Abstract
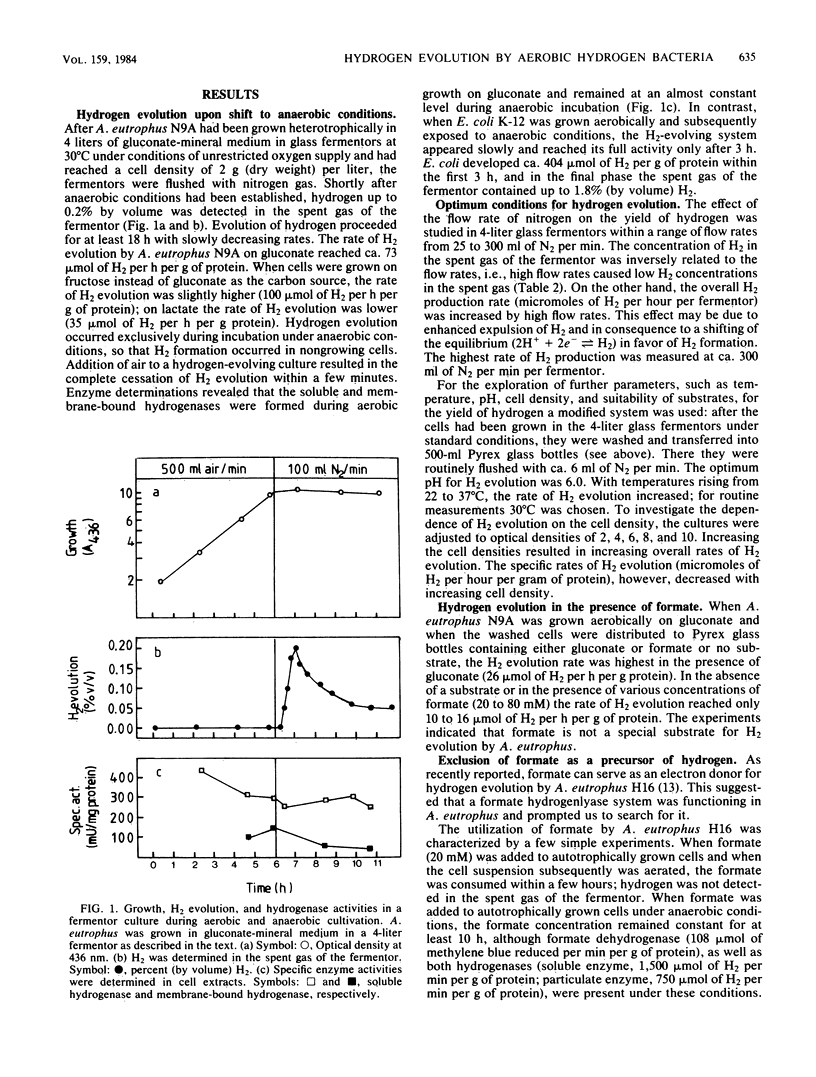
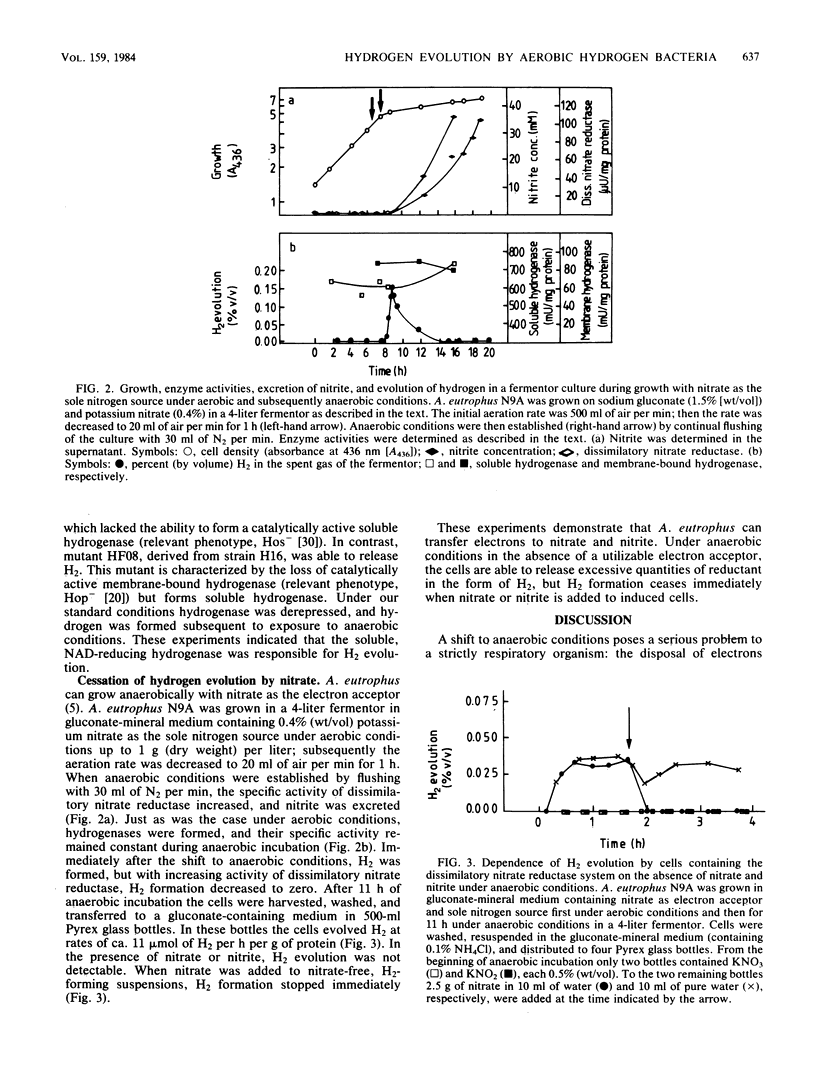
When strains and mutants of the strictly aerobic hydrogen-oxidizing bacterium Alcaligenes eutrophus are grown heterotrophically on gluconate or fructose and are subsequently exposed to anaerobic conditions in the presence of the organic substrates, molecular hydrogen is evolved. Hydrogen evolution started immediately after the suspension was flushed with nitrogen, reached maximum rates of 70 to 100 mumol of H2 per h per g of protein, and continued with slowly decreasing rates for at least 18 h. The addition of oxygen to an H2-evolving culture, as well as the addition of nitrate to cells (which had formed the dissimilatory nitrate reductase system during the preceding growth), caused immediate cessation of hydrogen evolution. Formate is not the source of H2 evolution. The rates of H2 evolution with formate as the substrate were lower than those with gluconate. The formate hydrogenlyase system was not detectable in intact cells or crude cell extracts. Rather the cytoplasmic, NAD-reducing hydrogenase is involved by catalyzing the release of excessive reducing equivalents under anaerobic conditions in the absence of suitable electron acceptors. This conclusion is based on the following experimental results. H2 is formed only by cells which had synthesized the hydrogenases during growth. Mutants lacking the membrane-bound hydrogenase were still able to evolve H2. Mutants lacking the NAD-reducing or both hydrogenases were unable to evolve H2.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Adams M. W., Mortenson L. E., Chen J. S. Hydrogenase. Biochim Biophys Acta. 1980 Dec;594(2-3):105–176. doi: 10.1016/0304-4173(80)90007-5. [DOI] [PubMed] [Google Scholar]
- Arp D. J., Burris R. H. Purification and properties of the particulate hydrogenase from the bacteroids of soybean root nodules. Biochim Biophys Acta. 1979 Oct 11;570(2):221–230. doi: 10.1016/0005-2744(79)90142-6. [DOI] [PubMed] [Google Scholar]
