Abstract
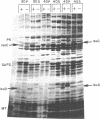
The isopropylmalate isomerase in Salmonella typhimurium is the second enzyme specific for leucine biosynthesis. It is a complex enzyme composed of two subunits which are coded for by two genes of the leucine operon, leuC and leuD. The two polypeptides have been shown to copurify through successive ammonium sulfate fractionations and have been identified on sodium dodecyl sulfate-polyacrylamide gels as having molecular weights of 51,000 (leuC gene product) and 23,500 (leuD gene product). They have also been shown to be fairly stable, since in vitro complementation of cell-free extracts of leuC and leuD mutant strains was demonstrated, with only a 40% loss of activity 16 h after preparation of the extracts. The native isopropylmalate isomerase was shown to have a Km for its substrate alpha-isopropylmalate of 3 x 10(-4)M.
Full text
PDF









Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bigelis R., Umbarger H. E. Purification of yeast alpha-isopropylmalate isomerase. High ionic strength hydrophobic chromatography. J Biol Chem. 1975 Jun 10;250(11):4315–4321. [PubMed] [Google Scholar]
- Bigelis R., Umbarger H. E. Yeast alpha-isopropylmalate isomerase. Factors affecting stability and enzyme activity. J Biol Chem. 1976 Jun 25;251(12):3545–3552. [PubMed] [Google Scholar]
- Burns R. O., Calvo J., Margolin P., Umbarger H. E. Expression of the leucine operon. J Bacteriol. 1966 Apr;91(4):1570–1576. doi: 10.1128/jb.91.4.1570-1576.1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calvo J. M., Worden H. E. A multisite-mutation map of the leucine operon of Salmonella typhimurium. Genetics. 1970 Feb;64(2):199–214. doi: 10.1093/genetics/64.2.199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crawford I. P. Gene rearrangements in the evolution of the tryptophan synthetic pathway. Bacteriol Rev. 1975 Jun;39(2):87–120. doi: 10.1128/br.39.2.87-120.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fultz P. N., Choung K. K., Kemper J. Construction and characterization of Salmonella typhimurium strains that accumulate and excrete alpha- and beta-isopropylmalate. J Bacteriol. 1980 May;142(2):513–520. doi: 10.1128/jb.142.2.513-520.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fultz P. N., Kwoh D. Y., Kemper J. Salmonella typhimurium newD and Escherichia coli leuC genes code for a functional isopropylmalate isomerase in Salmonella typhimurium-Escherichia coli hybrids. J Bacteriol. 1979 Mar;137(3):1253–1262. doi: 10.1128/jb.137.3.1253-1262.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- GROSS S. R., BURNS R. O., UMBARGER H. E. THE BIOSYNTHESIS OF LEUCINE. II. THE ENZYMIC ISOMERIZATION OF BETA-CARBOXY-BETA-HYDROXYISOCAPROATE AND ALPHA-HYDROXY-BETA-CARBOXYISOCAPROATE. Biochemistry. 1963 Sep-Oct;2:1046–1052. doi: 10.1021/bi00905a023. [DOI] [PubMed] [Google Scholar]
- Kashmiri S. V., Gross S. R. Mutations affecting the regulation of production of the enzymes of leucine synthesis in Neurospora. Genetics. 1970 Mar-Apr;64(3):423–440. [PMC free article] [PubMed] [Google Scholar]
- Kemper J. Evolution of a new gene substituting for the leuD gene of Salmonella typhimurium: characterization of supQ mutations. J Bacteriol. 1974 Sep;119(3):937–951. doi: 10.1128/jb.119.3.937-951.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kemper J. Evolution of a new gene substituting for the leuD gene of Salmonella typhimurium: origin and nature of supQ and newD mutations. J Bacteriol. 1974 Dec;120(3):1176–1185. doi: 10.1128/jb.120.3.1176-1185.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kemper J., Margolin P. Suppression by gene substitution for the leuD gene of Salmonella typhimurium. Genetics. 1969 Oct;63(2):263–279. doi: 10.1093/genetics/63.2.263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kohlhaw G., Leary T. R., Umbarger H. E. Alpha-isopropylmalate synthase from Salmonella typhimurium. Purification and properties. J Biol Chem. 1969 Apr 25;244(8):2218–2225. [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- MARGOLIN P. Genetic fine structure of the leucine operon in Salmonella. Genetics. 1963 Mar;48:441–457. doi: 10.1093/genetics/48.3.441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miozzari G. F., Yanofsky C. Gene fusion during the evolution of the tryptophan operon in enterobacteriaceae. Nature. 1979 Feb 8;277(5696):486–489. doi: 10.1038/277486a0. [DOI] [PubMed] [Google Scholar]
- Pinel J. P., Chorover S. L. Inhibition by arousal of epilepsy induced by chlorambucil in rats. Nature. 1972 Mar 31;236(5344):232–234. doi: 10.1038/236232a0. [DOI] [PubMed] [Google Scholar]
- Polacco J. C., Gross S. R. The product of the leu-3 cistron as a regulatory element for the production of the leucine biosynthetic enzymes of Neurospora. Genetics. 1973 Jul;74(3):443–459. doi: 10.1093/genetics/74.3.443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reichenbecher V. E., Jr, Gross S. R. Structural features of normal and complemented forms of the Neurospora isopropylmalate isomerase. J Bacteriol. 1978 Feb;133(2):802–810. doi: 10.1128/jb.133.2.802-810.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Somers J. M., Amzallag A., Middleton R. B. Genetic fine structure of the leucine operon of Escherichia coli K-12. J Bacteriol. 1973 Mar;113(3):1268–1272. doi: 10.1128/jb.113.3.1268-1272.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang H. L., Kessler D. P. Genetic analysis of the leucine region in Escherichia coli B-r: gene-enzyme assignments. J Bacteriol. 1974 Jan;117(1):63–72. doi: 10.1128/jb.117.1.63-72.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]