Abstract
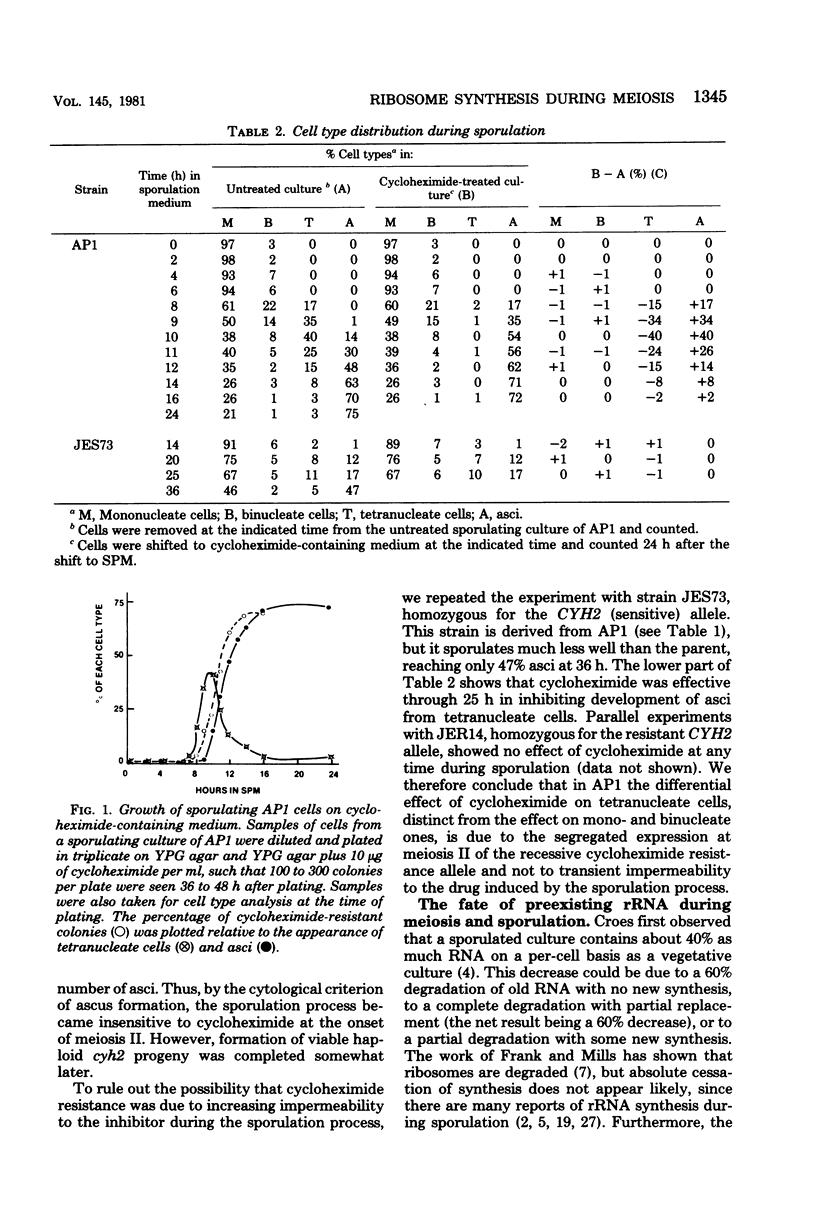
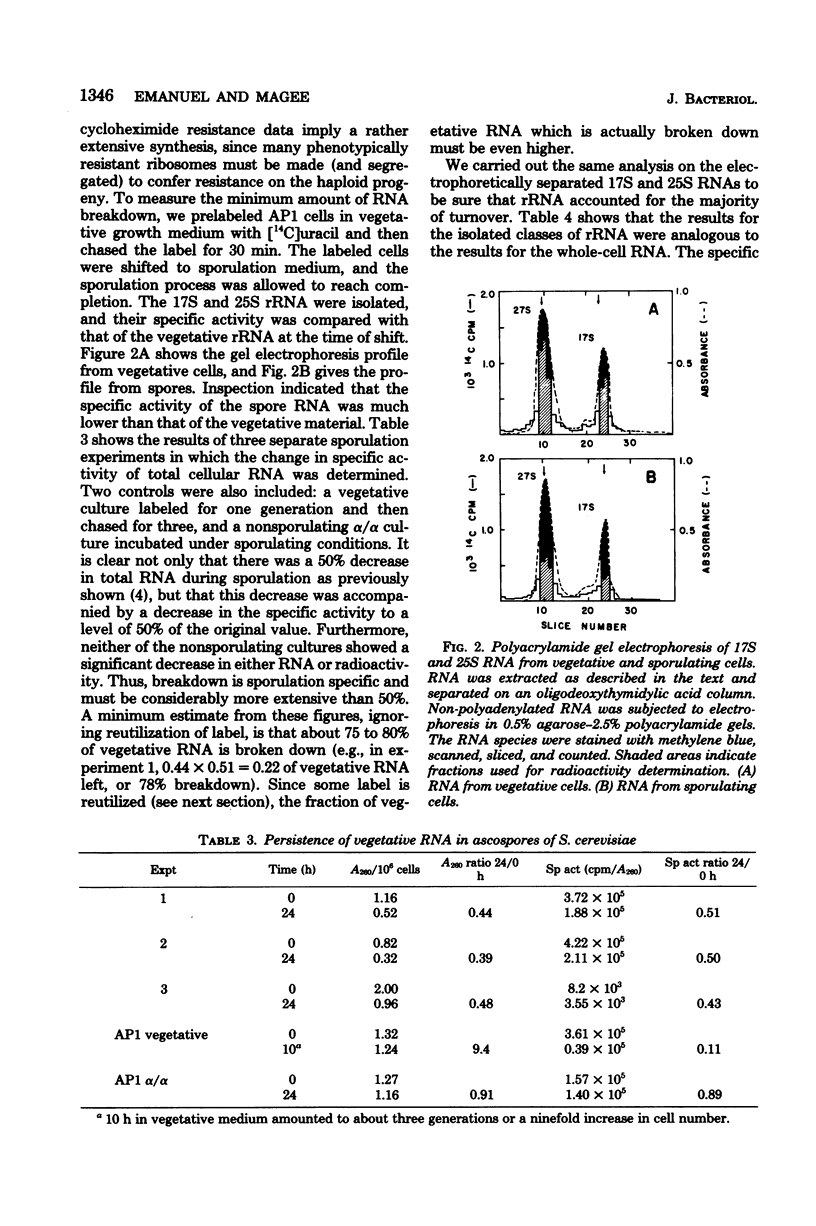
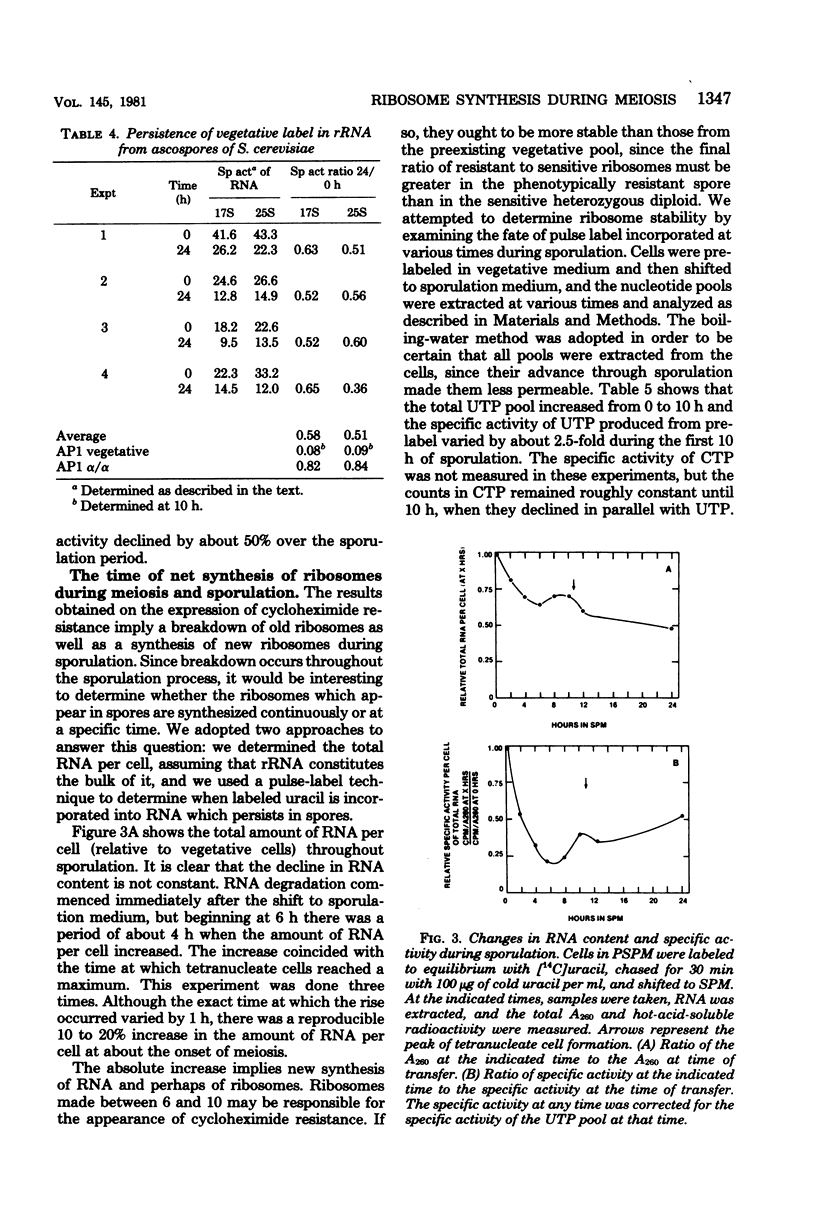
During meiosis and sporulation in Saccharomyces cerevisiae, the recessive genetic marker for cycloheximide resistance, believed to be due to an altered ribosomal protein (C. S. McLaughlin, p. 815-827, in M. Nomura et al., ed., Ribosomes, Cold Spring Harbor Laboratory, Cold Spring Harbor, N.Y.), is expressed as early as meiosis II. Ribosomal ribonucleic acid synthesis peaks near the time that cycloheximide resistance begins to appear. Less than 25% of the 17S and 25S ribonucleic acid of the vegetative cells persists in spores, but pulse-labeling studies indicate that greater than 90% of the stable ribonucleic acid made after 6 h survives in spores. These results indicate that the haploid daughter genomes begin to function near the time of meiosis II.
Full text
PDF





Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aviv H., Leder P. Purification of biologically active globin messenger RNA by chromatography on oligothymidylic acid-cellulose. Proc Natl Acad Sci U S A. 1972 Jun;69(6):1408–1412. doi: 10.1073/pnas.69.6.1408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chaffin W. L., Sogin S. J., Halvorson H. O. Nature of ribonucleic acid synthesis during early sporulation in Saccharomyces cerevisiae. J Bacteriol. 1974 Nov;120(2):872–879. doi: 10.1128/jb.120.2.872-879.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen A. W., Miller J. J. Proteolytic activity of intact yeast cells during sporulation. Can J Microbiol. 1968 Sep;14(9):957–963. doi: 10.1139/m68-159. [DOI] [PubMed] [Google Scholar]