- BULEN W. A., BURNS R. C., LECOMTE J. R. NITROGEN FIXATION: HYDROSULFITE AS ELECTRON DONOR WITH CELL-FREE PREPARATIONS OF AZOTOBACTER VINELANDII AND RHODOSPIRILLUM RUBRUM. Proc Natl Acad Sci U S A. 1965 Mar;53:532–539. doi: 10.1073/pnas.53.3.532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Davis D. H., Stanier R. Y., Doudoroff M., Mandel M. Taxonomic studies on some gram negative polarly flagellated "hydrogen bacteria" and related species. Arch Mikrobiol. 1970;70(1):1–13. doi: 10.1007/BF00691056. [DOI] [PubMed] [Google Scholar]
- Dixon R. O. Hydrogenase in legume root nodule bacteroids: occurrence and properties. Arch Mikrobiol. 1972;85(3):193–201. doi: 10.1007/BF00408844. [DOI] [PubMed] [Google Scholar]
- Friedrich B., Friedrich C. G., Meyer M., Schlegel H. G. Expression of hydrogenase in Alcaligenes spp. is altered by interspecific plasmid exchange. J Bacteriol. 1984 Apr;158(1):331–333. doi: 10.1128/jb.158.1.331-333.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Friedrich C. G., Friedrich B. Regulation of hydrogenase formation is temperature sensitive and plasmid coded in Alcaligenes eutrophus. J Bacteriol. 1983 Jan;153(1):176–181. doi: 10.1128/jb.153.1.176-181.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gogotov J. N., Schlegel H. G. N2-fixation by chemoautotrophic hydrogen bacteria. Arch Microbiol. 1974 May 20;97(4):359–362. doi: 10.1007/BF00403073. [DOI] [PubMed] [Google Scholar]
- LOWE R. H., EVANS H. J. PREPARATION AND SOME PROPERTIES OF A SOLUBLE NITRATE REDUCTASE FROM RHIZOBIUM JAPONICUM. Biochim Biophys Acta. 1964 Jun 1;85:377–389. doi: 10.1016/0926-6569(64)90301-3. [DOI] [PubMed] [Google Scholar]
- Mergeay M., Houba C., Gerits J. Extrachromosomal inheritance controlling resistance to cadmium, cobalt, copper and zinc ions: evidence from curing in a Pseudomonas [proceedings]. Arch Int Physiol Biochim. 1978 May;86(2):440–442. [PubMed] [Google Scholar]
- Pfitzner J. Ein Beitrag zum H2-O2-Oxidoreductasesystem von Hydrogenomonas eutropha Stamm H 16: Hydrogenasedefekte Mutanten. Zentralbl Bakteriol Orig A. 1974;228(1):121–127. [PubMed] [Google Scholar]
- Pinchukova E. E., Varfolomeev S. D. Obratimoe okislenie-vosstanovlenie NAD vodorodom, kataliziruemo gidrogenazoi Alcaligenes eutrophus Z-1. Biokhimiia. 1980 Aug;45(8):1405–1411. [PubMed] [Google Scholar]
