Abstract
To compare the diarrheagenic Escherichia coli (DEC) identifications obtained between traditional O serotyping and modern virulence gene detection assays, we developed a multiplex real-time PCR assay by detecting six specific virulence genes for enteropathogenic E. coli (EPEC), enterohemorrhagic E. coli (EHEC), enterotoxigenic E. coli (ETEC), and enteroinvasive E. coli (EIEC). Among 261 clinical diarrheal stool samples, a total of 137 suspected DEC (sDEC) isolates were identified by the use of commercially available antisera. The most prevalent serogroups were O1 (12/137; 8.7%), O25 (9/137; 6.5%), and O44 (9/137; 6.5%). The specific virulence genes for the 137 sDEC isolates were analyzed by the multiplex real-time PCR assay. Fifteen (10.9%) of 137 isolates were confirmed to be true DEC strains, indicating that the serotypic markers did not correlate with the specific virulence genes. ETEC (66.7%) was the most prevalent, followed by EIEC (20%) and EPEC (13.3%). No EHEC strains were identified in the specimens. Four novel serotypes were found in the study: two in EPEC strains (O111:H9 and O63:H6) and two in EIEC strains (O63:H9 and O169:H9). In conclusion, the real-time PCR assay considerably reduces the high false-positive rate from the use of serotyping alone, and thus, it is suggested that serogrouping-based methods are inadequate for the identification of DEC isolates, although they are useful for the identification of a limited number of serogroups. In addition, ETEC, EPEC, and EIEC strains were present in 5.7% (15/261) of the diarrheal patients in northern Taiwan in 2006.
Escherichia coli is a predominant member of the human intestinal flora. Some strains are rendered pathogenic by their ability to possess specific virulence factors, such as enterotoxin or adherent fimbriae, that are genetically encoded by plasmid, chromosome, and bacteriophage DNA (2, 14). These diarrheagenic E. coli (DEC) strains usually play important roles as the causes of endemic and epidemic human diseases, such as severe diarrhea, food poisoning, and similar outbreaks worldwide (9, 15). Infections with these kinds of pathogens have therefore been of increasing concern in the clinical diagnosis of diarrheal disease in recent years. These virulent organisms can be classified into five major categories on the basis of the nature of their infection and pathogenic mechanisms: they are enteropathogenic E. coli (EPEC), enterohemorrhagic E. coli (EHEC), enterotoxigenic E. coli (ETEC), enteroinvasive E. coli (EIEC), and enteroaggregative E. coli (EAEC) (14, 25). However, the current rates of infections by these important enteric pathogens are probably largely underestimated due to the limitations of existing clinical diagnostic methods to be able to distinguish them from normal nonpathogenic flora. In order to achieve the goal of epidemic prevention and control of DEC in Taiwan, we need a more reliable procedure to identify and categorize DEC isolates so that more reliable studies of DEC incidence rates can be conducted in the future.
Phenotypic assays and serotyping are the traditional methods available for the detection of DEC (7), and the former, based on virulence characteristics, remains the “gold standard” for EAEC detection. However, such assays are extremely time-consuming and laborious, and they need to be carried out and their results need to be interpreted by well-trained staff. As a result, many clinical laboratories are not quite capable of conducting this assay without difficulties. The serotype-based method has been widely used in clinical laboratories with commercial antisera or antisera prepared in-house. Although several serotypes are predominantly found by the use of serotyping and correlated with specific categories of DEC, not all of the isolates belonging to those serotypes are truly pathogenic (1, 7, 17). In addition, serotypic markers themselves do not confer pathogenicity but sometimes correlate very closely with pathogenicity. This antigen similarity may easily lead to false-positive results in the typing process. In Japan, only 17.5% of E. coli isolates agglutinated with one of the proposed DEC O antisera harboring pathogenic properties (25). It may therefore be insufficient and unreliable to define an isolate as truly pathogenic by using serotyping alone (24). Since several virulence factors have been identified in DEC strains, modern molecular detection methods, including PCR and DNA hybridization, have been developed (12, 21, 26). They are performed at the genetic level and directly detect genes for specific virulence factors, which themselves determine the pathogenicity. The presence of these genes is the evidence that renders the virulence and that can be used to categorize DEC strains harboring the genes into five groups. Among the molecular detection assays, real-time PCR is commonly used and can give rapid, sensitive, and specific results. Various DEC detection assays that have used real-time PCR have been reported in recent years (4, 5, 19).
In this study, we looked for the presence of ETEC, EPEC, EIEC, and EHEC strains and their serotypes in northern Taiwan. We also developed a multiplex real-time PCR system to identify the four categories of DEC strains and to clarify the relationship between the E. coli serotypic markers and the specific virulence factors. Four novel serotypes of DEC isolates were also identified in this study.
MATERIALS AND METHODS
Clinical specimens and culture process.
