Abstract
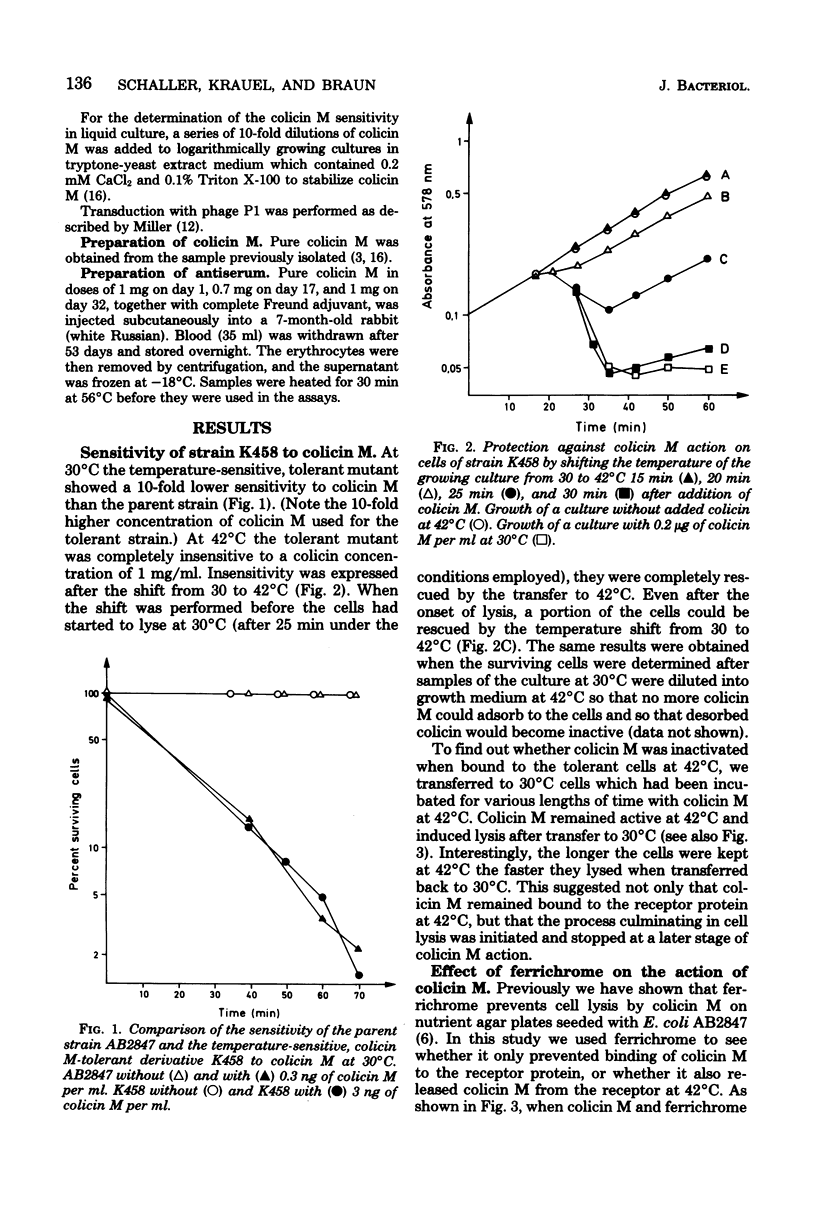
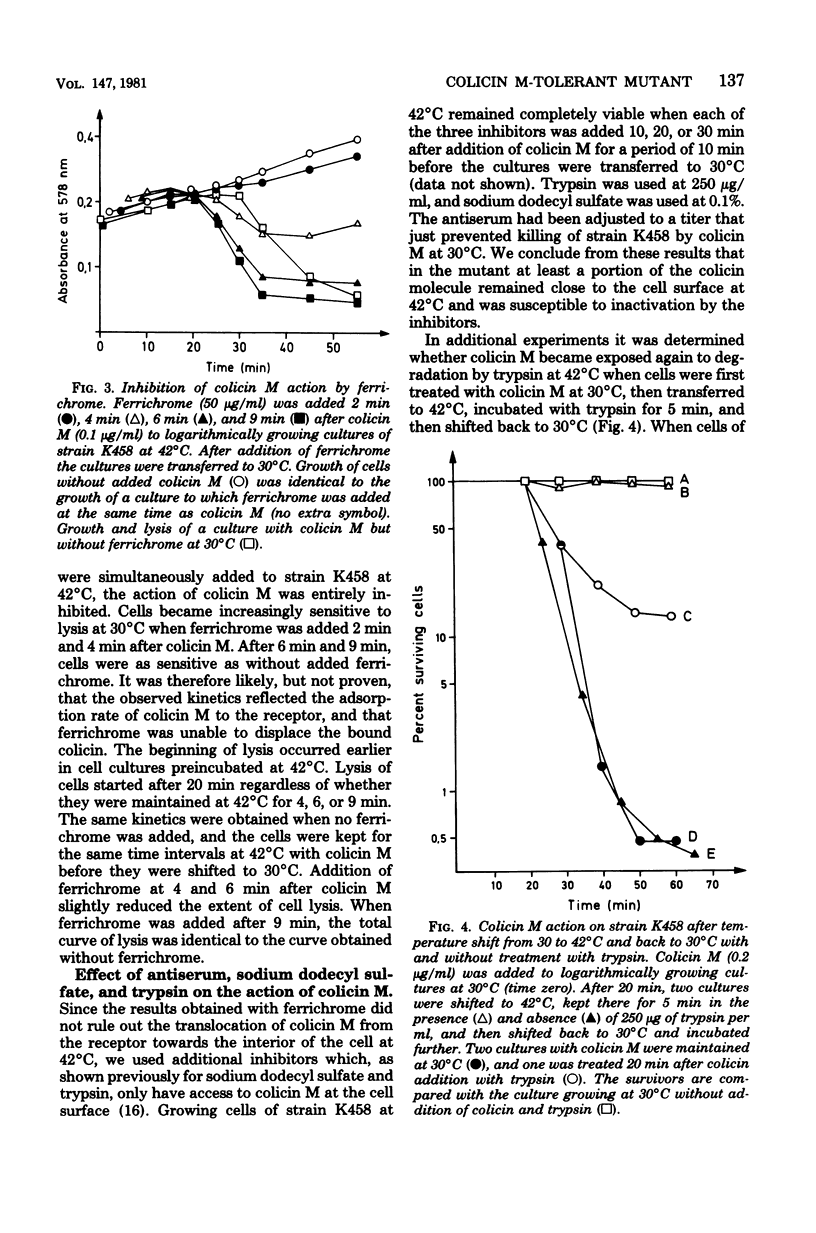
A mutant sensitive to colicin M at 30 degrees C and tolerant at 42 degrees C to high concentrations of colicin M was isolated from Escherichia coli K-12. A temperature shift from 30 to 42 degrees C rescued all cells up to the time they started to lyse at 30 degrees C (25 min after addition of colicin M). The growth rate at 42 degrees C remained unaffected by colicin M. AT 42 degrees C the cell-bound colicin M was inactivated by trypsin, sodium dodecyl sulfate, and antiserum against colicin M. Ferrichrome competed with colicin M at 42 degrees C only during the initial adsorption to the common receptor protein in the outer membrane. Since cells lysed earlier at 30 degrees C when they had been preincubated with colicin M at 42 degrees C, we conclude that the process leading finally to cell lysis is initiated at 42 degrees C and stops at a later stage of colicin M trypsin, dodecyl sulfate, and antiserum when cells were transferred from 30 to 42 degrees C, we assume that colicin M is translocated from its target site towards the cell surface. The mutation conferring tolerance was mapped close to the rpsL gene.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Braun V., Frenz J., Hantke K., Schaller K. Penetration of colicin M into cells of Escherichia coli. J Bacteriol. 1980 Apr;142(1):162–168. doi: 10.1128/jb.142.1.162-168.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Braun V., Schaller K., Wabl M. R. Isolation, characterization, and action of colicin M. Antimicrob Agents Chemother. 1974 May;5(5):520–533. doi: 10.1128/aac.5.5.520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Braun V., Schaller K., Wolff H. A common receptor protein for phage T5 and colicin M in the outer membrane of Escherichia coli B. Biochim Biophys Acta. 1973 Sep 27;323(1):87–97. doi: 10.1016/0005-2736(73)90433-1. [DOI] [PubMed] [Google Scholar]
