Abstract
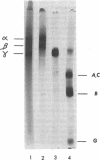
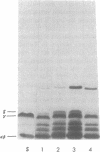
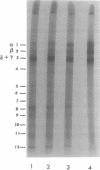
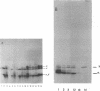
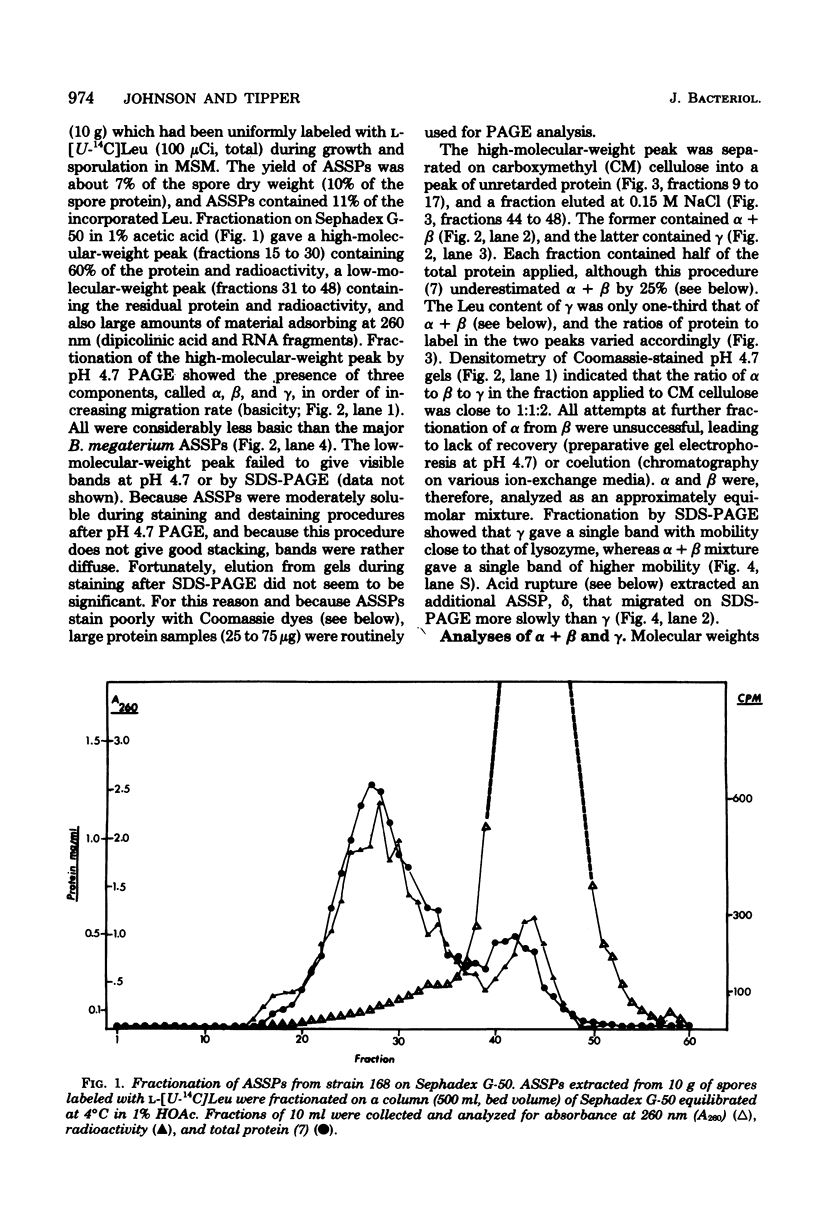
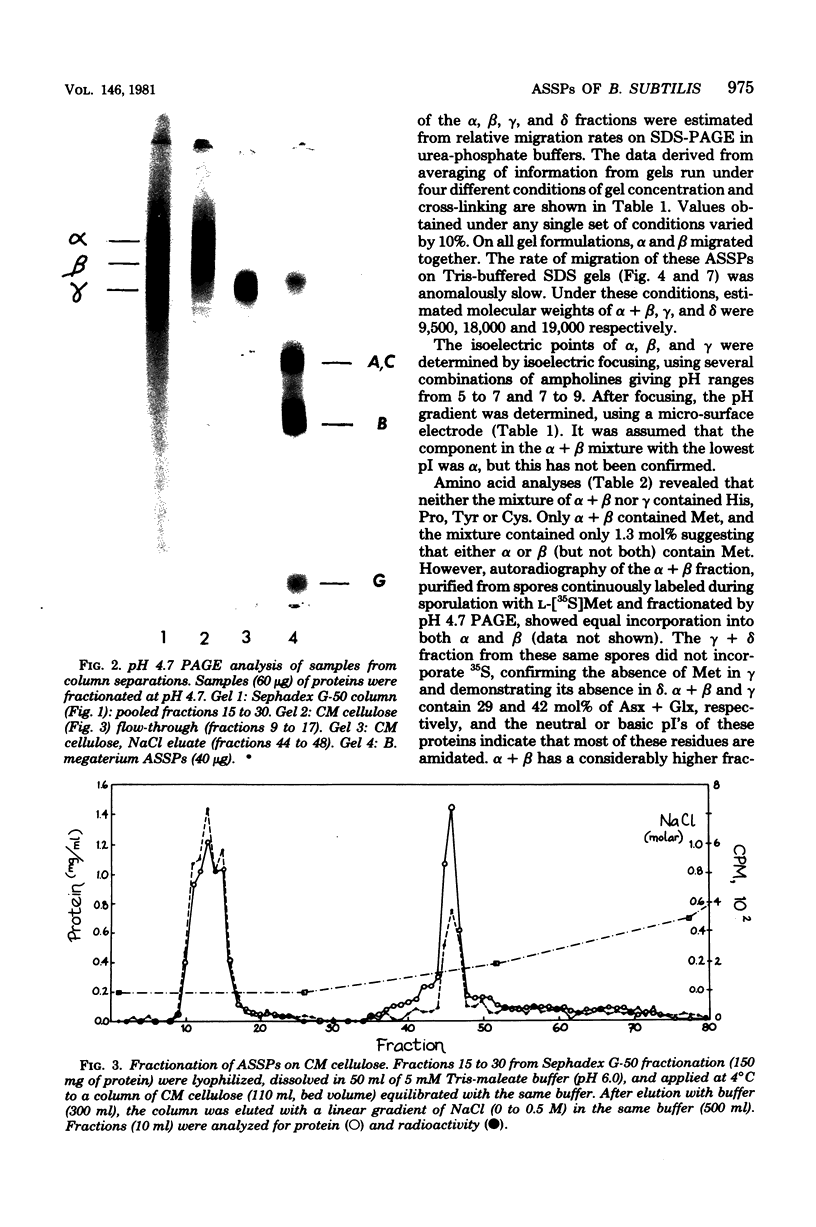
Acid-soluble spore proteins (ASSPs) comprise about 5% of the total protein of mature spores of different Bacillus subtilis strains. They consist of three abundant species, alpha, beta, and gamma, four less abundant species, and several minor species, alpha, beta, and gamma make up about 18, 18 and 36%, respectively, of the total ASSPs of strain 168, have molecular weights of 5,900, 5,9000, and 11,000, respectively, and resemble the major (A, C, and B) components of Bacillus megaterium ASSPs in several respects, including sensitivity to a specific B. megaterium spore endopeptidase. However, they have pI's of 6.58, 6.67, and 7.96, all lower than those of any of the B. megaterium ASSPs. Although strains varied in the proportions of different ASSPs, to overall patterns seen on gel electrophoresis are constant. ASSPs are located interior to the cortex, presumably in the spore cytoplasm, and are synthesized during sporulation and degraded during germination.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ames G. F., Nikaido K. Two-dimensional gel electrophoresis of membrane proteins. Biochemistry. 1976 Feb 10;15(3):616–623. doi: 10.1021/bi00648a026. [DOI] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Böhlen P., Stein S., Dairman W., Udenfriend S. Fluorometric assay of proteins in the nanogram range. Arch Biochem Biophys. 1973 Mar;155(1):213–220. doi: 10.1016/s0003-9861(73)80023-2. [DOI] [PubMed] [Google Scholar]
- Goldman R. C., Tipper D. J. Bacillus subtilis spore coats: complexity and purification of a unique polypeptide component. J Bacteriol. 1978 Sep;135(3):1091–1106. doi: 10.1128/jb.135.3.1091-1106.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldman R. C., Tipper D. J. Morphology and patterns of protein synthesis during sporulation of Bacillus subtilis Eryr spo(Ts) mutants. J Bacteriol. 1979 May;138(2):625–637. doi: 10.1128/jb.138.2.625-637.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Linn T., Losick R. The program of protein synthesis during sporulation in Bacillus subtilis. Cell. 1976 May;8(1):103–114. doi: 10.1016/0092-8674(76)90191-4. [DOI] [PubMed] [Google Scholar]
