Abstract
An active involvement of blood–brain barrier endothelial cell basement membranes in development of inflammatory lesions in the central nervous system (CNS) has not been considered to date. Here we investigated the molecular composition and possible function of the extracellular matrix encountered by extravasating T lymphocytes during experimental autoimmune encephalomyelitis (EAE).
Endothelial basement membranes contained laminin 8 (α4β1γ1) and/or 10 (α5β1γ1) and their expression was influenced by proinflammatory cytokines or angiostatic agents. T cells emigrating into the CNS during EAE encountered two biochemically distinct basement membranes, the endothelial (containing laminins 8 and 10) and the parenchymal (containing laminins 1 and 2) basement membranes. However, inflammatory cuffs occurred exclusively around endothelial basement membranes containing laminin 8, whereas in the presence of laminin 10 no infiltration was detectable. In vitro assays using encephalitogenic T cell lines revealed adhesion to laminins 8 and 10, whereas binding to laminins 1 and 2 could not be induced. Downregulation of integrin α6 on cerebral endothelium at sites of T cell infiltration, plus a high turnover of laminin 8 at these sites, suggested two possible roles for laminin 8 in the endothelial basement membrane: one at the level of the endothelial cells resulting in reduced adhesion and, thereby, increased penetrability of the monolayer; and secondly at the level of the T cells providing direct signals to the transmigrating cells.
Keywords: laminin, experimental autoimmune encephalomyelitis, endothelium, basement membranes, inflammation
Introduction
One of the major functional components of all basement membranes is the laminin family of glycoproteins which influence motility, proliferation, and differentiation of many cell types (Timpl and Brown 1994; Ekblom and Timpl 1996). Laminins are heterotrimers composed of an α, β, and γ chain. To date, 5 α, 3 β, and 3 γ chains have been reported which can combine to form up to 12 different laminin isoforms (Timpl et al. 1979; Hunter et al. 1989; Paulsson and Saladin 1989; Ehrig et al. 1990; Miner et al. 1995; Frieser et al. 1997; Koch et al. 1999). However, in vitro cellular interactions are well defined only for laminin 1 (Aumailley and Smyth 1998) and, to a lesser extent, for laminins 2 and 5, which have restricted distributions in vivo. Only two isoforms, laminins 8 (composed of laminin α4, β1, and γ1) and 10 (composed of laminin α5, β1, and γ1) are found in endothelial basement membranes of most tissues (Sorokin et al. 1994, Sorokin et al. 1997a; Frieser et al. 1997). Further, we have shown that laminin α4 expression by endothelial cells in vitro is strongly upregulated by proinflammatory cytokines such as interleukin (IL)-1 (Frieser et al. 1997), suggesting a role in inflammatory events. Blood vessels in the central nervous system (CNS) have also been reported to express laminins 1 and 2 (Jucker et al. 1996; Powell and Kleinman 1997; Tian et al. 1997), which are not detected in blood vessel basement membranes elsewhere and which may be related to their highly specialized structure. The differential expression of laminin isoforms under different endothelial cell activation states has prompted us to investigate the role of laminins in inflammatory events. We have used a murine experimental autoimmune encephalomyelitis (EAE), an inflammation model with a well defined immunopathology and clinically relevant similarity to the human disease, multiple sclerosis.
EAE can be induced in susceptible mouse strains by immunization with myelin proteins, myelin protein fragments, or by adoptive transfer of myelin reactive CD4+ T cell blasts (Engelhardt 1997). A critical step in this inflammation is the extravasation of leukocytes from the blood stream into the CNS parenchyma, which involves autoaggressive T cell adhesion to and migration through the endothelial cell monolayer of the postcapillary venules. However, after passage across the endothelial cell monolayer, leukocytes still face the endothelial cell basement membrane and the subjacent glia limitans consisting of astrocyte endfeet and associated basement membrane (Wolburg and Risau 1995).
Ultrastructurally, at least two basement membranes can be identified in association with larger blood vessels in the brain, an endothelial and an astroglial basement membrane (Wolburg and Risau 1995). In addition, the epithelium of the meninges coinvaginates with blood vessels from the surface of the brain and contributes to the astroglial basement membrane (Alcolado et al. 1988; Wolburg and Risau 1990; Zhang et al. 1990; Wolburg and Risau 1995). Collectively, the astroglial basement membrane and the meningeal epithelium basement membrane are known as the parenchymal basement membrane, as they delineate the border to the brain parenchyma. The endothelial cell and parenchymal basement membranes define the inner and outer limits of the perivascular space where leukocytes accumulate during acute EAE before infiltrating the brain parenchyma (Cross et al. 1993; Engelhardt et al. 1997). However, studies to date have not distinguished between penetration of the endothelial cell basement membrane and the process of parenchymal invasion across the glia limitans. There is evidence that suggests these two steps are distinct and independent of one another: in EAE induced in macrophage-depleted mice (Tran et al. 1998) or in tumor necrosis factor (TNF)-deficient mice (Körner et al. 1997), the inflammatory infiltrate becomes entrapped in the perivascular space and parenchymal infiltration is prevented, indicating that progression through the astrocyte basement membrane is functionally distinct from endothelial cell basement membrane transmigration.
In this study, we define the extracellular matrix barriers encountered by extravasating T cells and the steps involved in their transmigration using two different murine EAE models. Further, the use of encephalitogenic T cell lines in in vitro adhesion assays has permitted definition of cell–matrix interactions permissive or restrictive for T cell extravasation. Our data demonstrate that endothelial cell basement membranes containing laminin 8 are permissive for T cell transmigration, whereas those containing laminin 10 are restrictive for T cell transmigration. Penetration of the parenchymal basement membrane, characterized by the expression of laminins 1 and 2, which are not adhesive for T cells, occurs only after disruption of this outer barrier, probably via proteolysis and involving matrix metalloproteinases (MMPs; Graesser et al. 2000).
Materials and Methods
Animals
Female SJL/N and C57Bl/6 mice between 3 and 4 wk of age were obtained from Bomholdgard Breeding.
Induction of EAE and PLP T Cell Lines
EAE was induced in SJL/N mice by immunization with syngeneic spinal cord homogenate in CFA (Engelhardt et al. 1997) or C57Bl/6 mice using myelin oligodendrocyte glycoprotein peptide (Gardinier et al. 1992) as described previously. Animals were observed daily and neurological deficits were scaled as follows: 1+, flaccid tail; 2+, hind limb weakness; 3+, severe hind limb weakness; 4+, hind quarter paralysis; and 5+, forelimb weakness. At least three mice at each stage were examined.
Protein–lipid–protein (PLP)-specific T cell lines were established as described in detail previously (Engelhardt et al. 1997, Engelhardt et al. 1998).
Primary Antibodies
Primary antibodies to extracellular matrix and cell surface adhesion molecules used in immunofluorescence and cell adhesion studies are listed in Table .
