Abstract
West Nile virus (WNV) is a neurotropic flavivirus that causes encephalitis, most frequently in elderly and immunocompromised humans. Previous studies demonstrated that CD8+ T cells utilize perforin-dependent cytolytic mechanisms to limit WNV infection. Nonetheless, the phenotype of perforin-deficient CD8+ T cells was not as severe as that of an absence of CD8+ T cells, suggesting additional effector control mechanisms. In this study, we evaluated the contribution of Fas-Fas ligand (FasL) interactions to CD8+ T-cell-mediated control of WNV infection. Notably, the cell death receptor Fas was strongly upregulated on neurons in culture and in vivo after WNV infection. gld mice that were functionally deficient in FasL expression showed increased susceptibility to lethal WNV infection. Although antigen-specific priming of CD8+ T cells in peripheral lymphoid tissues was normal in gld mice, increased central nervous system (CNS) viral burdens and delayed clearance were observed. Moreover, the adoptive transfer of WNV-primed wild-type but not gld CD8+ T cells to recipient CD8−/− or gld mice efficiently limited infection in the CNS and enhanced survival rates. Overall, our data suggest that CD8+ T cells also utilize FasL effector mechanisms to contain WNV infection in Fas-expressing neurons in the CNS.
West Nile virus (WNV) is a mosquito-borne neurotropic flavivirus that is closely related to other viruses that cause significant human disease, including dengue virus, yellow fever virus, and Japanese, St. Louis, and tick-borne encephalitis viruses. WNV cycles enzootically between wild birds and mosquitoes but also causes neuroinvasive disease in humans, horses, and other vertebrate animals (12, 30). Most humans with symptomatic infections develop a mild febrile illness, but a subset of cases progress to meningitis, encephalitis, or an acute flaccid paralysis syndrome (38, 39). Severe WNV infection occurs more frequently in elderly and immunocompromised persons and in individuals homozygous for the CCR5Δ32 mutation than in members of the general population (5, 9, 26).
Studies with rodents have elucidated how the maturation and integrity of the immune system is required for resistance to WNV infection (reviewed in references 37 and 50). Initial WNV replication is believed to occur in subsets of skin dendritic cells after mosquito inoculation or subcutaneous infection (2, 13, 14). These infected cells migrate to draining lymph nodes, where viral amplification occurs, resulting in primary viremia and dissemination to other peripheral tissues (7). After several rounds of multiplication, WNV accumulates in blood and, through an uncertain mechanism that may involve inflammatory cytokines such as tumor necrosis factor alpha, crosses the blood-brain barrier to infect and injure parenchymal neurons in the central nervous system (CNS) (42, 51, 54).
CD8+ T cells have crucial functions in controlling acute infection by some neurotropic viruses (1, 41, 46, 52). In general, CD8+ T cells exert antiviral functions via two basic mechanisms: a noncytolytic pathway through the secretion of antiviral cytokines such as gamma interferon (IFN-γ) and tumor necrosis factor alpha and a cytolytic pathway through the use of perforin-granzyme molecules or Fas-FasL interactions (11, 15, 17, 25, 32, 35, 40). Interactions between Fas (CD95) on target infected cells and FasL (CD95L) on effector T and natural killer cells leads to cytolysis via the activation of a death domain and a caspase apoptosis cascade (19, 27, 40). Defects in either the perforin or Fas-FasL cytolytic pathway can adversely affect the control of several CNS viruses, including lymphocytic choriomeningitis, mouse hepatitis, and Theiler's viruses (17, 29, 34).
For WNV, a genetic or acquired deficiency of CD8+ T cells in mice results in increased mortality, uncontrolled infection, and viral persistence in the CNS (41, 52). Studies with perforin−/− mice infected with a virulent North American WNV isolate also showed increased CNS infection and mortality, although the phenotype was not as severe as that observed with the depletion or a deficiency of CD8+ T cells (43). In contrast, the infection of perforin-, Fas-, or FasL-deficient mice with the less virulent lineage II Sarafend strains of WNV had little significant effect on viral burden or mortality compared to that in wild-type mice (53). Because the adoptive transfer of IFN-γ−/− CD8+ T cells effectively restricts WNV infection (43, 44), we speculated that an alternate cytolytic mechanism contributes to the CD8+ T-cell-mediated control of CNS infection with virulent North American WNV isolates.
In this study, we directly assessed the contribution of Fas-Fas ligand (FasL) interactions to the CD8+ T-cell-mediated control of infection with a virulent, lineage I North American WNV strain. Although at the baseline, CNS neurons lack Fas expression, infection with WNV rapidly induced expression. Mice that were functionally deficient in FasL were susceptible to lethal WNV infection with increased viral burdens and persistence in the CNS. Moreover, adoptive transfer experiments showed that CD8+ T cells require functional FasL to limit WNV infection in the CNS. Combining these data with our previous results, we conclude that CD8+ T cells use independent cytolytic effector mechanisms to combat virulent WNV infection in the CNS.
MATERIALS AND METHODS
Viruses and cells.
The WNV strain (3000.0259) isolated in New York (2000) was passaged once in C6/36 cells as a stock virus as described previously (7). Baby hamster kidney fibroblast (BHK21) cells were used for determining WNV burdens in infected tissues by plaque assays as described previously (6).
Mouse experiments.
C57BL/6 strain (H-2b) inbred wild-type mice were obtained from Jackson Laboratory (Bar Harbor, ME). The congenic CD8−/− mice and gld mice (defective in the expression of functional FasL [48]) were obtained as a gift (T. Ley, Washington University School of Medicine). All experiments were performed in the animal facilities with the approval and under the guidelines of the Washington University Animal Studies Committee. Eight- to 10-week-old mice were inoculated subcutaneously via the footpad with 102 PFU of WNV per mouse in 1% heat-inactivated fetal bovine serum in Hanks balanced salt solution.
Tissue preparation and quantitation of virus growth.
For the analysis of virus growth in different tissues of infected mice, groups of wild-type and gld mice were infected with 102 PFU of WNV through the footpad and euthanized on day 2, 4, 6, 8, or 10 after infection. Mice were perfused with 10 ml of phosphate-buffered saline (PBS), organs were removed and homogenized using a BeadBeater apparatus, and virus titers were determined on BHK21 cells as previously described (7). To assess WNV persistence in the CNS, separate groups of mice were infected with 102 PFU of WNV. Brains and spinal cords were harvested from surviving mice on days 14, 21, 28, and 35 after infection, and viral burdens in the CNS tissues were analyzed by plaque assays. For pathological analysis, brains were harvested from infected mice after perfusion at day 10. Brains were fixed in 4% paraformaldehyde for 24 h at 4°C and embedded in paraffin as described previously (42).
