Abstract
Recent changes to the childhood immunization schedule in the United Kingdom have resulted in the inclusion of the 7-valent pneumococcal conjugate vaccine. However, the seroprevalence of pneumococcal antibodies in the population was unknown. To address this, we measured pneumococcal, age-specific immunoglobulin G (IgG) concentrations specific for nine serotypes by an assay run on the Bioplex platform, using 2,664 serum samples collected in England from 2000 to 2004. The lowest concentrations of IgG specific to all serotypes and the proportions of serotype-specific IgG concentrations of ≥0.35 μg/ml were observed in children aged <1 year. From 1 year on, there was a general increase in antibody levels with increasing age, and they remained high in adults. Maternal antibody was detected in young children aged <36 days but waned rapidly. Comparison of the age-specific seroprevalence of serotype-specific IgG to the serotype-specific incidence of invasive pneumococcal disease demonstrated a general inverse relationship for all age groups except the elderly. These data provide a baseline for natural immunity to the pneumococcal serotypes analyzed prior to the introduction of pneumococcal conjugate vaccine in the United Kingdom.
Streptococcus pneumoniae infection remains a significant public health issue globally. In the United Kingdom, the 7-valent pneumococcal conjugate vaccine Prevenar, which includes serotypes 4, 6B, 9V, 14, 18C, 19F, and 23F, was introduced into the childhood immunization schedule in September 2006 (4). Protection against S. pneumoniae infection is known to be mediated by the production of anti-capsular polysaccharide antibody, primarily of the immunoglobulin G (IgG) isotype (36, 38). The measurement of pneumococcal-capsular-serotype-specific IgG was established as a correlate of protection following conjugate vaccination, with a level of ≥0.35 μg/ml recommended as putatively protective by the World Health Organization (44), and hence was used together with a higher cutoff of 1.00 μg/ml as a laboratory marker to examine trends in natural immunity in this population. The level of ≥0.35 μg/ml was derived from an analysis of conjugate vaccine efficacy data from several clinical trials with young children (2, 20, 29). An important consideration when relating IgG levels to the protective correlate of ≥0.35 μg/ml is the assay used to obtain these data. This level is considered appropriate when using a third-generation enzyme-linked immunosorbent assay which incorporates adsorption with pneumococcal cell wall polysaccharide (CPS) (44). The age-specific baseline of pneumococcal-serotype-specific IgG in England is currently unknown.
Determination of the seroprevalence of naturally developing antibodies to a pathogen within a population has proved to be useful in providing data on the epidemiology of infection. Seroprevalence studies of poliomyelitis (11), rubella (5), and hepatitis A (9) provided a greater understanding of the epidemiology of each of these infections. In the 1980s and 1990s, studies of natural immunity to measles, mumps, rubella, and diphtheria informed future vaccination policy in the United Kingdom (24, 25). The determination of the level of antibodies to Haemophilus influenzae type b in children in England highlighted that a further catch-up campaign was required to boost immunity and reduce disease in those age groups at most risk (42). Seroprevalence studies can also provide indirect evidence for establishing correlates of protection, as demonstrated for meningococci (14, 41).
The aim of the study was to determine the age-specific seroprevalence of IgG specific for nine pneumococcal capsular polysaccharides (1, 2, 4, 6B, 9V, 14, 18C, 19F, and 23F) in a cross section of the population in England during 2000 to 2004 to establish a baseline prior to the introduction of Prevenar. Serotype-specific IgG concentrations were determined using a validated multiplex bead assay specific for nine pneumococcal capsular polysaccharides (23).
MATERIALS AND METHODS
Serum samples.
Serum samples (n = 2,664) obtained from the collection at the Health Protection Agency Seroepidemiology Unit (Preston, United Kingdom) from individuals aged 0 to 93 years between 2000 and 2004 were tested for pneumococcal-serotype-specific IgG concentrations. Residual serum samples from routine diagnostic testing are submitted to the Seroepidemiology Unit by up to 11 participating clinical laboratories in England as previously described (30).
Determination of serotype-specific IgG concentrations.
Serotype-specific IgG concentrations for serotypes 1, 4, 5, 6B, 9V, 14, 18C, 19F, and 23F were determined using a multiplex bead-based assay as described previously (23). This assay has been validated against the third-generation, internationally recognized enzyme-linked immunosorbent assay (43) and includes adsorption of sera with serotype 22F polysaccharide and CPS. All serotype-specific capsular polysaccharides were obtained from the ATCC or LGC Promochem; CPS was obtained from the Statens Serum Institut, Copenhagen, Denmark.
Disease incidence.
Age- and serotype-specific incidences of invasive pneumococcal disease (IPD) were estimated from cases serotyped by the Respiratory and Systemic Infection Laboratory (RSIL), Health Protection Agency, London, United Kingdom, between 2000 and 2004 and from population estimates from 2000 to 2004 from the Office for National Statistics, Newport, South Wales, United Kingdom. Since only 52% of all laboratory-confirmed cases of IPD (approximately 5,000 to 6,000 cases per year) reported to the Centre for Infections (London, United Kingdom) are serotyped and the proportion varies with age (up to 70% of IPD in patients aged <5 years are serotyped), the serotype-specific incidence in each group was estimated by multiplying the overall IPD incidence in each age group by the proportion of isolates serotyped in that age group that belonged to each serotype. There was no incidence of IPD due to serotype 5 recorded, and so this serotype was omitted from the analysis.