Table 1.
Specificities and Sources of Antibodies Used
| Specificity | Species | Antibody | Source/reference |
|---|---|---|---|
| Laminin α1 | Rat | 198 | Sorokin et al. 1992 |
| Laminin α1 | Rabbit | 317 | Durbeej et al. 1996 |
| Laminin α2 | Rat | 4H8-2 | Schuler and Sorokin 1995 |
| Laminin α4 | Rat | 341 | Ringelmann et al. 2000 |
| Laminin α4 | Rabbit | 377 | Sorokin et al. 2000 |
| Laminin α3 | Rabbit | Lamα3 | Galliano et al. 1995 |
| Laminin α5 | Rat | 4G6 | Sorokin et al. 1997a |
| Laminin α5 | Rabbit | 405 | Sorokin et al. 2000 |
| Laminin β1 | Rat | 3E10 | Sixt et al. 2001 |
| Laminin γ1 | Rabbit | N283 | Mayer et al. 1993 |
| Laminin β2 | Guinea pig | GP-5 | Patton et al. 1997 |
| Pan–laminin 1 | Rabbit | 308 | Sorokin et al. 1990; Ringelmann et al. 2000 |
| Integrin α6 | Rat | GoH3 | Sonnenberg et al. 1988 |
| Integrin α3 | Rabbit | 340 | Valles et al. 1996 |
| Integrin α3 | Rabbit | C-18 | Santa Cruz Biotechnology, Inc. |
| Integrin α7 | Rabbit | 242 | Echtermeyer et al. 1996; Mayer et al. 1997 |
| Integrin β1 | Hamster | Ha2/5 | Lenter and Vestweber 1994; BD PharMingen |
| Integrin β4 | Rat | 346-11A | Lenter and Vestweber 1994; BD PharMingen |
| β-Dystroglycan | Mouse | NCL-43DAG | Novo Castra |
Rat anti–mouse platelet endothelial cell adhesion molecule (PECAM)-1 (MEC13.3; Vecchi et al. 1994) was used as an endothelial cell marker (BD PharMingen); rat anti–mouse CD45 (leukocyte common antigen; Ly-5) was used to detect leukocytes (30-F11; BD PharMingen); Cy3 conjugate anti–mouse glial fibrillar acidic protein (GFAP; Sigma-Aldrich) was used as a marker for astrocyte endfeet; and Cy3-coupled anti–mouse smooth muscle actin (Sigma-Aldrich), which in the CNS can be used to distinguish arteries and arterioles from veins, capillaries, and postcapillary venules (Nehls and Drenckhahn 1991; Nehls et al. 1992; Boado and Pardridge 1994).
Immunofluorescence
Tissues were prepared and immunofluorescently stained as described previously (Sorokin et al. 1992). Bound antibodies were visualized using FITC- or Texas red–conjugated goat anti–rat, goat anti–guinea pig, or goat anti–rabbit IgG second antibodies (Dianova). In the case of anti–β-dystroglycan stainings, unfixed sections were used. Stained sections were examined using confocal laser scanning microscopy (model 1000; Bio-Rad Laboratories).
Isolation of Laminins 8 and 10
Mouse laminin 8 (composed of α4β1γ1 chains) was isolated from the conditioned media of 3T3 fibroblasts and the MC3T3-G2/PA6 preadipocyte cell line using a combination of an ion exchange chromatography (POROS 20 HQ column; Applied Biosystems) and immunoaffinity chromatography (CNBr-Sepharose) with a rat anti–mouse laminin γ1 chain monoclonal antibody raised in our laboratory (3E10). In brief, conditioned medium was diluted 1:3 in 50 mM Tris-HCl buffer, pH 8, and concentrated on an ion exchange column, POROS 20 HQ column. After dialysis against PBS, the protein solution was subjected to immunoaffinity chromatography with the rat monoclonal anti-laminin γ1 antibody (3E10). The affinity column was prepared by coupling 2 mg purified antibody to CNBr-Sepharose. Bound protein was eluted batchwise from the affinity material with 0.1 M triethylamine, pH 11.5, neutralized, and dialysed against 20 mM Tris-HCl, pH 8. Before final dialysis against PBS, gelatin-Sepharose was used to absorb fibronectin and a small POROS 20 HQ column was used for sample concentration.
Laminin 10 was isolated from human placenta by affinity chromatography using mouse anti–human γ1 (D18) (Sanes et al. 1990) and α5 (4C7) monoclonal antibodies (Leivo and Engvall 1988) as described previously (Lindblom et al. 1994; Sixt et al. 2001). Mouse anti–human laminin β2 (C4) antibody was used to immunoabsorb laminin β2–containing complexes where necessary. Before immunopurification, 50–100 gm human placenta was homogenized twice on ice in 300 ml 50 mM Tris-HCl, pH 7.5, plus 150 mM NaCl and protease inhibitors, and finally in 50 mM Tris-HCl, pH 7.5, plus 300 mM NaCl and 20 mM EDTA. After at least 3 h of extraction at 4°C with gentle rolling, the placenta extract was clarified by centrifugation and dialysed against PBS. Further purification steps were performed essentially as described for laminin 8.
The chain composition of the laminin 8 and 10 preparations was assessed by immunoblotting and ELISA using laminin chain–specific antibodies (Table ), whereas the purity of the preparations was assessed by silver-stained SDS-PAGE under reducing conditions. The laminin 8 preparation was found to contain only laminin α4, β1, and γ1 chains (Sixt et al. 2001), whereas the laminin isolated from human placenta contained predominantly laminin α5, β1, and γ1 chains (laminin 10), plus a weak laminin β2 chain band in silver staining (laminin 11) as shown in Sixt et al. 2001. Laminin 11 represented <5% of the human laminin preparation.
Endothelial Cell Lines
Mouse endothelial cell lines derived from brain capillaries (bEND3) and a skin hemangioma (sEND1) were as described previously (Frieser et al. 1997; Sorokin et al. 1994, Sorokin et al. 1997a). Both cells were grown in DME supplemented with 10% fetal calf serum at 37°C and 7.5% CO2 in a water-saturated environment.
Northern Blot Analysis
mRNA extraction and Northern blot analyses were performed as described previously (Sorokin et al. 1997a,Sorokin et al. 1997b). RNA was isolated from unactivated endothelial cell lines (bEND3 and sEND1), and cell lines activated for 4 h at 37°C with 75 U/ml TNF-α (BD PharMingen) or 10 ng/ml IL-1β (Biomol), or treated for 3 d with 4 μg/ml hydroxymethylprogesterone (Sigma-Aldrich) and freshly dissected control and EAE brains (clinical scores, +4/+5). Hybridizations were performed with the following 32P-labeled specific cDNA probes: laminin α1 covering nucleotides 7786–9286 (Schuler and Sorokin 1995); laminin α2 covering nucleotides 6420–6895 (Schuler and Sorokin 1995); laminin α4 covering nucleotides 4720–5311 (Frieser et al. 1997); laminin α5 covering nucleotides 3962–4623 (Sorokin et al. 1997b); or with a glyceraldehyde-3-phosphate dehydrogenase (GADPH)-specific probe (Sorokin et al. 1997b).
