Abstract
The soluble copper silicate (CS) MIC of 100 strains of methicillin-resistant Staphylococcus aureus and 100 strains of methicillin-susceptible S. aureus (MSSA) was 175 mg Cu/liter. Bactericidal and postantibiotic effects (≥1 h) were seen at 2× MIC and 4× MIC. The frequency of mutation was <10−9, and serial passage could not extend growth beyond 1.6× MIC.
Staphylococcus aureus is a commensal bacterium and also a major human pathogen. It may be carried transiently on the skin or for several months in the mucus membranes of the nasal passages. People who persistently carry S. aureus are at an increased risk of subsequent infection (12). Topical antibiotics, such as mupirocin or neomycin, have been used to eliminate S. aureus carriage in high-risk groups of hospitalized patients (6, 13, 14).
In nature copper silicate (CS; chrysolla) is an insoluble, amorphous blue-green mineral found within copper ore bodies. A novel, solubilized form of CS has been developed by Convé PLC as a topical antimicrobial that contains 0.28% (wt/wt) as copper, or 2,800 mg Cu/liter. The CS solution used in this study was manufactured by Patheon Inc. (Toronto, Canada) on 12 December 2005.
The 200 isolates of S. aureus and S. aureus ATCC 29213 used were obtained from the PathWest Laboratory Medicine WA (Nedlands, Western Australia, Australia) culture collection of bacteria isolated from normally sterile anatomical sites. All clinical isolates were identified by microbiology staff at PathWest Laboratory Medicine WA using standard techniques.
The MIC was assayed in Mueller-Hinton broth (MHB) using the Clinical and Laboratory Standards Institute (CLSI) guidelines for susceptibility testing of aerobic bacteria by broth microdilution (3). The copper concentration was taken as the known amount of copper (wt/wt) in the CS solution and did not necessarily reflect the free copper ions available in the MHB. The MIC for CS was remarkably consistent, as all S. aureus strains, including the control strain, S. aureus ATCC 29213, had an MIC of 175 mg Cu/liter. In comparison, the copper sulfate MIC for 20 strains (10 methicillin-resistant S. aureus [MRSA] strains and 10 methicillin-susceptible S. aureus [MSSA] strains) was 1 dilution higher, at 350 mg Cu/liter. The minimal bactericidal concentration (MBC) of CS ranged from 175 to 700 mg Cu/liter, with a 50% MBC (MBC50) and MBC90 of 350 mg Cu/liter for both MRSA and MSSA. There was no evidence of a more resistant subgroup within the total population tested.
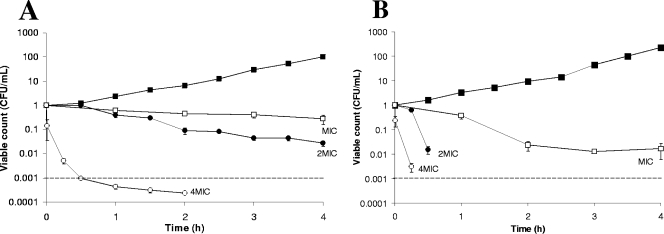
Time-kill kinetics showed that CS at the MIC was inhibitory. Bactericidal effects at 2× MIC were strain dependent, as only MRSA 101 was inhibited, while a total kill (99.9%) of S. aureus ATCC 29213 was achieved in 28 min (Fig. 1A and B). At 4× MIC, a total kill for both MRSA 101 and S. aureus ATCC 29213 occurred at 28 min and 10 min, respectively (Fig. 1A and B).
FIG. 1.
Time-kill curves of CS in MHB over a 4-h period with MRSA 101 (A) and S. aureus ATCC 29213 (B) and concentrations at MIC, 2× MIC, and 4× MIC. ▪, growth control; 2MIC, 2× MIC; 4MIC, 4× MIC; dashed lines, total kill. The experiments were conducted a minimum of three times and the results normalized. The standard error of each data point is shown.
