Abstract
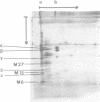
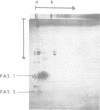
We have purified the F1-F0 adenosine triphosphatase complex from wild-type Streptococcus faecalis ATCC 9790 and an N,N'-dicyclohexylcarbodiimide (DCCD)-resistant mutant strain, SF-dcc-8. For preliminary purification of the complex, reconstituted F1-F0, prepared from isolated F1 adenosine triphosphatase and depleted membranes, was extracted with sodium deoxycholate and fractionated by salt precipitation. By means of two-dimensional gel electrophoresis, the F1-F0 complex was purified as a single, catalytically active band in the first dimension and then resolved into constituent subunits under denaturing conditions in the second dimension. The electrophoretic purification of F1-F0 removed a delta-less form of F1 as well as other impurities, including lipoteichoic acid. Both the DCCD-sensitive and the DCCD-resistant F1-F0 adenosine triphosphatase appeared to consist of eight proteins, five of which corresponded to the F1 subunits alpha, beta,, gamma, delta, and epsilon. The F0 sector proteins, designated M27, M15, and M6, had Mr values of 27,000, 15,000, and 6,000, respectively. There appear to be multiple copies of M6 in the complex. [14C]DCCD reacted specifically and covalently with M6 in the wild-type F1-F0 but failed to label the M6 protein in the complex from the DCCD-resistant strain. It is suggested that DCCD resistance in the SF-dcc-8 mutant may be due to a modification of the M6 protein which hinders access of DCCD to the reactive site.
Full text
PDF









Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- ABRAMS A., McNAMARA P., JOHNSON F. B. Adenosine triphosphatase in isolated bacterial cell membranes. J Biol Chem. 1960 Dec;235:3649–3662. [PubMed] [Google Scholar]
- Abrams A., Baron C., Schnebli H. P. The isolation of bacterial membrane ATPase and nectin. Methods Enzymol. 1974;32:428–439. doi: 10.1016/0076-6879(74)32042-3. [DOI] [PubMed] [Google Scholar]
- Abrams A., Baron C. The isolation and subunit structure of streptococcal membrane adenosine triphosphatase. Biochemistry. 1967 Jan;6(1):225–229. doi: 10.1021/bi00853a035. [DOI] [PubMed] [Google Scholar]
- Abrams A., Jensen C., Morris D. H. Role of Mg2+ ions in the subunit structure and membrane binding properties of bacterial energy transducing ATPase. Biochem Biophys Res Commun. 1976 Apr 5;69(3):804–811. doi: 10.1016/0006-291x(76)90946-3. [DOI] [PubMed] [Google Scholar]
- Abrams A., Morris D., Jensen C. Chymotryptic conversion of bacterial membrane ATPase to an active form with modified alpha chains and defective membrane binding properties. Biochemistry. 1976 Dec 14;15(25):5560–5566. doi: 10.1021/bi00670a021. [DOI] [PubMed] [Google Scholar]
- Abrams A., Smith J. B., Baron C. Carbodiimide-resistant membrane adenosine triphosphatase in mutants of Streptococcus faecalis. I. Studies of the mechanism of resistance. J Biol Chem. 1972 Mar 10;247(5):1484–1488. [PubMed] [Google Scholar]
- Abrams A. The release of bound adenosine triphosphatase from isolated bacterial membranes and the properties of the solubilized enzyme. J Biol Chem. 1965 Sep;240(9):3675–3681. [PubMed] [Google Scholar]
- Cohen N. S., Lee S. H., Brodie A. F. Purification and characteristics of hydrophobic membrane protein(s) required for DCCD sensitivity of ATPase in Mycobacterium phlei. J Supramol Struct. 1978;8(1):111–117. doi: 10.1002/jss.400080109. [DOI] [PubMed] [Google Scholar]
- Downie J. A., Gibson F., Cox G. B. Membrane adenosine triphosphatases of prokaryotic cells. Annu Rev Biochem. 1979;48:103–131. doi: 10.1146/annurev.bi.48.070179.000535. [DOI] [PubMed] [Google Scholar]
- Fillingame R. H. Identification of the dicyclohexylcarbodiimide-reactive protein component of the adenosine 5'-triphosphate energy-transducing system of Escherichia coli. J Bacteriol. 1975 Nov;124(2):870–883. doi: 10.1128/jb.124.2.870-883.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fillingame R. H. Purification of the carbodiimide-reactive protein component of the ATP energy-transducing system of Escherichia coli. J Biol Chem. 1976 Nov 10;251(21):6630–6637. [PubMed] [Google Scholar]
- Foster D. L., Fillingame R. H. Energy-transducing H+-ATPase of Escherichia coli. Purification, reconstitution, and subunit composition. J Biol Chem. 1979 Sep 10;254(17):8230–8236. [PubMed] [Google Scholar]
- Friedl P., Friedl C., Schairer H. U. The ATP synthetase of Escherichia coli K12: purification of the enzyme and reconstitution of energy-transducing activities. Eur J Biochem. 1979 Oct;100(1):175–180. doi: 10.1111/j.1432-1033.1979.tb02046.x. [DOI] [PubMed] [Google Scholar]
