Abstract
Persistent neurogenesis in an adult insect brain was recently shown to be stimulated by juvenile hormone (JH). This morphogenetic hormone was also shown to act on polyamine biosynthesis. To analyze the possible involvement of polyamines in the neurogenic action of JH, two series of experiments were carried out with adult female crickets, Acheta domesticus: (i) inhibition of the first key enzyme in polyamine biosynthesis, ornithine decarboxylase, with α-difluoromethylornithine (α-DFMO), and examination of the effects of this treatment on the neuroblast proliferation response to JH; and (ii) examination of the effects of putrescine supplementation on the mitotic index of JH-deprived and α-DFMO-treated females. In control females, α-DFMO treatment, as well as JH deprivation, greatly reduced neuroblast proliferation. Putrescine supplementation in α-DFMO-treated insects overcame the effects of α-DFMO, and allowed for detection of putrescine in the neural tissue and stimulation of brain neurogenesis. In JH-deprived females, α-DFMO treatment completely prevented the stimulatory action of JH on neuroblast proliferation and on brain putrescine levels. By contrast, putrescine feeding of JH-deprived animals was able to mimic the stimulatory effect of JH: brain putrescine levels increased and neuroblast proliferation was restored. To our knowledge, this report demonstrates for the first time that in vivo administration of putrescine can mimic the effects of a morphogenetic hormone on adult neuroblast proliferation, and shows the importance of polyamines, especially putrescine, in the transduction of JH message in neural tissue.
The pleiotropic action of juvenile hormone (JH) in insects has been widely demonstrated (1–3). However, JH receptors have not yet been characterized, and, despite numerous attempts, the precise molecular mechanisms of action of JH are far from being understood (1–3). Recently, persistent neurogenesis modulated by hormones was demonstrated in one of the main integrative areas of adult cricket brain, the mushroom bodies (4, 5).† Neuroblast proliferation was depressed in the absence of JH, whereas JH injection significantly stimulated the mitotic activity of the proliferative area. Although in adult females the most studied role of JH is action on fat body and ovaries to induce vitellogenesis, it is now apparent that neural tissue is also a target for JH action. It has also been shown that in the neural tissue, lack of JH depresses the activities of ornithine decarboxylase (ODC) and S-adenosylmethionine decarboxylase (AdoMetDC), the two key enzymes in polyamine biosynthesis, resulting in a significant lowering of polyamine titres, an effect that was reversed by JH injection (7). In the cricket, oviposition behavior requires the action of JH, and a strong reduction of oviposition results from inhibition of ODC (8). These findings allowed us to hypothesize that JH can act through polyamine metabolism. The naturally occurring polyamines, putrescine, spermidine, and spermine, are ubiquitous polycations that play a major role in cell proliferation and differentiation (9–11). Many hormones and extracellular signals are known to induce an early stimulation of the activity of ODC, the first regulator of polyamine biosynthesis, which provokes a correlative increase in tissue polyamine levels (12, 13).
To investigate the role of polyamines in the transduction of JH message leading to brain neuroblast proliferation, we examined the effects of JH and of putrescine supplementation on mitotic activity in mushroom bodies of adult female crickets that were intact or JH-deprived and treated or not with a specific and irreversible inhibitor of ODC, α-difluoromethylornithine (α-DFMO) (14). The results indicate that polyamines are involved in the mechanism of action of JH on neural tissue. They also demonstrate that among the polyamines, putrescine is specifically required for mushroom body neuroblast proliferation.
MATERIALS AND METHODS
Insects.
Acheta domesticus were reared under a long day photoperiod (16-h light/8-h dark) at 29°C and 55% relative humidity. They were fed bran, wheat germ, and ground rabbit chow; water was continuously available. Newly emerged adult females were isolated and reared as virgins. They were killed as 3- or 6-day-old adults—i.e., previtellogenic and fully mature respectively. However, because both periods of exposure showed the same trends, the data were combined for presentation.
Surgical Procedure, Hormone Injection, and Drug Administration.
Surgical removal of the corpora allata (allatectomy), the endocrine glands secreting JH, was performed during the last larval instar and resulted in adult females deprived of JH (15). JH III (Sigma) (100 μg/10 μl paraffin oil) was injected into allatectomized females on the day of adult emergence to counteract the effects of allatectomy. Injections were performed through an intersegmental membrane of the cricket abdomen.