The capacity of CS to cause a postantibiotic effect (PAE) in MRSA 101, MRSA 102, and S. aureus ATCC 29213 was investigated. MRSA 102 was included, as it had a MBC of 700 mg Cu/liter, 1 dilution higher than that of MRSA 101 and S. aureus ATCC 29213. After a 30-min exposure to CS at the MIC, growth of MRSA 101 and MRSA 102 was unaffected. However, S. aureus ATCC 29213 had an extended lag phase of 2 to 2.5 h, and the time taken for the 10-fold increase was significantly different (P < 0.05) from the that for the control (Table 1). When exposed to 2× MIC, all three strains took a significantly longer time than the control to increase 10-fold (Table 1). The PAE was considered to be significant only when the corresponding time for the 10-fold increase in the viable count was significantly different from that for the control (P < 0.05). Therefore, a significant PAE was seen at the MIC with S. aureus ATCC 29213 and with all three strains at twice the MIC (Table 1).
TABLE 1.
Time for a 10-fold increase and the calculated PAE for MRSA 101, MRSA 102, and S. aureus ATCC 29213 after 30-min exposure to CSa
| Isolate (exposure level) | Time (h) for 10-fold increase
|
PAE (h) | |
|---|---|---|---|
| Range | Avg ± SDb | ||
| MRSA 101 (control) | 2.18-2.78 | 2.39 ± 0.34 | |
| MRSA 101 (MIC) | 2.33-2.58 | 2.74 ± 0.12 | 0.35 |
| MRSA 101 (2× MIC) | 2.85-3.67 | 3.38 ± 0.46* | 0.99 |
| MRSA 102 (control) | 2.11-2.25 | 2.19 ± 0.07 | |
| MRSA 102 (MIC) | 1.11-2.98 | 1.95 ± 0.95 | −0.24 |
| MRSA 102 (2× MIC) | 3.32-6.31 | 4.72 ± 1.50# | 2.53 |
| ATCC 29213 (control) | 1.68-1.88 | 1.80 ± 0.10 | |
| ATCC 29213 (MIC) | 3.34-3.62 | 3.45 ± 0.15* | 1.65 |
| ATCC 29213 (2× MIC) | 3.41-4.47 | 3.90 ± 0.53* | 2.10 |
The experiments were conducted a minimum of three times.
*, P < 0.05 in two-tailed t test; #, P < 0.05 in one-tailed t test.
Spontaneous-resistance studies with mupirocin have shown that S. aureus produces a small population of resistant mutants, with MICs of 1 to 4 mg/liter, at a frequency of 1 to 4 per 109 bacterial cells (2). In similar studies with CS at 2× MIC and 4× MIC, no resistant mutants were detected (Table 2). In serial-passage experiments to induce tolerance or resistance, a twofold increase in the MIC was recorded for two of nine phenotypically distinct isolates capable of growth at 275 mg Cu/liter (1.6× MIC). Nevertheless, this did not represent a significant change in susceptibility as it was less than a fourfold difference. Thus, it appears that tolerance or resistance to CS cannot be readily induced in S. aureus.
TABLE 2.
Viable count and frequency of mutation of MRSA 101, MRSA 102, MSSA 1, and S. aureus ATCC 29213 with rifampin and CSa
| Isolate | Viable count (CFU/ml) | Avg FOMb ± SD with:
|
|
|---|---|---|---|
| Rifampin at 0.5 mg/liter | CS at 2× and 4× MIC | ||
| MRSA 101 | 1.90 × 109 | 5.26 × 10−8 ± 2.98 × 10−8 | <10−9 |
| MRSA 102 | 1.80 × 109 | 16.45 × 10−8 ± 8.79 × 10−8 | <10−9 |
| MSSA 1 | 2.35 × 109 | 16.60 × 10−8 ± 13.5 × 10−8 | <10−9 |
| ATCC 29213 | 2.05 × 109 | 11.27 × 10−8 ± 3.50 × 10−8 | <10−9 |
The experiments were conducted twice.
FOM, frequency of mutation.