- Pinkwart M., Schneider K., Schlegel H. G. Purification and properties of the membrane-bound hydrogenase from N2-fixing Alcaligenes latus. Biochim Biophys Acta. 1983 Jun 29;745(3):267–278. doi: 10.1016/0167-4838(83)90058-4. [DOI] [PubMed] [Google Scholar]
- Schink B., Schlegel H. G. The membrane-bound hydrogenase of Alcaligenes eutrophus. I. Solubilization, purification, and biochemical properties. Biochim Biophys Acta. 1979 Apr 12;567(2):315–324. doi: 10.1016/0005-2744(79)90117-7. [DOI] [PubMed] [Google Scholar]
- Schink B., Schlegel H. G. The membrane-bound hydrogenase of Alcaligenes eutrophus: II. Localization and immunological comparison with other hydrogenase systems. Antonie Van Leeuwenhoek. 1980;46(1):1–14. doi: 10.1007/BF00422224. [DOI] [PubMed] [Google Scholar]
- Schlegel H. G., Lafferty R., Krauss I. The isolation of mutants not accumulating poly-beta-hydroxybutyric acid. Arch Mikrobiol. 1970;71(3):283–294. doi: 10.1007/BF00410161. [DOI] [PubMed] [Google Scholar]
- Schneider K., Schlegel H. G., Jochim K. Effect of nickel on activity and subunit composition of purified hydrogenase from Nocardia opaca 1 b. Eur J Biochem. 1984 Feb 1;138(3):533–541. doi: 10.1111/j.1432-1033.1984.tb07948.x. [DOI] [PubMed] [Google Scholar]
- Schneider K., Schlegel H. G. Localization and stability of hydrogenases from aerobic hydrogen bacteria. Arch Microbiol. 1977 Apr 1;112(3):229–238. doi: 10.1007/BF00413086. [DOI] [PubMed] [Google Scholar]
- Schneider K., Schlegel H. G. Purification and properties of soluble hydrogenase from Alcaligenes eutrophus H 16. Biochim Biophys Acta. 1976 Nov 8;452(1):66–80. doi: 10.1016/0005-2744(76)90058-9. [DOI] [PubMed] [Google Scholar]
- WALDE E. [Studies on growth and synthesis of stored substance by Hydrogenomonas]. Arch Mikrobiol. 1962;43:109–137. [PubMed] [Google Scholar]
- Walker C. C., Yates M. G. The hydrogen cycle in nitrogen-fixing Azotobacter chroococcum. Biochimie. 1978;60(3):225–231. doi: 10.1016/s0300-9084(78)80818-9. [DOI] [PubMed] [Google Scholar]



