Abstract
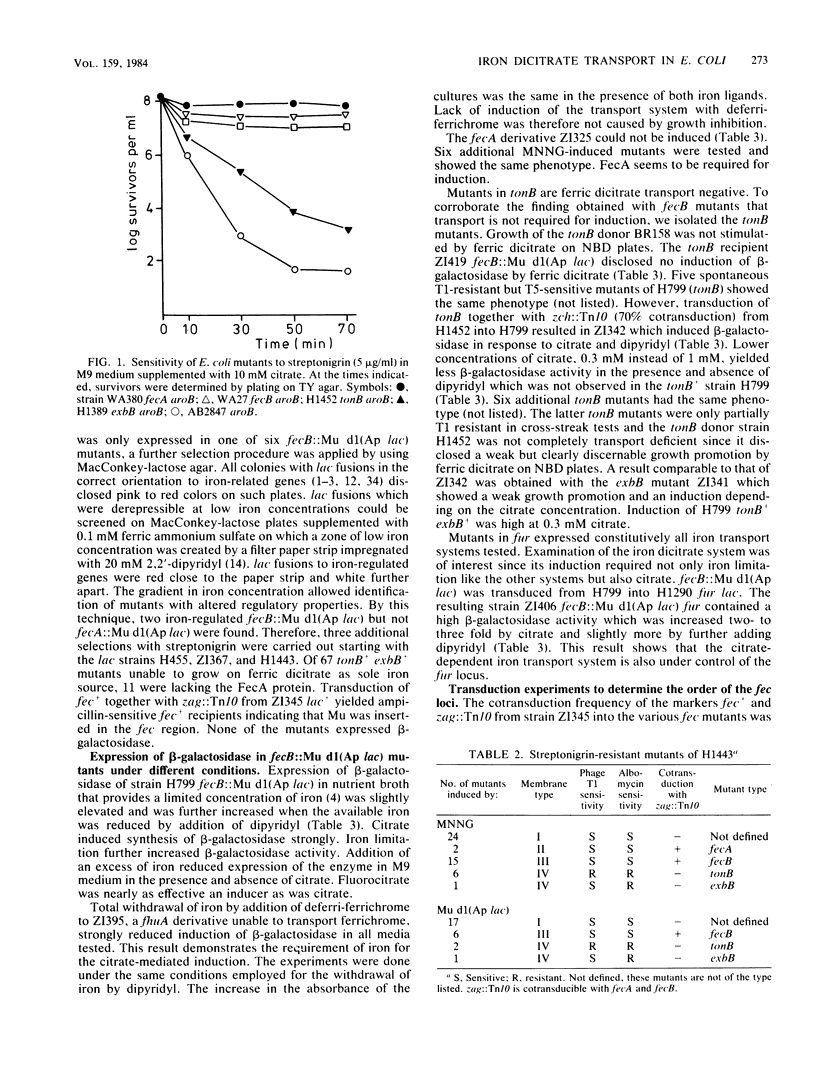
Streptonigrin was used to select mutants impaired in the citrate-dependent iron transport system of Escherichia coli K-12. Mutants in fecA and fecB could not transport iron via citrate. fecA-lac and fecB-lac operon fusions were constructed with the aid of phage Mu dl(Ap lac). Strains deficient in ferric dicitrate transport which were mutated in fecB were as inducible as transport-active strains. They expressed the FecA outer membrane protein and beta-galactosidase of the fecB-lac operon fusions. In contrast, all fecA::lac mutants and fecA mutants induced with N-methyl-N'-nitro-N-nitrosoguanidine did not respond to ferric dicitrate supplied in the growth medium. tonB fecB mutants which were lacking all tonB-related functions were not inducible. We conclude that binding of iron in the presence of citrate to the outer membrane receptor protein is required for induction of the transport system. In addition, the tonB gene has to be active. However, iron and citrate must not be transported into the cytoplasm for the induction process. These data support our previous conclusion of an exogenous induction mechanism. Mutants in fur expressed the transport system nearly constitutively. In wild-type cells limiting the iron concentration in the medium enhanced the expression of the transport system. Thus, the citrate-dependent iron transport system shares regulatory devices with the other iron transport systems in E. coli and, in addition, requires ferric dicitrate for induction.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Braun V., Burkhardt R. Regulation of the ColV plasmid-determined iron (III)-aerobactin transport system in Escherichia coli. J Bacteriol. 1982 Oct;152(1):223–231. doi: 10.1128/jb.152.1.223-231.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Braun V., Gross R., Köster W., Zimmermann L. Plasmid and chromosomal mutants in the iron(III)-aerobactin transport system of Escherichia coli. Use of streptonigrin for selection. Mol Gen Genet. 1983;192(1-2):131–139. doi: 10.1007/BF00327658. [DOI] [PubMed] [Google Scholar]
- Braun V., Hancock R. E., Hantke K., Hartmann A. Functional organization of the outer membrane of escherichia coli: phage and colicin receptors as components of iron uptake systems. J Supramol Struct. 1976;5(1):37–58. doi: 10.1002/jss.400050105. [DOI] [PubMed] [Google Scholar]
- Casadaban M. J., Cohen S. N. Lactose genes fused to exogenous promoters in one step using a Mu-lac bacteriophage: in vivo probe for transcriptional control sequences. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4530–4533. doi: 10.1073/pnas.76.9.4530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dunn J. T., Spiro R. G. The alpha 2-macroglobulin of human plasma. I. Isolation and composition. J Biol Chem. 1967 Dec 10;242(23):5549–5555. [PubMed] [Google Scholar]
