Abstract
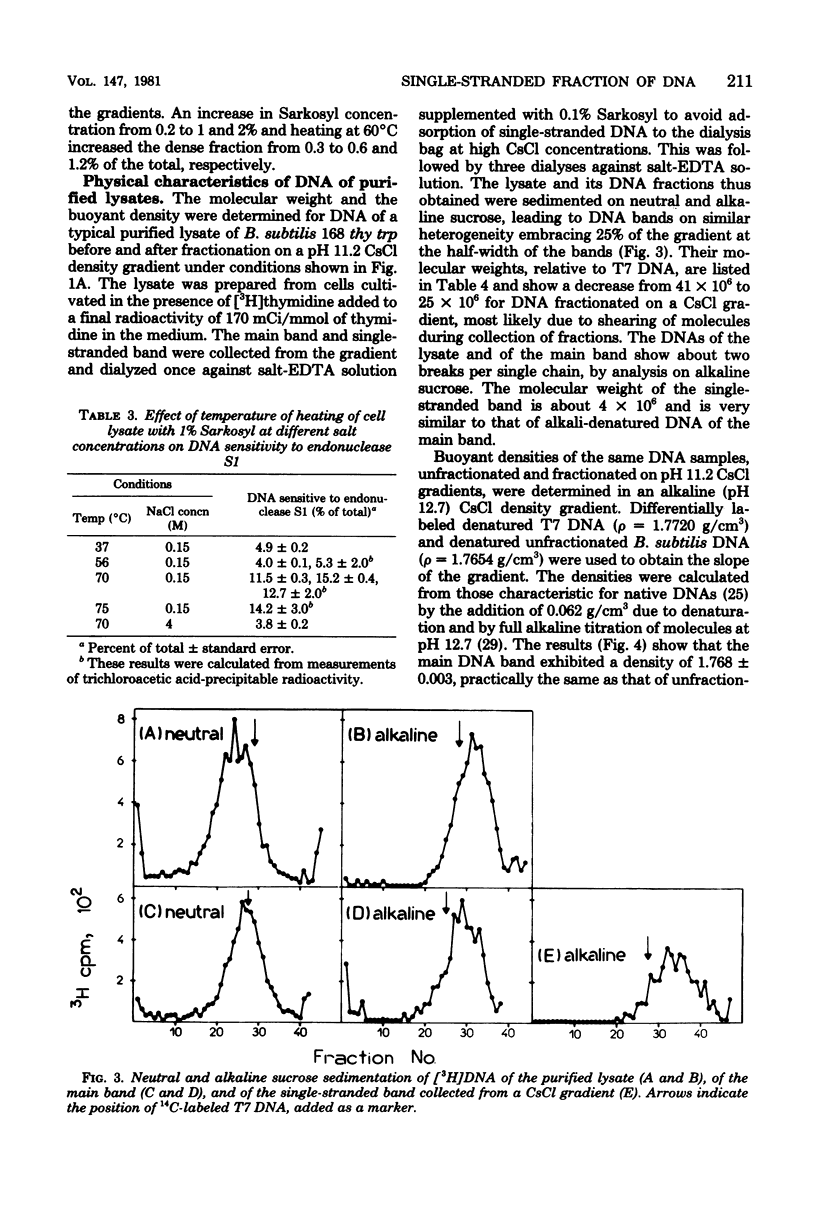
About 13% of the deoxyribonucleic acid (DNA) of various strains of Bacillus subtilis, independent of the stage of growth or competence for transformation, was rendered acid soluble by endonuclease S1. In a pH 11.2 CsCl gradient, 4% of the untreated DNA banded at the density typical for single-stranded molecules, whereas 9% of the remaining DNA (main band) was sensitive to endonuclease S1. Selective inhibition of DNA polymerase III, or of DNA-dependent ribonucleic acid polymerase, did not increase or abolish single-strandedness. The DNA purification procedure did affect the level of single-stranded DNA, indicating its binding to cell constituents containing ribonucleic acid, protein, and membranous material. The molecular weight of the single-stranded fraction resembled that of total denatured DNA, and its buoyant density in an alkaline CsCl gradient was centered partially at a density of 1.772 g/cm3 and partially at a density of 7.759 g/cm3. Incubation of DNA under conditions leading to renaturation of its single-stranded fraction led to an increase in transforming activity for the purA16+ marker (close to the origin of replication) relative to leu-8+ and metC3+ markers (located in the middle of the chromosome), indicating this region is the main source of the single-stranded fraction.
