Abstract
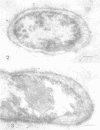
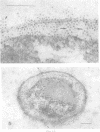
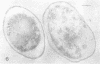
Lipopolysaccharide antigens were demonstrated on ultrathin sections of styrene-embedded Salmonella typhimurium by direct postembedding staining with ferritin-labeled antibodies. The antigenicity, partially masked in the embedding process, could be satisfactorily recovered by treatment of ultrathin sections with nonspecific protease. As judged from the reaction site of the ferritin-labeled antibodies, the lipopolysaccharides were localized in two zones. The broader zone of densely distributed ferritin molecules was superimposed over the whole outer cellwall, and a smaller zone revealing antigenicity was found over the cell membrane, which strongly supports the concept that the latter is the site of synthesis of lipopolysaccharides. The well-defined labeled areas between these two antigenic zones may be the routes whereby the synthesized polysaccharide molecules reach the cell wall.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Hama S., Katsumoto T., Takagi A. Investigations on the subcellular localization of O-antigens in Salmonella enteritidis by Means of Ferritin-conjugated antibody technique. Yonago Acta Med. 1973 Dec;17(3):207–215. [PubMed] [Google Scholar]
- Kulpa C. F., Jr, Leive L. Mode of insertion of lipopolysaccharide into the outer membrane of escherichia coli. J Bacteriol. 1976 Apr;126(1):467–477. doi: 10.1128/jb.126.1.467-477.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LUFT J. H. Improvements in epoxy resin embedding methods. J Biophys Biochem Cytol. 1961 Feb;9:409–414. doi: 10.1083/jcb.9.2.409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mühlradt P. F., Menzel J., Golecki J. R., Speth V. Outer membrane of salmonella. Sites of export of newly synthesised lipopolysaccharide on the bacterial surface. Eur J Biochem. 1973 Jun 15;35(3):471–481. doi: 10.1111/j.1432-1033.1973.tb02861.x. [DOI] [PubMed] [Google Scholar]
- Osborn M. J., Gander J. E., Parisi E. Mechanism of assembly of the outer membrane of Salmonella typhimurium. Site of synthesis of lipopolysaccharide. J Biol Chem. 1972 Jun 25;247(12):3973–3986. [PubMed] [Google Scholar]
- Shands J. W. Localization of Somatic Antigen on Gram-Negative Bacteria by Electron Microscopy. J Bacteriol. 1965 Jul;90(1):266–270. doi: 10.1128/jb.90.1.266-270.1965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shands J. W. Localization of somatic antigen on gram-negative bacteria using ferritin antibody conjugates. Ann N Y Acad Sci. 1966 Jun 30;133(2):292–298. doi: 10.1111/j.1749-6632.1966.tb52372.x. [DOI] [PubMed] [Google Scholar]
- Takamiya H., Bodemer W., Vogt A. Masking of protein antigen by modification of amino groups with carbobenzoxychloride (benzyl chloroformate) and demasking by treatment with nonspecific protease. J Histochem Cytochem. 1978 Nov;26(11):914–920. doi: 10.1177/26.11.82572. [DOI] [PubMed] [Google Scholar]