Abstract
Leprosy is caused by infection with Mycobacterium leprae. The immune response of leprosy patients can be highly diverse, ranging from strong cellular responses accompanied by an apparent deficit of M. leprae-specific antibodies to strong humoral responses with a deficit of cell-mediated responses. Leprosy takes many years to manifest, and this has precluded analyses of disease and immune response development in infected humans. In an attempt to better define development of the immune response during leprosy we have developed an M. leprae ear infection model. Intradermal inoculation of M. leprae into the ear supported not only infection but also the development of a chronic inflammatory response. The inflammatory response was localized, comprising a T-cell infiltration into the ear and congestion of cells in the draining lymph nodes. The development of local chronic inflammation was prevented by rifampin treatment. Importantly, and in contrast to subcutaneous M. leprae footpad infection, systemic M. leprae-specific gamma interferon and antibody responses were detected following intradermal ear infection. These results indicate the utility of intradermal ear infection for both induction and understanding of the immune response during M. leprae infection and the identification or testing of new leprosy treatments.
Mycobacterium leprae is the causative agent of leprosy. Over the last 20 years the World Health Organization has implemented an intense and coordinated worldwide multidrug therapy (MDT) program to treat leprosy. The MDT program has reduced the prevalence of M. leprae infection to <1 case per 10,000 in 90% of the endemic countries, where leprosy was considered to be a public health problem (39). Despite this program new-case detection rates over the last few years have remained stable at approximately 500,000 per year, and in some countries rates are actually increasing (11, 17, 18, 22). As a further complication, drug resistance may be emerging (10, 20, 24), and relapse rates in some areas are unacceptably high (16). These factors indicate that a greater understanding of M. leprae infection is required, with a goal of providing additional treatments that could eliminate leprosy.
Cases in which leprosy manifests are represented by a clinical spectrum of disease. Clinical, histopathological, and immunological criteria identify five forms of leprosy: tuberculoid (TT), borderline tuberculoid (BT), mid-borderline (BB), borderline lepromatous (BL), and lepromatous (LL) leprosy (28, 29). Paucibacillary (PB) patients, generally encompassing those with TT and BT forms of leprosy, demonstrate low or absent bacterial indices and specific cell-mediated immunity against M. leprae. In marked contrast, multibacillary patients (BB, BL, and LL leprosy) demonstrate high bacterial indices and high titers of anti-M. leprae antibodies and cell-mediated immunity against M. leprae is either modest or absent. Most leprosy patients develop immune responses somewhere between these extremes. Why M. leprae infection results in such polarized immune responses remains unclear.
Current animal models are limited and do not rapidly develop pathology following M. leprae infection, hampering the ability to study disease and immune response development. Armadillos can become naturally infected with M. leprae, and leprosy develops across a spectrum similar to that seen in humans (31, 36, 38). Armadillos are difficult to maintain, and the armadillo immune system is poorly defined, however, thereby limiting the use of armadillos as a leprosy model. The mouse footpad model, developed by Shepard in the 1960s, has previously been used for experimental investigation of M. leprae infection (32-34). While this model clearly demonstrates M. leprae growth, it requires over 6 months to yield results (2, 3). The ability of immune-competent mice to limit M. leprae growth in footpads, unlike the uncontrolled growth that occurs in immune-compromised mice, indicates that some protective immunity is induced in response to infection (1, 4, 12, 13, 21, 27). Following footpad infection there is, however, virtually no disease in the infected footpads of immune-competent mice and measurable systemic immune responses are generally not observed.
In an attempt to investigate the development of M. leprae infection, we tested the hypothesis that intradermal (i.d.) infection of the mouse ear would support infection and promote anti-M. leprae immune responses. Ears were chosen as the inoculation site because they are consistently cooler than the rest of the body and M. leprae bacilli grow only at cool temperatures. In addition, recent experiments comparing mouse ear and footpad infection models of cutaneous leishmaniasis have indicated differences in disease development and suggest that experimental dermal infection may better mimic typical human infection (6-8). Our data indicate that M. leprae bacilli not only grow within the ears but also stimulate a rapid and prolonged local inflammatory response. The inflammatory response presents as a T-cell infiltrate within the ear and a local lymphadenopathy, both of which are limited by treatment with the antimycobacterial drug rifampin. In addition, and in contrast with mice infected in the footpad, mice infected in the ear demonstrate M. leprae-specific cellular responses. Our data indicate that M. leprae infection of the mouse ear provides a system with which to evaluate antileprosy treatments and analyze the development of M. leprae-induced inflammatory responses.
MATERIALS AND METHODS
Mice.
Female C57BL/6 (B6) mice were purchased from Charles River Laboratories (Wilmington, MA) and maintained in specific-pathogen-free conditions in the animal facilities of the Infectious Disease Research Institute, Seattle, WA. All animal procedures were approved by the institutional animal care and use committee. Mice were inoculated at 6 to 10 weeks of age.