Full text
PDF









Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alberts B. M., Doty P. Characterization of a naturally occurring, cross-linked fraction of DNA. 1. Nature of the cross-linkage. J Mol Biol. 1968 Mar 14;32(2):379–403. doi: 10.1016/0022-2836(68)90017-x. [DOI] [PubMed] [Google Scholar]
- Anderson E. H. Growth Requirements of Virus-Resistant Mutants of Escherichia Coli Strain "B". Proc Natl Acad Sci U S A. 1946 May;32(5):120–128. doi: 10.1073/pnas.32.5.120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aubert J. P., Ryter A., Schaeffer P. Comportement de l'ADN des bactéries et des spores au cours d'un cycle sporal chez B. subtilis. Ann Inst Pasteur (Paris) 1968 Dec;115(6):990–1007. [PubMed] [Google Scholar]
- BURGI E., HERSHEY A. D. Sedimentation rate as a measure of molecular weight of DNA. Biophys J. 1963 Jul;3:309–321. doi: 10.1016/s0006-3495(63)86823-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown N. C. Inhibition of bacterial DNA replication by 6-(p-hydroxyphenylazo)-uracil: differential effect on repair and semi-conservative synthesis in Bacillus subtilis. J Mol Biol. 1971 Jul 14;59(1):1–16. doi: 10.1016/0022-2836(71)90409-8. [DOI] [PubMed] [Google Scholar]
- Burgi A. W., Robinton J., Carlson C. L. Studies on the folded chromosome of Escherichia coli. Cold Spring Harb Symp Quant Biol. 1974;38:43–51. doi: 10.1101/sqb.1974.038.01.007. [DOI] [PubMed] [Google Scholar]
- Cahn F. H., Fox M. S. Fractionation of transformable bacteria from ocompetent cultures of Bacillus subtilis on renografin gradients. J Bacteriol. 1968 Mar;95(3):867–875. doi: 10.1128/jb.95.3.867-875.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calvori C., Frontali L., Leoni L., Tecce G. Effect of rifamycin on protein synthesis. Nature. 1965 Jul 24;207(995):417–418. doi: 10.1038/207417a0. [DOI] [PubMed] [Google Scholar]
- Deddish P. A., Ravin A. W. Single-stranded regions in Streptococcus pneumoniae chromosomal deoxyribonucleic acid and their relation to transformation. J Bacteriol. 1979 Mar;137(3):1191–1199. doi: 10.1128/jb.137.3.1191-1199.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dodgson J. B., Wells R. D. Action of single-strand specific nucleases on model DNA heteroduplexes of defined size and sequence. Biochemistry. 1977 May 31;16(11):2374–2379. doi: 10.1021/bi00630a010. [DOI] [PubMed] [Google Scholar]
- Harford N. Bidirectional chromosome replication in Bacillus subtilis 168. J Bacteriol. 1975 Mar;121(3):835–847. doi: 10.1128/jb.121.3.835-847.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harris W. J., Barr G. C. Structural features of DNA in competent Bacillus subtilis. Mol Gen Genet. 1971;113(4):316–330. doi: 10.1007/BF00272332. [DOI] [PubMed] [Google Scholar]
- LeClerc J. E., Setlow J. K. Single-strand regions in the deoxyribonucleic acid of competent Haemophilus influenzae. J Bacteriol. 1975 Jun;122(3):1091–1102. doi: 10.1128/jb.122.3.1091-1102.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leibowitz P. J., Schaechter M. The attachment of the bacterial chromosome to the cell membrane. Int Rev Cytol. 1975;41:1–28. doi: 10.1016/s0074-7696(08)60964-x. [DOI] [PubMed] [Google Scholar]