Statistical analysis.
Data were stratified by the following age groups for the calculation of geometric mean concentrations (GMC) of serotype-specific IgG and 95% confidence intervals (95% CI): 0 to 0.09 year (<36 days), 0.1 to 0.49 year, 0.5 to 0.99 year, years 1 to 19 in 1-year age bands, 20 to 24.9 years, 25 to 34.9 years, 35 to 44.9 years, 45 to 54.9 years, 55 to 64.9 years, 65 to 74.9 years, and ≥75 years. The sample sizes per age group are given in Table 1. The numbers of individuals with IgG concentrations of ≥0.35 μg/ml for each serotype were also calculated for the same age groups.
TABLE 1.
Numbers of samples analyzed per age group from the seroepidemiology collection for England for 2000 to 2004
| Age group (yr) | No. of samples analyzed |
|---|---|
| 0 to 0.09 | 16 |
| 0.1 to 0.4 | 31 |
| 0.5 to 0.9 | 38 |
| 1.0 to 1.9 | 168 |
| 2.0 to 2.9 | 186 |
| 3.0 to 3.9 | 185 |
| 4.0 to 4.9 | 179 |
| 5.0 to 5.9 | 202 |
| 6.0 to 6.9 | 157 |
| 7.0 to 7.9 | 119 |
| 8.0 to 8.9 | 98 |
| 9.0 to 9.9 | 77 |
| 10.0 to 10.9 | 75 |
| 11.0 to 11.9 | 54 |
| 12.0 to 12.9 | 68 |
| 13.0 to 13.9 | 53 |
| 14.0 to 14.9 | 55 |
| 15.0 to 15.9 | 52 |
| 16.0 to 16.9 | 58 |
| 17.0 to 17.9 | 67 |
| 18.0 to 18.9 | 80 |
| 19.0 to 19.9 | 63 |