Drugs were dissolved in drinking water. Their concentrations were 2% α-DFMO (Merrell Dow Research Institute, Strasbourg) and 0.1% putrescine (Sigma). They were administered either alone or together from the day of adult emergence to the day of assays of mitotic index and polyamine titres. The solutions were renewed each day.
Polyamine Determination.
The cerebral ganglia (brain plus sub-oesophageal ganglion) were dissected out in saline. All fat body was carefully removed. Tissues were sonicated in 75 μl ice-cold 0.4 M perchloric acid (Merck) and centrifuged at 10,000 × g for 4 min at 4°C. The supernatants were collected and stored at −20°C until further analysis. One hundred microliters of 0.1 M NaOH was added to each pellet for protein determination according to the method of Bradford (16) using bovine serum albumin as a standard.
For polyamine determination, the tissue extracts and standards were dansylated overnight in the dark, at room temperature, to 40 μl of supernatant, and 10 μl of 5 × 10−6 M 1,7-diamino heptane (used as an internal standard), 200 μl of dansylchloride (5 mg/ml in acetone), and 100 μl of sodium carbonate (0.3 M in distilled water) were added. Next, the samples were mixed with 700 μl of distilled water, and they were vortex mixed and applied to a Waters Sep-Pak reverse-phase C18 cartridge. The Sep-Pak was washed with 4 ml of 20% methanol and the polyamines were eluted with 2 ml of 100% methanol. Separation and quantification of polyamines were performed by reverse-phase high performance liquid chromatography (17). The major polyamines were identified by their retention times compared with those of standards. Peak areas were automatically measured by an integrator. Mixed polyamine standards from 10 to 70 pmol were reacted and chromatographed to establish linear standard curves that served to determine the absolute amount of polyamines. The absolute limit of detection per injection was 1 pmol for dansylated spermidine and spermine and 7 pmol for putrescine. Two blank injections were routinely run between calibrations and sample analysis.
Hydrochloride salts of putrescine, spermidine, and spermine as well as 1,7-diamino heptane were purchased from Sigma. Solvents (chromasol grade) were obtained from Solvants Documentation Synthese (Peypin, France).
Polyamine levels were expressed in nmol/mg protein as mean values ± SEM.
Mitotic Index Determination.
Cerebral ganglia were quickly dissected out in saline, then fixed for 6 h in Carnoy’s fixative [absolute ethanol/chloroform/acetic acid, 6:3:1 (vol/vol)]. After three 24-h washes in 95% ethanol and three 24-h washes in 1-butanol, tissues were embedded in paraffin and cut in 6-μm serial sections. Sections were deparaffined, rehydrated, and treated for DNA staining according to the method of Feulgen-Rossenbeck (in ref. 18). DNA was hydrolyzed using 6 M HCl for 60 min at room temperature. Sections were counterstained in 0.4% indigo carmine in a saturated solution of picric acid, dehydrated, and mounted in DePeX (Merck). Analysis of mitoses was performed blind on serial sections of mushroom bodies. The mitotic index was expressed as the number of mitoses in mushroom bodies per brain, reported as mean values ± SEM.
Data Analysis.
Significance of differences was determined using Mann–Whitney U test (P < 0.05).
RESULTS
α-DFMO Prevents the Mitogenic Action of JH on Adult Cricket Neuroblasts.