The most intensive study of copper homeostasis and resistance in gram-positive bacteria has been with the enterococci (7, 8, 15). Nothing is known of copper homoeostasis or resistance in S. aureus, though genomic sequencing has identified a putative copA gene in both MRSA and MSSA (5, 10, 13). The copper sulfate MIC range for Enterococcus hirae and Enterococcus faecium has been recorded at 127 to 508 mg Cu/liter (1, 9), a very similar result to that recorded for S. aureus in this study with CS. In enterococci, acquired copper resistance has been linked to a transferable plasmid that harbors the copper resistance operon (tcrYAZB), which increased the copper MIC sevenfold to 1,525 mg Cu/liter and also transferred resistance to tetracycline, kanamycin, and streptomycin, either singly or in combination with resistance to macrolides and glycopeptides (7, 9). The plasmid also carried the transposable element Tn1546 (8). S. aureus has acquired vancomycin resistance from an interspecies transfer of the Tn1546 element from Enterococcus faecalis (16), but the possibility of the tcrYAZB operon being transferred in such a manner is unknown.
Mupirocin is the current agent of choice for MRSA eradication in colonized patients and health care workers (11). Widespread clinical use of mupirocin has resulted in mutants with low (8 to 256 mg Cu/liter) and high (≥512 mg Cu/liter) levels of resistance; the increasing prevalence of these mutants gives impetus to the need to find alternative topical antimicrobials for the control of MRSA carriage (4).
CS has in vitro antimicrobial activity against of S. aureus, including MRSA. The data in this study suggest that topical formulations of this antimicrobial may be useful in nasal decolonization therapy and treatment of dermatological infections. Convé PLC has produced a topical cream formulation (0.22% Cu, wt/wt) that has been well tolerated by over 350 people in phase 1 studies. However, clinical trials would need to be conducted to see if these promising in vitro results are a true indication of the potential of CS.
(These results were presented at the joint meeting of the 17th European Congress of Clinical and Infectious Diseases and the 25th International Congress of Chemotherapy, Munich, Germany, 31 March to 3 April 2007.)
Acknowledgments
This research was fully funded by Convé Ltd., a subsidiary of Convé PLC in which K. C. Carson is a shareholder.
Footnotes
Published ahead of print on 1 October 2007.
REFERENCES
- 1.Aarestrup, F. M., and H. Hasman. 2004. Susceptibility of different bacterial species isolated from food animals to copper sulphate, zinc chloride and antimicrobial substances used for disinfection. Vet. Microbiol. 100:83-89. [DOI] [PubMed] [Google Scholar]
- 2.Casewell, M., and R. Hill. 1985. In vitro activity of mupirocin ('pseudomonic acid') against clinical isolates of Staphylococcus aureus. J. Antimicrob. Chemother. 15:523-531. [DOI] [PubMed] [Google Scholar]
- 3.CLSI. 2006. Performance standards for antimicrobial testing; 16th informational supplement (M100-S16). Clinical and Laboratory Standards Institute, Wayne, PA.