M. leprae inoculations and rifampin treatment.
Live M. leprae bacilli (Thai-53 strain) were purified from the footpads of nu/nu mice at National Hansen's Disease Programs and shipped overnight on ice to the Infectious Disease Research Institute for inoculations (37). Heat-killed M. leprae bacilli were obtained by heating bacilli at 70°C for 1 h and then quenching on ice. Mice were inoculated with bacilli in a volume of 10 μl by i.d. injection into the ear pinnae or subcutaneous (s.c.) injection into the footpad. To assess growth, both ears were harvested and the bacilli were enumerated by direct microscopic counting of acid-fast bacilli according to the method of Shepard and McRae (35). In treatment experiments, mice were injected intraperitoneally with 0.5 mg rifampin (Sigma) or phosphate-buffered saline (PBS) at 1, 2, and 3 weeks after infection.
Histology.
Ears were fixed in formalin and sectioned. Slides were then stained with hematoxylin and eosin.
Cell preparations.
Single-cell suspensions were prepared from the spleen and lymph nodes (LN; auricular, axillary, inguinal, and popliteal). Spleens and LN were disrupted between frosted slides and erythrocytes removed by lysis in 1.66% NH4Cl solution. Single-cell suspensions were also prepared from ears. Ears were collected, rinsed with 70% ethanol, and allowed to air dry. Ears were then split into dorsal and ventral halves and floated on 1 ml RPMI 1640 (BioWhittaker, Walkersville, MD) supplemented with Liberase CI (Sigma, St. Louis, MO) for 1.5 h at 37°C. Enzymatic digestion was stopped by the addition of 1 ml RPMI 1640 supplemented with 0.05% DNase (Sigma). Digested ears were homogenized in 50 μm Medicon filters using a MediMachine (BD Biosciences, San Jose, CA), and the resultant homogenate was filtered through a 70-μm Filcon filter (BD Biosciences). Ear, LN, and spleen cells were washed at least twice before enumerating mononuclear cells using a hemocytometer.
Flow cytometry.
To stain for flow cytometry, cells were incubated with the anti-FcγII/IIIR antibody 2.4G2 and then with combinations of fluorescently conjugated anti-CD4 (clone GK1.5), anti-CD8a (clone 53-6.7), anti-CD11c (clone HL3), anti-CD44 (clone IM7), anti-F4/80 (clone BM8), anti-gamma interferon (IFN-γ; clone XMG1.2), anti-interleukin-10 (IL-10; clone JES5-16E3), anti-Ly6G/C (clone RB6-8C5), anti-NK1.1 (clone PK136), anti-pan-T-cell receptor-β (TCRβ)-chain (clone H57-597), and anti-TCRγδ-chain (clone GL3) (all BD Biosciences). For the elucidation of intracellular cytokine expression, cells were cultured at 37°C for 4 to 6 h in the presence of 0.5 μg/ml phorbol myristate acetate (PMA)-ionomycin (Sigma) and Golgi Plug (BD Biosciences). Cells were fixed and permeabilized in Cytofix/Cytoperm (BD Biosciences) before staining. Flow cytometry was performed using FACScalibur (BD Biosciences), and the data were analyzed with WinMDI 2.8.
Antigen stimulation assays.
Spleen mononuclear cells (2 × 105) were cultured in duplicate in a 96-well plate (Corning Incorporated, Corning, NY) in T-cell media (RPMI 1640 supplemented with 5% heat-inactivated fetal bovine serum and 50,000 U penicillin-streptomycin [all BioWhittaker]), in the presence of 10 μg/ml M. leprae cell wall antigen (kindly provided by John Spencer, Colorado State University) or recombinant ML2028 protein. ML2028 is the M. leprae homolog of Mycobacterium tuberculosis Rv1886c (83.3% identity), which induces the highly immunogenic and protective Ag85B (26). Culture supernatants were harvested after 72 h, and IFN-γ was assayed by enzyme-linked immunosorbent assay (ELISA) (clones R4-6A2 and XMG1.2; BD Biosciences), according to the manufacturer's instructions.
Antibody analyses.
Individual mouse sera were analyzed by ELISA. Briefly, ELISA plates (Nunc, Rochester, NY) were coated with 2 μg/ml antigen in 0.1 M bicarbonate buffer and blocked with 0.1% bovine serum albumin-PBS. Then, in consecutive order and following washes in PBS-Tween, serum samples (diluted 1/100), anti-mouse immunoglobulin G (IgG)-horseradish peroxidase (Southern Biotech, Birmingham, AL), and ABTS [2,2′-azinobis(3-ethylbenzthiazolinesulfonic acid]-H2O2 (Kirkegaard and Perry Laboratories, Gaithersburg, MD) were added to the plates. Plates were analyzed at 405 nm (ELX808; Bio-Tek Instruments Inc., Winooski, VT).
Bone marrow antibody-secreting cells (ASC) were enumerated using an antigen-specific enzyme-linked immunospot assay (15). Briefly, bone marrow cells were treated with 1.66% NH4Cl solution to lyse red blood cells, prior to plating onto antigen-coated (10 μg/ml) Multiscreen 96-well plates (Millipore, Billerica, MA). Cells were plated at 106 cells/well, and threefold serial dilutions were made before a 4-h incubation at 37°C. After incubation, plates were washed with 0.1% Tween 20 followed by distilled water. ASC were detected by incubation with horseradish peroxidase-conjugated anti-mouse IgG (heavy plus light chains) (Southern Biotech). Plates were developed with peroxidase-3-amino-9-ethylcarbazole (Vector Laboratories, Burlingame, CA) chromogen substrate and enumerated using ImmunoSpot software (CTL Analyzers LLC, Cleveland, OH).