- Davies J. K., Reeves P. Genetics of resistance to colicins in Escherichia coli K-12: cross-resistance among colicins of group B. J Bacteriol. 1975 Jul;123(1):96–101. doi: 10.1128/jb.123.1.96-101.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hantke K., Braun V. Functional interaction of the tonA/tonB receptor system in Escherichia coli. J Bacteriol. 1978 Jul;135(1):190–197. doi: 10.1128/jb.135.1.190-197.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hardy K. G. Colicinogeny and related phenomena. Bacteriol Rev. 1975 Dec;39(4):464–515. doi: 10.1128/br.39.4.464-515.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kadner R. J., Heller K., Coulton J. W., Braun V. Genetic control of hydroxamate-mediated iron uptake in Escherichia coli. J Bacteriol. 1980 Jul;143(1):256–264. doi: 10.1128/jb.143.1.256-264.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagel de Zwaig R., Luria S. E. Genetics and physiology of colicin-tolerant mutants of Escherichia coli. J Bacteriol. 1967 Oct;94(4):1112–1123. doi: 10.1128/jb.94.4.1112-1123.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nomura M. Colicins and related bacteriocins. Annu Rev Microbiol. 1967;21:257–284. doi: 10.1146/annurev.mi.21.100167.001353. [DOI] [PubMed] [Google Scholar]
- Nomura M., Witten C. Interaction of colicins with bacterial cells. 3. Colicin-tolerant mutations in Escherichia coli. J Bacteriol. 1967 Oct;94(4):1093–1111. doi: 10.1128/jb.94.4.1093-1111.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schaller K., Dreher R., Braun V. Structural and functional properties of colicin M. J Bacteriol. 1981 Apr;146(1):54–63. doi: 10.1128/jb.146.1.54-63.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]