| 20.0 to 24.9 | 88 |
| 25.0 to 34.9 | 62 |
| 35.0 to 44.9 | 112 |
| 45.0 to 54.9 | 81 |
| 55.0 to 64.9 | 96 |
| 65.0 to 74.9 | 81 |
| ≥75 | 63 |
| Total | 2,664 |
RESULTS
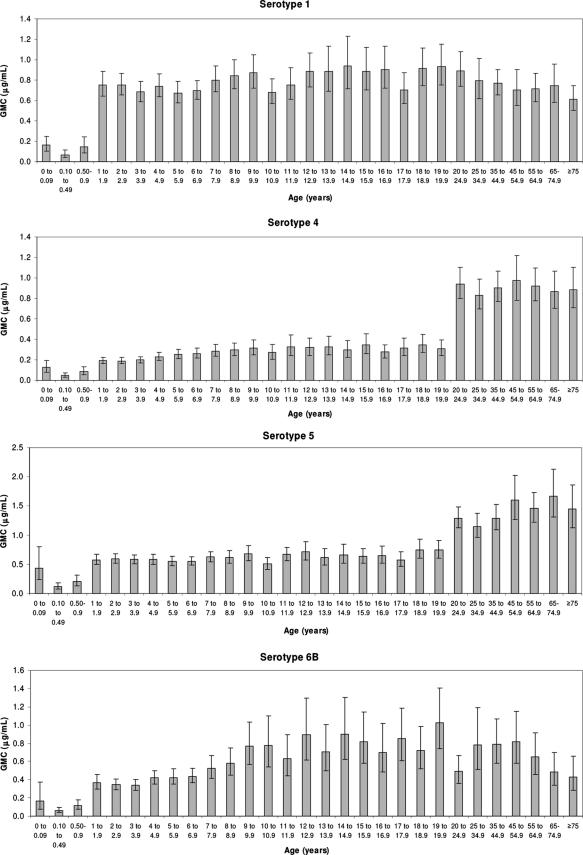
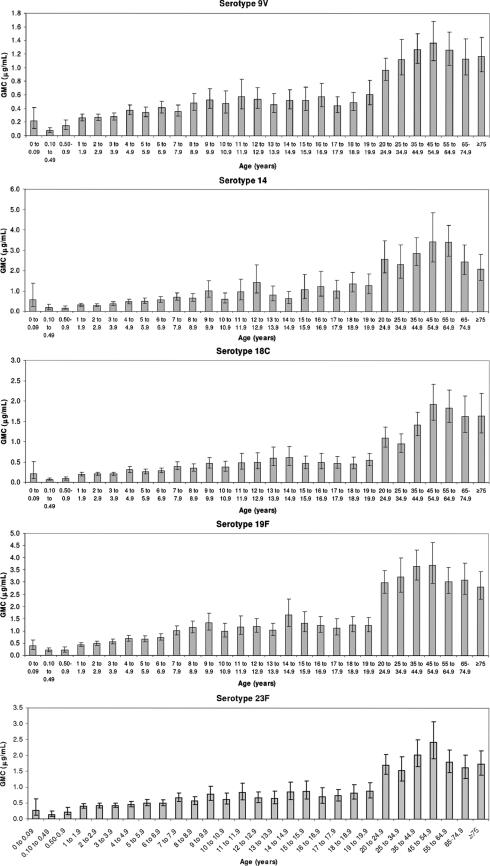
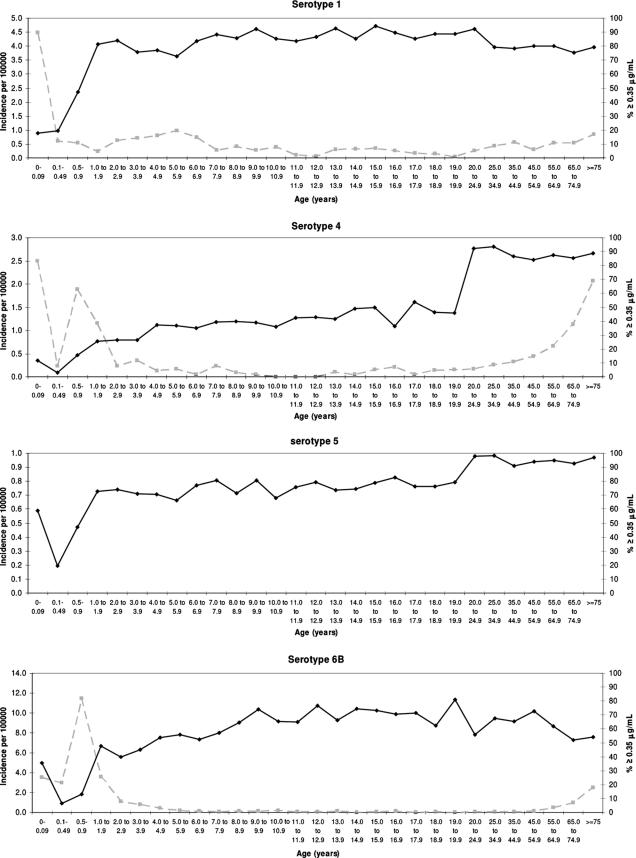
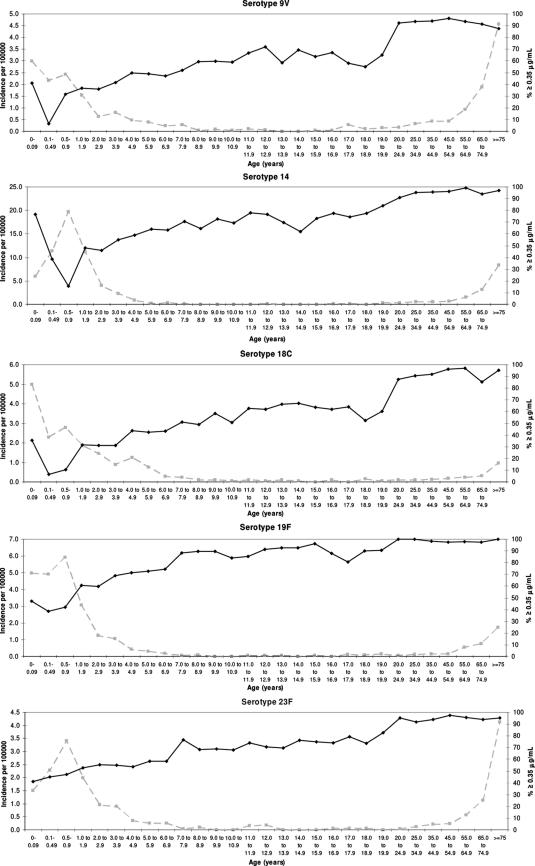
GMC of IgG and 95% CI are given in Fig. 1 for the age groups as detailed. Results were available for all serotypes for all samples except for one, which did not have a result for serotype 18C. The proportions of individuals within each age group with serotype-specific IgG concentrations of ≥0.35 μg/ml and the incidence of IPD due to individual serotypes are given in Fig. 2.
FIG. 1.
Age-specific pneumococcal serotype-specific IgG GMC (±95% CI) in England from 2000 to 2004. The y axis scales are different for each of the individual serotypes.
FIG. 2.
Age-specific incidence of laboratory-confirmed IPD in England from 2000 to 2004 (dashed gray line) and prevalence of serotype-specific IgG at concentrations of ≥0.35 μg/ml (solid black line). The incidence rates of IPD for individual serotypes are expressed on different scales. No incidence of IPD due to serotype 5 was confirmed from 2000 to 2005.