Stool samples from patients who had diarrhea, which was characterized by the occurrence of loose, watery, or bloody stools, were collected in Cary-Blair transport medium (BD Diagnostic Systems, Sparks, MD) from more than 30 clinics and hospitals in northern Taiwan, including the cities and counties of Taipei, Taoyuan, Hsin-Chu, Yilan, and Keelung. Rectal swabs were also taken from inbound travelers with diarrhea by quarantine officials at the Taiwan Taoyuan International Airport. After the samples arrived in our laboratory, they were cultured on sorbitol MacConkey agar (Creative Microbiologicals, Ltd., Taipei, Taiwan), followed by overnight incubation at 37°C. A total of 261 clinical specimens, gathered between 1 October 2005 and 30 December 2006, were studied. At least five separate lactose-fermenting and lactose-nonfermenting colonies from each specimen were then purified and confirmed to be E. coli by several biochemical reactions, including sugar utilization, lysine carboxylation, indole production, and the motility test. These purified E. coli isolates were further identified as described below.
Bacterial strains.
The DEC reference strains used as positive controls in this study were CCRC 15376 (an EHEC isolate) containing eae, stx1, and stx2; CCRC 15372 (an ETEC isolate) containing lt and st; CCRC 15536 (an EPEC isolate) containing eae; and ATCC 43893 (an EIEC isolate) containing ipaH. Other non-DEC enteric pathogens used in the specificity test were clinical isolates obtained from the Taiwan CDC, including flora E. coli, Shigella flexneri, Shigella sonnei, Salmonella enterica serovar Enteritidis, Salmonella enterica serovar Typhi, Salmonella enterica serovar Paratyphi, Bacillus cereus, Staphylococcus aureus, and Vibrio parahemolyticus. The isolates to be examined by the real-time PCR were E. coli isolates belonging to one of the 50 pathogenic O serogroups and were isolated by the Taiwan CDC from the 261 patients with cases of sporadic diarrhea.
Serotyping O and H antigens.
The O serotyping method was used to identify the isolates belonging to O serogroups defined commercially. Isolates that can agglutinate with one of the specific commercial pathogenic O antisera were defined as “suspected DEC” (sDEC) isolates, and they were collected for further testing to see if they carried any of the six virulence genes mentioned below. For O-antigen determination, we suspended the bacterial culture in 3 ml normal saline, heated the mixture to 100°C for 1 h, and used the boiled suspension as the antigenic mixture. We then mixed 1 drop of a specific O poly- or monovalent antiserum of “pathogenic E. coli immune sera” (Denka Seiken, Tokyo, Japan) with the antigen preparation on a glass slide for 1 min and observed the slide for agglutination.
For H-antigen determination, we passed the bacterial culture through the semisolid medium with a Craigie's tube to enhance the motile ability and then grew the culture in liquid broth. After the addition of a formalin solution to achieve a final concentration of 1%, the suspension could be used as an antigenic mixture and was mixed with specific H-antigen monovalent antiserum (Denka Seiken) in a plastic tube. The agglutination results could be observed after the tubes were kept in a 50°C water bath for 1 h.
Template DNA preparation.
The template DNA source for the real-time PCR was crude bacterial extract. A 0.5-ml overnight bacterial suspension was centrifuged for 3 min. The resulting pellet was resuspended in 0.5 ml PrepMan Ultra sample preparation reagent (Applied Biosystems, Foster City, CA), and the suspension was boiled for 5 min. We then centrifuged the suspension for 5 min and directly used the supernatant as the PCR template.
Real-time PCR amplification, oligonucleotide primers, and probes.
Six virulence factor genes of E. coli were selected to classify the DEC strains: they were lt and st, which encode heat-labile and heat-stable toxins, respectively; eae, which encodes intimin; stx1 and stx2, which encode Shiga-like toxins I and II, respectively; and ipaH, which is present at multiple loci on a large invasive plasmid. The detection targets, oligonucleotide primers, and probes used for the real-time PCR used in this study are listed in Table 1. The primers and probes for stx1, stx2 and eae, including stxA1 598, stxA1 1015, stxA2 679, stxA2 942, eaeAF, eaeAR, stxA1 FL724, stxA1 LC, ALL FL, ALL LC, eae FL, and eae LC, have been published elsewhere (4, 18). The primers for lt, st, and ipaH were designed by our laboratory; and their suitabilities were evaluated by TIB MOLBIOL, Berlin, Germany. No secondary structure was formed among the primers; and the oligonucleotide lengths, the amplicon sizes, and the melting temperatures ranged from 16 to 24 bases, 150 to 450 bp, and 50 to 62°C, respectively. The multiplex real-time PCR mixture was prepared in a total volume of 50 μl. Each PCR mixture contained the following: 4 μl of LightCycler FastStart DNA MasterPlus HybProbe 5×-concentrated reagent (Roche Diagnostics, Penzberg, Germany), 0.5 μM each forward and reverse primers, 0.2 μM each FL and LC probes, and 5 μl of the boiled bacterial extract. The PCR amplification program consisted of one 10-min denaturation step at 95°C, followed by a cycling step of 45 cycles. For multiplex detection, the cycling steps were as follows: heating at 95°C for 5 s, annealing at 50°C for 7 s (lt and st) or 56°C for 7 s (stx1 and stx2), and extension at 72°C for 12 s (lt and st) or 18 s (stx1 and stx2). For ipaH and eae, the cycling steps were as follows: heating at 95°C for 7 s, annealing at 50°C (ipaH) for 7 s or at 56°C for 7 s (eae), and extension at 72°C for 10 s (ipaH) or 16 s (eae).