Fas staining by immunofluorescence. (i) Brain sections.
Brain sections were costained for WNV and Fas antigens. Briefly, paraffin-embedded brain sections were dewaxed and rehydrated in PBS. Antigen retrieval was performed by incubating sections with 1% sodium dodecyl sulfate in Tris buffer solution (100 mM Tris, 138 mM NaCl, and 27 mM KCl, pH 7.4) for 5 min at room temperature. After rinsing sections with Tris buffer solution and incubating with blocking solution (DAKO, Carpinteria, CA), 10 μg/ml of anti-Fas antibody (BD Transduction Laboratories, Lexington, KY) was added for 1 h at room temperature. After additional rinsing, sections were costained with WNV-immune rat serum (1:100 dilution) for 1 h at room temperature. Fluorescence signals were detected after incubation with Alexa 488 (Molecular Probes Inc., Eugene, OR)- and Cy3 (Jackson Laboratories, West Grove, PA)-conjugated secondary antibodies. Sections were visualized by fluorescence microscopy with an Axiovert 200 microscope (Zeiss, Oberkochen, Germany) after counterstaining with 4′,6′-diamidino-2-phenylindole (DAPI).
(ii) Primary neurons.
Cortical neurons were generated as described previously (18). Briefly, embryos were harvested on day 15 of gestation and the brains were removed and placed in a 35-mm dish with Dulbecco's modified Eagle medium on ice. The cortical hemispheres were separated, placed in a 15-ml tube with Dulbecco's modified Eagle medium, and centrifuged at 300 rpm for 5 min at 4°C (Sorvall Legend). The cells were dissociated with 0.25% trypsin (Sigma Chemical, St Louis, MO) and 300 μl of DNase I (Invitrogen, Carlsbad, CA). After washing, 6 × 105 cells per well were plated onto poly-d-lysine and laminin (BD Biosciences, San Diego, CA)-coated coverslips in a 24-well plate and incubated at 37°C. After 24 h, the culture was fed with fresh Neurobasal medium supplemented with B27 (Invitrogen, Carlsbad, CA) for the enrichment of the neurons. Three days later, cortical neurons were infected with WNV at a multiplicity of infection (MOI) of 1. One day later, cells were fixed with 4% paraformaldehyde in PBS at 4°C for 15 min. After several washes in PBS and blocking in 5% normal goat serum in PBS, neurons were costained for Fas expression and WNV antigen as described above.
Adoptive transfer of wild-type or gld CD8+ T cells into CD8−/− or gld mice.
CD8+ T cells from WNV-infected mice were purified and transferred as described previously (43). Briefly, splenocytes were harvested from naïve or primed wild-type or gld mice on day 7 after WNV infection. CD8+ T cells were purified by negative selection using antibody-coated magnetic beads (Miltenyi Biotec, Auburn, CA) and transferred into CD8−/− mice (107 CD8+ T cells) or gld mice (3 × 106 CD8+ T cells) 1 day after WNV infection. Mice were either monitored for 28 days for survival or sacrificed on day 10 after initial infection for viral burden analysis.
Intracellular IFN-γ staining.
Intracellular IFN-γ staining of splenocytes was performed as described previously (31, 45). Briefly, splenocytes were harvested from wild-type and gld mice on day 7 after infection. After erythrocyte lysis, 106 splenocytes were stimulated with 0.2 μg/ml of an immunodominant Db-restricted WNV-specific NS4B peptide (SSVWNATTAI) (31) or 50 ng/ml phorbol myristate acid (PMA; Sigma Chemical) and 500 ng/ml ionomycin (Sigma Chemical) for 4 h at 37°C in the presence of Golgi plug (BD Biosciences). Splenocytes were cooled to 4°C and incubated with fluorescein isothiocyanate-conjugated CD8 or an isotype control antibody (BD Biosciences) for 30 min at 4°C. After washing in PBS containing 5% goat serum, splenocytes were fixed with 1% paraformaldehyde in PBS, permeabilized with saponin, and stained with an allophycocyanin-conjugated anti-IFN-γ antibody or an isotype control antibody (BD Biosciences) for 30 min at 4°C. After a final series of washes, cells were assessed by flow cytometry and the percentage of CD8+ T cells that expressed IFN-γ was determined using CellQuest software (BD Biosciences).
Addition of CD8+ T cells to WNV-infected neurons.
The incubation of purified CD8+ T cells with WNV-infected neurons was performed as described previously (43). Briefly, cortical neurons were infected at an MOI of 0.001. One hour after infection, unbound virus was removed with four washes in warm medium. Subsequently, purified naïve or WNV-primed CD8+ T cells from wild-type or gld mice were added at effector/target (E/T) ratios of 50:1 and 10:1. Supernatants were harvested at 48 h after infection, and the reduction in WNV production was measured by a viral plaque assay.
Data analysis.
All data were analyzed statistically using Prism software (GraphPad, San Diego, CA). Kaplan-Meier survival curves were analyzed by the log rank test. Differences in viral burdens in mice were analyzed by the Mann-Whitney test. Differences in viral burdens in neuron cultures were analyzed by an unpaired t test.
RESULTS
WNV induces expression of Fas in neurons.
Because prior studies had suggested a role for perforin-independent control of WNV infection in neurons by CD8+ T cells, we hypothesized that neurons may induce Fas expression to levels that would allow recognition and targeting by T cells. The engagement of Fas by FasL on effector cells activates the extrinsic pathway of apoptosis, culminating in caspase-3 activation and endonucleolytic cell death (3, 19, 28). Although Fas is normally expressed at low levels, if at all, on neurons, during physiologic stress its expression can be induced (20, 23, 24, 27).