- Curiale M. S., Petryna M. M., Mills D. Ribonucleic acid synthesized in meiotic cells of Saccharomyces cerevisiae: effect of culture medium pH. J Bacteriol. 1976 May;126(2):661–667. doi: 10.1128/jb.126.2.661-667.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dickinson H. G., Heslop-Harrison J. The ribosome cycle, nucleoli, and cytoplasmic nucleoloids in the meiocytes of Lilium. Protoplasma. 1970;69(2):189–200. doi: 10.1007/BF01280721. [DOI] [PubMed] [Google Scholar]
- Frank K. R., Mills D. Ribosome activity and degradation in meiotic cells of Saccharomyces cerevisiae. Mol Gen Genet. 1978 Mar 20;160(1):59–65. doi: 10.1007/BF00275119. [DOI] [PubMed] [Google Scholar]
- Galau G. A., Klein W. H., Davis M. M., Wold B. J., Britten R. J., Davidson E. H. Structural gene sets active in embryos and adult tissues of the sea urchin. Cell. 1976 Apr;7(4):487–505. doi: 10.1016/0092-8674(76)90200-2. [DOI] [PubMed] [Google Scholar]
- Gorenstein C., Warner J. R. Coordinate regulation of the synthesis of eukaryotic ribosomal proteins. Proc Natl Acad Sci U S A. 1976 May;73(5):1547–1551. doi: 10.1073/pnas.73.5.1547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harper J. F., Clancy M. J., Magee P. T. Properties of polyadenylate-associated ribonucleic acid from Saccharomyces cerevisiae ascospores. J Bacteriol. 1980 Aug;143(2):958–965. doi: 10.1128/jb.143.2.958-965.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hopper A. K., Magee P. T., Welch S. K., Friedman M., Hall B. D. Macromolecule synthesis and breakdown in relation to sporulation and meiosis in yeast. J Bacteriol. 1974 Aug;119(2):619–628. doi: 10.1128/jb.119.2.619-628.