TABLE 1.
Primers and probes for the real-time PCR used in this study
| Target gene | Primer or probe | Sequence (5′-3′) | Amplicon size (bp) | Tma (°C) of primer or probe | Reference or source |
|---|---|---|---|---|---|
| lt | LT S | TTT GTC AGA TAT GAT GAC GGA | 253 | 50.6 | This study |
| LT A | TCA CAC CAA AAT TAA CAC GA | 50.9 | This study | ||
| LT FL | ATT AGG CGT ATA CAG CCC TCA CCC-FL | 62.2 | This study | ||
| LT LC | 640-TAT GAA CAG GAG GTT TCT GCG TTA GGT G-p | 64.4 | This study | ||
| st | ST S | GAT GCT AAA CCA GTA GAG TCT TC | 152 | 50.7 | This study |
| ST rev short | CCG GTA CAA GCA GGA TT | 59.1 | This study | ||
| ST FL | AAA GTG GTC CTG AAA GCA TGA AT-FL | 57.5 | This study | ||
| ST LC | 705-GTA GCA ATT ACT GCT GTG AAT TGT GTT G-p | 61.3 | This study | ||
| stx1 | stxA1 598 | AGT CGT ACG GGG ATG CAG ATA AAT | 418 | 60.2 | 11 |
| stxA1 1015 | CCG GAC ACA TAG AAG GAA ACT CAT | 58.7 | 11 | ||
| stxA1 FL724 | CTG TCA CAG TAA CAA ACC GTA ACA TCG CTC-FL | 65.5 | 11 | ||
| stxA1 LC | 705-TGC CAC AGA CTG CGT CAG TGA GGT-p | 67.3 | 11 | ||
| stx2 | stxA2 679 | TTC CGG AAT GCA AAT CAG TC | 264 | 55.9 | 11 |
| stxA2 942 | CGA TAC TCC GGA AGC ACA TTG | 58.4 | 11 | ||
| ALL FL | MAG AGC AGT TCT GCG TTT TGT CAC TGT CA-FL | 65.6 | 11 | ||
| ALL LC | 640-AGC AGA AGC CTT ACG CTT CAG GC-p | 63.3 | 11 | ||
| eae | eae AF | GAC CCG GCA CAA GCA TAA GC | 384 | 61.5 | 12 |
| eae AR | CCA CCT GCA GCA ACA AGA GG | 61.2 | 12 | ||
| eae FL | CCT GGT CAG CAG ATC ATT TTG CCA CTC A-FL | 68.4 | 12 | ||
| eae LC | 640-ACT TCC CTT TGA ATA CAG TGC CTT ACC A-p | 63.4 | 12 | ||
| ipaH | ipaH-S | GAT AAT GAT ACC GGC GCT CTG | 221 | 57.1 | This study |
| ipaH-R | CGG AGG TCA TTT GCT GTC A | 56.5 | This study | ||
| ipaH FL | CCT TCC AGA CCA TGC TCG CAG AGA A-FL | 67.0 | This study | ||
| ipaH LC | 640-TCA GCT CTC CAC TGC CGT GAA GGA A-p | 67.9 | This study |
Tm, melting temperature.
Nucleotide sequence analysis of real-time PCR product.
The nucleotide sequences of the amplified DNA fragments in the target virulence genes were determined with an ABI PRISM 3100 genetic analyzer (Applied Biosystems). The sequences were further analyzed by evaluation of the sequences against those in the NCBI database with the BLAST program.
RESULTS
Classification of clinical E. coli isolates with pathogenic O serogroup antisera.
A total of 261 stool specimens from patients with sporadic diarrhea were tested for sDEC by using commercially available antisera. One hundred thirty-seven sDEC isolates (52%) belonging to 1 of the 50 pathogenic O serogroups were identified, as shown in Table 2. One or more isolates were found to be of 34 of the 50 O serogroups. The most prevalent serogroups among these isolates were O1 (12/137; 8.7%), O25 (9/137; 6.5%), and O44 (9/137; 6.5%).
TABLE 2.
O serogroups of the 137 sDEC isolates
| Serogroup | No. of isolates |
|---|---|
| O1 | 12 |
| O6 | 7 |
| O8 | 8 |
| O15 | 8 |
| O18 | 7 |
| O20 | 1 |
| O25 | 9 |
| O26 | 2 |
| O27 | 3 |
| O28ac | 1 |
| O29 | 1 |
| O44 | 9 |
| O55 | 4 |
| O63 | 2 |
| O74 | 2 |
| O86a | 7 |
| O91 | 3 |
| O103 | 2 |
| O111 | 1 |
| O112ac | 4 |
| O114 | 3 |
| O124 | 1 |
| O125 | 4 |
| O128 | 2 |
| O144 | 4 |
| O146 | 3 |
| O151 | 1 |
| O152 | 2 |
| O153 | 4 |
| O159 | 6 |
| O164 | 6 |
| O166 | 4 |
| O168 | 2 |
| O169 | 2 |
Sensitivity and specificity of the LightCycler real-time PCR assays for detection of target virulence genes in purified sDEC cultures.