- Ganfield M. C., Pieringer R. A. Phosphatidylkojibiosyl diglyceride. The covalently linked lipid constituent of the membrane lipoteichoic acid from Streptococcus faecalis (faecium) ATCC 9790. J Biol Chem. 1975 Jan 25;250(2):702–709. [PubMed] [Google Scholar]
- Glossmann H., Neville D. M., Jr Glycoproteins of cell surfaces. A comparative study of three different cell surfaces of the rat. J Biol Chem. 1971 Oct 25;246(20):6339–6346. [PubMed] [Google Scholar]
- Hare J. F. Purification and characterization of a dicyclohexylcarbodiimide-sensitive adenosine triphosphatase complex from membranes of Escherichia coli. Biochem Biophys Res Commun. 1975 Oct 27;66(4):1329–1337. doi: 10.1016/0006-291x(75)90505-7. [DOI] [PubMed] [Google Scholar]
- Harold F. M., Baarda J. R., Baron C., Abrams A. Inhibition of membrane-bound adenosine triphosphatase and of cation transport in Streptococcus faecalis by N,N'-dicyclohexylcarbodiimide. J Biol Chem. 1969 May 10;244(9):2261–2268. [PubMed] [Google Scholar]
- Harold F. M., Spitz E. Accumulation of arsenate, phosphate, and aspartate by Sreptococcus faecalis. J Bacteriol. 1975 Apr;122(1):266–277. doi: 10.1128/jb.122.1.266-277.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoppe J., Schairer H. U., Sebald W. The proteolipid of a mutant ATPase from Escherichia coli defective in H+-conduction contains a glycine instead of the carbodiimide-reactive aspartyl residue. FEBS Lett. 1980 Jan 1;109(1):107–111. doi: 10.1016/0014-5793(80)81321-4. [DOI] [PubMed] [Google Scholar]
- Kessler R. E., Shockman G. D. Precursor-product relationship of intracellular and extracellular lipoteichoic acids of Streptococcus faecium. J Bacteriol. 1979 Feb;137(2):869–877. doi: 10.1128/jb.137.2.869-877.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Leimgruber R. M., Jensen C., Abrams A. Accessibility of the alpha chains in membrane-bound and solubilized bacterial ATPase to chymotryptic cleavage. Biochem Biophys Res Commun. 1978 Mar 30;81(2):439–447. doi: 10.1016/0006-291x(78)91553-x. [DOI] [PubMed] [Google Scholar]
- Rosen B. P., Hasan S. M. Purification of an N,N'-dicyclohexylcarbodiimide-sensitive ATPase from Escherichia coli. FEBS Lett. 1979 Aug 15;104(2):339–342. doi: 10.1016/0014-5793(79)80847-9. [DOI] [PubMed] [Google Scholar]
- Schnebli H. P., Abrams A. Membrane adenosine triphosphatase from Streptococcus faecalis. Preparation and homogeneity. J Biol Chem. 1970 Mar 10;245(5):1115–1121. [PubMed] [Google Scholar]
- Schnebli H. P., Vatter A. E., Abrams A. Membrane adenosine triphosphatase from Streptococcus faecalis. Molecular weight, subunit structure, and amino acid composition. J Biol Chem. 1970 Mar 10;245(5):1122–1127. [PubMed] [Google Scholar]
- Schneider E., Altendorf K. Reconstitution of the purified proton conductor (F0) of the adenosine triphosphatase complex from Escherichia coli. FEBS Lett. 1980 Jul 28;116(2):173–176. doi: 10.1016/0014-5793(80)80636-3. [DOI] [PubMed] [Google Scholar]
- Sebald W., Machleidt W., Wachter E. N,N'-dicyclohexylcarbodiimide binds specifically to a single glutamyl residue of the proteolipid subunit of the mitochondrial adenosinetriphosphatases from Neurospora crassa and Saccharomyces cerevisiae. Proc Natl Acad Sci U S A. 1980 Feb;77(2):785–789. doi: 10.1073/pnas.77.2.785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simoni R. D., Postma P. W. The energetics of bacterial active transport. Annu Rev Biochem. 1975;44:523–554. doi: 10.1146/annurev.bi.44.070175.002515. [DOI] [PubMed] [Google Scholar]
- Sone N., Yoshida M., Hirata H., Kagawa Y. Purification and properties of a dicyclohexylcarbodiimide-sensitive adenosine triphosphatase from a thermophilic bacterium. J Biol Chem. 1975 Oct 10;250(19):7917–7923. [PubMed] [Google Scholar]
- Sone N., Yoshida M., Hirata H., Kagawa Y. Resolution of the membrane moiety of the H+-ATPase complex into two kinds of subunits. Proc Natl Acad Sci U S A. 1978 Sep;75(9):4219–4223. doi: 10.1073/pnas.75.9.4219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Todd R. D., Griesenbeck T. A., Douglas M. G. The yeast mitochondrial adenosine triphosphatase complex. Subunit stoichiometry and physical characterization. J Biol Chem. 1980 Jun 10;255(11):5461–5467. [PubMed] [Google Scholar]
- Wachter E., Schmid R., Deckers G., Altendorf K. Amino acid replacement in dicyclohexylcarbodiimide-reactive proteins from mutant strains of Escherichia coli defective in the energy-transducing ATPase complex. FEBS Lett. 1980 May 5;113(2):265–270. doi: 10.1016/0014-5793(80)80606-5. [DOI] [PubMed] [Google Scholar]
- Weber K., Osborn M. The reliability of molecular weight determinations by dodecyl sulfate-polyacrylamide gel electrophoresis. J Biol Chem. 1969 Aug 25;244(16):4406–4412. [PubMed] [Google Scholar]