In Situ Hybridization
In situ hybridization of E16 and E18 mouse embryos and newborn and adult brains was performed as described previously (Sorokin et al. 1997a), using the cDNA probes for laminin α1, α2, and α4 chains described above for Northern blot analysis, plus an endothelial cell–specific probe, flk-1 (Breier et al. 1992).
Adhesion Assays
In vitro cell attachment assays were performed as described previously (Goodman et al. 1987; Schuler and Sorokin 1995), using laminins 8 and 10 prepared as described above, laminin 1 isolated from the Engelbreth-Holm-Swarm (EHS) mouse tumor (Paulsson et al. 1987), and laminin 2 isolated from mouse hearts (Paulsson and Saladin 1989) as substrates. PLP encephalotigenic T cell lines (1 d after restimulation) were washed with PBS and 5 × 104 cells were added per well in 100 μl adhesion buffer (RPMI, 0.5% BSA, 10 mM Hepes, pH 7.5). The number of adherent cells after 30 min of incubation at 37°C without shear stress was determined by colorimetric analysis of lysosomal hexosaminidase (OD405; Landegren 1984). Since leukocytes are activated by chemokines and cytokines are released at sites of inflammation in vivo, attachment assays were performed with nonactivated and PMA-activated (100 ng/ml) cells.
To assess which cellular receptors mediated interactions with extracellular matrix molecules, inhibition studies were carried out using specific antibodies to integrin subunits (see Table ). Cells were preincubated with given concentrations of antibodies against integrin subunits or control antibodies for 30 min before addition to protein-coated microtiter plates. Experiments were carried out with different laminin isoforms plated at a concentration of 20 μg/ml. The experimental procedure was otherwise as described in detail elsewhere (Frieser et al. 1996).
The percentage of cells which bound specifically to the coating substrate was determined as follows: ([OD405 of total bound cells − OD405 of BSA bound cells]/OD405 of 50,000 applied) × 100 = percentage of specific binding.
Results
Basement Membranes Associated with Brain Blood Vessels
In the noninflamed brain the cellular and extracellular matrix layers of the blood vessels appear as a single structure at the light microscope level and are distinguishable only by electron microscopy. However, during early stages of inflammation or in animals with low clinical EAE scores (1+ to 2+), leukocytes accumulate in the perivascular space between the endothelial and the astrocyte endfeet basement membranes, rendering them distinguishable at the light microscope level and allowing the clear identification of the localization of laminin chains (Fig. 1, A–C). In EAE, local inflammations develop around blood vessels, hence only a fraction of blood vessels develop a perivascular cuff, allowing analysis of vessels with and without infiltrates in the same section (Fig. 1B and Fig. C). Double staining with a pan–laminin 1 antibody, which recognizes laminin α1, β1, and γ1 chains equally well and hence any laminin containing at least one of these chains and anti–PECAM-1 (Fig. 1 B) or the leukocyte marker, anti-CD45 (Fig. 1 C), reveals the PECAM-1–positive endothelial cell monolayer and the separate inner endothelial and outer parenchymal basement membranes at sites of infiltration (Fig. 1 B, inset), but not where no infiltration has occurred (Fig. 1B and Fig. C).
Figure 1.
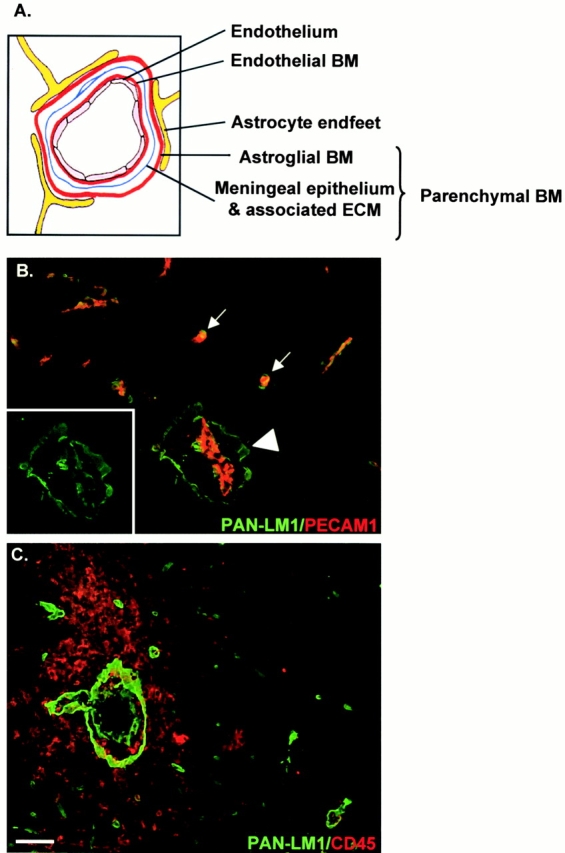
Cell layers and basement membranes which occur in association with blood vessels in the CNS. (A) Schematic representation of blood vessels showing the inner endothelial cell layer and its basement membrane (BM), bordered by epithelial meningeal cells and associated extracellular matrix and outer astroglial basement membrane and astrocyte endfeet. Collectively, the meningeal component and astroglial basement membrane are termed the parenchymal basement membrane. (B) Double staining of EAE brains (clinical scores +1 to +2) for PECAM-1 (red) and pan–laminin 1 antibody (PAN-LM1, green) which recognizes laminin α1, β1, and γ1 chains and thereby many laminin isoforms. Inset shows pan–laminin 1 staining alone. At sites of mononuclear infiltration the internal endothelial cell layer, bordered by its basement membrane, is distinct from the outer parenchymal basement membrane (arrowhead), whereas where no infiltration has occurred these two basement membranes are indistinguishable (arrows). (C) Double staining of EAE brains (clinical scores +3 to +4) with the leukocyte marker, CD45 and pan–laminin 1 antibody, demonstrating infiltrating mononuclear cells accumulating in the perivascular space and penetrating the outer parenchymal basement membrane. Bar, 50 μm.