Statistics.
The P values were determined using the Student t test.
RESULTS
Short-term assessment of inflammatory responses during M. leprae infection.
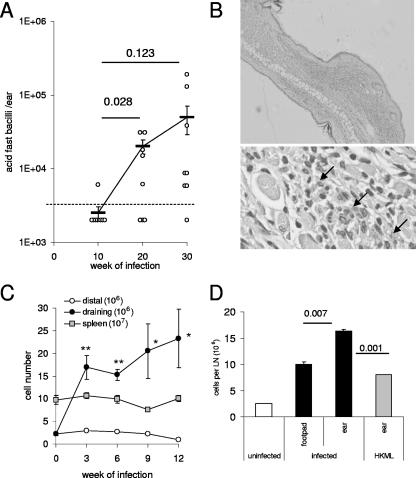
To establish if ears can support M. leprae infection and be used to develop a more rapid system with which to investigate experimental leprosy, we inoculated mice in the ears with M. leprae bacilli. M. leprae bacilli could not be detected in ears collected 10 weeks after infection (detectable bacilli were recovered from only 12.5% of the inoculated ears), whereas significantly more bacilli were counted in ears collected 20 weeks (62.5%, P = 0.028) or 30 weeks (90%, P = 0.123) after infection (Fig. 1A). Histological analyses of M. leprae-infected ears revealed that bacilli could be observed within the dermal layer, along with significant numbers of infiltrating cells (Fig. 1B). These data indicate that the ear can support M. leprae growth as well as a local immune response.
FIG. 1.
Local lymphadenopathy following inoculation in each ear with M. leprae bacilli. (A) Mice were infected with 5 × 105 M. leprae bacilli, and the numbers of M. leprae bacilli in 8 to 12 infected ears (four to six mice) at each time were determined. Each point represents the number of bacilli detected in an individual ear, with the bar representing the mean number of bacilli at each time and the dashed line indicating the limit of detection of this assay. (B) Hematoxylin- and eosin-stained sections of ears infected for 30 weeks. Some of the M. leprae bacilli are indicated by the arrows. (C) LN (each mouse with two DLN or six distal nodes) and spleen cell numbers are shown as the means for three mice per infected time or of eight total mice (two per time) to derive an uninfected value. *, P < 0.05; **, P < 0.01. (D) Additional mice were inoculated in the ears with 1 × 106 heat-killed M. leprae (HKML) or in the footpads with 1 × 106 live M. leprae bacilli, and DLN were examined 15 weeks later. Cell numbers are shown as the means and standard errors of the means for two groups comprising four to six DLN. Results are representative of three to five similar experiments.
To investigate pathogenesis and immune responses during leprosy, we analyzed local inflammation during M. leprae ear infection in relatively short-term experiments (12 to 15 weeks, compared to 6 to 12 months required following footpad inoculation). Draining LN (DLN) from infected mice contained significantly more cells than distal LN from the same infected mice or LN from uninfected mice (week 0), with DLN cell numbers progressively increasing during the early weeks of infection (Fig. 1B). In contrast to the DLN, the distal LN and spleens of infected and uninfected mice were similar in size and cell number (Fig. 1B). These data indicate that inoculation of M. leprae into the ear dermis stimulates a local inflammatory response.
To contrast the ear and footpad routes of infection, mice were infected by i.d. inoculation of bacilli into the ear or s.c. inoculation of bacilli into the footpad. Fifteen weeks later DLN cell numbers in mice inoculated via either route were increased (P < 0.001 for either ear- or footpad-infected mice versus uninfected mice; Fig. 1C). The increase, however, was greater following ear infection than footpad infection (P = 0.007; Fig. 1C). Experiments were also conducted with an additional group of mice inoculated in the ears with heat-killed M. leprae. Although DLN from heat-killed M. leprae-inoculated mice were also enlarged, they contained significantly less cells than DLN from infected mice (Fig. 1C; P = 0.001). These observations indicate that infection of the ear stimulates a larger inflammatory response than infection of the footpad and that the magnitude of DLN hyperplasia is related to infection.
DLN cell populations in infected mice.