Analysis of the nine pneumococcal-serotype-specific profiles revealed distinct populations in terms of the IgG concentrations observed. For the majority of the serotypes (4, 5, 9V, 18C, 19F, and 23F), both the observed IgG GMC and the numbers of individuals with IgG concentrations of ≥0.35 μg/ml were low in children of <1 year of age. An increase was observed in older children to a level that remained relatively constant up to the age of 19 years. For those aged ≥20 years, further increases in the IgG concentrations were observed. Data were analyzed to determine the percentage of individuals with IgG concentrations of ≥1.00 μg/ml, and trends for all serotypes were similar to those reported for IgG concentrations of ≥0.35 μg/ml, but at a lower magnitude (data not shown). Serotypes 1, 6B, and 14 had different profiles. Both serotype 1- and 6B-specific concentrations were low in children aged <1 year and increased in children aged ≥1 year to a level that remained constant in older children and adults. Antibody concentrations for serotype 14 demonstrated a general age-dependent increase, with the greatest concentrations observed in adults, which is similar to what occurred with the other serotypes. Infants aged less than 0.1 year had significantly greater IgG GMC for all serotypes (P < 0.05), except 23F (P = 0.17), than children aged 0.1 to 0.49 year (Fig. 1).
The peak incidences of IPD occurred in infants aged 0 to 0.09 year (<36 days) for serotypes 1, 4, and 18C; infants aged 0.5 to 0.9 year for serotypes 6B, 14, and 19F; and adults aged ≥75 years for serotypes 9V and 23F (Fig. 2). The incidence rate of IPD observed is low in children aged ≥2 years and remains low in adults until an increase is observed in older adults, with rates in those aged ≥75 years similar to or greater than the peak in young children for serotypes 4, 9V, and 23F. Comparison of the incidence rate of IPD to the prevalence of serotype-specific IgG at concentrations of ≥0.35 μg/ml demonstrates a broadly inverse relationship. The peak incidence of IPD in children is associated with the lowest prevalence of serotype-specific IgG at concentrations of ≥0.35 μg/ml. As the incidence rate of IPD declines with increasing age, the prevalence of serotype-specific IgG at concentrations of ≥0.35 μg/ml increases and remains high in adults. No IPD due to serotype 5 was reported for the period 2000 to 2004 in England.
DISCUSSION
The data reported here detail the seroprevalence of IgG antibodies specific for capsular serotypes 1, 4, 5, 6B, 9V, 14, 18C, 19F, and 23F in a cross section of the population of England from 2000 to 2004. To our knowledge, this is the first report of the prevalence of serotype-specific pneumococcal antibodies using an assay which incorporates both CPS and 22F adsorption. The lowest concentrations of IgG, specific to all serotypes, were observed in children aged less than 1 year. The antibody profiles could then be broadly split into three groups: (i) similar antibody concentrations in patients from 1 to 19 years of age and an increase in adults ≥20 years of age (serotypes 4, 5, and 23F), (ii) increasing antibody concentrations with increasing age from 1 year to 19 years of age and a further increase in adults ≥20 years of age (serotypes 9V, 14, 18C, and 19F), and (iii) similar antibody concentrations from ≥1 year of age (serotypes 1 and 6B). Analysis of these data with respect to a threshold antibody level of ≥0.35 μg/ml shows profiles similar to those observed for the GMC.
The IPD incidence was determined for each age group, and differing profiles were demonstrated for individual serotypes. The number of cases of IPD in infants aged 0 to 0.09 year (<36 days) was small; thus, the incidence rates in this age group vary by serotype. This age group includes neonates acquiring IPD intrapartum. The incidence of IPD is next influenced by the acquisition of carriage in older infants. A general decline in IPD incidence is observed and remains low until there is an increase in older adults. Comparison of the proportion of individuals with serotype-specific IgG concentrations of ≥0.35 μg/ml with the age-specific IPD incidence for each serotype shows, in general, an inverse relationship. This relationship is similar to that reported for serogroup C meningococci (14, 31, 41). The seroprevalence studies performed for meningococci were able to propose and add indirect evidence for a surrogate of protection, namely, serum bactericidal antibody (14, 41). A level of ≥0.35 μg/ml of serotype-specific IgG has been proposed as a correlate of protection for pneumococci in studies of pneumococcal-conjugate-vaccine-induced immunity (37, 44). The relevance of this threshold to natural immunity has not been established. However, the prevalence of serotype-specific IgG at concentrations of ≥0.35 μg/ml did show an inverse relationship to serotype-specific IPD for the majority of the serotypes analyzed, suggesting that this may be an appropriate threshold to use in analyses in these types of studies. Of note for serotypes 4, 5, 9V, 18C, and 23F was the sharp increase in serotype-specific IgG antibody at around 20 years of age. One may hypothesize that this is due to the commencement of parenthood and, therefore, direct interaction with infants and young children, who most frequently carry the pneumococci (17).
Analysis of the number of serum samples with antibody concentrations of ≥0.5 μg/ml for serotype 6B and ≥5.0 μg/ml for serotype 14 suggested correlates for the prevention of otitis media and carriage, respectively (12, 18), and showed age distribution profiles similar to those observed for concentrations of ≥0.35 μg/ml, with an obvious difference in magnitude for the analysis of serotype 14. A maximum of approximately 40% of those aged 45 to 54.9 years had serotype 14-specific IgG concentrations of ≥5.0 μg/ml (data not shown).