- Pandey N. K., Aronson A. I. Properties of the Bacillus subtilis spore coat. J Bacteriol. 1979 Mar;137(3):1208–1218. doi: 10.1128/jb.137.3.1208-1218.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- REISFELD R. A., LEWIS U. J., WILLIAMS D. E. Disk electrophoresis of basic proteins and peptides on polyacrylamide gels. Nature. 1962 Jul 21;195:281–283. doi: 10.1038/195281a0. [DOI] [PubMed] [Google Scholar]
- ROE J. H. The determination of sugar in blood and spinal fluid with anthrone reagent. J Biol Chem. 1955 Jan;212(1):335–343. [PubMed] [Google Scholar]
- Rotman Y., Fields M. L. A modified reagent for dipicolinic acid analysis. Anal Biochem. 1968 Jan;22(1):168–168. doi: 10.1016/0003-2697(68)90272-8. [DOI] [PubMed] [Google Scholar]
- Setlow P. Identification and localization of the major proteins degraded during germination of Bacillus megaterium spores. J Biol Chem. 1975 Oct 25;250(20):8159–8167. [PubMed] [Google Scholar]
- Setlow P., Ozols J. Covalent structure of protein A. A low molecular weight protein degraded during germination of Bacillus megaterium spores. J Biol Chem. 1979 Dec 10;254(23):11938–11942. [PubMed] [Google Scholar]
- Setlow P., Ozols J. Covalent structure of protein C. A second major low molecular weight protein degraded during germination of Bacillus megaterium spores. J Biol Chem. 1980 Sep 25;255(18):8413–8416. [PubMed] [Google Scholar]
- Setlow P., Ozols J. The complete covalent structure of protein B. The third major protein degraded during germination of Bacillus megaterium spores. J Biol Chem. 1980 Nov 10;255(21):10445–10450. [PubMed] [Google Scholar]
- Setlow P., Primus G. Protein metabolism during germination of Bacillus megaterium spores. I. Protein synthesis and amino acid metabolism. J Biol Chem. 1975 Jan 25;250(2):623–630. [PubMed] [Google Scholar]
