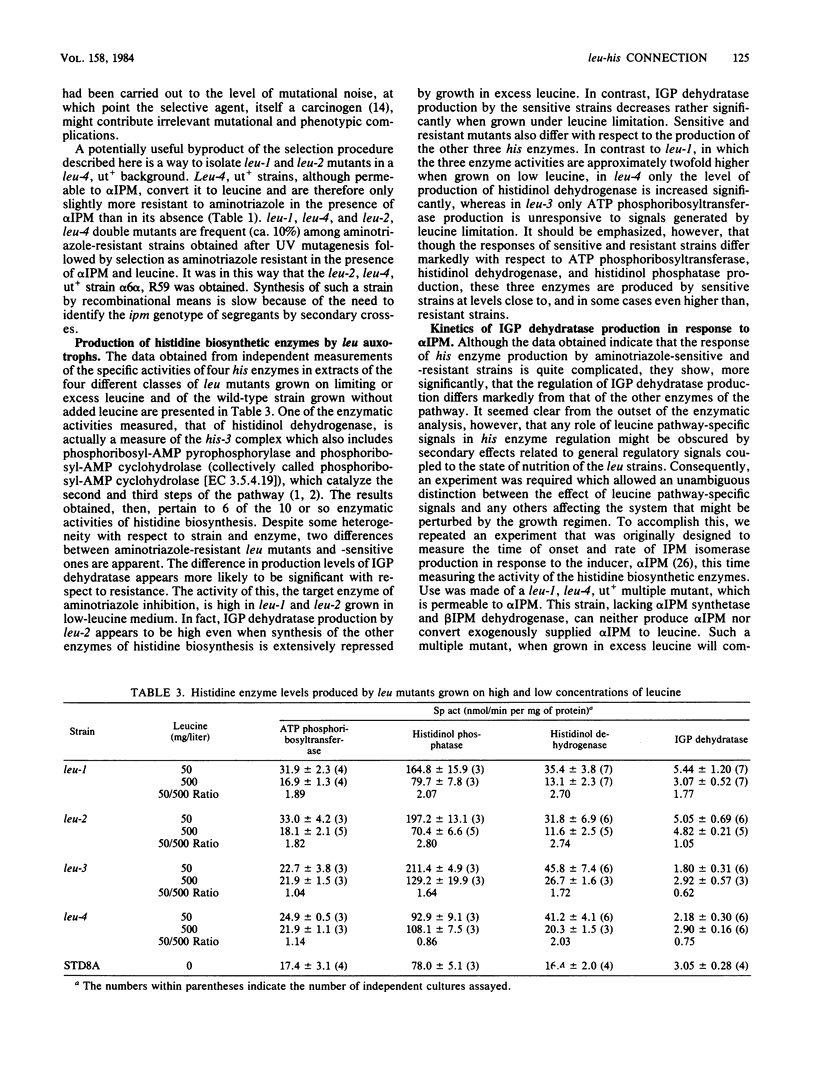
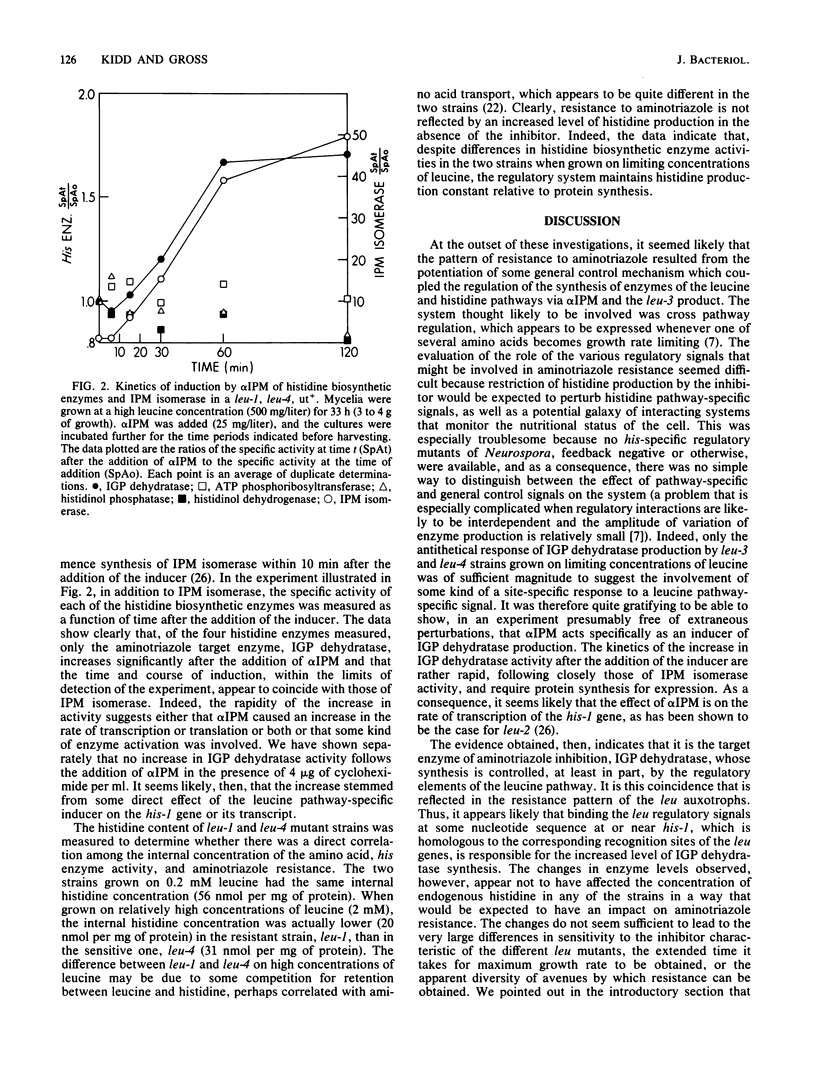
Leucine auxotrophs of Neurospora fall into two discrete categories with respect to sensitivity to the herbicide, 3-amino-1,2,4-triazole. The pattern of resistance corresponds exactly to the ability to produce the leucine pathway control elements, alpha-isopropylmalate and the leu-3 product. An analysis of the regulatory response of the production of enzymes of histidine biosynthesis to alpha-isopropylmalate implicates the control elements of the leucine pathway as important components of the mechanism governing the production of the target enzyme of aminotriazole inhibition, imidazoleglycerol-phosphate dehydratase (EC 4.2.1.19). The evidence suggests that the regulatory interconnection between the two pathways is direct and is independent of other general integrating regulatory mechanisms which appear to be operative in both pathways. A general method for isolating leu-1 and leu-2, as well as other regulatory mutants, is described, which takes advantage of the specificity of the resistance to the inhibitor. Use of analogous systems is prescribed for the analysis of other regulatory interconnections which, like this one, might not be anticipated directly from structural or biosynthetic considerations.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- AHMED A., CASE M. E., GILES N. H. THE NATURE OF COMPLEMENTATION AMONG MUTANTS IN THE HISTIDINE-3 REGION OF NEUROSPORA CRASSA. Brookhaven Symp Biol. 1964 Dec;17:53–65. [PubMed] [Google Scholar]
- AMES B. N. The biosynthesis of histidine; D-erythro-imidazoleglycerol phosphate dehydrase. J Biol Chem. 1957 Sep;228(1):131–143. [PubMed] [Google Scholar]
- Ahmed A. Organization of the histidine-3 region of Neurospora. Mol Gen Genet. 1968;103(2):185–193. doi: 10.1007/BF00427145. [DOI] [PubMed] [Google Scholar]