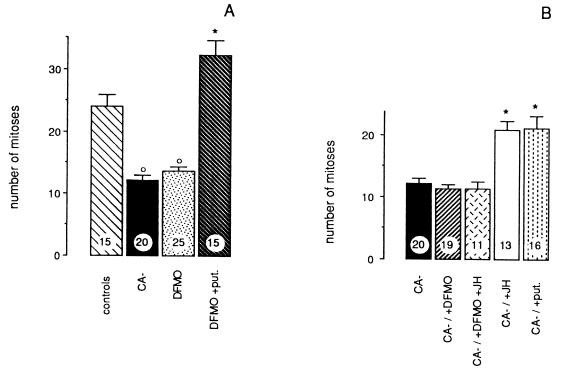
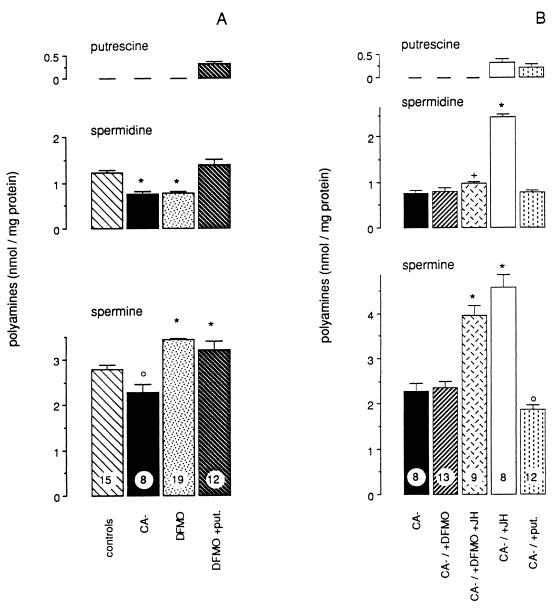
In adult females deprived of JH, the mushroom body neuroblasts exhibited a reduced mitotic activity. After allatectomy the mitotic index markedly and significantly decreased to 51,7% (P < 0.0001) relative to values in control females (Fig. 1A). Injections of JH into females lacking hormone markedly enhanced cell multiplication (P < 0.0001) and restored neurogenesis similar to that of control animals (Fig. 1B). Taken together, these observations clearly demonstrated the stimulatory action of JH on neuroblast proliferation. Similarly, lack of JH in allatectomized females produced a marked reduction of spermidine and spermine titres (Fig. 2A). It was previously shown (17) that, in the neural tissue, putrescine levels remained under the detection limit of our method, whereas spermidine and spermine were easily measured. As seen in Fig. 2A, spermidine and spermine levels were significantly lower in hormone-deprived females than in the controls (P < 0.0001 and P < 0.008, respectively). JH injection into allatectomized animals greatly increased polyamine levels (Fig. 2B). As a result of a stimulation of their biosynthesis, spermidine and spermine levels were higher (P < 0.001), as well as that of putrescine, which then became detectable.
Figure 1.
(A) Number of mitotic cells in control females (controls), allatectomized females (CA−), α-DFMO-treated females (DFMO), and α-DFMO-fed females supplemented with putrescine (DFMO + put.). Treatments were administered from adult emergence to the day of assay. Data are the mean values ± SEM. Sample size is indicated in the columns. Columns with the same sign are not significantly different. (B) Number of mitotic cells in allatectomized females (CA−) submitted to different experimental treatments. Putrescine (put.) or α-DFMO (DFMO) were given in drinking water from emergence onwards. JH III (100 μg/10 μl paraffin oil) was injected at emergence into females, which were provided with α-DFMO (DFMO + JH) or not (JH). Data are the mean values ± SEM. Sample size is indicated in the columns. Columns with the same sign are not significantly different.
Figure 2.
(A) Brain polyamine (putrescine, spermidine, and spermine) titres in control females (controls), allatectomized females (CA−), α-DFMO-treated females (DFMO), and α-DFMO-fed females supplemented with putrescine (DFMO + put.). Data are the mean values ± SEM. Sample size is indicated in the spermine columns. Columns with the same sign are not significantly different. (B) Brain polyamine (putrescine, spermidine, and spermine) titres in allatectomized females (CA−) submitted to various experimental treatments. Putrescine (put.) or α-DFMO (DFMO) were given in drinking water from emergence onwards. JH III (100 μg/10 μl paraffin oil) was injected at emergence into females, which were provided with α-DFMO (DFMO + JH) or not (JH). Data are the mean values ± SEM. Sample size is indicated in the spermine columns. Columns with the same sign are not significantly different.
Continuous treatment (up to 10 days) with α-DFMO, an inhibitor of ODC, was previously shown not to alter JH biosynthesis and vitellogenesis (8). However, intact females treated with α-DFMO presented a significant reduction of their mitotic index (P < 0.0001), which dropped to 58% of the values recorded in controls (Fig. 1A). At the same time, the neural tissue of α-DFMO-treated females showed a significant reduction of spermidine levels (P < 0.0001), while spermine titres increased (P < 0.0005) (Fig. 2A). From these data, it appears that allatectomy or α-DFMO treatment led to a decrease of brain spermidine titre that was correlated with a strong reduction of neuroblast proliferation.