The virulence characteristics used to define individual groups of DEC were as follows: ETEC, the presence of the lt or st gene; EHEC, the presence of the eae and the stx1 or the stx2 gene, or both; EPEC, the presence of the eae gene and the absence of the stx genes; and EIEC, the presence of the ipaH gene. DEC isolates carrying one of the genes mentioned above were reliably classified into four categories and could be compared with the isolates described previously after the H-antigen determination was completed.
The targets of ETEC determination, lt and st, could be detected simultaneously in one reaction mixture, as could the targets of EHEC, stx1 and stx2 (Fig. 1). However, the other two genes, eae and ipaH, were detected in separate reaction tubes. Due to the same primer annealing temperature, the detection of eae could be performed in the same run with the targets of EHEC, and ipaH could be performed in the same run with the targets of ETEC. The entire real-time detection assay, including template DNA preparation, could be completed within 1.5 h. To determine the lower detection limits of the six target genes, serially diluted DNA suspensions for each DEC reference strain were used as the PCR templates. The fluorescence signals of each of the six target genes showed a rather conservative lower limit of 10 CFU per assay. We also validated the specificity of the assay by using different categories of DEC reference strains as positive controls and two flora E. coli isolates and eight other enteric pathogens as negative controls. The results of the test for specificity are shown in Table 3. Each of the four DEC reference strains gave specific positive signals. In addition, no other crossover signals, except the signal generated from the same sequences of the invasive plasmids of EIEC and Shigella spp., were observed.
FIG. 1.
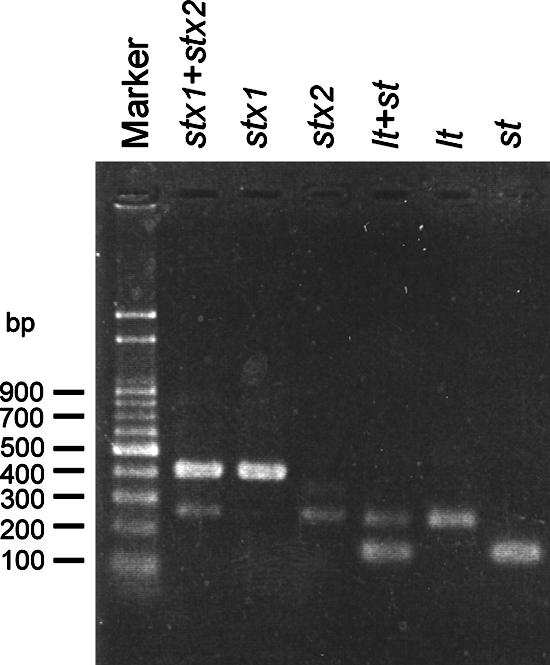
Electrophoresis gel for the multiplex real-time PCRs. The lanes are unnumbered but consist of lanes 1 to 7 from left to right, respectively: lane 1, marker; lanes 2 to 4, detection of EHEC by multiplex reaction for stx1 and stx2, single reaction for stx1, and single reaction for stx2, respectively; lanes 5 to 7, detection of ETEC by multiplex reaction for lt and st, single reaction for lt, and single reaction for st, respectively. The DNA templates used for lanes 2 to 4 and lanes 5 to 7 were from E. coli CCRC 15376 and CCRC 15372, respectively.
TABLE 3.
Specificity of the real-time PCR assay
| Organism | Result for the following target genes detected by the real-time PCR assay:
|
|||||
|---|---|---|---|---|---|---|
| lt | st | stx1 | stx2 | eae | ipaH | |
| Escherichia coli CCRC 15376 (O157:H7; EHEC) | − | − | + | + | + | − |
| Escherichia coli CCRC 15372 (O78:H11; ETEC) | + | + | − | − | − | − |
| Escherichia coli CCRC 15536 (O142:H6; EPEC) | − | − | − | − | + | − |
| Escherichia coli ATCC 43893 (O124:NM; EIEC) | − | − | − | − | − | + |
| Flora E. coli 1 | − | − | − | − | − | − |
| Flora E. coli 2 | − | − | − | − | − | − |
| Shigella flexneri | − | − | − | − | − | + |
| Shigella sonnei | − | − | − | − | − | + |
| Salmonella enterica serovar Enteritidis | − | − | − | − | − | − |
| Salmonella enterica serovar Typhi | − | − | − | − | − | − |
| Salmonella enterica serovar Paratyphi | − | − | − | − | − | − |
| Bacillus cereus | − | − | − | − | − | − |
| Staphylococcus aureus | − | − | − | − | − | − |
| Vibrio parahemolyticus | − | − | − | − | − | − |
Detection of tDEC strains by real-time PCR.