We evaluated the expression of Fas on neurons in vitro and in vivo before and after WNV infection (Fig. 1). Primary cortical neurons from C57BL/6 mice were generated, infected with WNV for 24 h, and stained for Fas expression. Although uninfected neurons did not express Fas (Fig. 1A, panels b and d), almost all WNV-infected neurons expressed Fas at 24 h after infection (Fig. 1A, panels e, f, and h). To confirm that WNV induced Fas expression on neurons in vivo, brains were harvested from wild-type mice on day 10 after infection and costained for Fas and WNV antigen. Similar to those in our primary neuron cultures, the CNS neurons from uninfected mice did not express Fas (Fig. 1B, panels j and l, and Table 1). In contrast, Fas expression was induced in vivo on a large percentage (80%) of WNV-infected neurons in the cerebral cortex (Fig. 1B, panels m, n, and p, and Table 1). Fas expression was also found in WNV-infected neurons in the hippocampus and brain stem (data not shown). However, in the brains of infected mice, Fas was not induced in neurons that stained negatively for WNV antigen (Fig. 1B, panels q, r, and t). Thus, WNV infection rapidly induces Fas expression on different neuron populations in the CNS.
FIG. 1.
Fas expression in neurons after WNV infection. (A) Fas expression in WNV-infected cortical neurons. Cortical neurons were infected with WNV (MOI of 1), and 1 day later, neurons were stained for Fas (green) and WNV antigen (red). Uninfected neurons did not express Fas (upper panels a to d), whereas infected neurons did (lower panels e to h). p.i., postinfection. (B) Fas expression in brain sections. Brain tissue from wild-type mice was harvested on day 10 after infection with 102 PFU of WNV, sectioned, and stained for Fas expression. Neurons from uninfected mice were negative for Fas (upper panels i to l), whereas WNV-infected neurons stained positively for Fas expression (middle panels m to p). In WNV-infected mice, regions of the brain that stained negatively for WNV lacked the induction of Fas (lower panels q to t). Typical sections from cortex tissue selected after the review of more than five different brains are shown. WNV antigen and Fas expression are indicated by red and green fluorescence, respectively. DAPI was used as a nuclear stain.
TABLE 1.
Fas expression in neurons in the brains of wild-type micea
| Condition | No. of WNV+ neurons/high-power field | No. of Fas+ neurons/high-power field | % of WNV+ Fas+ neurons |
|---|---|---|---|
| Uninfected | <1 | <1 | <1 |
| Infected | 29 ± 16 | 23 ± 14 | 79 ± 18 |
Wild-type mice were infected with 102 PFU of WNV by footpad inoculation. Brains were harvested at day 10 after infection, sectioned, and costained for WNV and Fas antigens. Numbers of WNV+ and Fas+ neurons per high-power field (magnification, ×400) were determined by fluorescence microscopy. The percentage of WNV+ Fas+ neurons was calculated by dividing the number of WNV+ Fas+ neurons by the total number of WNV+ neurons. Fas was expressed only in neurons that were infected with WNV, although not all WNV-infected neurons expressed Fas. The results were generated after evaluating 12 different fields from three to five different mice per group. Values for infected neurons are means ± standard deviations.
Functional FasL is required to protect mice from lethal WNV infection.
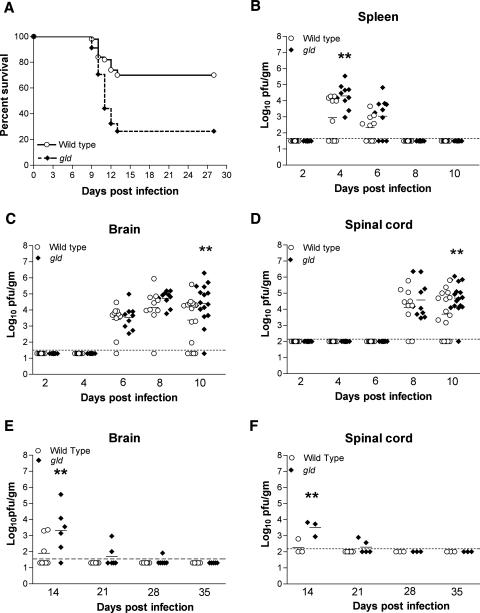
To directly assess the significance of Fas-FasL interactions in the control of WNV infection, we compared the survival rates of wild-type and congenic gld mice after subcutaneous infection with 102 PFU of WNV. gld mice contain a point mutation in the FasL gene, rendering it nonfunctional (48). By day 8 after WNV infection, all mice showed clinical signs of infection, including reduced activity, weight loss, and hair ruffling. However, survival rates of gld mice were markedly lower, 26% compared to 70% for congenic wild-type mice (Fig. 2A) (P < 0.0001), although there was no difference in the mean time to death (wild-type mice, 10.9 ± 0.3 days; gld mice, 10.8 ± 0.2 days). Mortality analysis showed that an absence of functional FasL caused more severe WNV infection with an adverse clinical outcome.
FIG. 2.
Survival and viral titer analysis for wild-type and gld mice infected with WNV. (A) Kaplan-Meier survival curves. Wild-type (n = 50) and gld (n = 34) mice were infected with 102 PFU of WNV and monitored for mortality for 28 days. Survival differences as judged by the log rank test were statistically significant (P < 0.0001). (B to E) WNV tissue burdens in wild-type and gld mice. Infectious WNV levels in tissues from spleens (B), brains (C and E), and spinal cords (D and F) of wild-type and gld mice were measured using a viral plaque assay on BHK21 cells after tissues were harvested at the indicated time points. Data are expressed as log numbers of PFU per gram of tissue and reflect results for 10 to 12 mice per time point between days 2 and 10 and 3 to 8 mice per time point between days 14 and 35. For viral burden experiments, the dotted line represents the limit of sensitivity of viral detection and asterisks indicate statistically significant (P < 0.05) differences between wild-type and gld mice as judged by the Mann-Whitney test.
WNV burdens are increased in gld mice.
Previous studies showed that the high mortality of CD8−/− mice after WNV infection was due to increased peripheral and CNS viral burdens (41). To understand how an absence of FasL made mice more susceptible to WNV infection, the levels of infectious virus in tissues were measured by viral plaque assays. Wild-type and gld mice were infected with 102 PFU, and the viral loads were quantitated on days 2, 4, 6, 8, and 10 after infection.
(i) Peripheral tissues.