To assess the involvement of polyamines in the mitogenic action of JH, allatectomized females were concomitantly fed α-DFMO and injected with JH. In allatectomized females, α-DFMO treatment alone did not affect neuroblast proliferation or polyamine titres (Figs. 1B and 2B). JH injection into α-DFMO-treated allatectomized females failed to induce any significant increase of the mitotic index (Fig. 1B). Similarly, the increase of putrescine and spermidine levels in neural tissue, previously described after hormone injection, could not be observed (Fig. 2B), and only spermine titres became significantly higher (P < 0.0001) as a consequence of the stimulatory action of α-DFMO and JH on AdoMetDC. As mentioned above, α-DFMO treatment of control females did not suppress the vitellogenic action of JH (8), and, after JH injection, ovarian development took place in the allatectomized, α-DFMO-treated females. We also found that JH was able to stimulate polyamine biosynthesis in the fat body of α-DFMO-fed insects (data not shown). From these experiments we conclude that α-DFMO prevents the action of JH on polyamine biosynthesis and mitosis specifically in neural tissue.
Putrescine Induces an Increase of Neurogenesis Independently of JH.
In intact females treated with α-DFMO (Fig. 2A), putrescine supplementation counterbalanced the decrease in spermidine titres (P < 0.0001) to values similar to the levels of controls; moreover, it induced the appearance of detectable amounts of putrescine in the neural tissue (Fig. 2A). In addition, putrescine treatment markedly stimulated the mitotic activity of mushroom body neuroblasts (Fig. 1A). The mitotic index values were more than 2-fold higher (P < 0.001) than those observed after α-DFMO treatment alone, and even higher than in control animals (P < 0.001).
Putrescine administration to allatectomized females did not markedly affect spermidine and spermine titres (Fig. 2B). However, putrescine levels increased to detectable amounts similar to those observed after JH injection. Moreover, neuroblast proliferation was greatly increased by putrescine treatment in the absence of JH (Fig. 1B). In putrescine-treated allatectomized females, the mitotic index was similar to that observed after JH injection and was significantly higher (P < 0.0001) than in control allatectomized animals.
Altogether, these results strongly suggest that putrescine, acting independently of JH, is able to stimulate neurogenesis and is responsible for the mitogenic action of JH in neural tissue.
DISCUSSION
α-DFMO and Polyamine Metabolism.
We have previously shown in A. domesticus that in vitro addition of α-DFMO completely inhibited the activity of ODC in nervous tissue and fat body (19). Administered in vivo, α-DFMO induced effects that are similar to those obtained in vertebrates. It is now well known that α-DFMO does not only suppress ornithine conversion into putrescine, and consequently reducing spermidine titres, but also increases the synthesis of AdoMetDC—the second key enzyme in polyamine biosynthesis—in response to the decline of spermidine levels. Thus the available spermidine is converted to spermine (20). Moreover, as putrescine exerts an inhibitory effect on spermine synthase, the disappearance of putrescine stimulates spermine synthesis and, in the absence of putrescine, polyamine metabolism is shifted to the formation of spermine (21, 22). All these effects were observed in drug-treated intact female crickets. In crickets as in mammals (23), administration of putrescine to α-DFMO-treated animals completely overcame the effects of α-DFMO; moreover, it led to the appearance of putrescine in neural tissue. This is in good agreement with our present knowledge of polyamine metabolism since several studies indicated that de novo synthesis can be replaced by polyamine uptake from the environment (24), and putrescine as well as spermidine and spermine can easily be detected in the cricket hemolymph (25).
We have previously demonstrated that JH stimulated the activities of ODC and AdoMetDC, which resulted, in neural tissue, in the appearance of putrescine and in an increase in spermidine and spermine levels (7). When JH and α-DFMO were concomitantly applied, the stimulatory action of JH on ODC was abolished. Thus, spermidine titres remained low, but spermine levels increased most likely due to a stimulation of the AdoMetDC activity in response to the α-DFMO and JH treatment. Allatectomized females fed putrescine exhibited detectable amounts of putrescine in their neural tissue, whereas spermidine and spermine levels remained unchanged (Fig. 2B) as a consequence of the low activity of AdoMetDC due to the lack of JH (7).