For the purpose of differentiating the sDEC isolates, classifying them into the four groups, and finding the rates of positivity for the six genes among the strains, we tested the 137 isolates by the multiplex real-time PCR assay. Fifteen (10.9%) isolates carrying one of the six virulence genes were true DEC (tDEC) strains, as shown in Table 4. We classified 10 of the 15 isolates as ETEC due to their carriage of the st gene, another 2 isolates with the eae gene but without the stx genes as EPEC, and 3 isolates with the ipaH gene as EIEC. ETEC strains (10/15; 66.7%) were the most prevalent among the isolates tested, followed by EIEC (3/15; 20%) and EPEC (2/15; 13.3%) strains. No EHEC isolates were detected among the clinical specimens tested. The rate of isolation of the tDEC isolates among the 261 clinical specimens was 5.7% (15/261). The rates of isolation of ETEC, EPEC, and EIEC strains among the 137 serogroup-defined sDEC isolates were 7.3% (10/137), 1.5% (2/137), and 2.2% (3/137), respectively.
TABLE 4.
Categorization of the 15 virulence gene-harboring DEC isolates by the real-time PCR assay
| Isolate | Source of specimen | Result for the following target genes detected by the real-time PCR assay:
|
Category | |||||
|---|---|---|---|---|---|---|---|---|
| lt | st | stx1 | stx2 | eae | ipaH | |||
| 809P | Inbound travelera | − | + | − | − | − | − | ETEC |
| 7864 | Indigenous patientb | − | + | − | − | − | − | ETEC |
| 86482 | Indigenous patient | − | + | − | − | − | − | ETEC |
| 559P | Inbound traveler | − | + | − | − | − | − | ETEC |
| 463P | Inbound traveler | − | − | − | − | + | − | EPEC |
| J63 | Emergency patientc | − | + | − | − | − | − | ETEC |
| J80 | Emergency patient | − | + | − | − | − | − | ETEC |
| J91 | Emergency patient | − | + | − | − | − | − | ETEC |
| 896P | Inbound traveler | − | + | − | − | − | − | ETEC |
| 051P | Inbound traveler | − | + | − | − | − | − | ETEC |
| J94 | Emergency patient | − | + | − | − | − | − | ETEC |
| 008629 | Indigenous patient | − | − | − | − | − | + | EIEC |
| 019882 | Indigenous patient | − | − | − | − | − | + | EIEC |
| 019871 | Indigenous patient | − | − | − | − | − | + | EIEC |
| J113 | Emergency patient | − | − | − | − | + | − | EPEC |
Inbound traveler, inbound travelers with diarrhea spotted at the Taoyuan International Airport.
Indigenous patient, patients not considered inbound travelers or emergency patients.
Emergency patient, patients visiting emergency rooms at hospitals.
Serotypes of the virulence gene-positive tDEC isolates.
We further determined the H antigens of the 15 tDEC isolates to determine their serotypes, as shown in Table 5. The 10 ETEC isolates belonging to six different O serogroups (i.e., serogroups O1, O6, O15, O25, O44, and O159) could be further classified into eight different serotypes. Two isolates each were found to be of serotypes O15:H11 and O25:NM (where NM is nonmotile). The three EIEC isolates belonging to serogroups O63, O124, and O169 were found to be of three different serotypes. The two EPEC isolates belonged to serogroups O63 and O111.
TABLE 5.
Serotypes of the 15 tDEC isolates
| Organism category | No. of isolates | Serotypes (no. of isolates) |
|---|---|---|
| ETEC | 10 | O15:H11 (2), O15:H16 (1), O1:H6 (1), O25:NM (2), O159:H34 (1), O44:H18 (1), O6:H16 (1), O1:NM (1) |
| EPEC | 2 | O111:H9 (1), O63:H6 (1) |
| EIEC | 3 | O124:NM (1), O169:H9 (1), O63:H7 (1) |
DISCUSSION
Since it is not easy to distinguish DEC strains from the normal nonpathogenic flora due to the limitations of existing clinical diagnostic methods, the current incidence rates of infections caused by DEC have probably been largely underestimated. Due to heightened public concern and a limited number of complete epidemiological studies on DEC isolates in Taiwan, we have tried to set up a DEC reference laboratory at the Taiwan CDC. Over the past decade, various molecular detection assays based on virulence factors for the identification DEC strains have been developed and evaluated in different laboratories (3, 4, 12, 22, 27). It is also believed that the characteristics of several specific virulence genes are sufficient to be the basis of the identification of five categories of DEC strains. In this study, we have determined the incidence rate and serotypes of DEC isolates causing infections in northern Taiwan and successfully developed a multiplex real-time PCR assay to identify four categories of DEC strains by detecting six virulence genes. This useful assay is based on gene levels and provided reliable results in a short time. Its sensitivity and specificity were evaluated and proved to be as effective as those of assays described previously (5, 19). In addition, novel serotypes of DEC isolates were also found in this study.