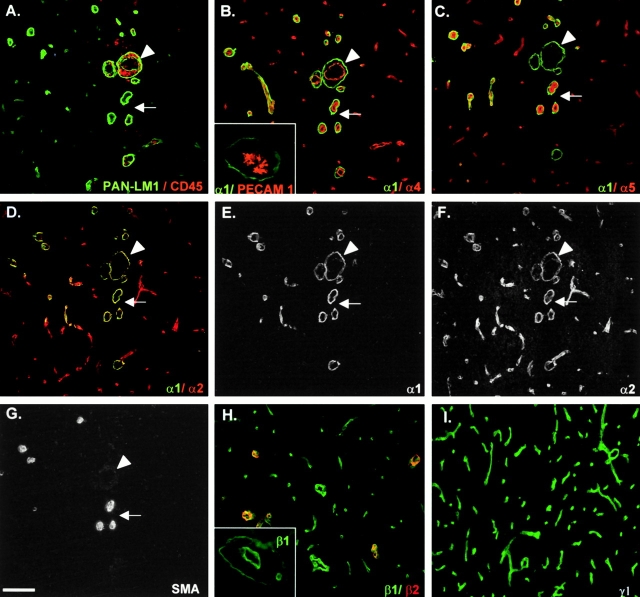
Stainings with specific monoclonal and/or polyclonal antibodies for the five known laminin α chains (α1–5) showed that all α chains except laminin α3 are expressed in association with blood vessels. However, the different laminin α chains were not colocalized in the same basement membrane and they had different cellular origins (Fig. 2 and Fig. 3). Fig. 2 shows consecutive sections of an inflamed brain double stained for pan–laminin 1 and CD45 to define sites of mononuclear infiltration (Fig. 2 A), double stained for individual laminin α chains (Fig. 2, B–F), or stained for α-smooth muscle actin alone (Fig. 2 G). Only laminin α4 and α5 occurred in the inner endothelial cell basement membrane (Fig. 2B and Fig. C), whereas laminin α1 and α2 were restricted to the parenchymal basement membrane (Fig. 2, D–F). The inset in Fig. 2 B shows a double staining for PECAM-1 and laminin α1, revealing clear separation of immunoreactivity, which was also observed for laminin α2/PECAM-1 double stainings. One of the most striking findings was the absence of laminin α5 in the endothelial cell basement membrane where mononuclear infiltration had occurred (compare Fig. 2A and Fig. C). Spinal cords and brains of mice afflicted with EAE with clinical scores of 1+ to 5+ showed essentially the same pattern of results. Recruitment of inflammatory cells, as defined by CD45 reactivity, was consistently localized around blood vessels where only laminin α4 could be detected in the endothelial cell basement membrane (compare Fig. 2A and Fig. B). No infiltrate was observed around blood vessels where both laminin α4 and α5 were detected (compare Fig. 2, A–C). Further, mononuclear infiltration occurred at sites where the parenchymal basement membrane contained both laminin α1 and α2 chains (Fig. 2, D–F), but where no smooth muscle actin reactivity was detectable (Fig. 2 G).
Figure 2.
Confocal microscopy analysis of consecutive sections of EAE brains (clinical score +1 to +2) double stained for pan–laminin 1 (PAN-LM1) and CD45 (A), laminins α1 and α4 (B), laminins α1 and α5 (C), laminins α1 and α2 (D–F), or α-smooth muscle actin (SMA) alone (G). Inset in B shows double staining for PECAM-1 and laminin α1 at sites of monoclear infiltration. Superimposed images are shown in A–D; individual images for laminins α1 and α2 are shown in E and F, respectively. Arrowheads in all sections mark the same blood vessel where mononuclear infiltration has occurred, revealing the presence of laminin α4 alone in the endothelial cell basement membrane. The arrows mark vessels where no infiltration has occurred, revealing the presence of laminin α4 and α5 in the endothelial cell basement membrane and α-smooth muscle actin reactivity (G). Note the presence of laminin α1 and α2 in parenchymal basement membrane only (B–F) and the restricted distribution of laminin α1 (D and E). Double staining for laminin β1 and β2 (H) and for laminin γ1 alone (I) revealed the presence of laminin β1 and γ1 chains in association with all blood vessels, whereas laminin β2 is restricted to larger vessels. Inset in H shows laminin β1 staining at a site of mononuclear infiltration, localizing this chain to endothelial and parenchymal basement membranes. The same pattern of results was observed for laminin γ1. Bar, 80 μm.
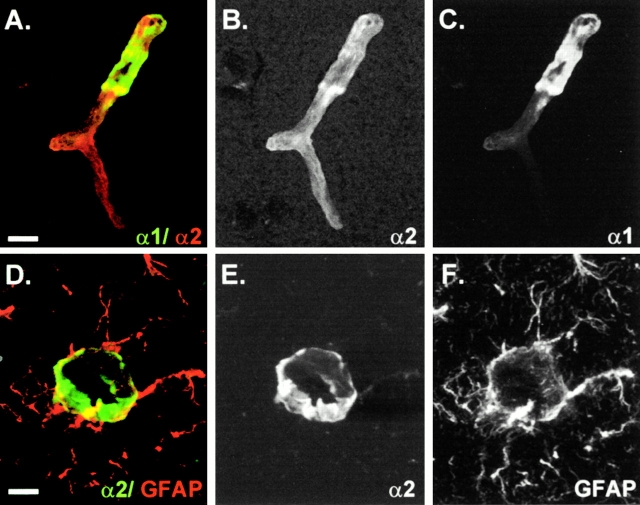
Figure 3.
Confocal microscopy analysis of individual blood vessels in the CNS double stained for laminin α1 and α2 (A–C) or laminin α2 and GFAP (D–F). Superimposed images are shown in A and D; individual images are shown in B, C, E, and F. Note the restricted distribution of laminin α1 which disappears at the precapillary level (A–C). Laminin α2 localizes at the GFAP-positive astrocyte endfeet (D and E). Bars: (A–C) 8 μm; (D–F) 12 μm.
Laminin α2 immunoreactivity was more widespread than that of laminin α1 and was detected in association with all blood vessels within the brain (Fig. 2, D–F). This is well illustrated in Fig. 3A–C, which shows laminin α1/α2 double staining of a single microvessel, revealing the more extensive staining for laminin α2 and the restriction of laminin α1 to a portion of the vessel. The close association of the laminin α2 and GFAP (Fig. 3, D–F) suggests that laminin α2 is expressed by astrocytes and that the protein is deposited at their endfeet. Laminin α1 chain immunoreactivity was restricted to vessels with diameters >10 μm throughout the CNS (Fig. 2D and Fig. E) and was contiguous with pia mater epithelium covering the brain, disappearing abruptly in the precapillary region of arteries and veins (Fig. 3A and Fig. C). No laminin α1 staining was detected in association with capillaries (Fig. 2 E).
To define the laminin isoforms localized in endothelial and parenchymal basement membranes, stainings were also performed for laminin β1, β2, and γ1 chains, revealing the presence of laminin β1 (Fig. 2 H) and γ1 (Fig. 2 I) in endothelial and parenchymal basement membranes (Fig. 2 H, inset), whereas laminin β2 was restricted to the smooth muscle layer of larger blood vessels (Fig. 2 H). This suggests that laminin 8 (α4β1γ1) and 10 (α5β1γ1) are the main laminin isoforms in the endothelial cell basement membrane, whereas laminin 1 (α1β1γ1) and 2 (α2β1γ1) occur in the parenchymal basement membrane.