Requirement of Putrescine for the Transduction of JH Message and its Mitogenic Action.
The necessity of polyamines for cell growth has been known for some time (26). Subsequently, the effects of polyamine depletion through α-DFMO action were first examined in vitro in mammalian cell cultures, demonstrating that putrescine and spermidine deprivation altered DNA and protein synthesis and decreased cell multiplication (14, 27). Only a few investigations have addressed the effects of in vivo administration of α-DFMO on cell proliferation. Inhibition of ODC by α-DFMO suppressed the normal rate of small intestinal mucosal growth in rats (28). Postnatal administration of α-DFMO depleted the number of cerebellar granule cells in rats (29, 30) and reduced the numerical density of granule cells in the dorsal cochlear nucleus of hamster (31). Our data closely agree with these findings in that α-DFMO-fed intact crickets showed a strong reduction of neuroblast proliferation, although a basal rate of cell renewal still remained in both α-DFMO-treated intact animals and in allatectomized females.
The persistence and even the increase of cell proliferation in the presence of α-DFMO and putrescine—i.e., without activation of ODC—indicates that it is not the ODC stimulation but the availability of putrescine that is the major inducer of neuroblast proliferation. In cell culture, putrescine has been shown to be mitogenic in a variety of mammalian cells (32, 33). Similarly, from studies of regenerating planaria, a relationship between putrescine content and the rate of cell proliferation has been observed (34). We demonstrated further that an increase in brain putrescine content, without any change in spermidine of spermine levels, was able to mimic JH stimulation of neuroblast proliferation in allatectomized females fed α-DFMO. It must be emphasized that it is only when putrescine was detectable in neural tissue that a strong increase of the mitotic index could be observed in the proliferative area of the mushroom body. Since in JH-deprived females α-DFMO inhibited the stimulatory action of JH injection on neuroblast proliferation, we can conclude that JH action on neurogenesis requires polyamine biosynthesis.
A few in vitro studies have previously shown that α-DFMO inhibited the stimulatory action of hormones and mitogenic factors. In human breast cancer cell culture, α-DFMO can prevent the growth stimulation induced by oestradiol (35). Similarly, variants of PC12 cells with altered ODC expression did not properly respond to nerve growth factor (36). In an intestinal crypt cell line, gastrin increased the ODC mRNA levels and polyamine titres together with c-myc mRNA expression. The nuclear protooncogene c-myc participates in the regulation of ODC and cell development (37, 38) and α-DFMO prevented the expression of c-myc in cells treated with gastrin (39). The expression of c-myc is stimulated by numerous mitogenic signals like oestrogens (40), or prolactin which markedly enhanced the expression of c-myc and max (41). Recent data demonstrated that c-Myc (42) and/or the c-Myc–Max protein complex (43) play a major role in the transcriptional modulation of the ODC gene, which was recognized as a protooncogene (44). However, as polyamines have specific structural determinants that can influence B to Z conformational changes of DNA (45–47), they, in turn, participate in the regulation of protooncogene expression (48–50). Further evidence that a hormone increases polyamine concentration at a site that regulates gene expression is found in breast cancer cells, in which the mitotic cyclin B1 is a target where the effect of oestradiol was linked to cellular polyamine concentration (51, 52).
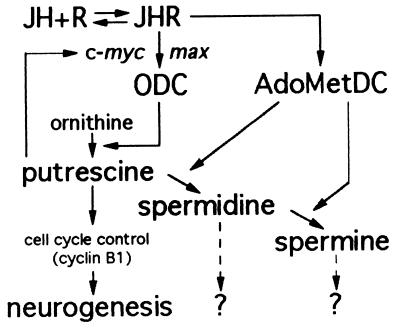
Our in vivo results showed that polyamines and especially putrescine play a role in the transduction of the JH signals leading to neuroblast division. From the data mainly collected in mammalian cell cultures, it can be hypothesized that similar mechanisms regulate JH action on adult cricket neurogenesis. According to the scheme proposed in Fig. 3, JH could act on the protooncogenes c-myc and max and induce activation of the ODC gene; the resulting putrescine production could regulate protooncogene expression and/or act on cyclins involved in cell cycle control. Spermidine and spermine could be implicated in cell differentiation, as they are known to promote microtubule formation (53) and to act on cytoskeletal proteins (49). In vitro studies on neuroblast cultures are now required to further develop experimental evidences that support the hypothetical scheme proposed for JH mitogenic activity.