- Setlow P. Protein metabolism during germination of Bacillus megaterium spores. II. Degradation of pre-existing and newly synthesized protein. J Biol Chem. 1975 Jan 25;250(2):631–637. [PubMed] [Google Scholar]
- Setlow P. Purification and characterization of additional low-molecular-weight basic proteins degraded during germination of Bacillus megaterium spores. J Bacteriol. 1978 Oct;136(1):331–340. doi: 10.1128/jb.136.1.331-340.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Setlow P. Purification and properties of a specific proteolytic enzyme present in spores of Bacillus magaterium. J Biol Chem. 1976 Dec 25;251(24):7853–7862. [PubMed] [Google Scholar]
- Setlow P. Purification and properties of some unique low molecular weight basic proteins degraded during germination of Bacillus megaterium spores. J Biol Chem. 1975 Oct 25;250(20):8168–8173. [PubMed] [Google Scholar]
- Smucker R. A., Pfister R. M. Liquid nitrogen cryo-impacting: a new concept for cell disruption. Appl Microbiol. 1975 Sep;30(3):445–449. doi: 10.1128/am.30.3.445-449.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sonenshein A. L., Cami B., Brevet J., Cote R. Isolation and characterization of rifampin-resistant and streptolydigin-resistant mutants of Bacillus subtilis with altered sporulation properties. J Bacteriol. 1974 Oct;120(1):253–265. doi: 10.1128/jb.120.1.253-265.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tipper D. J., Johnson C. W., Ginther C. L., Leighton T., Wittmann H. G. Erythromycin resistant mutations in Bacillus subtilis cause temperature sensitive sporulation. Mol Gen Genet. 1977 Jan 18;150(2):147–159. doi: 10.1007/BF00695395. [DOI] [PubMed] [Google Scholar]
- Weber K., Osborn M. The reliability of molecular weight determinations by dodecyl sulfate-polyacrylamide gel electrophoresis. J Biol Chem. 1969 Aug 25;244(16):4406–4412. [PubMed] [Google Scholar]
- Yuan K., Johnson W. C., Tipper D. J., Setlow P. Comparison of various properties of low-molecular-weight proteins from dormant spores of several Bacillus species. J Bacteriol. 1981 Jun;146(3):965–971. doi: 10.1128/jb.146.3.965-971.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]