We observed no difference in the kinetics and magnitude of viremia between wild-type and gld mice (data not shown). Infectious virus was detected in the spleens of both groups 4 days after infection. However, only 60% of wild-type mice (6 of 10) had levels above the limit of sensitivity. In contrast, 100% of gld mice (10 of 10) had measurable viral loads (∼103 to 105 PFU/g) in the spleen (Fig. 2B) (P = 0.01). By day 6, the difference in WNV titers was no longer evident, as the percentages of animals with splenic infection and the average titers were similar (P > 0.2). By day 8, WNV infection had been effectively cleared from both wild-type and gld mice. Interestingly, these results contrast with prior data from CD8−/− and perforin−/− mice, which failed to efficiently clear viral infection from the spleen (41, 43). Thus, Fas-FasL interactions apparently are less necessary for the clearance of WNV infection from peripheral lymphoid tissues.
(ii) CNS tissues.
Similar levels of WNV (∼104 to 105 PFU/g) were detected in the brains of gld and wild-type mice through day 8 after infection. However, by day 10, the WNV burden in gld mice was on average 10-fold higher (104.4 compared to 103.4 PFU/g; P = 0.04) (Fig. 2C). Analogous results for the spinal cords were observed, with similar viral burdens through day 8 but higher levels in gld mice on day 10 (104.7 compared to 103.7 PFU/g; P = 0.02) (Fig. 2D). Because of the higher viral titers in the CNS tissues of gld mice on day 10 after infection, we evaluated whether there was a difference in the susceptibilities of different neuronal populations to WNV infection. WNV antigen was detected only in neurons, with the cerebral cortex, brain stem, and hippocampus as principal sites of infection in the brain (data not shown). These virologic experiments suggest that FasL has an essential role in controlling WNV infection in the CNS.
Clearance of WNV from the CNS is delayed in gld mice.
A lack of CD8+ T cells or perforin granules resulted in WNV persistence in the CNS up to 5 weeks after infection (41, 43). To assess the effect of FasL on the kinetics of viral clearance from the CNS, infectious virus levels in the surviving wild-type and gld mice were measured (Fig. 2E and F). Low levels (102 to 103 PFU/g) of infectious virus were detected on day 14 in the CNS tissues of ∼35% of wild-type mice (three of eight brains and one of three spinal cords), and no infectious virus was recovered from the CNS at 21 days or after. Higher levels (∼102 to 105 PFU/g) were detected in the CNS tissues of gld mice on days 14 (five of six brains and three of three spinal cords) and 21 (two of five brains and two of five spinal cords). Nonetheless, by day 28, only one of six gld mice had detectable CNS infection, and at later time points, infectious WNV was not recovered. Thus, an absence of functional FasL delayed the clearance of WNV infection from the CNS.
CD8+ T-cell priming is not impaired in gld mice.
Based on the increased mortality, elevated viral burdens, and delayed clearance in gld mice, we hypothesized that FasL contributed to the CD8+ T-cell mediated control of WNV infection in the brain and spinal cord. However, because gld mice have some abnormalities in T-cell development due to the absence of precursor T-cell apoptosis at specific stages (47), we confirmed that CD8+ T-cell priming occurred normally. The levels of IFN-γ-secreting CD8+ T cells from gld and wild-type mice were compared after ex vivo restimulation with an immunodominant Db-restricted WNV peptide (31) or with a nonspecific agonist, phorbol ester and ionomycin. As expected, mock-infected wild-type or gld mice showed no or low levels of intracellular IFN-γ staining in CD8+ T cells after restimulation with the NS4B peptide or PMA and ionomycin, respectively (Fig. 3A). In contrast, on day 7 we observed a significant increase in IFN-γ production by splenic CD8+ T cells from both wild-type and gld mice after antigen-specific or nonspecific reactivation (P ≤ 0.02), suggesting a lack of requirement for functional Fas ligand in the initial priming of WNV-specific CD8+ T cells. There was no significant difference in the percentages of IFN-γ+ CD8+ T cells from wild-type and gld mice after exposure to a WNV-specific peptide (P = 0.5), although there was a twofold increase in the absolute number of IFN-γ+ CD8+ T cells from gld mice after treatment with PMA and ionomycin (Fig. 3B and C) (P = 0.01). This difference was consistent with the increased number of splenic CD8+ T cells in naïve gld mice compared to that in wild-type mice at this stage (data not shown). Overall, these experiments demonstrate that the lack of functional Fas ligand did not impair in vivo priming of CD8+ T cells after WNV infection.
FIG. 3.
IFN-γ production by WNV-primed CD8+ T cells from wild-type (WT) and gld mice. Uninfected or WNV-infected splenocytes from wild-type or gld mice were harvested on day 7 and stimulated ex vivo with an immunodominant Db-restricted NS4B peptide (SSVWNATTAI) or phorbol ester and ionomycin for 4 h. Cells were costained for CD8 and IFN-γ and analyzed by flow cytometry. (A) Representative flow cytometry profiles of intracellular IFN-γ staining in the absence of the peptide (upper panels) or in the presence of the peptide (middle panels) or phorbol ester (PMA) and ionomycin (bottom panels). The value in the top right corner indicates the percentage of CD8+ T cells that expressed IFN-γ after restimulation. (B and C) Total numbers of IFN-γ-producing CD8+ T cells after stimulation with NS4B peptides (B) or phorbol ester and ionomycin (C) were calculated. Each symbol represents data from an individual mouse, and lines indicate mean values. The numbers of IFN-γ+ CD8+ T cells in wild-type mice and those in gld mice after antigen-specific stimulation were not statistically different (P > 0.4), although there was a small increase (P = 0.01; asterisks) in the numbers of IFN-γ+ CD8+ T cells in gld mice after phorbol ester and ionomycin stimulation.
Adoptive transfer of primed CD8+ T cells increases survival.
Although a deficiency of Fas ligand was associated with increased CNS viral burden and mortality, other immune cells (e.g., CD4+ T or NK cells) may possibly use Fas-FasL interactions to promote the killing of virus-infected target cells. To establish a direct role for FasL in the CD8+ T-cell-mediated control of WNV infection, adoptive transfer studies were performed with primed CD8+ T cells harvested from WNV-infected wild-type or gld mice on day 7 after purification (∼95%) by negative selection using antibody-coated magnetic beads (data not shown). CD8+ T cells (107) were adoptively transferred into congenic CD8−/− mice 1 day after WNV infection, and survival was evaluated. Although both wild-type and gld primed CD8+ T cells protected CD8−/− mice (Fig. 4A) (P < 0.0001), we observed a trend towards decreased protection with the gld cells (67% compared to 93% survival rate; P = 0.1). As an independent demonstration of the differential effect of FasL on CD8+ T-cell effector function, we transferred 3 × 106 WNV-primed or naïve wild-type or gld CD8+ T cells into gld mice 1 day after infection and evaluated survival (Table 2). The transfer of naïve wild-type or gld CD8+ T cells did not protect gld mice (20 to 33% survival rate; P > 0.5). In contrast, the transfer of primed wild-type CD8+ T cells protected 80% of gld mice from lethal WNV infection, whereas the transfer of primed gld CD8+ T cells protected only 57% of gld mice. Together, these experiments suggest that CD8+ T cells require FasL for the optimal control of lethal WNV infection.