In older adults, those aged ≥65 years, the proportions of individuals with antibody levels of ≥0.35 μg/ml was high for all serotypes except 6B, for which only approximately 50% of individuals exceeded this threshold. However, the incidence of IPD in these age groups showed an increase compared to that in younger adults, and for serotypes 4, 9V, and 23F, the incidence matched the peak observed in young children. There was no reduction in GMC observed in these age groups. The main protective mechanism against pneumococci is antibody-mediated opsonophagocytosis, and hence, although serotype-specific IgG levels remain high in older adults, these data do not directly reflect the functionality of the antibodies via this mechanism of protection. Lower functional antibody levels have been observed in older adults (>65 years) than in younger adults (18 to 45 years) in response to one dose of pneumococcal polysaccharide vaccine (8). The reduction in functional antibody activity in vaccinated elderly individuals has also been shown to highly correlate with decreased IgG antibody avidity (33). Adults residing in the United Kingdom aged ≥65 years are eligible for pneumococcal polysaccharide immunization, and over the period of 1993 to 2004, the uptake of vaccination was 29% (http://www.hpa.org.uk/infections/topics_az/pneumococcal/vaccine/uptake_monitoring.htm). This may account for the sustained antibody levels observed in the older adults, but it has been reported that immunization of this age group with the polysaccharide vaccine does not prevent community-acquired pneumonia or death due to pneumococcal infection (7).
Infants aged less than 0.1 year (<36 days) were observed to have greater serotype-specific IgG GMC for all serotypes, except 23F, than those aged 0.1 to 0.4 year, suggesting the presence of maternal antibody. The reported estimate for the half-life of antipneumococcal antibodies following transplacental transmission is 35 days (34). However, the concentration of antibodies observed in those aged less than 0.1 year is lower than that in the age groups which contain potential mothers. It is known that transplacental transfer of IgG1 is more efficient than that of IgG2 (1, 10); therefore, this finding is not surprising. The data generated for serotype-specific IgG concentrations in young infants are comparable to those of previous studies (2, 19, 26, 32, 35). However, they are lower than the antibody levels reported from pneumococcal vaccine studies in England (13) and studies of American Indian infants (28), which may be accounted for by differences in assay procedures or the carriage rates of the population.
The development of natural immunity to pneumococci is likely due to exposure to carriers and/or to exposure to non-IPDs such as otitis media or sinusitis. Other bacterial species may also induce cross-reacting antibodies to certain serotypes (6, 21). The mean prevalence of carriage of pneumococci in children aged 0 to 2 years in a mixed urban/rural setting in Hertfordshire was reported to be 52% (17). Carriage also occurred more than once in 86% of children aged 0 to 2 years, who were swabbed monthly over a 10-month period, compared to 36% of individuals aged ≥18 years. The most-prevalent serotypes carried were 6B, 19F, 6A, and 14. The rate of pneumococcal carriage in adults aged ≥65 years has been observed to be low, with 6 of 399 individuals found to be positive in a recent study (R. George and E. Miller, unpublished data). An increase in pneumococcal-serotype-specific IgG to serotypes 9V, 14, 18C, 19F, and 23F was observed in adults in that study over the 10 months associated with an episode of carriage with those serotypes (12). Interestingly, for serotype 6B, carriage was not associated with an increase in serotype-specific IgG, although this may be different in those individuals primed with pneumococcal conjugate vaccine. Carriage has been shown to induce antibody specific for serotypes 11A and 14 in children aged 6 months (40). A further factor to consider in the development of natural immunity is the differing durations of carriage observed for individual serotypes. Serotype 1 is rarely detected in carriage studies, whereas serotypes 6B, 19F, and 23F are three of the most commonly carried serotypes (3, 17, 39, 40). Hence, the differing rates of carriage, the subsequent development of age-specific immunity generated, and the invasive disease potential of serotypes all contribute to the observed serotype-specific incidence of IPD.
Recent reports have suggested that the epidemiology of invasive disease due to individual serotypes fluctuates greatly (15), and it is hypothesized that this is linked to the level of immunity within a population. A cyclical nature to the epidemiology of serotypes 1 and 5 has been suggested by data from Sweden (16, 27) and Chile (22). It is likely that the level of serotype-specific immunity within a population is related to these cycles of IPD caused by individual serotypes. The data reported in this study are from all age groups from 2000 to 2004. It would be of interest to focus on one age group, for example, young children, and measure the levels of immunity over an appropriate duration of time. Although this study demonstrated a sharp increase in serotype 1-specific IgG from the age of 0.4 year, associated with a decrease in incidence, this was not seen in a Finnish study performed between 1994 and 1997 with children aged 2 to 24 months (40), reflecting possible geographical and temporal differences.
This report demonstrates the age-specific incidence of IPD and seroprevalence of serotype-specific IgG concentrations in England and provides a baseline seroprevalence of serotype-specific IgG prior to the introduction of the 7-valent pneumococcal conjugate vaccine to the childhood immunization schedule in the United Kingdom in September 2006 (4). Although a general inverse relationship between serotype-specific IgG and the incidence of disease was demonstrated for infants, young children, and adults, this was not true for the elderly. Measurement of the functional activity of these antibodies in this age group would be of interest.
Acknowledgments
We acknowledge Andrew Vyse, Centre for Infections, Health Protection Agency, Colindale, London, United Kingdom, for the selection of serum samples and Louise Hesketh, Health Protection Agency Seroepidemiology Unit, Preston, United Kingdom, for maintaining the serum sample collection.