Previous studies have reported that the identification of DEC isolates by serotyping may lead to false-positive results and that the reliability of serotyping is limited to certain strains or serogroups. We have thus evaluated the different identification results obtained between the real-time PCR and assays with commercial pathogenic O antisera. Real-time PCR tests were performed with a total of 137 sDEC isolates of the specific commercially defined O serogroups. Among the 137 isolates, 10, 2, and 3 were positive for the st, eae, and ipaH genes, respectively. ETEC (66.7%), EIEC (20%), and EPEC (13.3%) isolates were successfully identified. The tDEC isolation rate among the 261 specimens was 5.7%, and the virulence gene positivity rate for the 137 serogroup-defined isolates was 10.9%. The prevalence of these DEC strains in northern Taiwan could be compared with those determined in other studies (7, 16, 25). It also showed that many false-positive results might occur when only somatic antigens are used to characterize DEC strains. Although EHEC isolates, an important group of DEC strains that cause severe outbreaks, have been found to be members of many serogroups worldwide (10), especially serogroup O157 in Japan (20, 28), none of the clinical isolates in our study were found to be EHEC. In fact, only one imported case infected with EHEC serotype O157:H7 has been reported to the Taiwan CDC since 2001 (29). The rate of identification of human EHEC strains in Taiwan is particularly low compared with those in other Asian countries. Since it has been reported that the Shiga-like toxin-encoding plasmids or phages may be unstable (11), we cannot rule out the possibility that some of the isolates became negative for virulence genes during subculture or storage.
The 10 clinical ETEC isolates from this study belong to the following six O serogroups: O15 (n = 3 isolates), O1 (n = 2), O25 (n = 2), O159 (n = 1), O44 (n = 1), and O6 (n = 1). The respective virulence gene positivity rates for the strains of the six O serogroups identified above were 37.5% (3/8), 16.7% (2/12), 22.2% (2/9), 16.7% (1/6), 11.1% (1/9), and 14.3% (1/7), respectively. Compared with the percentage of ETEC strains identified in the study of Tamaki et al. (25), the incidences of two of the six O serogroups, serogroups O15 and O1, were significantly different. The rates of identification of ETEC strains of these two serogroups that we found were 37.5% and 16.7%, respectively, whereas the rates identified by Tamaki et al. (25) were 0% and 0%, respectively. However, the classification results of the remaining four O serogroups showed only slight differences. The rates of positivity for ETEC strains of serogroup O6, O25, O44, and O159 in our study were 14.3%, 22.2%, 11.1%, and 16.7%, respectively, while the corresponding rates were 8.1%, 12.5%, 13.3%, and 25% in the study of Tamaki et al. (25). The ETEC rate in our sDEC isolates was also similar to that in the study of Tamaki et al. (25): 7.3% (10/137) and 7.0% (79/1,130), respectively.
The World Health Organization has considered isolates in the following 12 O serogroups to be EPEC strains (7): O26, O55, O86, O111, O114, O119, O125, O126, O127, O128, O142, and O158. Other published reports have also proposed the similarly important O serogroups to be EPEC (13) and studied the EPEC positivity rates for these O serogroups (7, 25). Scotland et al. (23) also reported that a high percentage (62.3%) of strains of serogroups O26, O55, O111, and O128 in the United Kingdom were EPEC. Among the 23 sDEC isolates in our study, including strains of 7 of the 12 EPEC serogroups described above (serogroups O26, O55, O86a, O111, O114, O125, and O128), only 1 strain (serogroup O111) belonged to EPEC. The positivity rate was 4.4% (1/23), which is much lower than that reported previously (16.9%, 62%, and 18.6% in references 25, 7, and 24, respectively). We did not identify any isolates of serogroups O26, O55, and O128 as EPEC. This suggests that the rate of positivity of EPEC isolates in each of the 12 proposed O serogroups might vary between countries. Another EPEC isolate identified in this study turned out to belong to serogroup O63. The rates of EPEC isolates of serogroups O111 and O63 were 100% (one of one isolate) and 50% (one of two isolates), respectively. The observed result was similar to those reported previously that serogroup O111 is strongly associated with several DEC pathotypes (6, 8). In addition, the serotypes of two of the EPEC isolates in our study (serotypes O111:H9 and O63:H6) were both newly identified novel ones.
The three EIEC isolates that we identified were also compared with those described previously. The EIEC serogroup O124 strain in our study was the only one similar to what was reported by Levine (13), including serogroups O28ac, O29, O124, O136, O143, O144, O152, O164, and O167. The rate of positivity for EIEC isolates in this serogroup was 100% (one of one isolate), but this serogroup was not reported by either Tamaki et al. (25) (zero of five isolates) or Sunabe and Honma (zero of three isolates) (24). The other two serogroups, O63 and O169, have not been categorized as EIEC strains in the past. The rates of EIEC in these two serogroups were both 50% (one of two isolates) in our study. The findings also suggest that EIEC might have different serogroup patterns in various areas. Among the serotypes of the three isolates, O124:NM was the typical serogroup of EIEC strains reported previously. EIEC isolates of serogroups O63:H9 and O169:H9 have never been reported elsewhere and thus were also novel types.
According to our data, there are particularly high virulence gene carriage rates in some of the commercially defined O serogroups. The rates for three serogroups, serogroups O63, O111, and O124, were all 100%. So the identification results were conserved between the serotyping and real-time PCR methods for these three serogroups. The results reveal that the identification results by the serotyping method were reliable for serogroups O63, O111, and O124. The high rates of false-positive results may still exist for other serogroups when only the serotyping method is used to identify DEC strains. In addition, even for strains of the same O serogroup, different categories of DEC strains were observed. Due to the differences between the categorization results for the isolates belonging to the same O serogroup previously reported by others and in our studies, serotyping may be useful for the identification of DEC strains of limited serogroups (such as serogroups O63, O111, and O124) but not in classifying them into the five virulence categories.