- Frost G. E., Rosenberg H. The inducible citrate-dependent iron transport system in Escherichia coli K12. Biochim Biophys Acta. 1973 Nov 30;330(1):90–101. doi: 10.1016/0005-2736(73)90287-3. [DOI] [PubMed] [Google Scholar]
- Hall B. G. Chromosomal mutation for citrate utilization by Escherichia coli K-12. J Bacteriol. 1982 Jul;151(1):269–273. doi: 10.1128/jb.151.1.269-273.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hancock R. E., Hantke K., Braun V. Iron transport of Escherichia coli K-12: involvement of the colicin B receptor and of a citrate-inducible protein. J Bacteriol. 1976 Sep;127(3):1370–1375. doi: 10.1128/jb.127.3.1370-1375.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hancock R. W., Braun V. Nature of the energy requirement for the irreversible adsorption of bacteriophages T1 and phi80 to Escherichia coli. J Bacteriol. 1976 Feb;125(2):409–415. doi: 10.1128/jb.125.2.409-415.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hantke K., Braun V. Functional interaction of the tonA/tonB receptor system in Escherichia coli. J Bacteriol. 1978 Jul;135(1):190–197. doi: 10.1128/jb.135.1.190-197.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hantke K. Identification of an iron uptake system specific for coprogen and rhodotorulic acid in Escherichia coli K12. Mol Gen Genet. 1983;191(2):301–306. doi: 10.1007/BF00334830. [DOI] [PubMed] [Google Scholar]
- Hantke K. Regulation of ferric iron transport in Escherichia coli K12: isolation of a constitutive mutant. Mol Gen Genet. 1981;182(2):288–292. doi: 10.1007/BF00269672. [DOI] [PubMed] [Google Scholar]
- Harley J. B., Fetterolf C. J., Bello C. A., Flaks J. G. Streptonigrin toxicity in Escherichia coli: oxygen dependence and the role of the intracellular oxidation--reduction state. Can J Microbiol. 1982 May;28(5):545–552. doi: 10.1139/m82-082. [DOI] [PubMed] [Google Scholar]
- Hassan H. M., Fridovich I. Enzymatic defenses against the toxicity of oxygen and of streptonigrin in Escherichia coli. J Bacteriol. 1977 Mar;129(3):1574–1583. doi: 10.1128/jb.129.3.1574-1583.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hazelbauer G. L., Harayama S. Sensory transduction in bacterial chemotaxis. Int Rev Cytol. 1983;81:33–70. doi: 10.1016/s0074-7696(08)62334-7. [DOI] [PubMed] [Google Scholar]
- Hu M., Deonier R. C. Mapping of IS1 elements flanking the argF gene region on the Escherichia coli K-12 chromosome. Mol Gen Genet. 1981;181(2):222–229. doi: 10.1007/BF00268430. [DOI] [PubMed] [Google Scholar]