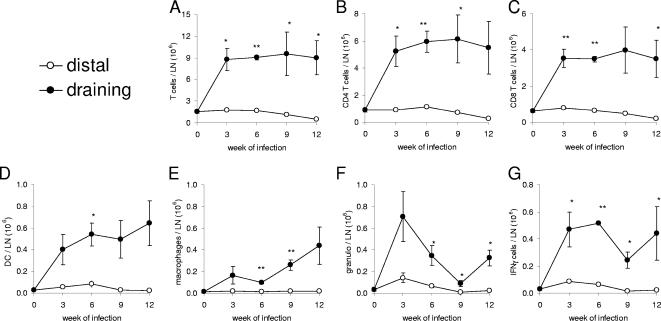
To examine if the increase in DLN cell numbers in infected mice was due to the expansion of all LN cells or the selective expansion of a particular cell type, DLN cell composition was determined by flow cytometry. These investigations indicated that, compared with either distal LN from infected mice or LN from uninfected mice, there were no dramatic alterations in the DLN cell composition of M. leprae-infected mice (data not shown). Rather, the numbers of all cell types examined (αβ CD4 and CD8 T cells, dendritic cells, granulocytes, and macrophages) significantly increased during infection (Fig. 2). While the expansion of most cell types occurred early during infection and was maintained as infection progressed, the number of granulocytes peaked at 3 weeks following infection and then slowly declined (Fig. 2F). Vβ chain use within the T-cell populations was not altered (data not shown), demonstrating that selective outgrowth of particular T cells during infection did not occur. DLN cells capable of producing IFN-γ also increased in number during infection (Fig. 2G). These data indicate that M. leprae ear infection stimulates a prolonged expansion of cells, including IFN-γ-producing cells, within DLN.
FIG. 2.
T-cell expansion within DLN during M. leprae infection. Mice were infected in the ear with 1 × 106 live M. leprae bacilli, LN were collected at various times thereafter, and single-cell suspensions were subjected to flow cytometry to determine and enumerate various cell types. (A) αβ T cells (TCRβ+); (B) CD4 T cells (CD4+, TCRβ+); (C) CD8 T cells (CD8+, TCRβ+); (D) dendritic cells (DC) (CD11c+); (E) macrophages (F4/80+); (F) granulocytes (granulo) (Ly6G/C+); and (G) IFN-γ-producing cells (IFN-γ+ following PMA-ionomycin stimulation). Results are shown as the means and standard errors of the means for three mice per time and are representative of two individual experiments. *, P < 0.05; **, P < 0.01 (versus uninfected or distal LN).
Cell infiltration of M. leprae-infected ears.
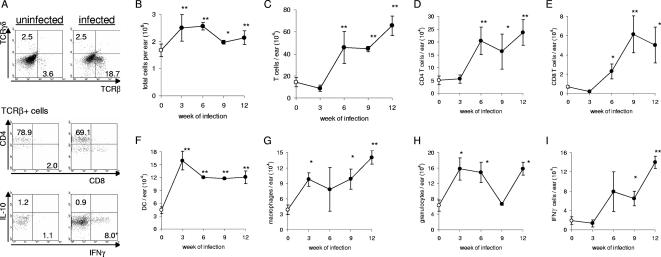
To more closely examine the site of infection, cells were extracted from M. leprae-infected ears. Infected ears yielded significantly more mononuclear cells than uninfected ears (Fig. 3A). Flow cytometry of the cells extracted from the ears demonstrated an influx of αβ T cells following infection (Fig. 3A), while the percentage of nonclassical T cells in infected ears was not altered (γδ T cells and NK1.1-expressing NK T cells; Fig. 3A and data not shown). The numbers of both CD4 and CD8 T cells increased as infection progressed, with CD4 T cells outnumbering CD8 T cells approximately 2 to 1 (Fig. 3). Many of the infiltrating T cells displayed an activated phenotype (CD44hi; data not shown) and produced IFN-γ, but not IL-10, upon mitogen stimulation (Fig. 3A). The number of IFN-γ-producing cells in the ears increased as infection progressed (Fig. 3I). These results indicate that M. leprae infection of the ear promotes an influx of IFN-γ-producing T cells and stimulates a persistent local inflammatory response.
FIG. 3.
T cells infiltrate the ear during M. leprae infection. Mice were infected in the ear with 1 × 106 live M. leprae bacilli, and ears were collected at regular intervals to provide single-cell suspensions. Cell suspensions were subjected to flow cytometry. (A) Representative flow cytometry plots. Various cell types were enumerated as (B) total cells, (C) αβ T cells (TCRβ+), (D) CD4 T cells (CD4+, TCRβ+), (E) CD8 T cells (CD8+, TCRβ+), (F) dendritic cells (DC) (CD11c+), (G) macrophages (F4/80+), (H) granulocytes (Ly6G/C+), and (I) IFN-γ-producing cells (IFN-γ+ following PMA-ionomycin stimulation). Results are shown as the means and standard errors of the means for three mice per time (results for uninfected mice are plotted at week 0) and are representative of two or three similar experiments. *, P < 0.05; **, P < 0.01 (versus uninfected mice).
Rifampin treatment prevents M. leprae-induced local inflammation.
