Abstract
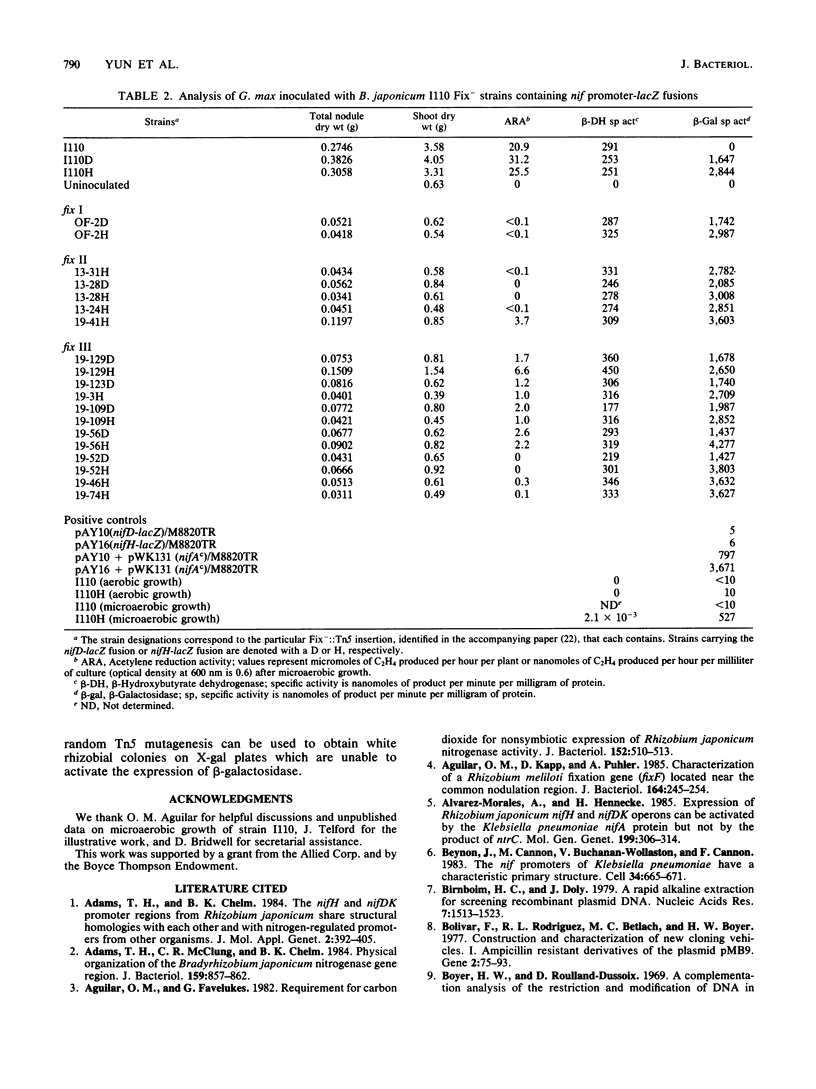
DNA fragments containing either the nifD or nifH promoter and 5' structural gene sequences from Bradyrhizobium japonicum I110 were fused in frame to the lacZ gene. Stable integration of these nif promoter-lacZ fusions by homologous double reciprocal crossover into a symbiotically nonessential region of the B. japonicum chromosome provided an easy assay for the effects of potential nif regulatory mutants. The level of beta-galactosidase activity expressed from these two nif promoter-lacZ fusions was assayed in bacteroids of B. japonicum I110 wild type and Fix mutants generated by transposon Tn5 mutagenesis and identified in the accompanying paper. No nif-positive regulatory mutants were identified from among an array of Fix- mutants in which Tn5 was inserted 9 kilobase pairs upstream of the nifDK operon and within the 18-kilobase-pair region separating the nifDK and nifH operons. This result indicates that there are no genes in these regions involved in the regulation of nitrogenase structural gene expression. Interestingly, the level of beta-galactosidase activity expressed from the nifH promoter was twice that expressed from the nifD promoter, suggesting that the normal cellular level of the nifH gene product in bacteroids is in a 2:1 ratio with the nifD gene product instead of in the 1:1 stoichiometry of the nitrogenase enzyme complex.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Adams T. H., Chelm B. K. The nifH and nifDK promoter regions from Rhizobium japonicum share structural homologies with each other and with nitrogen-regulated promoters from other organisms. J Mol Appl Genet. 1984;2(4):392–405. [PubMed] [Google Scholar]
- Adams T. H., McClung C. R., Chelm B. K. Physical organization of the Bradyrhizobium japonicum nitrogenase gene region. J Bacteriol. 1984 Sep;159(3):857–862. doi: 10.1128/jb.159.3.857-862.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aguilar O. M., Favelukes G. Requirement for carbon dioxide for nonsymbiotic expression of Rhizobium japonicum nitrogenase activity. J Bacteriol. 1982 Oct;152(1):510–513. doi: 10.1128/jb.152.1.510-513.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aguilar O. M., Kapp D., Pühler A. Characterization of a Rhizobium meliloti fixation gene (fixF) located near the common nodulation region. J Bacteriol. 1985 Oct;164(1):245–254. doi: 10.1128/jb.164.1.245-254.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alvarez-Morales A., Hennecke H. Expression of Rhizobium japonicum nifH and nifDK operons can be activated by the Klebsiella pneumonia nifA protein but not by the product of ntrC. Mol Gen Genet. 1985;199(2):306–314. doi: 10.1007/BF00330273. [DOI] [PubMed] [Google Scholar]
- Beynon J., Cannon M., Buchanan-Wollaston V., Cannon F. The nif promoters of Klebsiella pneumoniae have a characteristic primary structure. Cell. 1983 Sep;34(2):665–671. doi: 10.1016/0092-8674(83)90399-9. [DOI] [PubMed] [Google Scholar]
- Birnboim H. C., Doly J. A rapid alkaline extraction procedure for screening recombinant plasmid DNA. Nucleic Acids Res. 1979 Nov 24;7(6):1513–1523. doi: 10.1093/nar/7.6.1513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bolivar F., Rodriguez R. L., Betlach M. C., Boyer H. W. Construction and characterization of new cloning vehicles. I. Ampicillin-resistant derivatives of the plasmid pMB9. Gene. 1977;2(2):75–93. doi: 10.1016/0378-1119(77)90074-9. [DOI] [PubMed] [Google Scholar]
- Boyer H. W., Roulland-Dussoix D. A complementation analysis of the restriction and modification of DNA in Escherichia coli. J Mol Biol. 1969 May 14;41(3):459–472. doi: 10.1016/0022-2836(69)90288-5. [DOI] [PubMed] [Google Scholar]