- Hussein S., Hantke K., Braun V. Citrate-dependent iron transport system in Escherichia coli K-12. Eur J Biochem. 1981 Jul;117(2):431–437. doi: 10.1111/j.1432-1033.1981.tb06357.x. [DOI] [PubMed] [Google Scholar]
- Koshland D. E., Jr A response regulator model in a simple sensory system. Science. 1977 Jun 3;196(4294):1055–1063. doi: 10.1126/science.870969. [DOI] [PubMed] [Google Scholar]
- Lugtenberg B., Meijers J., Peters R., van der Hoek P., van Alphen L. Electrophoretic resolution of the "major outer membrane protein" of Escherichia coli K12 into four bands. FEBS Lett. 1975 Oct 15;58(1):254–258. doi: 10.1016/0014-5793(75)80272-9. [DOI] [PubMed] [Google Scholar]
- Lütgens M., Gottschalk G. Why a co-substrate is required for anaerobic growth of Escherichia coli on citrate. J Gen Microbiol. 1980 Jul;119(1):63–70. doi: 10.1099/00221287-119-1-63. [DOI] [PubMed] [Google Scholar]
- Neilands J. B. Microbial envelope proteins related to iron. Annu Rev Microbiol. 1982;36:285–309. doi: 10.1146/annurev.mi.36.100182.001441. [DOI] [PubMed] [Google Scholar]
- Reynolds C. H., Silver S. Citrate utilization by Escherichia coli: plasmid- and chromosome-encoded systems. J Bacteriol. 1983 Dec;156(3):1019–1024. doi: 10.1128/jb.156.3.1019-1024.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Udenfriend S., Stein S., Böhlen P., Dairman W., Leimgruber W., Weigele M. Fluorescamine: a reagent for assay of amino acids, peptides, proteins, and primary amines in the picomole range. Science. 1972 Nov 24;178(4063):871–872. doi: 10.1126/science.178.4063.871. [DOI] [PubMed] [Google Scholar]
- Wagegg W., Braun V. Ferric citrate transport in Escherichia coli requires outer membrane receptor protein fecA. J Bacteriol. 1981 Jan;145(1):156–163. doi: 10.1128/jb.145.1.156-163.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woodrow G. C., Langman L., Young I. G., Gibson F. Mutations affecting the citrate-dependent iron uptake system in Escherichia coli. J Bacteriol. 1978 Mar;133(3):1524–1526. doi: 10.1128/jb.133.3.1524-1526.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Worsham P. L., Konisky J. Use of cir-lac operon fusions to study transcriptional regulation of the colicin Ia receptor in Escherichia coli K-12. J Bacteriol. 1981 Jan;145(1):647–650. doi: 10.1128/jb.145.1.647-650.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yeowell H. N., White J. R. Iron requirement in the bactericidal mechanism of streptonigrin. Antimicrob Agents Chemother. 1982 Dec;22(6):961–968. doi: 10.1128/aac.22.6.961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- York M. K., Stodolsky M. Characterization of P1argF derivatives from Escherichia coli K12 transduction. I. IS1 elements flank the argF gene segment. Mol Gen Genet. 1981;181(2):230–240. doi: 10.1007/BF00268431. [DOI] [PubMed] [Google Scholar]