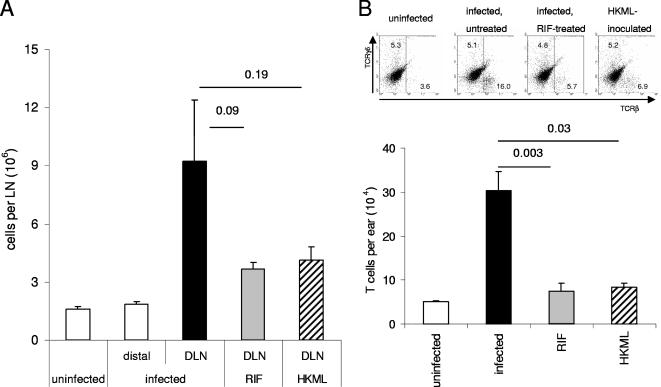
Having established that M. leprae infection of the ear causes local inflammation, we wanted to determine if this inflammation could be interrupted. Rifampin has antimycobacterial properties, and this drug is commonly used to treat leprosy (5, 14, 23). We hypothesized that rifampin treatment would prevent bacillus growth and therefore limit development of local inflammation. Mice were infected in the ears and treated with rifampin shortly after infection. In agreement with our hypothesis, although not significant in this experiment, fewer cells were recovered from DLN of infected, rifampin-treated mice than from DLN of infected, untreated mice (Fig. 4A). The DLN cell numbers for infected, rifampin-treated mice were similar to those for heat-killed M. leprae-inoculated mice (Fig. 4A). Similarly, when the ear cell infiltrates of these mice were examined, we found that infected, rifampin-treated mice had a significantly reduced T-cell infiltration compared to infected, untreated mice (Fig. 4B). Taken together, these data indicate that the local inflammation observed following M. leprae ear infection can be limited by antimycobacterial drug treatment.
FIG. 4.
Rifampin (RIF) treatment during M. leprae infection interrupts local inflammation. Mice were inoculated in each ear with 106 live M. leprae or heat-killed M. leprae (HKML) bacilli. Infected mice were then treated with 0.5 mg rifampin or PBS (untreated) at 1, 2, and 3 weeks after infection. Twelve weeks after inoculation LN and ears were collected. (A) Total cells in draining and distal LN were enumerated. (B) Representative fluorescence-activated cell sorter plots identifying γδ and αβ T cells within the ear are shown, and αβ T cells in the ears were enumerated. Results are shown as the means and standard errors of the means for three mice per group. Results are representative of two similar experiments.
Infection of the ear induces specific anti-M. leprae responses.
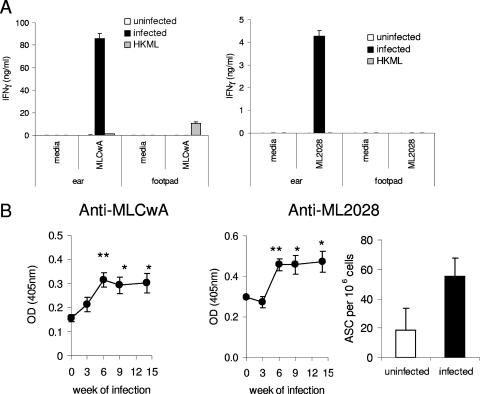
Infection of the ear stimulated a larger local inflammatory response than infection of the footpad, suggesting that ear infection is more immune stimulatory than footpad infection. To further test this observation, spleen cells from the M. leprae-infected mice were incubated with M. leprae cell wall antigens and the culture supernatants analyzed. In these experiments, spleen cells from mice infected in the footpad did not exhibit M. leprae-specific IFN-γ recall responses 15 weeks after infection (Fig. 5A). In marked contrast, spleen cells from mice infected in the ear produced IFN-γ, but not IL-4, in response to M. leprae cell wall antigens 15 weeks after infection (Fig. 5A and data not shown). While spleen cells from mice inoculated in the ears with heat-killed M. leprae also exhibited IFN-γ recall responses, these responses were significantly lower than the responses of the infected mice (P < 0.01). These results demonstrate that, following ear infection but not footpad infection, specific anti-M. leprae cellular responses are induced. We explored the antigen specificity of these responses by stimulating cells from mice infected in the ears with the recombinantly expressed M. leprae antigen ML2028. Cells from mice infected in the ears, but not from mice infected in the footpad or inoculated with heat-killed M. leprae, secreted IFN-γ in response to ML2028 (Fig. 5A). Thus, infection in the ear primes antigen-specific T cells more easily than infection in the footpad.
FIG. 5.
Ear infection, but not footpad infection, stimulates M. leprae-specific cellular and humoral responses. Mice were infected in the ear or footpad with 1 × 106 M. leprae bacilli for 15 weeks. (A) Spleen cell suspensions were cultured in the presence of M. leprae cell wall antigens (MLCwA) or ML2028, and IFN-γ content in the supernatants was determined by ELISA. Results are shown as the means and standard deviations for three mice per group. HKML, heat-killed M. leprae. (B) Sera were collected from mice infected in the ear dermis and IgG responses against M. leprae cell wall antigens or ML2028 determined. Results are shown as the means and standard errors of the means (SEM) for five mice per group. Results are representative of two or three similar experiments. *, P < 0.05; **, P < 0.01 (versus uninfected values). The numbers of anti-ML2028-secreting plasma cells in the bone marrow were also assessed in a single experiment, and results are shown as the means and SEM of eight mice per group. OD, optical density.