Figure 3.
Schematic representation of a possible mechanism of action of JH on adult neuroblast proliferation. As for vertebrate hormones, JH could act on the protooncogenes c-myc and max (39–41) and induce the activation of ODC gene (42, 43). The resulting putrescine production could regulate protooncogene expression (47–50) and/or act on cyclins involved in cell cycle control (51, 52).
Recent evidence, gathered in different tissues, suggests at least two different sites for the action of JH: the nucleus and the cell membrane (2, 3). The presence of receptors for JH on follicle cell membranes in the ovary, and the hormone action via a cascade including protein kinase C, has now been established (54, 55). In the cricket, α-DFMO treatment of allatectomized females did not prevent the gonadotropic action of JH injection, which led to an increase of fat body spermidine titres and to vitellogenesis. Because vitellogenin synthesis was stimulated and vitellogenin uptake into the oocytes occurred, it can be concluded that polyamines are not involved in the gonadotropic action of JH and that the prevention of JH action through inhibition of polyamine biosynthesis is specific to neural tissue and to neuroblast proliferation.
To our knowledge, these results are the first to demonstrate that putrescine administered in vivo can mimic the stimulation of the morphogenetic hormone, JH, on adult neuroblast proliferation. The present study on a mode of action of JH through a modulation of putrescine titres offers new basis for understanding JH nuclear action, and points out the role of polyamines in the transduction of JH message in neural tissue.
Acknowledgments
We acknowledge Drs. K.G. Davey, E. Hölttä, L. Pradayrol and N. Seiler for their helpful comments during the early writing of the manuscript. We are grateful to Drs. B. Stay, A. Woodhead, and G.R. Wyatt for critically reading the manuscript. We thank the Merrell Dow Research Institute (Strasbourg, France) for the generous gift of α-DFMO. This work was supported by the Centre National de la Recherche Scientifique.
ABBREVIATIONS
- JH
juvenile hormone
- ODC
ornithine decarboxylase
- AdoMetDC
S-adenosylmethionine decarboxylase
- α-DFMO
α-difluoromethylornithine
Footnotes
We detected 5-bromo-2′-deoxyuridine (BrdU) labeled cells in the optic lobes, but we did not determine their neuronal or glial origin (6).
References
- 1.Jones G. Annu Rev Entomol. 1995;40:147–169. doi: 10.1146/annurev.en.40.010195.001051. [DOI] [PubMed] [Google Scholar]
- 2.Riddiford L. In: Metamorphosis: Postembryonic Reprogramming of Gene Expression in Amphibian and Insect Cells. Gilbert L I, Tata J R, Atkinson B G, editors. New York: Academic; 1996. pp. 223–251. [Google Scholar]
- 3.Wyatt G R, Davey K G. Adv Insect Physiol. 1996;26:1–155. [Google Scholar]
- 4.Cayre M, Strambi C, Strambi A. Nature (London) 1994;368:57–59. [Google Scholar]
- 5.Cayre M, Strambi C, Charpin P, Augier R, Strambi A. Arch Insect Biochem Physiol. 1997;35:85–97. [Google Scholar]
- 6.Cayre M, Strambi C, Charpin P, Augier R, Meyer M R, Edwards J S, Strambi A. J Comp Neurol. 1996;371:300–310. doi: 10.1002/(SICI)1096-9861(19960722)371:2<300::AID-CNE9>3.0.CO;2-6. [DOI] [PubMed] [Google Scholar]