Cellular Origins of Laminin Chains
In situ hybridization experiments were performed to define the cellular origin of the laminin α chains. The low turnover of most laminin α chains in adult tissues (data not shown) made it necessary to compare the expression patterns for the individual laminin α chains in embryonic and/or newborn brains (Fig. 4). In E16, E18, and, more weakly, in newborn mouse brains, laminin α1 mRNA was not expressed by endothelial cells, but rather by the leptomeningeal cells of the pia mater that encase the brain and are infolded from the brain surface (Fig. 4A and Fig. B), correlating well with the protein distribution data. In situ hybridization of E16 and E18 mouse brains revealed a strong laminin α2 mRNA signal in the meninges (data for E16 is shown in Fig. 4 C), which is most likely to be a glial product (Sixt, M., and L. Sorokin, manuscript in preparation). Laminin α4 mRNA was expressed in all endothelial cells of embryonic, newborn, and adult vessels (data for E16 brain are shown in Fig. 4E and Fig. F), suggesting a high turnover of this molecule even in the mature blood vessel basement membrane. Our previous studies have shown that laminin α5 mRNA appears in endothelium of the brain (and other tissues) only 3–4 wk after birth and remains weak in the adult brain endothelium (Sorokin et al. 1997a).
Figure 4.
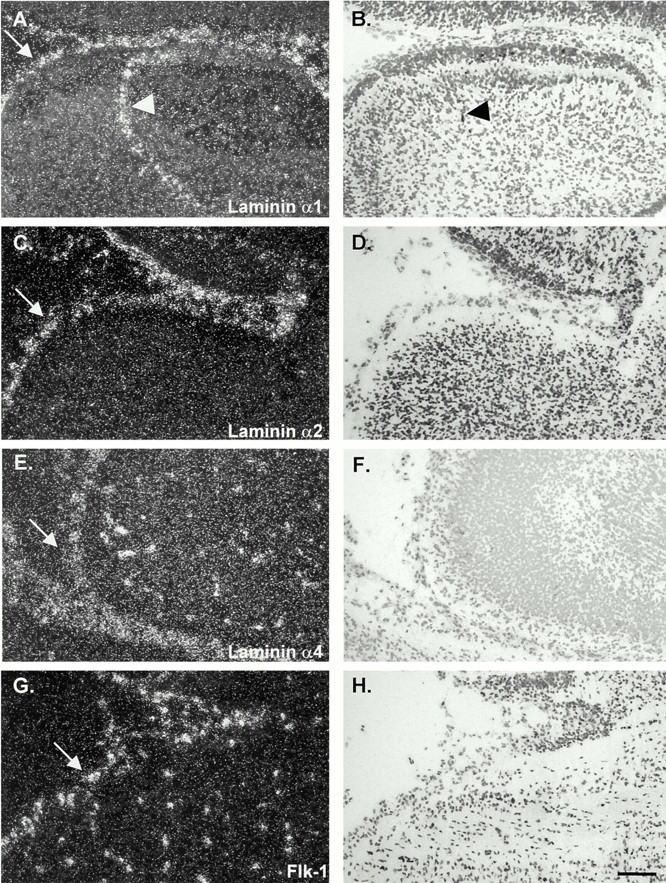
In situ hybridization of consecutive sections of E16 embryonic mouse brains with probes specific for laminin α1 (A and B), α2, (C and D), α4 (E and F), and an endothelial cell–specific probe, flk-1 (G and H). Data shown are for similar areas, encompassing meninges (arrows) and a portion of the cortex of the cerebellum. Similar results were observed for E18 embryos and, in the case of laminin α4, also for newborn and adult tissues. Laminin α1 mRNA was expressed by the leptomeningeal cells of the pia mater that encase the brain and are infolded from the brain surface (arrowheads in A and B); laminin α2 mRNA was restricted to the meninges (C and D), whereas laminin α4 mRNA was detected in endothelial cells within and outside the brain (E and F) in a similar pattern to that observed with the endothelial cell–specific probe, flk-1 (G and H). Bar, 25 μm.
Laminin Receptor Expression in Normal and Inflamed Blood Vessels
Immunoreactivity for the major laminin-binding receptors (α6β1, α3β1, α7β1, α6β4, and dystroglycan; Delwel and Sonnenberg 1996; Talts et al. 1999; Kikkawa et al. 2000; Kortesmaa et al. 2000) was analyzed in inflamed and noninflamed blood vessels. Due to the absence of antibodies to α-dystroglycan which function well in immunofluorescence of tissues other than skeletal muscle, it was necessary to utilize a β-dystroglycan–specific antibody for investigations of α-dystroglycan localization. β- and α-dystroglycan are products of the same gene and colocalize in most tissues (Henry and Campbell 1998). However, there are reports of β-dystroglycan expression in the absence of α-dystroglycan (Saito et al. 1999; Losasso et al. 2000).
Integrin α6 and β-dystroglycan were prominent in CNS blood vessels (Fig. 5, A–C), whereas no staining was observed for integrin α3, α7, and β4 subunits (data not shown; Pinkstaff et al. 1999). Confocal microscopy revealed a mutually exclusive expression of integrin α6 and β-dystroglycan on all blood vessels except capillaries (Fig. 5, A–C). It was not possible to perform double staining for integrin α6 and PECAM-1, as both were rat antibodies; however, comparison of the staining patterns for the two molecules revealed identical patterns. This suggests expression of integrin α6 on PECAM-1–positive endothelium in close association with laminin α4 and α5, but not with the laminin α1 and α2 chains of the parenchymal basement membranes (double staining for laminin α1 and integrin α6 is shown in Fig. 5, D–F). This pattern was even more distinct in EAE brains, where the presence of a perivascular cuff allowed clear identification of integrin α6 expression on endothelium in close association with the laminin α4– and/or α5–containing basement membrane (data for integrin α6 is shown in Fig. 5 F, inset). In contrast, β-dystroglycan immunostaining was restricted to astrocyte endfeet, in close association with GFAP staining (not shown) and laminin α2 immunoreactivity, and was distinct from that of laminin α4 and α5 (reconstruction of serial optical sections for laminin α4/β-dystroglycan double staining is shown in Fig. 5, G–I).
Figure 5.