Having demonstrated that mice infected in the ear with M. leprae develop local T-cell inflammation and systemic cellular responses, we questioned if these mice also developed anti-M. leprae humoral responses. Sera were collected during the early stages of infection, and antibody responses were assessed by ELISA. We could not detect IgM responses against PGL-I (data not shown), in agreement with previous results demonstrating that this M. leprae glycolipid is a poor immunogen in B6 mice (20a). Weak, but significant, IgG responses against M. leprae cell wall antigens and ML2028 were detected in sera from infected mice, and these responses increased as infection progressed (Fig. 5). Due to the low antibody responses, as measured by ELISA, after 6 weeks of infection, we also enumerated the number of long-lived ASC in the bone marrow of infected mice. More anti-ML2028-secreting plasma cells could be found in the bone marrow of infected mice than in that of uninfected mice (Fig. 5B). These data indicate that, in addition to T-cell priming, successful B-cell priming and antibody production occur following M. leprae infection of the ear.
DISCUSSION
Leprosy exhibits highly divergent immune responses and pathologies (28, 30). Little is known as to what influences the differential development of the strong cellular response of PB patients or the weak/absent cellular response of multibacillary patients. Animal models of leprosy could provide important insight, but the lack of animal models that demonstrate robust immune responses during M. leprae infection has limited the ability to understand leprosy pathogenesis. The M. leprae mouse footpad infection model was developed by Shepard in the 1960s to assess bacterial growth, but this model requires a significant length of time and demonstrates only minor pathology (32-34). Recent studies of experimental cutaneous leishmaniasis have indicated that i.d. inoculation of Leishmania major into the ear promotes disease symptoms (6-8). We report here that following M. leprae infection of the ear there is a marked DLN hyperplasia and a cell infiltration at the inoculation site, indicating the induction of inflammatory responses. In agreement, mice infected by i.d. inoculation in the ear demonstrate M. leprae-specific cellular responses, in stark contrast to mice infected by s.c. inoculation in the footpad. Mice infected in the ear also produce anti-M. leprae antibodies. Thus, following ear infection, both B- and T-cell responses are rapidly detected, indicating that the ear model can provide insight into leprosy pathogenesis and development of the immune response.
Mice infected by i.d. inoculation into the ear developed systemic IFN-γ responses against M. leprae antigens (M. leprae cell wall antigens and ML2028). This was in marked contrast with mice infected by s.c. inoculation in the footpad, which did not demonstrate IFN-γ recall responses. Several recent studies investigating experimental cutaneous leishmaniasis have identified differences in the development of L. major infection when infection of the ear is compared with infection of the footpad (6-8). It is unclear why there are differences in the resulting immune responses of mice infected in these different sites. One possibility is that the presence of different antigen-presenting-cell populations in the ear and footpad results in different qualities of M. leprae antigen presentation (9), and this is worthy of further examination.
PB leprosy patients demonstrate well-defined skin lesions that, upon biopsy, demonstrate a granulomatous inflammation comprising more CD4 than CD8 T cells (25). Mice infected with M. leprae in the ears quickly developed a localized inflammatory response comprising DLN hyperplasia and recruitment of more CD4 T cells than CD8 T cells to the inoculation site. Although macroscopic lesions were not observed on M. leprae-infected ears, the cell types of the ear infiltrate are consistent with those of PB leprosy lesions. It remains to be determined if these cells form granulomas within the infected ears, but these results indicate that the ear infection model can be used to delineate the critical mediators of local inflammatory responses during leprosy.
The early treatment of M. leprae infection is critical for prevention of tissue and nerve damage. The ear infection model described here provides results more rapidly than the footpad model, which typically investigates bacillus growth and is currently used for the testing of M. leprae drug candidates (19). We have demonstrated that rifampin can be used to limit the local inflammation that develops following ear infection. It is likely that M. leprae ear infection can be used to more rapidly assess other antimycobacterial drugs that could be used alone or in conjunction with current MDT protocols for the treatment of leprosy. In addition, we have demonstrated immune responses to both crude M. leprae cell wall antigens and the recombinant antigen ML2028 following M. leprae ear infection. ML2028 induces the M. leprae homolog of M. tuberculosis Ag85B, a secreted antigen that has previously been demonstrated to confer protection against experimental leprosy infection (26). Thus, the ear infection system may be invaluable for the identification of additional M. leprae antigens that can be used in a vaccine to protect against M. leprae infection.
The results reported here demonstrate several immune parameters (DLN enlargement, ear infiltration, cellular responses, and antibody responses) that can be analyzed relatively rapidly following M. leprae ear infection and used to improve our understanding of the immune response during leprosy. These parameters will also be helpful for the identification and assessment of new treatments for leprosy.
Acknowledgments
This paper is dedicated to John Dawson, without whom this work would not have been possible. We thank Yasuyuki Goto for critical reading of the manuscript.
This work was supported by a grant from the American Leprosy Missions (S.G.R.) and NIAID grant AI45725 (T.P.G.).
Editor: J. L. Flynn
Footnotes
Published ahead of print on 27 August 2007.