In conclusion, the use of virulence gene-based assays is recommended for the detection of DEC strains, regardless of the serogrouping results. The real-time PCR assay that we have developed could simultaneously identify and classify DEC strains reliably. As a national laboratory in Taiwan, we believe that a complete epidemiological study of DEC strains is essential. The results obtained in this study are epidemiologically significant and useful for the prevention of DEC infections and the control of DEC strains in Taiwan. With the more timely characterization of DEC, the results might influence patient care in the future.
Acknowledgments
This work was supported by grants DOH95-DC-2027 and DOH96-DC-2011 from the Centers for Disease Control, Department of Health, Taiwan.
Footnotes
Published ahead of print on 29 August 2007.
REFERENCE
- 1.Alam, M., A. H. Nur, S. Ahsan, G. P. Pazhani, K. Tamura, T. Ramamurthy, D. J. Gomes, S. R. Rahman, A. Islam, F. Akhtar, S. Shinoda, H. Watanabe, S. M. Faruque, and G. B. Nair. 2006. Phenotypic and molecular characteristics of Escherichia coli isolated from aquatic environment of Bangladesh. Microbiol. Immunol. 50:359-370. [DOI] [PubMed] [Google Scholar]
- 2.Aranda, K. R., U. Fagundes-Neto, and I. C. Scaletsky. 2004. Evaluation of multiplex PCRs for diagnosis of infection with diarrheagenic Escherichia coli and Shigella spp. J. Clin. Microbiol. 42:5849-5853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bekal, S., R. Brousseau, L. Masson, G. Prefontaine, J. Fairbrother, and J. Harel. 2003. Rapid identification of Escherichia coli pathotypes by virulence gene detection with DNA microarrays. J. Clin. Microbiol. 41:2113-2125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bellin, T., M. Pulz, A. Matussek, H. G. Hempen, and F. Gunzer. 2001. Rapid detection of enterohemorrhagic Escherichia coli by real-time PCR with fluorescent hybridization probes. J. Clin. Microbiol. 39:370-374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bischoff, C., J. Luthy, M. Altwegg, and F. Baggi. 2005. Rapid detection of diarrheagenic E. coli by real-time PCR. J. Microbiol. Methods 61:335-341. [DOI] [PubMed] [Google Scholar]
- 6.Blanco, J., M. Blanco, J. E. Blanco, A. Mora, E. A. Gonzalez, M. I. Bernardez, M. P. Alonso, A. Coira, A. Rodriguez, J. Rey, J. M. Alonso, and M. A. Usera. 2003. Verotoxin-producing Escherichia coli in Spain: prevalence, serotypes, and virulence genes of O157:H7 and non-O157 VTEC in ruminants, raw beef products, and humans. Exp. Biol. Med. (Maywood) 228:345-351. [DOI] [PubMed] [Google Scholar]
- 7.Campos, L. C., M. R. Franzolin, and L. R. Trabulsi. 2004. Diarrheagenic Escherichia coli categories among the traditional enteropathogenic E. coli O serogroups—a review. Mem. Inst. Oswaldo Cruz 99:545-552. [DOI] [PubMed] [Google Scholar]
- 8.Campos, L. C., T. S. Whittam, T. A. Gomes, J. R. Andrade, and L. R. Trabulsi. 1994. Escherichia coli serogroup O111 includes several clones of diarrheagenic strains with different virulence properties. Infect. Immun. 62:3282-3288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Franzolin, M. R., R. C. Alves, R. Keller, T. A. Gomes, L. Beutin, M. L. Barreto, C. Milroy, A. Strina, H. Ribeiro, and L. R. Trabulsi. 2005. Prevalence of diarrheagenic Escherichia coli in children with diarrhea in Salvador, Bahia, Brazil. Mem. Inst. Oswaldo Cruz 100:359-363. [DOI] [PubMed] [Google Scholar]
- 10.Griffin, P. M., and R. V. Tauxe. 1991. The epidemiology of infections caused by Escherichia coli O157:H7, other enterohemorrhagic E. coli, and the associated hemolytic uremic syndrome. Epidemiol. Rev. 13:60-98. [DOI] [PubMed] [Google Scholar]
- 11.Karch, H., T. Meyer, H. Russmann, and J. Heesemann. 1992. Frequent loss of Shiga-like toxin genes in clinical isolates of Escherichia coli upon subcultivation. Infect. Immun. 60:3464-3467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kimata, K., T. Shima, M. Shimizu, D. Tanaka, J. Isobe, Y. Gyobu, M. Watahiki, and Y. Nagai. 2005. Rapid categorization of pathogenic Escherichia coli by multiplex PCR. Microbiol. Immunol. 49:485-492. [DOI] [PubMed] [Google Scholar]
- 13.Levine, M. M. 1987. Escherichia coli that cause diarrhea: enterotoxigenic, enteropathogenic, enteroinvasive, enterohemorrhagic, and enteroadherent. J. Infect. Dis. 155:377-389. [DOI] [PubMed] [Google Scholar]