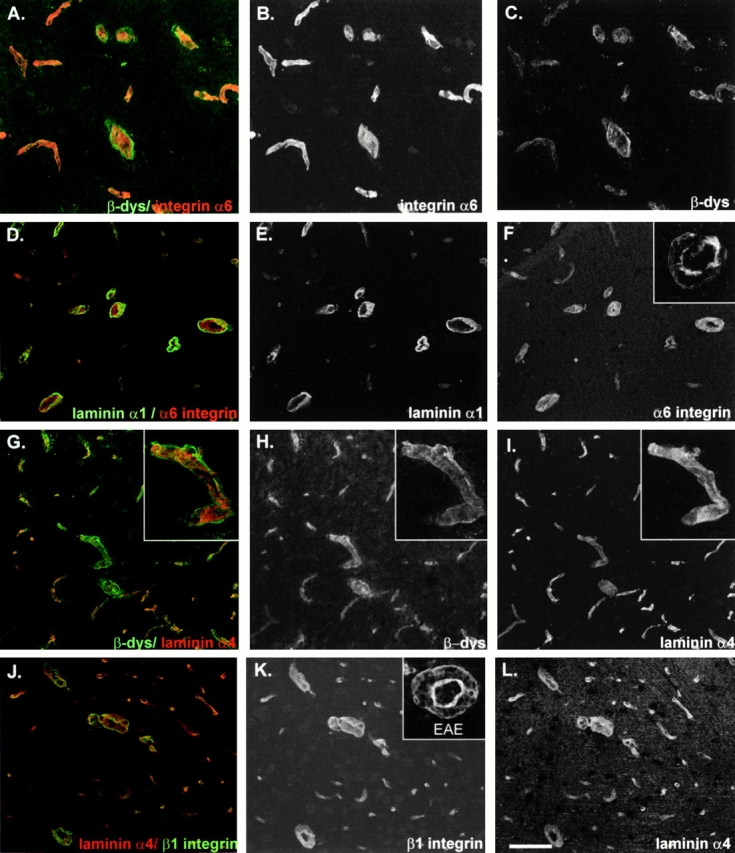
Confocal microscopy analysis for the expression of integrin α6 and β-dystroglycan in mouse brain blood vessels. A–C show double staining for integrin α6 and β-dystroglycan (β-dys) showing mutually exclusive distributions. D–F show double staining for integrin α6 and the parenchymal basement membrane component, laminin α1, localizing integrin α6 on the inner endothelial cell layer; inset in F shows integrin α6 expression at sites of mononuclear infiltration revealing discontinuous staining. G–I show double staining for β-dystroglycan and the endothelial cell basement membrane–specific laminin α4, demonstrating β-dystroglycan expression on astrocyte endfeet; the insets in G–I show a higher magnification of a single vessel. J–L show double staining for integrin β1 and laminin α4 showing overlapping distribution patterns; the inset in K shows integrin β1 expression at sites of mononuclear infiltration, demonstrating concentration on the inner endothelial cell layer, but also on infiltration cells, with weaker expression on the astrocyte endfeet. Bars: (A–C) 80 μm; (D–L) 100 μm.
At early stages of EAE and in control brains, all blood vessels showed an intense and continuous staining for integrin α6 (Fig. 5D and Fig. F), whereas at advanced stages of EAE integrin α6 immunoreactivity on endothelium was significantly less intense and discontinuous (Fig. 5 F, inset). β-Dystroglycan did not appear to be altered in its expression pattern or intensity. However, at sites of leukocyte infiltration this was difficult to assess because of high background staining of this mouse monoclonal antibody in EAE brains (data not shown).
Integrin β1 immunoreactivity was more widespread than that of integrin α6 as it occurred on both endothelium and astrocyte endfeet (compare Fig. 5F and Fig. K), and was strong on endothelium and weaker on astrocyte endfeet (Fig. 5, J–L). In EAE brains, venules with perivascular inflammatory cells staining for integrin β1 were not altered (Fig. 5 K, inset).
Northern Blot Analysis—Whole Brains
To investigate the possibility that laminin α5 mRNA is downregulated in EAE brains, Northern blot analysis was performed on whole brains of control and EAE mice (clinical score 4+/ 5+) using laminin α1–, α2–, α4–, and α5–specific probes. Laminin α1 and α2 mRNA were not detectable in either control or EAE brains, whereas the 12-kb laminin α5 mRNA was detectable after 6 d of exposure and a distinct 6.5-kb laminin α4 mRNA signal was seen after 24 h of exposure (Fig. 6 A). No difference was apparent between control and EAE brains.
Figure 6.
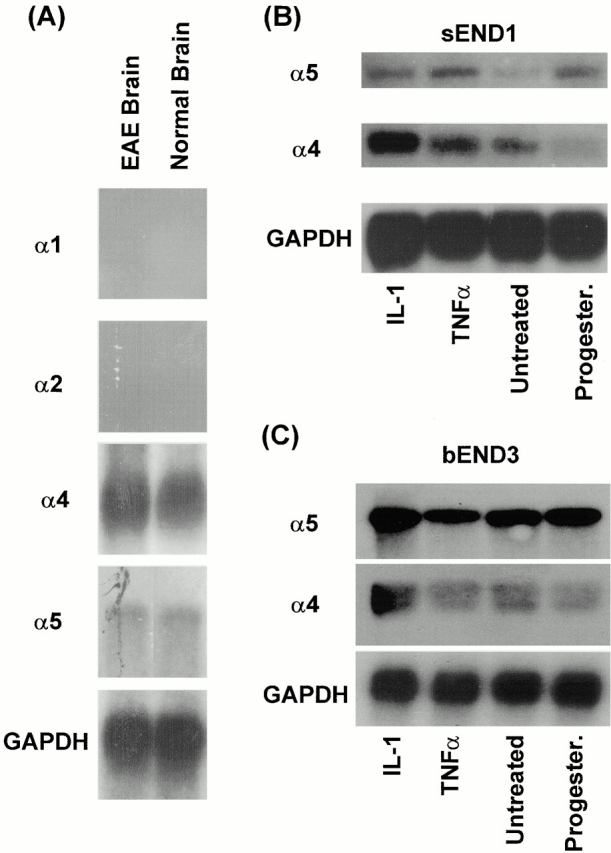
Northern blot analysis for the expression of laminin α1, α2, α4, and α5 mRNA in control and EAE (clinical score 4+/5+) mouse brains (A), and for laminins α4 and α5 mRNA in sEND1 (B) and bEND3 (C) mouse endothelial cell lines. Endothelial cell lines were either untreated, activated with IL-1 or transforming growth factor α (TNFα), or treated with hydroxymethylprogesterone (Progester.). Data in A for laminin α1 and α2 is after 2 wk of exposure, for laminin α5 after 6 d of exposure, and for laminin α4 after 24 h of exposure.
Northern Blot Analyses of Cultured Endothelial Cells
To test whether laminin isoform expression in microvascular endothelium is regulated by proinflammatory cytokines present at sites of mononuclear infiltration (for review see Sedgwick et al. 2000), mouse brain capillary–derived (bEND3) and vein-derived (sEND1) endothelial cell lines were investigated by Northern blot analysis.