FIG. 4.
Survival rates and viral burdens after adoptive transfer of WNV-primed CD8+ T cells. CD8+ T cells were purified from naïve or WNV-primed wild-type and gld mice and transferred into either CD8−/− or gld mice 1 day after infection. (A) Survival curves for CD8−/− mice after the adoptive transfer of 107 primed wild-type or gld CD8+ T cells. Differences in the survival curves for CD8−/− mice that received no T cells and for CD8−/− mice that received primed wild-type or gld cells were statistically significant (P ≤ 0.001). The number of mice in each group was 10 to 12. (B and C) Viral burden analysis of spleen, brain, and spinal cord tissues of recipient CD8−/− mice on day 10 after the adoptive transfer of 107 (B) or 3 × 106 (C) naïve or primed wild-type or gld CD8+ T cells.
TABLE 2.
Survival after adoptive transfer of CD8+ T cells into gld micea
| Inoculum | No. of surviving mice/total no. in group | % Survival | P value |
|---|---|---|---|
| No T cells | 9/34 | 26 | |
| Naïve wild-type CD8+ T cells | 2/6 | 33 | 0.57 |
| Naïve gld CD8+ T cells | 1/5 | 20 | 0.79 |
| Primed wild-type CD8+ T cells | 8/10 | 80 | 0.003** |
| Primed gld CD8+ T cells | 8/14 | 57 | 0.02** |
Purified naïve or WNV-primed CD8+ T cells (3 × 106) from wild-type or gld mice were adoptively transferred into gld mice intraperitoneally at 24 h postinfection. Mice were monitored for survival for 28 days after infection. P values were calculated by the log rank test. Asterisks indicate values that are statistically significant.
To assess the virologic basis for the differential protection by wild-type and gld CD8+ T cells, spleen and CNS tissues were harvested from recipient CD8−/− mice on day 9 after the adoptive transfer (Fig. 4B). The adoptive transfer of 107 naïve wild-type CD8+ T cells showed no control of infection, as high levels of WNV were present in the spleens, brains, and spinal cords of all recipient CD8−/− mice. In contrast, the transfer of 107 WNV-primed wild-type CD8+ T cells completely cleared WNV infection from the spleens and CNS tissues. Although the transfer of primed gld CD8+ T cells was sufficient to clear virus from the spleens, WNV was detected in the brains and spinal cords of 67% of recipient mice (two of three). However, the viral burdens in CD8−/− mice receiving gld CD8+ T cells were lower than those in mice receiving naïve wild-type cells (∼104 PFU/g versus 106.6 PFU/g; P = 0.03).
Although the transfer of 107 WNV-primed wild-type but not gld CD8+ T cells resulted in no detectable virus on day 10 after infection, we were concerned that this high dose of primed T cells effectively aborted the infection in peripheral tissues in some of the animals so that WNV may never have disseminated into the brains. To clarify further the role of gld on CD8+ T-cell clearance from the CNS, fewer (3 × 106) WNV-primed CD8+ T cells were also transferred (Fig. 4C). By day 9 after the transfer of wild-type cells, WNV was cleared completely from the spleens but not from the CNS tissues of recipient CD8−/− mice, as 67% (two of three) had reduced but measurable levels (∼105 PFU/g) in the brains and spinal cords. The transfer of 3 × 106 primed gld CD8+ T cells controlled WNV less efficiently, with persistent infection in the spleens of two of three mice (∼103 to 104 PFU/g) and higher viral titers in the CNS tissues that approached those in CD8−/− mice (105 to 107 PFU/g). Taken together, the adoptive transfer experiments demonstrate that a deficiency of functional FasL on CD8+ T cells impairs the clearance of WNV infection from both the spleen and CNS.
Addition of wild-type and gld CD8+ T cells to WNV-infected neurons.
To demonstrate directly that CD8+ T cells use FasL to control WNV-infected neurons, we performed a viral clearance assay with mouse primary cortical neurons. One hour after infection, WNV-primed wild-type or gld CD8+ T cells were added to neurons at two different E/T ratios. At 48 h, the level of infectious virus in the supernatants was measured by plaque assays. WNV-primed wild-type CD8+ T cells significantly reduced (P ≤ 0.001) infectious virus yields from cortical neurons at E/T ratios of both 50:1 (∼260-fold reduction) and 10:1 (∼33-fold reduction) compared to yields from neurons with naïve CD8+ T cells (Fig. 5). Although WNV-primed gld CD8+ T cells also reduced infectious virus production (P ≤ 0.005) compared to that in cultures of neurons with naïve CD8+ T cells, they were less efficient (18-fold reduction; P = 0.03) than primed wild-type CD8+ T cells at the higher E/T ratio. However, at the lower E/T ratio, no significant difference (P = 0.14) in virus production in cultures with primed wild-type CD8+ T cells and those with gld CD8+ T cells was observed. Overall, these experiments suggest that WNV-primed CD8+ T cells can control neuronal infection in vitro, in part through FasL-dependent mechanisms.
FIG. 5.
Control of WNV infection in primary neurons by primed CD8+ T cells. Cortical neurons were infected with WNV at an MOI of 0.001. After 1 h, purified naïve or WNV-primed CD8+ T cells from wild-type (WT) and gld mice were added at an E/T ratio of 50:1 or 10:1. After 48 h, supernatants were harvested and WNV production was measured by a plaque assay. Statistically significant reductions in WNV production compared to that in cultures of neurons without the addition of T cells are indicated by asterisks (**, P ≤ 0.005; *, P ≤ 0.05), and ns indicates that differences were not statistically significant.