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Magee P. T., Hopper A. K. Protein synthesis in relation to sporulation and meiosis in yeast. J Bacteriol. 1974 Sep;119(3):952–960. doi: 10.1128/jb.119.3.952-960.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCusker J. H., Haber J. E. Efficient sporulation of yeast in media buffered near pH6. J Bacteriol. 1977 Oct;132(1):180–185. doi: 10.1128/jb.132.1.180-185.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mills D. Effect of pH on adenine and amino acid uptake during sporulation in Saccharomyces cerevisiae. J Bacteriol. 1972 Oct;112(1):519–526. doi: 10.1128/jb.112.1.519-526.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moens P. B., Esposito R. E., Esposito M. S. Aberrant nuclear behavior at meiosis and anucleate spore formation by sporulation-deficient (SPO) mutants of Saccharomyces cerevisiae. Exp Cell Res. 1974 Jan;83(1):166–174. doi: 10.1016/0014-4827(74)90700-9. [DOI] [PubMed] [Google Scholar]
- Moens P. B. Fine structure of ascospore development in the yeast Saccharomyces cerevisiae. Can J Microbiol. 1971 Apr;17(4):507–510. doi: 10.1139/m71-084. [DOI] [PubMed] [Google Scholar]
- Pearson N. J., Haber J. E. Changes in regulation of ribosome synthesis during different stages of the life cycle of Saccharomyces cerevisiae. Mol Gen Genet. 1977 Dec 14;158(1):81–91. doi: 10.1007/BF00455122. [DOI] [PubMed] [Google Scholar]
- Petes T. D., Hereford L. M., Skryabin K. G. Characterization of two types of yeast ribosomal DNA genes. J Bacteriol. 1978 Apr;134(1):295–305. doi: 10.1128/jb.134.1.295-305.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rao S. S., Grollman A. P. Cycloheximide resistance in yeast: a property of the 60s ribosomal subunit. Biochem Biophys Res Commun. 1967 Dec 15;29(5):696–704. doi: 10.1016/0006-291x(67)90273-2. [DOI] [PubMed] [Google Scholar]