Consistent with the in vivo results, both endothelial cell lines expressed laminin α4 and α5 mRNA (Fig. 6B and Fig. C). Laminin α4 mRNA was upregulated in sEND1 after activation with TNF-α and IL-1 (Fig. 6 B), whereas treatment with the angiostatic agent, hydroxymethylprogesterone, decreased expression (Fig. 6 B). Interestingly, laminin α5 mRNA was also upregulated by TNF-α and to lesser extent by IL-1, whereas the 12-kb laminin α5 mRNA increased compared with the controls after hydroxymethylprogesterone treatment (Fig. 6 B). bEND3 responded less intensely than sEND1 to either TNF or IL-1 activation, consistent with its capillary origin (Fig. 6 C).
We have shown in a previous study that bEND3 and sEND1 do not express laminin α1 mRNA (Sorokin et al. 1994); experiments performed here with specific probes for laminin α2 and α3 mRNA also revealed no signal, even after 6 d of exposure (data not shown).
Adhesion of Encephalitogenic T Cell Lines to Purified Laminins
To investigate whether encephalitogenic T cells were capable of interacting with endothelial cell laminins, SJL/N mouse–derived T cell lines specific for the encephalitogenic peptide (amino acids 139–153) of PLP were tested for adhesion to mouse laminins 1, 2, and 8, and human laminin 10. T cells adhered weakly to the laminin α5 chain containing laminin 10 (11% of total cells added) and exhibited minimal binding to the laminin α4 chain containing laminin 8 (5%; Fig. 7 A). The parenchymal basement membrane laminins 1 and 2 were not adhesive, even at high coating concentrations (Fig. 7 A).
Figure 7.
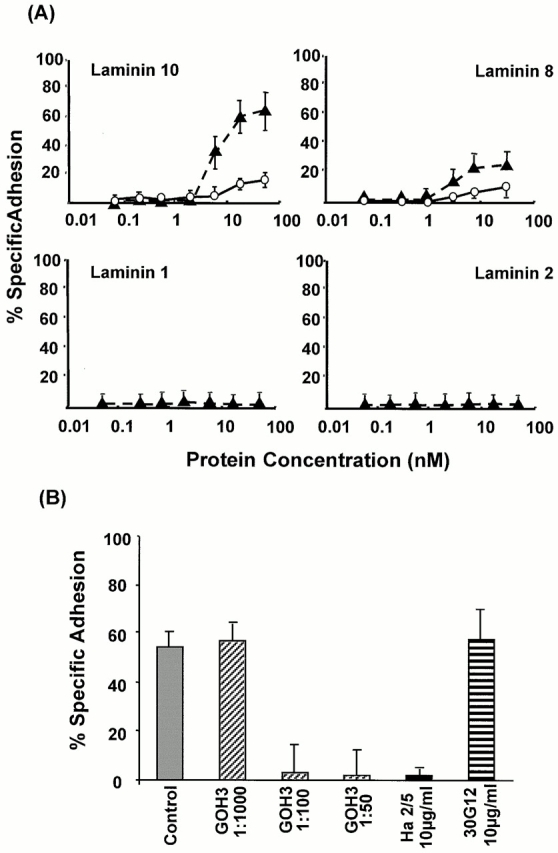
(A) Specific binding of nonactivated (○) and PMA-activated (▴) PLP-specific, encephalitogenic T cell lines to increasing concentrations of the endothelial cell basement membrane laminins 10 and 8, and the parenchymal basement membrane laminins 1 and 2. Saturable T cell binding occurred only to laminins 10 and 8; binding to laminins 1 and 2 could not be induced, even under maximal integrin activation conditions (10 mM Mn2+). (B) Binding to 30 nM laminin 10 was completely inhibited by antiintegrin α6 (GoH3) or antiintegrin β1 (Ha2/5), whereas anti-CD45 antibody (30G12) had no effect. The same pattern of results was observed for laminin 8. Values represent means of at least six experiments ± SD.
Upon stimulation of T cells with PMA, encephalitogenic T cells bound principally to laminin 10 (60%) and showed increased binding to laminin 8 (20%; Fig. 7 A). However, no specific binding to laminins 1 or 2 could be induced, even under conditions that maximally activate integrins (10 mM Mn2+). The differential adhesion of the T cells to the different laminin isoforms was not due to either the quality of the laminin preparations or their ability to bind to the culture plates, as C2C12 myoblasts where found to bind significantly (50–100% cell adhesion) to all four laminin isoforms (Karosi, S., O. Wendler, and L. Sorokin, manuscript in preparation). To ensure that the different purified laminin isoforms bound with the same efficiency to the plastic plates used in cell adhesion assays, the laminins were labeled with 125I and coated under the same conditions as applied for cell binding assays. It was shown that all four laminin isoforms used in this study bound to the plastic surfaces in a comparable and concentration-dependent manner (data not shown).
Adhesion of encephalitogenic T cells to laminins 8 and 10 was inhibited in a concentration-dependent manner with the function-blocking antibodies to integrin α6 chain (GoH3) and the mouse integrin β1 chain (Ha2/5; data for laminin 10 is shown in Fig. 7 B), indicating α6β1-mediated binding to both laminin 8 and 10.
Discussion
We have shown previously that different mouse endothelial cell lines express predominantly either laminin α4 or α5 and that this expression pattern can be altered by the growth or activation state of the cells (Sorokin et al. 1994, Sorokin et al. 1997a; Frieser et al. 1997), suggesting that the laminin isoforms containing these chains have different functions in endothelial cell basement membranes. The data presented here support this hypothesis. Further, we have shown for the first time that mononuclear cells infiltrating the CNS during the course of EAE encounter two biochemically and functionally distinct basement membranes, the endothelial and parenchymal basement membranes. To date, most studies of EAE have concentrated on leukocyte–endothelial cell interactions, revealing α4β1 and vascular cell adhesion molecule 1 as significant players (Engelhardt et al. 1998; Laschinger and Engelhardt 2000), or the subsequent penetration of the CNS, with little consideration that after penetrating the endothelial cell monolayer infiltrating cells still face two formidable and distinct extracellular barriers. The localization of a perivascular cuff of infiltrating mononuclear cells surrounding blood vessels at early stages of EAE allowed clear identification of the endothelial cell and parenchymal basement membranes, demonstrating that endothelial cell basement membranes contain laminin α4 and/or α5 chains which are produced by endothelial cells, whereas parenchymal basement membranes contain laminin α1 and α2 produced by leptomeningeal cells and astrocytes, respectively (see Fig. 8 for summary of data). Interestingly, mononuclear recruitment in the perivascular space correlated with the laminin composition of the endothelial cell basement membrane: recruitment of T cells occurred exclusively around endothelial cell basement membranes containing laminin α4, but not laminin α5, suggesting that laminins containing the laminin α4 chain are permissive for T cell infiltration, whereas those containing the laminin α5 chains are restrictive or inhibitory (Fig. 8). In vitro studies demonstrated that this is probably due to high affinity integrin α6β1–mediated binding of encephalitogenic T cells to laminin 10, which has been shown in other cells not to be conducive to migration, whereas the labile, low affinity binding to laminin 8 is more likely to support cell migration (Goodman et al. 1989; von der Mark et al. 1999; Pedraza et al. 2000). Whether this is the case with the encephalitogenic T cell lines is currently under investigation.