- Barthelmess I. B., Curtis C. F., Kacser H. Control of the flux to arginine in Neurospora crassa: de-repression of the last three enzymes of the arginine pathway. J Mol Biol. 1974 Aug 5;87(2):303–316. doi: 10.1016/0022-2836(74)90151-x. [DOI] [PubMed] [Google Scholar]
- Barthelmess I. B. Mutants affecting amino acid cross-pathway control in Neurospora crassa. Genet Res. 1982 Apr;39(2):169–185. doi: 10.1017/s0016672300020863. [DOI] [PubMed] [Google Scholar]
- Carsiotis M., Jones R. F., Wesseling A. C. Cross-pathway regulation: histidine-mediated control of histidine, tryptophan, and arginine biosynthetic enzymes in Neurospora crassa. J Bacteriol. 1974 Sep;119(3):893–898. doi: 10.1128/jb.119.3.893-898.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Donahue T. F., Daves R. S., Lucchini G., Fink G. R. A short nucleotide sequence required for regulation of HIS4 by the general control system of yeast. Cell. 1983 Jan;32(1):89–98. doi: 10.1016/0092-8674(83)90499-3. [DOI] [PubMed] [Google Scholar]
- Gross S. R. The regulation of synthesis of leucine biosynthetic enzymes in Neurospora. Proc Natl Acad Sci U S A. 1965 Dec;54(6):1538–1546. doi: 10.1073/pnas.54.6.1538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hilton J. L., Kearney P. C., Ames B. N. Mode of action of the herbicide, 3-amino-1,2,4-triazole(amitrole): inhibition of an enzyme of histidine biosynthesis. Arch Biochem Biophys. 1965 Dec;112(3):544–547. doi: 10.1016/0003-9861(65)90093-7. [DOI] [PubMed] [Google Scholar]
- Hulanicka D., Klopotowski T., Bagdasarian G. Inhibition of aminoimidazole ribotide biosynthesis in Salmonella typhimurium by aminotriazole. Acta Biochim Pol. 1969;16(2):127–133. [PubMed] [Google Scholar]
- Klopotowski T., Wiater A. Synergism of aminotriazole and phosphate on the inhibition of yeast imidazole glycerol phosphate dehydratase. Arch Biochem Biophys. 1965 Dec;112(3):562–566. doi: 10.1016/0003-9861(65)90096-2. [DOI] [PubMed] [Google Scholar]