DISCUSSION
Previous studies have shown that CD8+ T cells utilize perforin to clear WNV from infected neurons (41, 43). However, the phenotypes of perforin−/− and CD8−/− mice were distinct, and thus, additional CD8+ T-cell-dependent clearance mechanisms were hypothesized. In this study, we tested the contribution of the Fas-FasL recognition pathway to CD8+ T-cell-mediated control of WNV infection. gld mice that lack functional expression of FasL had elevated viral titers in the CNS tissues resulting in increased mortality compared to that among wild-type mice. In the few gld mice that survived infection, persistent WNV infection was detected for several weeks. Adoptive transfer studies suggested that the increased susceptibility associated with functional FasL deficiency was due to defects in CD8+ T-cell clearance mechanisms. Nonetheless, the viral burdens and survival phenotypes in gld mice were more subtle than those observed previously in perforin−/− and CD8−/− mice (41, 43). Thus, while CD8+ T cells utilize multiple effector mechanisms to clear WNV from neurons in the CNS, perforin-mediated control appears to be more dominant than the Fas-FasL pathway.
Our results showing increased lethality and CNS viral burdens in gld mice infected with a virulent lineage I North American WNV isolate contrast with those of a prior study that used the less virulent lineage II Sarafend WNV strain (53). In that report, similar levels of mortality and of viral infection in the brains in gld and Fas−/− mice are described. An independent publication from the same group stated that gld mice showed paradoxically delayed and reduced mortality after infection with the closely related Murray Valley encephalitis virus (21). In other nonflavivirus viral disease models, including those with a neurotropic strain of mouse hepatitis virus, influenza virus, Semliki Forest virus, and lymphocytic choriomeningitis virus, an absence of Fas-FasL interaction, by itself, had little effect on viral burden (16, 29, 49). However, an increased-susceptibility phenotype was observed with mouse hepatitis virus if both Fas-FasL and perforin functions were absent. One possible explanation for the disparity in results among models may be the differences in virulence of the individual viruses. The cytopathic potential of a virus in the CNS may define whether cytolytic T-cell responses are beneficial or pathogenic. For a highly neurovirulent virus (e.g., New York WNV), cytolytic T-cell clearance that limits CNS dissemination may outweigh the pathological effects of immune system-targeted neuronal injury.
FasL is normally expressed and upregulated on activated T cells and natural killer cells. FasL binding to Fas may have direct antiviral effects by inducing apoptosis of infected target cells (27). In our experiments, a deficiency of FasL did not dramatically alter antigen-specific CD8+ T-cell priming or the kinetics of clearance of WNV from the spleens of gld mice. In contrast, efficient clearance of WNV from the spleen requires CD8+ T cells and perforin molecules (43). Although Fas-FasL interactions had a subordinate effect on splenic clearance, our infection experiments with gld mice and our adoptive transfer studies with primed gld CD8+ T cells demonstrate an essential role for Fas-FasL interactions in the clearance of WNV infection from the CNS. Thus, FasL interactions may have a slightly more tissue-restricted effect against WNV than perforin.
FasL-mediated control of WNV in the CNS by CD8+ T cells requires neuronal expression of Fas. Basally, Fas is not expressed on neurons, but it can be upregulated by inflammatory stimuli (8, 10, 22, 24) or viral infection (23, 24, 36). Indeed, the functional significance of this finding was suggested by prior studies with reovirus showing that apoptosis of cortical neurons was inhibited by soluble Fas molecules (4, 33). Our experiments show directly the induction of Fas on WNV-infected cortical neurons in culture and in vivo. Although the mechanism of induction remains unexplored, some have suggested that Fas expression on neurons can be stimulated in vitro after treatment with IFN-γ (23, 25). Experiments with the adoptive transfer of WNV-primed gld or wild-type CD8+ T cells into IFN-γ receptor−/− mice are planned to test this hypothesis directly.
In summary, our study demonstrates that CD8+ T cells require functional FasL to optimally control WNV infection in the CNS. CD8+ T cells utilize multiple effector mechanisms, including Fas-FasL interactions, to control WNV infection and the injury of neurons. A more complete understanding of the effector mechanisms responsible for the T-cell clearance of WNV infection may inform novel vaccine strategies that stimulate robust cell-mediated immune responses against highly virulent CNS viruses.
Acknowledgments
We thank T. Ley for the gld and CD8−/− mice and T. Hansen for critical comments.
The work was supported by funds from Washington University School of Medicine and the NIH (U54 AI057160 to the Midwest Regional Center of Excellence for Biodefense and Emerging Infectious Diseases Research).
Footnotes
Published ahead of print on 5 September 2007.
REFERENCES
- 1.Bergmann, C. C., N. W. Marten, D. R. Hinton, B. Parra, and S. A. Stohlman. 2001. CD8 T cell mediated immunity to neurotropic MHV infection. Adv. Exp. Med. Biol. 494:299-308. [DOI] [PubMed] [Google Scholar]
- 2.Byrne, S. N., G. M. Halliday, L. J. Johnston, and N. J. King. 2001. Interleukin-1beta but not tumor necrosis factor is involved in West Nile virus-induced Langerhans cell migration from the skin in C57BL/6 mice. J. Investig. Dermatol. 117:702-709. [DOI] [PubMed] [Google Scholar]
- 3.Choi, C., and E. N. Benveniste. 2004. Fas ligand/Fas system in the brain: regulator of immune and apoptotic responses. Brain Res. Brain Res. Rev. 44:65-81. [DOI] [PubMed] [Google Scholar]