- Castilho B. A., Olfson P., Casadaban M. J. Plasmid insertion mutagenesis and lac gene fusion with mini-mu bacteriophage transposons. J Bacteriol. 1984 May;158(2):488–495. doi: 10.1128/jb.158.2.488-495.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chinault A. C., Carbon J. Overlap hybridization screening: isolation and characterization of overlapping DNA fragments surrounding the leu2 gene on yeast chromosome III. Gene. 1979 Feb;5(2):111–126. doi: 10.1016/0378-1119(79)90097-0. [DOI] [PubMed] [Google Scholar]
- Dhaese P., De Greve H., Decraemer H., Schell J., Van Montagu M. Rapid mapping of transposon insertion and deletion mutations in the large Ti-plasmids of Agrobacterium tumefaciens. Nucleic Acids Res. 1979 Dec 11;7(7):1837–1849. doi: 10.1093/nar/7.7.1837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dixon R. A. The genetic complexity of nitrogen fixation. The ninth Fleming lecture. J Gen Microbiol. 1984 Nov;130(11):2745–2755. doi: 10.1099/00221287-130-11-2745. [DOI] [PubMed] [Google Scholar]
- Hardy R. W., Holsten R. D., Jackson E. K., Burns R. C. The acetylene-ethylene assay for n(2) fixation: laboratory and field evaluation. Plant Physiol. 1968 Aug;43(8):1185–1207. doi: 10.1104/pp.43.8.1185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Magasanik B. Genetic control of nitrogen assimilation in bacteria. Annu Rev Genet. 1982;16:135–168. doi: 10.1146/annurev.ge.16.120182.001031. [DOI] [PubMed] [Google Scholar]
- Noti J. D., Folkerts O., Turken A. N., Szalay A. A. Organization and characterization of genes essential for symbiotic nitrogen fixation from Bradyrhizobium japonicum I110. J Bacteriol. 1986 Sep;167(3):774–783. doi: 10.1128/jb.167.3.774-783.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roberts G. P., Brill W. J. Genetics and regulation of nitrogen fixation. Annu Rev Microbiol. 1981;35:207–235. doi: 10.1146/annurev.mi.35.100181.001231. [DOI] [PubMed] [Google Scholar]
- Ruvkun G. B., Sundaresan V., Ausubel F. M. Directed transposon Tn5 mutagenesis and complementation analysis of Rhizobium meliloti symbiotic nitrogen fixation genes. Cell. 1982 Jun;29(2):551–559. doi: 10.1016/0092-8674(82)90171-4. [DOI] [PubMed] [Google Scholar]
- Sundaresan V., Jones J. D., Ow D. W., Ausubel F. M. Klebsiella pneumoniae nifA product activates the Rhizobium meliloti nitrogenase promoter. Nature. 1983 Feb 24;301(5902):728–732. doi: 10.1038/301728a0. [DOI] [PubMed] [Google Scholar]
- Sundaresan V., Ow D. W., Ausubel F. M. Activation of Klebsiella pneumoniae and Rhizobium meliloti nitrogenase promoters by gln (ntr) regulatory proteins. Proc Natl Acad Sci U S A. 1983 Jul;80(13):4030–4034. doi: 10.1073/pnas.80.13.4030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Szeto W. W., Zimmerman J. L., Sundaresan V., Ausubel F. M. A Rhizobium meliloti symbiotic regulatory gene. Cell. 1984 Apr;36(4):1035–1043. doi: 10.1016/0092-8674(84)90053-9. [DOI] [PubMed] [Google Scholar]
- Wong P. P., Evans H. J. Poly-beta-hydroxybutyrate Utilization by Soybean (Glycine max Merr.) Nodules and Assessment of Its Role in Maintenance of Nitrogenase Activity. Plant Physiol. 1971 Jun;47(6):750–755. doi: 10.1104/pp.47.6.750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yun A. C., Szalay A. A. Structural genes of dinitrogenase and dinitrogenase reductase are transcribed from two separate promoters in the broad host range cowpea Rhizobium strain IRc78. Proc Natl Acad Sci U S A. 1984 Dec;81(23):7358–7362. doi: 10.1073/pnas.81.23.7358. [DOI] [PMC free article] [PubMed] [Google Scholar]