Abstract
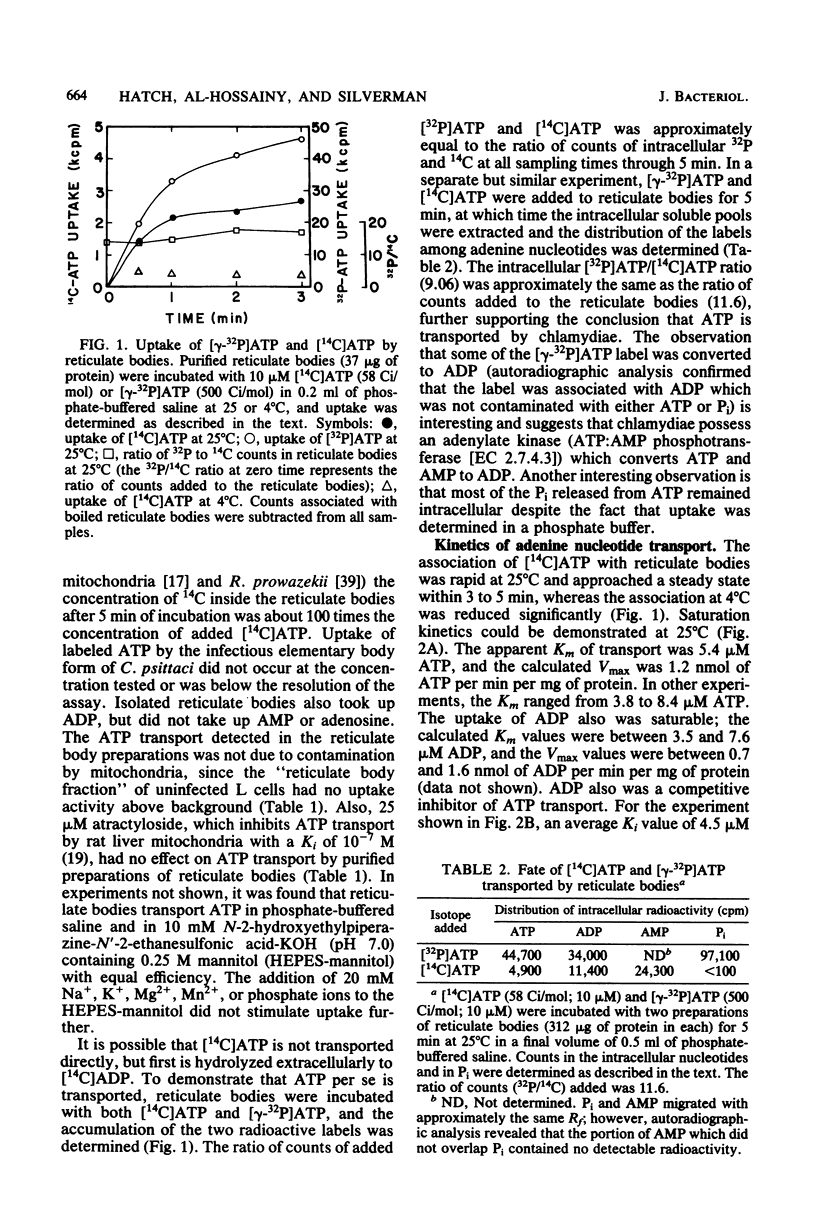
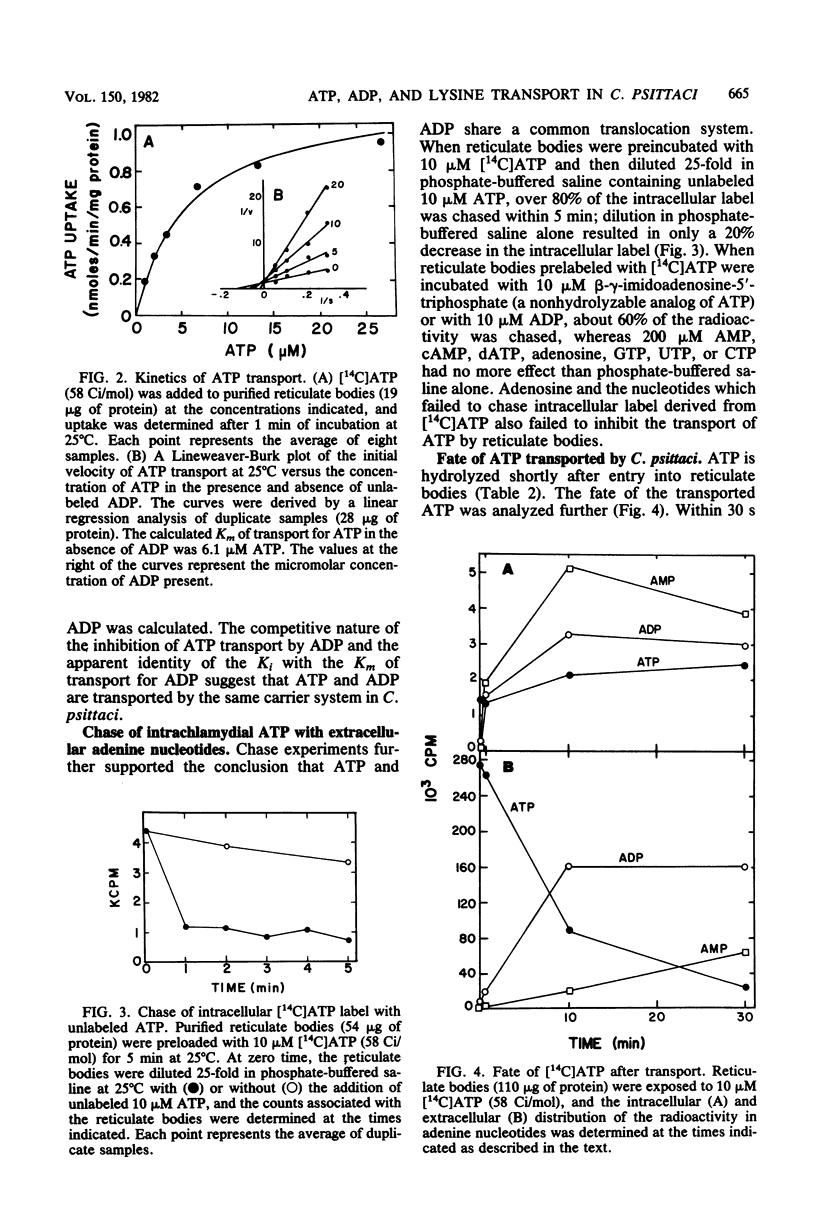
Isolated reticulate bodies of Chlamydia psittaci were found to transport ATP and ADP by an ATP-ADP exchange mechanism. ATP uptake activity was not detected in elementary bodies. The apparent Km of transport for both ATP and ADP was approximately 5 microM, and the calculated Vmax for both was about 1 nmol of nucleotide transported per min per mg of protein. ADP competitively inhibited ATP transport with a Ki of 4.5 microM. Other nucleotides tested had no effect on the uptake of ATP. A magnesium-dependent, oligomycin-sensitive ATPase (ATP phosphohydrolase, EC 3.6.1.3) was associated with reticulate bodies, and most of the transported ATP was hydrolyzed to ADP, which was exchanged for additional, extracellular nucleotide. Some ADP was hydrolyzed to AMP, which exited the cells slowly. Lysine was transported against the electrochemical gradient by reticulate bodies in the presence of ATP. Oligomycin and carbonyl cyanide p-trifluoromethoxyphenylhydrazone inhibited ATP-dependent lysine transport. Lysine exited reticulate bodies when the reticulate bodies were incubated in the presence of ADP, carbonyl cyanide p-trifluoromethoxyphenylhydrazone, or a reduced concentration of ATP. The results support the concept that chlamydiae are energy parasites which are capable of drawing upon the adenine nucleotides of their hosts, hydrolyzing ATP, and establishing an energized membrane.