- Roth R., Halvorson H. O. Sporulation of yeast harvested during logarithmic growth. J Bacteriol. 1969 May;98(2):831–832. doi: 10.1128/jb.98.2.831-832.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rousseau P., Halvorson H. O. Macromolecular synthesis during the germanation of Saccharomyces cerevisiae spores. J Bacteriol. 1973 Mar;113(3):1289–1295. doi: 10.1128/jb.113.3.1289-1295.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siersma P. W., Chiang K. S. Conservation and degradation of cytoplasmic and chloroplast ribosomes in Chlamydomonas reinhardtii. J Mol Biol. 1971 May 28;58(1):167–185. doi: 10.1016/0022-2836(71)90239-7. [DOI] [PubMed] [Google Scholar]
- Stern H., Hotta Y. Biochemical controls of meiosis. Annu Rev Genet. 1973;7:37–66. doi: 10.1146/annurev.ge.07.120173.000345. [DOI] [PubMed] [Google Scholar]
- Warner J. R., Udem S. A. Temperature sensitive mutations affecting ribosome synthesis in Saccharomyces cerevisiae. J Mol Biol. 1972 Mar 28;65(2):243–257. doi: 10.1016/0022-2836(72)90280-x. [DOI] [PubMed] [Google Scholar]
- Wejksnora P., Haber J. E. Influence of pH on the rate of ribosomal ribonucleic acid synthesis during sporulation in Saccharomyces cerevisiae. J Bacteriol. 1976 Jul;127(1):128–134. doi: 10.1128/jb.127.1.128-134.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zinker S., Warner J. R. The ribosomal proteins of Saccharomyces cerevisiae. Phosphorylated and exchangeable proteins. J Biol Chem. 1976 Mar 25;251(6):1799–1807. [PubMed] [Google Scholar]