REFERENCES
- 1.Adams, L. B., T. P. Gillis, D. H. Hwang, and J. L. Krahenbuhl. 1997. Effects of essential fatty acid deficiency on prostaglandin E2 production and cell-mediated immunity in a mouse model of leprosy. Infect. Immun. 65:1152-1157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Adams, L. B., C. K. Job, and J. L. Krahenbuhl. 2000. Role of inducible nitric oxide synthase in resistance to Mycobacterium leprae in mice. Infect. Immun. 68:5462-5465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Adams, L. B., D. M. Scollard, N. A. Ray, A. M. Cooper, A. A. Frank, I. M. Orme, and J. L. Krahenbuhl. 2002. The study of Mycobacterium leprae infection in interferon-gamma gene-disrupted mice as a model to explore the immunopathologic spectrum of leprosy. J. Infect. Dis. 185(Suppl. 1):S1-S8. [DOI] [PubMed] [Google Scholar]
- 4.Azouaou, N., R. H. Gelber, K. Abel, D. T. Sasaki, L. P. Murray, R. M. Locksley, and N. Mohagheghpour. 1993. Reconstitution of Mycobacterium leprae immunity in severe combined immunodeficient mice using a T-cell line. Int. J. Lepr. Other Mycobact. Dis. 61:398-405. [PubMed] [Google Scholar]
- 5.Bakker, M. I., M. Hatta, A. Kwenang, B. H. Van Benthem, S. M. Van Beers, P. R. Klatser, and L. Oskam. 2005. Prevention of leprosy using rifampicin as chemoprophylaxis. Am. J. Trop. Med. Hyg. 72:443-448. [PubMed] [Google Scholar]
- 6.Baldwin, T. M., C. Elso, J. Curtis, L. Buckingham, and E. Handman. 2003. The site of Leishmania major infection determines disease severity and immune responses. Infect. Immun. 71:6830-6834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Belkaid, Y., K. F. Hoffmann, S. Mendez, S. Kamhawi, M. C. Udey, T. A. Wynn, and D. L. Sacks. 2001. The role of interleukin (IL)-10 in the persistence of Leishmania major in the skin after healing and the therapeutic potential of anti-IL-10 receptor antibody for sterile cure. J. Exp. Med. 194:1497-1506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Belkaid, Y., S. Mendez, R. Lira, N. Kadambi, G. Milon, and D. Sacks. 2000. A natural model of Leishmania major infection reveals a prolonged “silent” phase of parasite amplification in the skin before the onset of lesion formation and immunity. J. Immunol. 165:969-977. [DOI] [PubMed] [Google Scholar]
- 9.Berman, B., V. L. Chen, D. S. France, W. I. Dotz, and G. Petroni. 1983. Anatomical mapping of epidermal Langerhans cell densities in adults. Br. J. Dermatol. 109:553-558. [DOI] [PubMed] [Google Scholar]
- 10.Cambau, E., P. Bonnafous, E. Perani, W. Sougakoff, B. Ji, and V. Jarlier. 2002. Molecular detection of rifampin and ofloxacin resistance for patients who experience relapse of multibacillary leprosy. Clin. Infect. Dis. 34:39-45. [DOI] [PubMed] [Google Scholar]
- 11.Cellona, R. V., M. F. Balagon, E. C. de la Cruz, J. A. Burgos, R. M. Abalos, G. P. Walsh, R. Topolski, R. H. Gelber, and D. S. Walsh. 2003. Long-term efficacy of 2 year WHO multiple drug therapy (MDT) in multibacillary (MB) leprosy patients. Int. J. Lepr. Other Mycobact. Dis. 71:308-319. [DOI] [PubMed] [Google Scholar]
- 12.Colston, M. J., and G. R. Hilson. 1976. Growth of Mycobacterium leprae and M. marinum in congenitally athymic (nude) mice. Nature 262:399-401. [DOI] [PubMed] [Google Scholar]
- 13.Converse, P. J., V. L. Haines, A. Wondimu, L. E. Craig, and W. M. Meyers. 1995. Infection of SCID mice with Mycobacterium leprae and control with antigen-activated “immune” human peripheral blood mononuclear cells. Infect. Immun. 63:1047-1054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Dhople, A. M. 2000. Search for newer antileprosy drugs. Indian J. Lepr. 72:5-20. [PubMed] [Google Scholar]
- 15.Erickson, L. D., B. G. Durell, L. A. Vogel, B. P. O'Connor, M. Cascalho, T. Yasui, H. Kikutani, and R. J. Noelle. 2002. Short-circuiting long-lived humoral immunity by the heightened engagement of CD40. J. Clin. Investig. 109:613-620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Gelber, R. H., V. F. Balagon, and R. V. Cellona. 2004. The relapse rate in MB leprosy patients treated with 2-years of WHO-MDT is not low. Int. J. Lepr. Other Mycobact. Dis. 72:493-500. [DOI] [PubMed] [Google Scholar]
- 17.Girdhar, B. K., A. Girdhar, and A. Kumar. 2000. Relapses in multibacillary leprosy patients: effect of length of therapy. Lepr. Rev. 71:144-153. [DOI] [PubMed] [Google Scholar]
- 18.Jamet, P., and B. Ji. 1994. Relapse after long-term follow up of multibacillary patients treated by WHO multidrug regimen. Int. J. Lepr. Other Mycobact. Dis. 62:622. [PubMed] [Google Scholar]