Full text
PDF






Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- ALLEN E. G., BOVARNICK M. R. Association of reduced diphosphopyridine nucleotide cytochrome c reductase activity with meningopneumonitis virus. J Exp Med. 1957 Jun 1;105(6):539–547. doi: 10.1084/jem.105.6.539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Atkinson D. E. The energy charge of the adenylate pool as a regulatory parameter. Interaction with feedback modifiers. Biochemistry. 1968 Nov;7(11):4030–4034. doi: 10.1021/bi00851a033. [DOI] [PubMed] [Google Scholar]
- BOSE S. K., GEST H. PROPERTIES OF ADENOSINE TRIPHOSPHATASE IN A PHOTOSYNTHETIC BACTERIUM. Biochim Biophys Acta. 1965 Jan;96:159–162. doi: 10.1016/0005-2787(65)90621-0. [DOI] [PubMed] [Google Scholar]
- Ceballos M. M., Hatch T. P. Use of HeLa cell guanine nucleotides by Chlamydia psittaci. Infect Immun. 1979 Jul;25(1):98–102. doi: 10.1128/iai.25.1.98-102.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doddema H. J., Claesen C. A., Kell D. B., van der Drift C., Vogels G. D. An adenine nucleotide translocase in the procaryote Methanobacterium thermoautotrophicum. Biochem Biophys Res Commun. 1980 Aug 14;95(3):1288–1293. doi: 10.1016/0006-291x(80)91613-7. [DOI] [PubMed] [Google Scholar]
- Duee E. D., Vignais P. V. Echange entre adenine-nucleotides extra- et intramitochondriaux. Biochim Biophys Acta. 1965 Aug 24;107(1):184–188. doi: 10.1016/0304-4165(65)90419-8. [DOI] [PubMed] [Google Scholar]
- Gill S. D., Stewart R. B. Effect of metabolic inhibitors on the production of Chlamydia psittaci by infected L cells. Can J Microbiol. 1970 Nov;16(11):1079–1085. doi: 10.1139/m70-182. [DOI] [PubMed] [Google Scholar]
- Gill S. D., Stewart R. B. Glucose requirements of L cells infected with Chlamydia psittaci. Can J Microbiol. 1970 Oct;16(10):997–1001. doi: 10.1139/m70-169. [DOI] [PubMed] [Google Scholar]
- Harold F. M., Papineau D. Cation transport and electrogenesis by Streptococcus faecalis. II. Proton and sodium extrusion. J Membr Biol. 1972;8(1):45–62. doi: 10.1007/BF01868094. [DOI] [PubMed] [Google Scholar]
- Hatch T. P. Competition between Chlamydia psittaci and L cells for host isoleucine pools: a limiting factor in chlamydial multiplication. Infect Immun. 1975 Jul;12(1):211–220. doi: 10.1128/iai.12.1.211-220.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hatch T. P. Utilization of L-cell nucleoside triphosphates by Chlamydia psittaci for ribonucleic acid synthesis. J Bacteriol. 1975 May;122(2):393–400. doi: 10.1128/jb.122.2.393-400.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hatch T. P., Vance D. W., Jr, Al-Hossainy E. Identification of a major envelope protein in Chlamydia spp. J Bacteriol. 1981 Apr;146(1):426–429. doi: 10.1128/jb.146.1.426-429.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heefner D. L., Harold F. M. ATP-linked sodium transport in Streptococcus faecalis. I. The sodium circulation. J Biol Chem. 1980 Dec 10;255(23):11396–11402. [PubMed] [Google Scholar]
- Holian A., Wilson D. F. Relationship of transmembrane pH and electrical gradients with respiration and adenosine 5'-triphosphate synthesis in mitochondria. Biochemistry. 1980 Sep 2;19(18):4213–4221. doi: 10.1021/bi00559a012. [DOI] [PubMed] [Google Scholar]
- Johansson B. C., Baltscheffsky M., Baltscheffsky H. Purification and properties of coupling factor (Ca2+-dependent adenosine triphosphatase) from Rhodospirillum rubrum. Eur J Biochem. 1973 Dec 3;40(1):109–117. doi: 10.1111/j.1432-1033.1973.tb03174.x. [DOI] [PubMed] [Google Scholar]