- 4.Clarke, P., and K. L. Tyler. 2003. Reovirus-induced apoptosis: a minireview. Apoptosis 8:141-150. [DOI] [PubMed] [Google Scholar]
- 5.Diamond, M. S., and R. S. Klein. 2006. A genetic basis for human susceptibility to West Nile virus. Trends Microbiol. 14:287-289. [DOI] [PubMed] [Google Scholar]
- 6.Diamond, M. S., T. Roberts, D. Edgil, B. Lu, J. Ernst, and E. Harris. 2000. Modulation of dengue virus infection in human cells by alpha, beta, and gamma interferons. J. Virol. 74:4957-4966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Diamond, M. S., B. Shrestha, A. Marri, D. Mahan, and M. Engle. 2003. B cells and antibody play critical roles in the immediate defense of disseminated infection by West Nile encephalitis virus. J. Virol. 77:2578-2586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Dowling, P., G. Shang, S. Raval, J. Menonna, S. Cook, and W. Husar. 1996. Involvement of the CD95 (APO-1/Fas) receptor/ligand system in multiple sclerosis brain. J. Exp. Med. 184:1513-1518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Glass, W. G., D. H. McDermott, J. K. Lim, S. Lekhong, S. F. Yu, W. A. Frank, J. Pape, R. C. Cheshier, and P. M. Murphy. 2006. CCR5 deficiency increases risk of symptomatic West Nile virus infection. J. Exp. Med. 203:35-40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Grosjean, M. B., P. M. Lenzlinger, P. F. Stahel, I. Yatsiv, E. Shohami, O. Trentz, T. Kossmann, and M. C. Morganti-Kossmann. 2007. Immunohistochemical characterization of Fas (CD95) and Fas ligand (FasL/CD95L) expression in the injured brain: relationship with neuronal cell death and inflammatory mediators. Histol. Histopathol. 22:235-250. [DOI] [PubMed] [Google Scholar]
- 11.Harty, J. T., A. R. Tvinnereim, and D. W. White. 2000. CD8+ T cell effector mechanisms in resistance to infection. Annu. Rev. Immunol. 18:275-308. [DOI] [PubMed] [Google Scholar]
- 12.Hubalek, Z., and J. Halouzka. 1999. West Nile fever: a reemerging mosquito-borne viral disease in Europe. Emerg. Infect. Dis. 5:643-650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Johnston, L. J., G. M. Halliday, and N. J. King. 2000. Langerhans cells migrate to local lymph nodes following cutaneous infection with an arbovirus. J. Investig. Dermatol. 114:560-568. [DOI] [PubMed] [Google Scholar]
- 14.Johnston, L. J., G. M. Halliday, and N. J. King. 1996. Phenotypic changes in Langerhans' cells after infection with arboviruses: a role in the immune response to epidermally acquired viral infection? J. Virol. 70:4761-4766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kagi, D., B. Ledermann, K. Burki, P. Seiler, B. Odermatt, K. J. Olsen, E. R. Podack, R. M. Zinkernagel, and H. Hengartner. 1994. Cytotoxicity mediated by T cells and natural killer cells is greatly impaired in perforin-deficient mice. Nature 369:31-37. [DOI] [PubMed] [Google Scholar]
- 16.Kagi, D., P. Seiler, J. Pavlovic, B. Ledermann, K. Burki, R. M. Zinkernagel, and H. Hengartner. 1995. The roles of perforin- and Fas-dependent cytotoxicity in protection against cytopathic and noncytopathic viruses. Eur. J. Immunol. 25:3256-3262. [DOI] [PubMed] [Google Scholar]
- 17.Kagi, D., F. Vignaux, B. Ledermann, K. Burki, V. Depraetere, S. Nagata, H. Hengartner, and P. Golstein. 1994. Fas and perforin pathways as major mechanisms of T cell-mediated cytotoxicity. Science 265:528-530. [DOI] [PubMed] [Google Scholar]
- 18.Klein, R. S., E. Lin, B. Zhang, A. D. Luster, J. Tollett, M. A. Samuel, M. Engle, and M. S. Diamond. 2005. Neuronal CXCL10 directs CD8+ T-cell recruitment and control of West Nile virus encephalitis. J. Virol. 79:11457-11466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Krzyzowska, M., J. Cymerys, A. Winnicka, and M. Niemialtowski. 2006. Involvement of Fas and FasL in ectromelia virus-induced apoptosis in mouse brain. Virus Res. 115:141-149. [DOI] [PubMed] [Google Scholar]
- 20.Lambert, C., A. M. Landau, and J. Desbarats. 2003. Fas—beyond death: a regenerative role for Fas in the nervous system. Apoptosis 8:551-562. [DOI] [PubMed] [Google Scholar]
- 21.Licon Luna, R. M., E. Lee, A. Mullbacher, R. V. Blanden, R. Langman, and M. Lobigs. 2002. Lack of both Fas ligand and perforin protects from flavivirus-mediated encephalitis in mice. J. Virol. 76:3202-3211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Matsuyama, T., R. Hata, M. Tagaya, Y. Yamamoto, T. Nakajima, J. Furuyama, A. Wanaka, and M. Sugita. 1994. Fas antigen mRNA induction in postischemic murine brain. Brain Res. 657:342-346. [DOI] [PubMed] [Google Scholar]
- 23.Medana, I. M., A. Gallimore, A. Oxenius, M. M. Martinic, H. Wekerle, and H. Neumann. 2000. MHC class I-restricted killing of neurons by virus-specific CD8+ T lymphocytes is effected through the Fas/FasL, but not the perforin pathway. Eur. J. Immunol. 30:3623-3633. [DOI] [PubMed] [Google Scholar]
- 24.Mori, I., F. Goshima, Y. Imai, S. Kohsaka, T. Sugiyama, T. Yoshida, T. Yokochi, Y. Nishiyama, and Y. Kimura. 2002. Olfactory receptor neurons prevent dissemination of neurovirulent influenza A virus into the brain by undergoing virus-induced apoptosis. J. Gen. Virol. 83:2109-2116. [DOI] [PubMed] [Google Scholar]
- 25.Mullbacher, A., M. Lobigs, R. T. Hla, T. Tran, T. Stehle, and M. M. Simon. 2002. Antigen-dependent release of IFN-gamma by cytotoxic T cells up-regulates Fas on target cells and facilitates exocytosis-independent specific target cell lysis. J. Immunol. 169:145-150. [DOI] [PubMed] [Google Scholar]
- 26.Murray, K., S. Baraniuk, M. Resnick, R. Arafat, C. Kilborn, K. Cain, R. Shallenberger, T. L. York, D. Martinez, J. S. Hellums, D. Hellums, M. Malkoff, N. Elgawley, W. McNeely, S. A. Khuwaja, and R. B. Tesh. 2006. Risk factors for encephalitis and death from West Nile virus infection. Epidemiol. Infect. 134:1325-1332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Nagata, S. 1997. Apoptosis by death factor. Cell 88:355-365. [DOI] [PubMed] [Google Scholar]