Footnotes
Published ahead of print on 19 September 2007.
REFERENCES
- 1.Baril, L., D. E. Briles, P. Crozier, J. D. King, S. K. Hollingshead, T. F. Murphy, and J. B. McCormick. 2004. Natural materno-fetal transfer of antibodies to PspA and to PsaA. Clin. Exp. Immunol. 135:474-477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Black, S., H. Shinefield, B. Fireman, E. Lewis, P. Ray, J. R. Hansen, L. Elvin, K. M. Ensor, J. Hackell, G. Siber, F. Malinoski, D. Madore, I. Chang, R. Kohberger, W. Watson, R. Austrian, K. Edwards, and Northern California Kaiser Permanente Vaccine Study Center Group. 2000. Efficacy, safety and immunogenicity of heptavalent pneumococcal conjugate vaccine in children. Pediatr. Infect. Dis. J. 19:187-195. [DOI] [PubMed] [Google Scholar]
- 3.Brueggemann, A. B., D. T. Griffiths, E. Meats, T. Peto, D. W. Crook, and B. G. Spratt. 2003. Clonal relationships between invasive and carriage Streptococcus pneumoniae and serotype- and clone-specific differences in invasive disease potential. J. Infect. Dis. 187:1424-1432. [DOI] [PubMed] [Google Scholar]
- 4.Chief Medical Officer, Chief Nursing Officer, and Chief Pharmaceutical Officer, Department of Health, London, United Kingdom. 2006. Important changes to the childhood immunisation programme. Professional letters PL/CMO/2006/1, PL/CNO/2006/1, and PL/CPHO/2006/1. Department of Health, London, United Kingdom.
- 5.Cockburn, W. C. 1969. World aspects of the epidemiology of rubella. Am. J. Dis. Child. 118:112-122. [DOI] [PubMed] [Google Scholar]
- 6.Crumrine, M. H., G. W. Fischer, and M. W. Balk. 1979. Immunochemical cross-reactions between type III group B streptococcus and type 14 Streptococcus pneumoniae. Infect. Immun. 25:960-963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Dear, K., J. Holden, R. Andrews, and D. Tatham. 2003. Vaccines for preventing pneumococcal infection in adults. Cochrane Database Syst. Rev. 4:CD000422. [DOI] [PubMed] [Google Scholar]
- 8.Devaster, J. M., I. Leroux-Roels, G. Leroux-Roels, P. Vandepapeliere, Y. Horsmans, I. Henckaerts, and J. Poolman. 2006. Inferior humoral response in elderly versus young adults to the 23-valent polysaccharide vaccine, p. 244. Abstr. 5th Int. Symp. Pneumococci Pneumococcal Dis., Alice Springs, Australia, 2 to 6 April 2006.
- 9.Dienstag, J. L., W. Szmuness, C. E. Stevens, and R. H. Purcell. 1978. Hepatitis A virus infection: new insights from seroepidemiologic studies. J. Infect. Dis. 137:328-340. [DOI] [PubMed] [Google Scholar]
- 10.Englund, J. A., W. P. Glezen, C. Turner, J. Harvey, C. Thompson, and G. R. Siber. 1995. Transplacental antibody transfer following maternal immunization with polysaccharide and conjugate Haemophilus influenzae type b vaccines. J. Infect. Dis. 171:99-105. [DOI] [PubMed] [Google Scholar]
- 11.Fallon, R. J. 1956. Serological epidemiology of poliomyelitis; distribution of immunity to poliomyelitis virus. Lancet 270:65-69. [DOI] [PubMed] [Google Scholar]
- 12.Goldblatt, D., M. Hussain, N. Andrews, L. Ashton, C. Virta, A. Melegaro, R. Pebody, R. George, A. Soininen, J. Edmunds, N. Gay, H. Kayhty, and E. Miller. 2005. Antibody responses to nasopharyngeal carriage of Streptococcus pneumoniae in adults: a longitudinal household study. J. Infect. Dis. 192:387-393. [DOI] [PubMed] [Google Scholar]
- 13.Goldblatt, D., J. Southern, L. Ashton, P. Richmond, P. Burbidge, J. Tasevska, A. Crowley-Luke, N. Andrews, R. Morris, R. Borrow, K. Cartwright, and E. Miller. 2006. Immunogenicity and boosting after a reduced number of doses of a pneumococcal conjugate vaccine in infants and toddlers. Pediatr. Infect. Dis. J. 25:312-319. [DOI] [PubMed] [Google Scholar]
- 14.Goldschneider, I., E. C. Gotschlich, and M. S. Artenstein. 1969. Human immunity to the meningococcus: II. Development of natural immunity. J. Exp. Med. 129:1327-1348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hausdorff, W. P. 2007. The roles of pneumococcal serotypes 1 and 5 in paediatric disease. Vaccine 25:2406-2412. [DOI] [PubMed] [Google Scholar]
- 16.Hedlund, J., M. Sorberg, B. Henriques Normark, and G. Kronvall. 2003. Capsular types and antibiotic susceptibility of invasive Streptococcus pneumoniae among children in Sweden. Scand. J. Infect. Dis. 35:452-458. [DOI] [PubMed] [Google Scholar]