- 14.Nataro, J. P., and J. B. Kaper. 1998. Diarrheagenic Escherichia coli. Clin. Microbiol. Rev. 11:142-201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Nguyen, T. V., P. Le Van, C. Le Huy, K. N. Gia, and A. Weintraub. 2005. Detection and characterization of diarrheagenic Escherichia coli from young children in Hanoi, Vietnam. J. Clin. Microbiol. 43:755-760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Nishikawa, Y., Z. Zhou, A. Hase, J. Ogasawara, T. Kitase, N. Abe, H. Nakamura, T. Wada, E. Ishii, and K. Haruki. 2002. Diarrheagenic Escherichia coli isolated from stools of sporadic cases of diarrheal illness in Osaka City, Japan between 1997 and 2000: prevalence of enteroaggregative E. coli heat-stable enterotoxin 1 gene-possessing E. coli. Jpn. J. Infect. Dis. 55:183-190. [PubMed] [Google Scholar]
- 17.Paciorek, J. 2002. Virulence properties of Escherichia coli fecal strains isolated in Poland from healthy children and strains belonging to serogroups O18, O26, O44, O86, O126 and O127 isolated from children with diarrhea. J. Med. Microbiol. 51:548-556. [DOI] [PubMed] [Google Scholar]
- 18.Reischl, U., M. T. Youssef, J. Kilwinski, N. Lehn, W. L. Zhang, H. Karch, and N. A. Strockbine. 2002. Real-time fluorescence PCR assays for detection and characterization of Shiga toxin, intimin, and enterohemolysin genes from Shiga toxin-producing Escherichia coli. J. Clin. Microbiol. 40:2555-2565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Reischl, U., M. T. Youssef, H. Wolf, E. Hyytia-Trees, and N. A. Strockbine. 2004. Real-time fluorescence PCR assays for detection and characterization of heat-labile I and heat-stable I enterotoxin genes from enterotoxigenic Escherichia coli. J. Clin. Microbiol. 42:4092-4100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sakuma, M., M. Urashima, and N. Okabe. 2006. Verocytotoxin-producing Escherichia coli, Japan, 1999-2004. Emerg. Infect. Dis. 12:323-325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Scaletsky, I. C., S. H. Fabbricotti, K. R. Aranda, M. B. Morais, and U. Fagundes-Neto. 2002. Comparison of DNA hybridization and PCR assays for detection of putative pathogenic enteroadherent Escherichia coli. J. Clin. Microbiol. 40:1254-1258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Scotland, S. M., H. R. Smith, T. Cheasty, B. Said, G. A. Willshaw, N. Stokes, and B. Rowe. 1996. Use of gene probes and adhesion tests to characterize Escherichia coli belonging to enteropathogenic serogroups isolated in the United Kingdom. J. Med. Microbiol. 44:438-443. [DOI] [PubMed] [Google Scholar]
- 23.Scotland, S. M., G. A. Willshaw, H. R. Smith, B. Said, N. Stokes, and B. Rowe. 1993. Virulence properties of Escherichia coli strains belonging to serogroups O26, O55, O111 and O128 isolated in the United Kingdom in 1991 from patients with diarrhea. Epidemiol. Infect. 111:429-438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Sunabe, T., and Y. Honma. 1998. Relationship between O-serogroup and presence of pathogenic factor genes in Escherichia coli. Microbiol. Immunol. 42:845-849. [DOI] [PubMed] [Google Scholar]
- 25.Tamaki, Y., H. Narimatsu, T. Miyazato, N. Nakasone, N. Higa, C. Toma, and M. Iwanaga. 2005. The relationship between O-antigens and pathogenic genes of diarrhea-associated Escherichia coli. Jpn. J. Infect. Dis. 58:65-69. [PubMed] [Google Scholar]
- 26.Teng, L. J., P. R. Hsueh, S. J. Liaw, S. W. Ho, and J. C. Tsai. 2004. Genetic detection of diarrheagenic Escherichia coli isolated from children with sporadic diarrhea. J. Microbiol. Immunol. Infect. 37:327-334. [PubMed] [Google Scholar]
- 27.Toma, C., Y. Lu, N. Higa, N. Nakasone, I. Chinen, A. Baschkier, M. Rivas, and M. Iwanaga. 2003. Multiplex PCR assay for identification of human diarrheagenic Escherichia coli. J. Clin. Microbiol. 41:2669-2671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Watanabe, Y., K. Ozasa, J. H. Mermin, P. M. Griffin, K. Masuda, S. Imashuku, and T. Sawada. 1999. Factory outbreak of Escherichia coli O157:H7 infection in Japan. Emerg. Infect. Dis. 5:424-428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wu, F. T., T. Y. Tsai, C. F. Hsu, T. M. Pan, H. Y. Chen, and I. J. Su. 2005. Isolation and identification of Escherichia coli O157:H7 in a Taiwanese patient with bloody diarrhea and acute renal failure. J. Formos. Med. Assoc. 104:206-209. [PubMed] [Google Scholar]