- 28.Nagata, S. 1999. Fas ligand-induced apoptosis. Annu. Rev. Genet. 33:29-55. [DOI] [PubMed] [Google Scholar]
- 29.Parra, B., M. T. Lin, S. A. Stohlman, C. C. Bergmann, R. Atkinson, and D. R. Hinton. 2000. Contributions of Fas-Fas ligand interactions to the pathogenesis of mouse hepatitis virus in the central nervous system. J. Virol. 74:2447-2450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Petersen, L. R., A. A. Marfin, and D. J. Gubler. 2003. West Nile virus. JAMA 290:524-528. [DOI] [PubMed] [Google Scholar]
- 31.Purtha, W. E., N. Myers, V. Mitaksov, E. Sitati, J. Connolly, D. H. Fremont, T. H. Hansen, and M. S. Diamond. 2007. Antigen-specific cytotoxic T lymphocytes protect against lethal West Nile virus encephalitis. Eur. J. Immunol. 37:1845-1854. [DOI] [PubMed] [Google Scholar]
- 32.Regner, M., M. Lobigs, R. V. Blanden, and A. Mullbacher. 2001. Effector cytolotic function but not IFN-gamma production in cytotoxic T cells triggered by virus-infected target cells in vitro. Scand. J. Immunol. 54:366-374. [DOI] [PubMed] [Google Scholar]
- 33.Richardson-Burns, S. M., D. J. Kominsky, and K. L. Tyler. 2002. Reovirus-induced neuronal apoptosis is mediated by caspase 3 and is associated with the activation of death receptors. J. Neurovirol. 8:365-380. [DOI] [PubMed] [Google Scholar]
- 34.Rossi, C. P., A. McAllister, M. Tanguy, D. Kagi, and M. Brahic. 1998. Theiler's virus infection of perforin-deficient mice. J. Virol. 72:4515-4519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Russell, J. H., and T. J. Ley. 2002. Lymphocyte-mediated cytotoxicity. Annu. Rev. Immunol. 20:323-370. [DOI] [PubMed] [Google Scholar]
- 36.Sabri, F., A. De Milito, R. Pirskanen, I. Elovaara, L. Hagberg, P. Cinque, R. Price, and F. Chiodi. 2001. Elevated levels of soluble Fas and Fas ligand in cerebrospinal fluid of patients with AIDS dementia complex. J. Neuroimmunol. 114:197-206. [DOI] [PubMed] [Google Scholar]
- 37.Samuel, M. A., and M. S. Diamond. 2006. Pathogenesis of West Nile virus infection: a balance between virulence, innate and adaptive immunity, and viral evasion. J. Virol. 80:9349-9360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Sejvar, J. J., A. V. Bode, A. A. Marfin, G. L. Campbell, J. Pape, B. J. Biggerstaff, and L. R. Petersen. 2006. West Nile virus-associated flaccid paralysis outcome. Emerg. Infect. Dis. 12:514-516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Sejvar, J. J., M. B. Haddad, B. C. Tierney, G. L. Campbell, A. A. Marfin, J. A. Van Gerpen, A. Fleischauer, A. A. Leis, D. S. Stokic, and L. R. Petersen. 2003. Neurologic manifestations and outcome of West Nile virus infection. JAMA 290:511-515. [DOI] [PubMed] [Google Scholar]
- 40.Shresta, S., C. T. Pham, D. A. Thomas, T. A. Graubert, and T. J. Ley. 1998. How do cytotoxic lymphocytes kill their targets? Curr. Opin. Immunol. 10:581-587. [DOI] [PubMed] [Google Scholar]
- 41.Shrestha, B., and M. S. Diamond. 2004. The role of CD8+ T cells in the control of West Nile virus infection. J. Virol. 78:8312-8321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Shrestha, B., D. I. Gottlieb, and M. S. Diamond. 2003. Infection and injury of neurons by West Nile encephalitis virus. J. Virol. 77:13203-13213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Shrestha, B., M. A. Samuel, and M. S. Diamond. 2006. CD8+ T cells require perforin to clear West Nile virus from infected neurons. J. Virol. 80:119-129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Shrestha, B., T. Wang, M. A. Samuel, K. Whitby, J. Craft, E. Fikrig, and M. S. Diamond. 2006. Gamma interferon plays a crucial early antiviral role in protection against West Nile virus infection. J. Virol. 80:5338-5348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Sitati, E., and M. S. Diamond. 2006. CD4+ T-cell responses are required for clearance of West Nile virus from the central nervous system. J. Virol. 80:12060-12069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Stohlman, S. A., C. C. Bergmann, R. C. van der Veen, and D. R. Hinton. 1995. Mouse hepatitis virus-specific cytotoxic T lymphocytes protect from lethal infection without eliminating virus from the central nervous system. J. Virol. 69:684-694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Suda, T., and S. Nagata. 1997. Why do defects in the Fas-Fas ligand system cause autoimmunity? J. Allergy Clin. Immunol. 100:S97-S101. [DOI] [PubMed] [Google Scholar]
- 48.Takahashi, T., M. Tanaka, C. I. Brannan, N. A. Jenkins, N. G. Copeland, T. Suda, and S. Nagata. 1994. Generalized lymphoproliferative disease in mice, caused by a point mutation in the Fas ligand. Cell 76:969-976. [DOI] [PubMed] [Google Scholar]
- 49.Topham, D. J., R. A. Tripp, and P. C. Doherty. 1997. CD8+ T cells clear influenza virus by perforin or Fas-dependent processes. J. Immunol. 159:5197-5200. [PubMed] [Google Scholar]
- 50.Wang, T., and E. Fikrig. 2004. Immunity to West Nile virus. Curr. Opin. Immunol. 16:519-523. [DOI] [PubMed] [Google Scholar]
- 51.Wang, T., T. Town, L. Alexopoulou, J. F. Anderson, E. Fikrig, and R. A. Flavell. 2004. Toll-like receptor 3 mediates West Nile virus entry into the brain causing lethal encephalitis. Nat. Med. 10:1366-1373. [DOI] [PubMed] [Google Scholar]
- 52.Wang, Y., M. Lobigs, E. Lee, and A. Mullbacher. 2003. CD8+ T cells mediate recovery and immunopathology in West Nile virus encephalitis. J. Virol. 77:13323-13334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Wang, Y., M. Lobigs, E. Lee, and A. Mullbacher. 2004. Exocytosis and Fas mediated cytolytic mechanisms exert protection from West Nile virus induced encephalitis in mice. Immunol. Cell Biol. 82:170-173. [DOI] [PubMed] [Google Scholar]
- 54.Xiao, S. Y., H. Guzman, H. Zhang, A. P. Travassos da Rosa, and R. B. Tesh. 2001. West Nile virus infection in the golden hamster (Mesocricetus auratus): a model for West Nile encephalitis. Emerg. Infect. Dis. 7:714-721. [DOI] [PMC free article] [PubMed] [Google Scholar]