- 17.Hussain, M., A. Melegaro, R. G. Pebody, R. George, W. J. Edmunds, R. Talukdar, S. A. Martin, A. Efstratiou, and E. Miller. 2005. A longitudinal household study of Streptococcus pneumoniae nasopharyngeal carriage in a UK setting. Epidemiol. Infect. 133:891-898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Jokinen, J. T., H. Ahman, T. M. Kilpi, P. H. Makela, and M. H. Kayhty. 2004. Concentration of antipneumococcal antibodies as a serological correlate of protection: an application to acute otitis media. J. Infect. Dis. 190:545-550. [DOI] [PubMed] [Google Scholar]
- 19.Kayhty, H., H. Ahman, K. Eriksson, M. Sorberg, and L. Nilsson. 2005. Immunogenicity and tolerability of a heptavalent pneumococcal conjugate vaccine administered at 3, 5 and 12 months of age. Pediatr. Infect. Dis. J. 24:108-114. [DOI] [PubMed] [Google Scholar]
- 20.Klugman, K. P., S. A. Madhi, R. E. Huebner, R. Kohberger, N. Mbelle, N. Pierce, and Vaccine Trialists Group. 2003. A trial of a 9-valent pneumococcal conjugate vaccine in children with and those without HIV infection. N. Engl. J. Med. 349:1341-1348. [DOI] [PubMed] [Google Scholar]
- 21.Lagergård, T., and P. Branefors. 1983. Nature of cross-reactivity between Haemophilus influenzae types a and b and Streptococcus pneumoniae types 6A and 6B. Acta Pathol. Microbiol. Immunol. Scand. Sect. C 91:371-376. [PubMed] [Google Scholar]
- 22.Lagos, R., A. Muñoz, O. San Martín, I. Heitmann, H. Loyola, and M. M. Levine. 2006. Secular variations in age incidence (Inc) and serotypes (St) causing invasive pneumococcal disease (IPD) in children 0 to 59 months of age (MoA) in the metropolitan region (MR), Chile, p. 164. Abstr. 5th Int. Symp. Pneumococci Pneumococcal Dis., Alice Springs, Australia, 2 to 6 April 2006.
- 23.Lal, G., P. Balmer, E. Stanford, S. Martin, R. Warrington, and R. Borrow. 2005. Development and validation of a nonaplex assay for the simultaneous quantitation of antibodies to nine Streptococcus pneumoniae serotypes. J. Immunol. Methods 296:135-147. [DOI] [PubMed] [Google Scholar]
- 24.Miller, E., M. Rush, P. Morgan-Capner, D. Hutchinson, and L. Hindle. 1994. Immunity to diphtheria in adults in England. BMJ 308:598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Morgan-Capner, P., J. Wright, C. L. Miller, and E. Miller. 1988. Surveillance of antibody to measles, mumps and rubella by age. BMJ 297:770-772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Moss, S. J., A. C. Fenton, R. Borrow, P. Balmer, J. Smith, and A. R. Gennery. 2006. Immunogenicity of a conjugate meningococcal group C (MCC) administered to term infants according to the current UK immunisation schedule at the same time as a seven valent conjugate pneumococcal vaccine (7VCPnc), p. 210. Abstr. 24th Meet. Eur. Soc. Paediatr. Infect. Dis., Basel, Switzerland, 3 to 5 May 2006.
- 27.Normark, B. H., A. Ortqvist, M. Kalin, B. Olsson-Liljequist, J. Hedlund, S. B. Svenson, and G. Källenius. 2001. Changes in serotype distribution may hamper efficacy of pneumococcal conjugate vaccines in children. Scand. J. Infect. Dis. 33:848-850. [DOI] [PubMed] [Google Scholar]
- 28.O'Brien, K. L., J. Moisi, L. H. Moulton, D. Madore, A. Elick, R. Reid, R. Weatherholtz, E. Millar, D. Hu, J. Hackell, R. Kohberger, G. Siber, and M. Santosham. 2007. Predictors of pneumococcal conjugate vaccine immunogenicity among infants and toddlers in an American Indian PnCRM7 efficacy trial. J. Infect. Dis. 196:104-114. [DOI] [PubMed] [Google Scholar]
- 29.O'Brien, K. L., L. H. Moulton, R. Reid, R. Weatherholtz, J. Oski, L. Brown, G. Kumar, A. Parkinson, D. Hu, J. Hackell, I. Chang, R. Kohberger, G. Siber, and M. Santosham. 2003. Efficacy and safety of seven-valent conjugate pneumococcal vaccine in American Indian children: group randomised trial. Lancet 362:355-361. [DOI] [PubMed] [Google Scholar]
- 30.Osborne, K., N. J. Gay, L. Hesketh, P. Morgan-Capner, and E. Miller. 2000. Ten years of serological surveillance in England and Wales: methods, results, implications and action. Int. J. Epidemiol. 29:362-368. [DOI] [PubMed] [Google Scholar]