- 19.Ji, B., A. Chauffour, K. Andries, and V. Jarlier. 2006. Bactericidal activities of R207910 and other newer antimicrobial agents against Mycobacterium leprae in mice. Antimicrob. Agents Chemother. 50:1558-1560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ji, B., P. Jamet, S. Sow, E. G. Perani, I. Traore, and J. H. Grosset. 1997. High relapse rate among lepromatous leprosy patients treated with rifampin plus ofloxacin daily for 4 weeks. Antimicrob. Agents Chemother. 41:1953-1956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20a.Koster, F. T., C. Teuscher, P. Matzner, E. Umland, D. Yanagihara, P. J. Brennan, and K. S. Tung. 1986. Strain variations in the murine cellular immune response to the phenolic glycolipid I antigen of Mycobacterium leprae. Infect. Immun. 51:495-500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Krahenbuhl, J., and L. B. Adams. 2000. Exploitation of gene knockout mice models to study the pathogenesis of leprosy. Lepr. Rev. 71(Suppl.):S170-S175. [DOI] [PubMed] [Google Scholar]
- 22.Lockwood, D. N. 2002. Leprosy elimination—a virtual phenomenon or a reality? BMJ 324:1516-1518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lockwood, D. N. 1997. Rifampicin/minocycline and ofloxacin (ROM) for single lesions—what is the evidence? Lepr. Rev. 68:299-300. [DOI] [PubMed] [Google Scholar]
- 24.Maeda, S., M. Matsuoka, N. Nakata, M. Kai, Y. Maeda, K. Hashimoto, H. Kimura, K. Kobayashi, and Y. Kashiwabara. 2001. Multidrug resistant Mycobacterium leprae from patients with leprosy. Antimicrob. Agents Chemother. 45:3635-3639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Modlin, R. L., J. Melancon-Kaplan, S. M. Young, C. Pirmez, H. Kino, J. Convit, T. H. Rea, and B. R. Bloom. 1988. Learning from lesions: patterns of tissue inflammation in leprosy. Proc. Natl. Acad. Sci. USA 85:1213-1217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ohara, N., M. Matsuoka, H. Nomaguchi, M. Naito, and T. Yamada. 2001. Protective responses against experimental Mycobacterium leprae infection in mice induced by recombinant bacillus Calmette-Guerin over-producing three putative protective antigen candidates. Vaccine 19:1906-1910. [DOI] [PubMed] [Google Scholar]
- 27.Rees, R. J., M. F. Waters, A. G. Weddell, and E. Palmer. 1967. Experimental lepromatous leprosy. Nature 215:599-602. [DOI] [PubMed] [Google Scholar]
- 28.Ridley, D. S., and W. H. Jopling. 1966. Classification of leprosy according to immunity. A five-group system. Int. J. Lepr. Other Mycobact. Dis. 34:255-273. [PubMed] [Google Scholar]
- 29.Scollard, D. M. 2004. Classification of leprosy: a full color spectrum, or black and white? Int. J. Lepr. Other Mycobact. Dis. 72:166-168. [DOI] [PubMed] [Google Scholar]
- 30.Scollard, D. M., L. B. Adams, T. P. Gillis, J. L. Krahenbuhl, R. W. Truman, and D. L. Williams. 2006. The continuing challenges of leprosy. Clin. Microbiol. Rev. 19:338-381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Scollard, D. M., G. W. Lathrop, and R. W. Truman. 1996. Infection of distal peripheral nerves by M. leprae in infected armadillos; an experimental model of nerve involvement in leprosy. Int. J. Lepr. Other Mycobact. Dis. 64:146-151. [PubMed] [Google Scholar]
- 32.Shepard, C. C. 1960. The experimental disease that follows the injection of human leprosy bacilli into foot-pads of mice. J. Exp. Med. 112:445-454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Shepard, C. C. 1962. Multiplication of Mycobacterium leprae in the foot-pad of the mouse. Int. J. Lepr. 30:291-306. [PubMed] [Google Scholar]
- 34.Shepard, C. C., and R. S. Guinto. 1963. Immunological identification of foot-pad isolates as Mycobacterium leprae by lepromin reactivity in leprosy patients. J. Exp. Med. 118:195-204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Shepard, C. C., and D. H. McRae. 1968. A method for counting acid-fast bacteria. Int. J. Lepr. Other Mycobact. Dis. 36:78-82. [PubMed] [Google Scholar]
- 36.Storrs, E. E., G. P. Walsh, H. P. Burchfield, and C. H. Binford. 1974. Leprosy in the armadillo: new model for biomedical research. Science 183:851-852. [DOI] [PubMed] [Google Scholar]
- 37.Truman, R. W., and J. L. Krahenbuhl. 2001. Viable M. leprae as a research reagent. Int. J. Lepr. Other Mycobact. Dis. 69:1-12. [PubMed] [Google Scholar]
- 38.Truman, R. W., M. J. Morales, E. J. Shannon, and R. C. Hastings. 1986. Evaluation of monitoring antibodies to PGL-I in armadillos experimentally infected with M. leprae. Int. J. Lepr. Other Mycobact. Dis. 54:556-559. [PubMed] [Google Scholar]
- 39.World Health Organization. 2005. Global leprosy situation, 2005. Wkly. Epidemiol. Rec. 80:289-295. [PubMed] [Google Scholar]