- 4.Deshpande, L. M., A. M. Fix, M. A. Pfaller, R. N. Jones, and the SENTRY Antimicrobial Surveillance Program Participants Group. 2002. Emerging elevated mupirocin resistance rates among staphylococcal isolates in the SENTRY Antimicrobial Surveillance Program (2000): correlations of results from disk diffusion, Etest and reference dilution methods. Diag. Microbiol. Infect. Dis. 42:283-290. [DOI] [PubMed] [Google Scholar]
- 5.Diep, B. A., S. R. Gill, R. F. Chang, T. H. Phan, J. H. Chen, M. G. Davidson, F. Lin, J. Lin, H. A. Carleton, E. F. Mongodin, G. F. Sensabaugh, and F. Perdreau-Remington. 2006. Complete genome sequence of USA300, an epidemic clone of community-acquired methicillin-resistant Staphylococcus aureus. Lancet 367:731-739. [DOI] [PubMed] [Google Scholar]
- 6.Farr, B. M. 2002. Mupirocin to prevent S. aureus infections. N. Engl. J. Med. 346:1905-1906. [DOI] [PubMed] [Google Scholar]
- 7.Hasman, H. 2005. The tcrB gene is part of the tcrYAZB operon conferring copper resistance in Enterococcus faecium and Enterococcus faecalis. Microbiology 151:3019-3025. [DOI] [PubMed] [Google Scholar]
- 8.Hasman, H., and F. M. Aarestrup. 2005. Relationship between copper, glycopeptide, and macrolide resistance among Enterococcus faecium strains isolated from pigs in Denmark between 1997 and 2003. Antimicrob. Agents Chemother. 49:454-456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hasman, H., and F. M. Aarestrup. 2002. tcrB, a gene conferring transferable copper resistance in Enterococcus faecium: occurrence, transferability, and linkage to macrolide and glycopeptide resistance. Antimicrob. Agents Chemother. 46:1410-1416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Holden, M. T. G., E. J. Feil, J. A. Lindsay, S. J. Peacock, N. P. J. Day, M. C. Enright, T. J. Foster, C. E. Moore, L. Hurst, R. Atkin, A. Barron, N. Bason, S. D. Bentley, C. Chillingworth, T. Chillingworth, C. Churcher, L. Clark, C. Corton, A. Cronin, J. Doggett, L. Dowd, T. Feltwell, Z. Hance, B. Harris, H. Hauser, S. Holroyd, K. Jagels, K. D. James, N. Lennard, A. Line, R. Mayes, S. Moule, K. Mungall, D. Ormond, M. A. Quail, E. Rabbinowitsch, K. Rutherford, M. Sanders, S. Sharp, M. Simmonds, K. Stevens, S. Whitehead, B. G. Barrell, B. G. Spratt, and J. Parkhill. 2004. Complete genomes of two clinical Staphylococcus aureus strains: evidence for the rapid evolution of virulence and drug resistance. Proc. Natl. Acad. Sci. USA 101:9786-9791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hurdle, J. G., A. J. O'Neill, and I. Chopra. 2005. Prospects for aminoacyl-tRNA synthetase inhibitors as new antimicrobial agents. Antimicrob. Agents Chemther. 49:4821-4833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kluytmans, J., A. van Belkum, and H. Verbrugh. 1997. Nasal carriage of Staphylococcus aureus: epidemiology, underlying mechanisms, and associated risks. Clin. Microbiol. Rev. 10:505-520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kuroda, M., T. Ohta, I. Uchiyama, T. Baba, H. Yuzawa, I. Kobayashi, L. Cui, A. Oguchi, K.-I. Aoki, Y. Nagai, J. Lian, T. Ito, M. Kanamori, H. Matsumaru, A. Maruyama, H. Murakami, A. Hosoyama, Y. Mizutani-Ui, N. K. Takahashi, T. Sawano, R.-I. Inoue, C. Kaito, K. Sekimizu, H. Hirakawa, S. Kuhara, S. Goto, J. Yabuzaki, M. Kanehisa, A. Yamashita, K. Oshima, K. Furuya, C. Yoshino, T. Shiba, M. Hattori, N. Ogasawara, H. Hayashi, and K. Hiramatsu. 2001. Whole genome sequencing of methicillin-resistant Staphylococcus aureus. Lancet 357:1225-1240. [DOI] [PubMed] [Google Scholar]
- 14.Lowy, F. 1998. Staphylococcus aureus infections. N. Engl. J. Med. 339:520-532. [DOI] [PubMed] [Google Scholar]
- 15.Odermatt, A., H. Suter, R. Krapf, and M. Solioz. 1993. Primary structure of two P-type ATPases involved in copper homeostasis in Enterococcus hirae. J. Biol. Chem. 268:12775-12779. [PubMed] [Google Scholar]
- 16.Weigel, L. M., D. B. Clewell, S. R. Gill, N. C. Clark, L. K. McDougal, S. E. Flannagan, J. F. Kolonay, J. Shetty, G. E. Killgore, and F. C. Tenover. 2003. Genetic analysis of a high-level vancomycin-resistant isolate of Staphylococcus aureus. Science 301:1569-1571. [DOI] [PubMed] [Google Scholar]