- 7.Cayre M, Strambi C, Tirard A, Renucci M, Charpin P, Augier R, Strambi A. Comp Biochem Physiol. 1995;222:241–250. [Google Scholar]
- 8.Cayre M, Strambi C, Charpin P, Augier R, Renucci M, Strambi A. Behav Neurosci. 1996;110:1117–1125. [PubMed] [Google Scholar]
- 9.Heby O. Differentiation. 1981;79:1–20. doi: 10.1111/j.1432-0436.1981.tb01123.x. [DOI] [PubMed] [Google Scholar]
- 10.Tabor C W, Tabor H. Annu Rev Biochem. 1984;53:749–790. doi: 10.1146/annurev.bi.53.070184.003533. [DOI] [PubMed] [Google Scholar]
- 11.Pegg A E. Cancer Res. 1988;48:759–774. [PubMed] [Google Scholar]
- 12.Feinstein S C, Dana S L, McConlogue L, Shooter E M, Coffino P. Proc Natl Acad Sci USA. 1985;82:5761–5765. doi: 10.1073/pnas.82.17.5761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Russell D H. In: The Physiology of Polyamines. Bachrach U, Heimer Y M, editors. Boca Raton, FL: CRC; 1989. pp. 255–280. [Google Scholar]
- 14.Mamont P S, Böhlen P, McCann P P, Bey F, Schuber F, Tardif C. Proc Natl Acad Sci USA. 1976;73:1626–1630. doi: 10.1073/pnas.73.5.1626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Renucci M, Strambi C, Strambi A, Augier R, Charpin P. Gen Comp Endocrinol. 1990;78:137–149. doi: 10.1016/0016-6480(90)90054-p. [DOI] [PubMed] [Google Scholar]
- 16.Bradford M M. Anal Biochem. 1976;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- 17.Strambi C, Faure P, Renucci M, Charpin P, Augier R, Tirard A, Strambi A. Arch Insect Biochem Physiol. 1993;24:203–217. [Google Scholar]
- 18.Gabe M. Techniques Histologiques. Paris: Masson & Cie; 1968. [Google Scholar]
- 19.Cayre M, Tirard A, Charpin P, Augier R, Strambi C, Strambi A. Arch Insect Biochem Physiol. 1993;24:219–228. [Google Scholar]
- 20.Shiharata A, Pegg A E. J Biol Chem. 1986;261:13833–13837. [PubMed] [Google Scholar]
- 21.Williams-Ashman H, Pegg A E. In: Polyamines in Biology and Medicine. Morris D R, Marton L J, editors. New York: Dekker; 1981. pp. 43–72. [Google Scholar]
- 22.Seiler N, Heby O. Acta Biochim Biophys Hung. 1988;23:1–36. [PubMed] [Google Scholar]
- 23.Pegg A E. Biochem J. 1984;224:29–38. doi: 10.1042/bj2240029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Seiler N, Dezeure F. Int J Biochem. 1990;22:211–218. doi: 10.1016/0020-711x(90)90332-w. [DOI] [PubMed] [Google Scholar]
- 25.Strambi C, Tirard A, Renucci M, Faure P, Charpin P, Augier R, Strambi A. In: Insect Juvenile Hormone Research: Fundamental and Applied Approaches. Mauchamp B, Couillaud F, Baehr J C, editors. Paris: INRA; 1992. pp. 193–206. [Google Scholar]
- 26.Herbst E J, Snell E E. J Biol Chem. 1949;181:47–54. [PubMed] [Google Scholar]
- 27.Rudkin B B, Mamont P S, Seiler N. Biochem J. 1984;217:731–741. doi: 10.1042/bj2170731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Wang J Y, McCormack S A, Viar M J, Johnson L R. Am J Physiol. 1991;261:G504–G511. doi: 10.1152/ajpgi.1991.261.3.G504. [DOI] [PubMed] [Google Scholar]
- 29.Bartolome J V, Schweitzer L, Slotkin T A, Nadler J V. Neuroscience. 1985;15:203–213. doi: 10.1016/0306-4522(85)90132-0. [DOI] [PubMed] [Google Scholar]
- 30.Schweitzer L, Nadler J V, Slotkin T A. Neuroscience. 1988;27:453–464. doi: 10.1016/0306-4522(88)90280-1. [DOI] [PubMed] [Google Scholar]