- 31.Pollard, A. J., J. Ochnio, M. Ho, M. Callaghan, M. Bigham, and S. Dobsong. 2004. Disease susceptibility to ST11 complex meningococci bearing serogroup C or W135 polysaccharide capsules, North America. Emerg. Infect. Dis. 10:1812-1815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Rennels, M. B., K. M. Edwards, H. L. Keyserling, K. S. Reisinger, D. A. Hogerman, D. V. Madore, I. Chang, P. R. Paradiso, F. J. Malinoski, and A. Kimura. 1998. Safety and immunogenicity of heptavalent pneumococcal vaccine conjugated to CRM197 in United States infants. Pediatrics 101:604-611. [DOI] [PubMed] [Google Scholar]
- 33.Romero-Steiner, S., D. M. Musher, M. S. Cetron, L. B. Pais, J. E. Groover, A. E. Fiore, B. D. Plikaytis, and G. M. Carlone. 1999. Reduction in functional antibody activity against Streptococcus pneumoniae in vaccinated elderly individuals highly correlates with decreased IgG antibody avidity. Clin. Infect. Dis. 29:281-288. [DOI] [PubMed] [Google Scholar]
- 34.Shahid, N. S., M. C. Steinhoff, S. S. Hoque, T. Begum, C. Thompson, and G. R. Siber. 1995. Serum, breast milk and infant antibody after maternal immunisation with pneumococcal vaccine. Lancet 346:1252-1257. [DOI] [PubMed] [Google Scholar]
- 35.Shinefield, H. R., S. Black, P. Ray, I. Chang, N. Lewis, B. Fireman, J. Hackell, P. R. Paradiso, G. Siber, R. Kohberger, D. V. Madore, F. J. Malinowski, A. Kimura, C. Le, I. Landaw, J. Aquilar, and J. Hansen. 1999. Safety and immunogenicity of heptavalent pneumococcal CRM197 conjugate vaccine in infants and toddlers. Pediatr. Infect. Dis. J. 18:757-763. [DOI] [PubMed] [Google Scholar]
- 36.Shurin, P. A., J. M. Rehmus, C. E. Johnson, C. D. Marchant, S. A. Carlin, D. M. Super, G. F. Van Hare, P. K. Jones, D. M. Ambrosino, and G. Siber. 1993. Bacterial polysaccharide immune globulin for prophylaxis of acute otitis media in high-risk children. J. Pediatr. 123:801-810. [DOI] [PubMed] [Google Scholar]
- 37.Siber, G. R., I. Chang, S. Baker, P. Fernsten, K. L. O'Brien, M. Santosham, K. P. Klugman, S. A. Madhi, P. Paradiso, and R. Kohberger. 2007. Estimating the protective concentration of anti-pneumococcal capsular polysaccharide antibodies. Vaccine 25:3816-3826. [DOI] [PubMed] [Google Scholar]
- 38.Siber, G. R., C. Thompson, G. R. Reid, J. Almeido-Hill, B. Zacher, M. Wolff, and M. Santosham. 1992. Evaluation of bacterial polysaccharide immune globulin for the treatment or prevention of Haemophilus influenzae type b and pneumococcal disease. J. Infect. Dis. 165(Suppl. 1):S129-S133. [DOI] [PubMed] [Google Scholar]
- 39.Smith, T., D. Lehmann, J. Montgomery, M. Gratten, I. D. Riley, and M. P. Alpers. 1993. Acquisition and invasiveness of different serotypes of Streptococcus pneumoniae in young children. Epidemiol. Infect. 111:27-39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Soininen, A., K. Pursiainen, T. Kilpi, and H. Kayhty. 2001. Natural development of antibodies to pneumococcal capsular polysaccharides depends on the serotype: association with pneumococcal carriage and acute otitis media in young children. J. Infect. Dis. 184:569-576. [DOI] [PubMed] [Google Scholar]
- 41.Trotter, C., R. Borrow, N. Andrews, and E. Miller. 2003. Seroprevalence of meningococcal serogroup C bactericidal antibody in England and Wales in the pre-vaccination era. Vaccine 21:1094-1098. [DOI] [PubMed] [Google Scholar]
- 42.Trotter, C. L., J. McVernon, N. J. Andrews, M. Burrage, and M. E. Ramsay. 2003. Antibody to Haemophilus influenzae type b after routine and catch-up vaccination. Lancet 361:1523-1524. [DOI] [PubMed] [Google Scholar]
- 43.WHO Pneumococcal Serology Reference Laboratories and Department of Pathology, University of Alabama at Birmingham. 4 February 2007, accession date. Training manual for enzyme linked immunosorbent assay for the quantitation of Streptococcus pneumoniae serotype specific IgG (Pn PS ELISA). WHO Pneumococcal Serology Reference Laboratories at the Institute of Child Health, University College London, London, England, and Department of Pathology, University of Alabama at Birmingham, Birmingham. http://www.vaccine.uab.edu/ELISA%20Protocol.pdf.
- 44.World Health Organization. 2005. Recommendations for the production and control of pneumococcal conjugate vaccines. WHO Tech. Rep. Ser. 927(Annex 2):64-98. [Google Scholar]