- Klingenberg M. The ADP-ATP translocation in mitochondria, a membrane potential controlled transport. J Membr Biol. 1980 Sep 30;56(2):97–105. doi: 10.1007/BF01875961. [DOI] [PubMed] [Google Scholar]
- MOULDER J. W., GRISSO D. L., BRUBAKER R. R. ENZYMES OF GLUCOSE CATABOLISM IN A MEMBER OF THE PSITTACOSIS GROUP. J Bacteriol. 1965 Mar;89:810–812. doi: 10.1128/jb.89.3.810-812.1965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niven D. F., Hamilton W. A. Valinomycin-induced amino acid uptake by Staphylococcus aureus. FEBS Lett. 1973 Dec 1;37(2):244–248. doi: 10.1016/0014-5793(73)80470-3. [DOI] [PubMed] [Google Scholar]
- Pfaff E., Klingenberg M. Adenine nucleotide translocation of mitochondria. 1. Specificity and control. Eur J Biochem. 1968 Oct 17;6(1):66–79. doi: 10.1111/j.1432-1033.1968.tb00420.x. [DOI] [PubMed] [Google Scholar]
- Pfaff E., Klingenberg M., Heldt H. W. Unspecific permeation and specific exchange of adenine nucleotides in liver mitochondria. Biochim Biophys Acta. 1965 Jun 15;104(1):312–315. doi: 10.1016/0304-4165(65)90258-8. [DOI] [PubMed] [Google Scholar]
- RANDERATH E., RANDERATH K. RESOLUTION OF COMPLEX NUCLEOTIDE MIXTURES BY TWO-DIMENSIONAL ANION-EXCHANGE THIN-LAYER CHROMATOGRAPHY. J Chromatogr. 1964 Oct;16:126–129. doi: 10.1016/s0021-9673(01)82446-8. [DOI] [PubMed] [Google Scholar]
- Sarov I., Becker Y. Deoxyribonucleic acid-dependent ribonucleic acid polymerase activity in purified trachoma elementary bodies: effect of sodium chloride on ribonucleic acid transcription. J Bacteriol. 1971 Sep;107(3):593–598. doi: 10.1128/jb.107.3.593-598.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith D. K., Winkler H. H. Characterization of a lysine-specific active transport system in Rickettsia prowazeki. J Bacteriol. 1977 Mar;129(3):1349–1355. doi: 10.1128/jb.129.3.1349-1355.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Souverijn J. H., Huisman L. A., Rosing J., Kemp A., Jr Comparison of ADP and ATP as substrates for the adenine nucleotide translocator in rat-liver mitochondria. Biochim Biophys Acta. 1973 May 30;305(2):185–198. doi: 10.1016/0005-2728(73)90168-0. [DOI] [PubMed] [Google Scholar]
- Vender J., Moulder J. W. Initial step in catabolism of glucose by the meningopneumonitis agent. J Bacteriol. 1967 Oct;94(4):867–869. doi: 10.1128/jb.94.4.867-869.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vignais P. V. Molecular and physiological aspects of adenine nucleotide transport in mitochondria. Biochim Biophys Acta. 1976 Apr 30;456(1):1–38. doi: 10.1016/0304-4173(76)90007-0. [DOI] [PubMed] [Google Scholar]
- Weiss E., Wilson N. N. Role of exogenous adenosine triphosphate in catabolic and synthetic activities of Chlamydia psittaci. J Bacteriol. 1969 Feb;97(2):719–724. doi: 10.1128/jb.97.2.719-724.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams J. C. Adenine nucleotide degradation by the obligate intracellular bacterium Rickettsia typhi. Infect Immun. 1980 Apr;28(1):74–81. doi: 10.1128/iai.28.1.74-81.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Winkler H. H., Bygrave F. L., Lehninger A. L. Characterization of the atractyloside-sensitive adenine nucleotide transport system in rat liver mitochondria. J Biol Chem. 1968 Jan 10;243(1):20–28. [PubMed] [Google Scholar]
- Winkler H. H. Rickettsial cell water and membrane permeability determined by a micro space technique. Appl Environ Microbiol. 1976 Jan;31(1):146–149. doi: 10.1128/aem.31.1.146-149.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]