- 31.Schweitzer L, Bell J M, Slotkin T A. Neuroscience. 1987;23:1123–1132. doi: 10.1016/0306-4522(87)90186-2. [DOI] [PubMed] [Google Scholar]
- 32.Pohjanpeltö P, Raina A. Nat New Biol. 1972;235:247–249. doi: 10.1038/newbio235247a0. [DOI] [PubMed] [Google Scholar]
- 33.Goyns M H, Hopkins C R. Cell Mol Biol. 1982;28:61–66. [PubMed] [Google Scholar]
- 34.Salo E, Baguna J. J Exp Zool. 1989;250:150–161. [Google Scholar]
- 35.Hoggard N, Green C D. Mol Cell Endocrinol. 1986;46:71–78. doi: 10.1016/0303-7207(86)90071-7. [DOI] [PubMed] [Google Scholar]
- 36.Marshall L G, Feinstein S C. Exp Cell Res. 1995;216:93–100. doi: 10.1006/excr.1995.1012. [DOI] [PubMed] [Google Scholar]
- 37.Spencer C A, Groudine M. Adv Cancer Res. 1991;56:1–48. doi: 10.1016/s0065-230x(08)60476-5. [DOI] [PubMed] [Google Scholar]
- 38.Marcu K B, Bossone S A, Patel A J. Annu Rev Biochem. 1992;61:809–860. doi: 10.1146/annurev.bi.61.070192.004113. [DOI] [PubMed] [Google Scholar]
- 39.Wang J Y, Wang H, Johnson L. Am J Physiol. 1995;269:C1474–C1481. doi: 10.1152/ajpcell.1995.269.6.C1474. [DOI] [PubMed] [Google Scholar]
- 40.Dubik D, Shiu R P. Oncogene. 1992;7:1587–1594. [PubMed] [Google Scholar]
- 41.Wang S, Bartolome J V, Schanberg S M. J Neurosci. 1996;16:836–842. doi: 10.1523/JNEUROSCI.16-02-00836.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Bello-Fernandez C, Packham G, Cleveland J L. Proc Natl Acad Sci USA. 1993;90:7804–7808. doi: 10.1073/pnas.90.16.7804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Pena A, Reddy C D, Wu S, Hickok N J, Reddy E P, Yumet G, Soprano D R, Soprano K J. J Biol Chem. 1993;268:27277–27285. [PubMed] [Google Scholar]
- 44.Auvinen M, Paasinen A, Andersson L C, Hölttä E. Nature (London) 1992;360:355–358. doi: 10.1038/360355a0. [DOI] [PubMed] [Google Scholar]
- 45.Behe M, Felsenfeld G. Proc Natl Acad Sci USA. 1981;78:1619–1623. doi: 10.1073/pnas.78.3.1619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Tofilon P J, Oredsson S M, Seen D F, Marton L M. Science. 1982;217:1044–1046. doi: 10.1126/science.6810463. [DOI] [PubMed] [Google Scholar]
- 47.Panagiotidis C A, Artandi S, Calame K, Silverstein S J. Nucleic Acids Res. 1995;23:1800–1809. doi: 10.1093/nar/23.10.1800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Celano P, Baylin S B, Casero R A., Jr J Biol Chem. 1989;264:8922–8927. [PubMed] [Google Scholar]
- 49.Hölttä E, Auvinen M, Andersson L C. J Cell Biol. 1993;122:903–914. doi: 10.1083/jcb.122.4.903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Tabib A, Bachrach U. Biochem Biophys Res Commun. 1994;202:720–727. doi: 10.1006/bbrc.1994.1990. [DOI] [PubMed] [Google Scholar]
- 51.Thomas T, Thomas T J. Cancer Res. 1994;54:1077–1084. [PubMed] [Google Scholar]
- 52.Thomas T, Gallo M A, Klinge C M, Thomas T J. J Steroid Biochem Mol Biol. 1995;54:89–99. doi: 10.1016/0960-0760(95)00126-k. [DOI] [PubMed] [Google Scholar]
- 53.Anderson P J, Bardocz S, Campos R, Brown D L. Biochem Biophys Res Commun. 1985;137:147–154. doi: 10.1016/0006-291x(85)91000-9. [DOI] [PubMed] [Google Scholar]
- 54.Ilenchuck T T, Davey K G. Can J Biochem Cell Biol. 1985;63:102–106. [Google Scholar]
- 55.Sevala V L, Davey K G. Invertebr Reprod Dev. 1993;24:39–46. [Google Scholar]