- MARMUR J., DOTY P. Thermal renaturation of deoxyribonucleic acids. J Mol Biol. 1961 Oct;3:585–594. doi: 10.1016/s0022-2836(61)80023-5. [DOI] [PubMed] [Google Scholar]
- Marvin D. A., Hohn B. Filamentous bacterial viruses. Bacteriol Rev. 1969 Jun;33(2):172–209. doi: 10.1128/br.33.2.172-209.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ogasawara N., Seiki M., Yoshikawa H. Effect of novobiocin on initiation of DNA replication in Bacillus subtilis. Nature. 1979 Oct 25;281(5733):702–704. doi: 10.1038/281702a0. [DOI] [PubMed] [Google Scholar]
- Oishi M. Studies of DNA replication in vivo. I. Isolation of the first intermediate of DNA replication in bacteria as single-stranded DNA. Proc Natl Acad Sci U S A. 1968 May;60(1):329–336. doi: 10.1073/pnas.60.1.329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paetkau V., Langman L., Miller R. C., Jr The origin of nascent single-stranded fragments in replicating TM DNA. J Mol Biol. 1975 Nov 15;98(4):719–737. doi: 10.1016/s0022-2836(75)80006-4. [DOI] [PubMed] [Google Scholar]
- Piechowska M., Fox M. S. Fate of transforming deoxyribonucleate in Bacillus subtilis. J Bacteriol. 1971 Nov;108(2):680–689. doi: 10.1128/jb.108.2.680-689.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Piechowska M. Jednołańcuchowe frakcje dwułańcuchowego DNA. Postepy Biochem. 1977;23(2):139–156. [PubMed] [Google Scholar]
- SCHILDKRAUT C. L., MARMUR J., DOTY P. Determination of the base composition of deoxyribonucleic acid from its buoyant density in CsCl. J Mol Biol. 1962 Jun;4:430–443. doi: 10.1016/s0022-2836(62)80100-4. [DOI] [PubMed] [Google Scholar]
- STUDIER F. W. SEDIMENTATION STUDIES OF THE SIZE AND SHAPE OF DNA. J Mol Biol. 1965 Feb;11:373–390. doi: 10.1016/s0022-2836(65)80064-x. [DOI] [PubMed] [Google Scholar]
- Soltyk A., Shugar D., Piechowska M. Heterologous deoxyribonucleic acid uptake and complexing with cellular constituents in competent Bacillus subtilis. J Bacteriol. 1975 Dec;124(3):1429–1438. doi: 10.1128/jb.124.3.1429-1438.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Toyoda H., Yamaguchi K., Yoshikawa H. Chromosome-membrane association in Bacillus subtilis. IV. Further purification of DNA-membrane complex by using a combination of centrifugation and electrophoresis. J Bacteriol. 1978 Nov;136(2):799–802. doi: 10.1128/jb.136.2.799-802.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- VINOGRAD J., MORRIS J., DAVIDSON N., DOVE W. F., Jr The bouyant behavior of viral and bacterial DNA in alkaline CsCl. Proc Natl Acad Sci U S A. 1963 Jan 15;49:12–17. doi: 10.1073/pnas.49.1.12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vogt V. M. Purification and further properties of single-strand-specific nuclease from Aspergillus oryzae. Eur J Biochem. 1973 Feb 15;33(1):192–200. doi: 10.1111/j.1432-1033.1973.tb02669.x. [DOI] [PubMed] [Google Scholar]
- Wanka F., Brouns R. M., Aelen J. M., Eygensteyn A., Eygensteyn J. The origin of nascent single-stranded DNA extracted from mammalian cells. Nucleic Acids Res. 1977 Jun;4(6):2083–2097. doi: 10.1093/nar/4.6.2083. [DOI] [PMC free article] [PubMed] [Google Scholar]