- Kumar C. C., Padmanaban G. 3-Amino-1,2,4-triazole is an inhibitor of protein synthesis on mitoribosomes in Neurospora crassa. Biochim Biophys Acta. 1980 Apr 30;607(2):339–349. doi: 10.1016/0005-2787(80)90086-6. [DOI] [PubMed] [Google Scholar]
- LESTER H. E., GROSS S. R. Efficient method for selection of auxotrophic mutants of Neurospora. Science. 1959 Feb 27;129(3348):572–572. doi: 10.1126/science.129.3348.572. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- MARGOLIASH E., NOVOGRODSKY A. A study of the inhibition of catalase by 3-amino-1:2:4:-triazole. Biochem J. 1958 Mar;68(3):468–475. doi: 10.1042/bj0680468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olshan A. R., Gross S. R. Role of the leu-3 cistron in the regulation of the synthesis of isoleucine and valine biosynthetic enzymes of Neurospora. J Bacteriol. 1974 May;118(2):374–384. doi: 10.1128/jb.118.2.374-384.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perkins D. D., Radford A., Newmeyer D., Björkman M. Chromosomal loci of Neurospora crassa. Microbiol Rev. 1982 Dec;46(4):426–570. doi: 10.1128/mr.46.4.426-570.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perkins D. D. The manifestation of chromosome rearrangements in unordered asci of Neurospora. Genetics. 1974 Jul;77(3):459–489. doi: 10.1093/genetics/77.3.459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Polacco J. C., Gross S. R. The product of the leu-3 cistron as a regulatory element for the production of the leucine biosynthetic enzymes of Neurospora. Genetics. 1973 Jul;74(3):443–459. doi: 10.1093/genetics/74.3.443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reichenbecher V. E., Jr, Fischer M., Gross S. R. Regulation of isopropylmalate isomerase synthesis in Neurospora crassa. J Bacteriol. 1978 Feb;133(2):794–801. doi: 10.1128/jb.133.2.794-801.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]