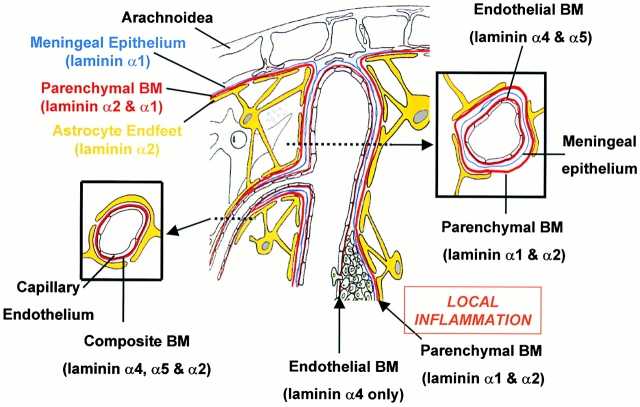
Figure 8.
Illustration of cell layers, basement membranes (BM), and their laminin composition of CNS blood vessels with and without an inflammatory cuff. Larger blood vessels consist of an inner endothelial cell layer with a basement membrane (containing laminins α4 and α5), bordered by the meningeal epithelium and its basement membrane (containing laminin α1) and an outer astroglial basement membrane (containing laminin α2) and astrocyte endfeet. The meningeal and astroglial basement membranes are collectively termed the parenchymal basement membrane as they delineate the border to the brain parenchyma. Only at sites of local inflammation are the endothelial and parenchymal basement membranes distinguishable and define the inner and outer limits of the perivascular space where leukocytes accumulate before infiltrating the brain parenchyma. Examination of such sites demonstrates that mononuclear infiltration occurs across endothelial basement membranes containing only the laminin α4 and bordered by a parenchymal basement membrane containing laminin α1 and α2. The basement membrane of microvessels where no epithelial meningeal contribution occurs appear to have a composite basement membrane containing the endothelial cell laminins, laminin α4 and α5, and laminin α2 produced by the astrocytes and deposited at their endfeet.
Integrin α6β1 has been reported to be a specific receptor for laminin 1 on several cell types, including T cells (Shimizu et al. 1990; van de Wiel-van Kemenade et al. 1992), and in some cells also mediates binding to laminin 2 (Chang et al. 1995). However, despite constitutive surface expression of this integrin, the encephalitogenic T cell lines could not be induced to bind the parenchymal basement membrane laminins 1 and 2. Even maximal cell (PMA) and integrin (Mn2+) activation conditions were ineffective in inducing binding to either of these laminins, demonstrating that the presence of the α6β1 receptor is insufficient for ligand binding. The data also suggest that T cell transmigration of the parenchymal basement membrane is fundamentally different from transmigration of the endothelial cell basement membrane. This has been suggested by the results of several other studies which have shown that additional activating agents or comigrating bystander cells are necessary for the final entry of T cells into the brain parenchyma. In macrophage-depleted (Tran et al. 1998) and TNF-α−/− (Körner et al. 1997; Riminton et al. 1998) mice, infiltrating T cells cross the endothelium and its basement membrane, but remain in the perivascular space even during peak stages of EAE. Furthermore, GFAP knockout mice, which show a disrupted association of the astrocyte endfeet with blood vessels, exhibit an early spreading of the infiltrating cells into the CNS parenchyma during EAE (Liedtke et al. 1998). Our data suggest that the parenchymal barrier breaks down in lesions only at advanced stages of inflammation in a process which is likely to be dependent on the proteolytic activity of MMPs. MMPs have been extensively studied in EAE (Chandler et al. 1997; Madri and Graesser 2000), demonstrating a significant role for MMPMT-1/MMP2 (Graesser et al. 1998, Graesser et al. 2000) and MMP9 (Dubois et al. 1999; Lichtinghagen et al. 1999; Hartung and Kieseier 2000) in T cell entry into and residency in the parenchyma of the CNS, as well as in demyelination. Although protease inhibitors have been shown to reduce the severity or delay the onset of EAE, to date no protease inhibitor has been shown to completely ablate T cell migration into the perivascular space or the brain parenchyma (Clements et al. 1997; Graesser et al. 2000). Until now it was not clear that emigrating T cells face more than just the subendothelial cell basement membrane. Therefore, the results of the MMP inhibitor experiments were interpreted as reduced transmigration across the endothelial cell basement membrane. However, they can equally well be explained by inhibition of transmigration across the parenchymal basement membrane. It now remains to be determined whether the main targets of MMP activity are components of the endothelial cell or the parenchymal basement membrane.
One of the major laminin receptors, integrin α6β1, was localized predominantly on the endothelial cells, where it is likely to mediate interactions with the endothelial cell laminins 8 and 10, whereas astrocyte endfeet appear to utilize a different receptor for interactions with the parenchymal laminins. β-Dystroglycan occurred predominantly on astrocyte endfeet where no α6 integrin was detected, suggesting that α-dystroglycan may be the laminin receptor on the astrocyte endfeet mediating binding to the parenchymal laminins 1 and 2. We have tested two α-dystroglycan antibodies, IIH6 and VIA4-1, both of which showed extremely weak staining of the CNS blood vessels (data not shown). Hence, it cannot be excluded from the data represented here that β-dystroglycan alone occurs on the astrocyte endfeet as has been shown in other tissues (Saito et al. 1999; Losasso et al. 2000). We and others have shown that during the course of EAE, integrin α6β1 is downregulated on the endothelial cells at sites of infiltration (Defilippi et al. 1992; Sobel et al. 1998). This, in combination with our observed high turnover of laminin α4 in endothelial cell basement membranes in the brain and the selective upregulation of laminin α4 expression by cytokines such as TNF-α, which have been shown to play a role in EAE, may lead to a loosening of the endothelial cell–basement membrane interaction, resulting in the reported “rounding up” of endothelial cells observed at sites of T cell infiltration in EAE (Wolburg et al. 1999) and further facilitating the infiltration process.
The clear identification of the endothelial cell and parenchymal basement membranes possible in the present study and characterization of their laminin isoform content clarifies confusion in the literature concerning laminin distribution in blood vessels in the CNS. Both laminin α1 and α2 have been reported to be expressed by brain blood vessels in noninflamed brains (Sanes et al. 1990; Jucker et al. 1996; Virtanen et al. 2000). However, in the absence of an inflammation, as shown here, the endothelial and parenchymal basement membranes lie in such close proximity that at the light microscopy level they may be mistaken for a single structure, leading to the misconception that laminins α1 and α2 are present in endothelial cell basement membranes. Further discrepancies may also result from the fact that the laminin α1 monoclonal antibodies used in earlier studies were subsequently shown to recognize the laminin α5 chain (Virtanen et al. 1995; Jucker et al. 1996; Tiger et al. 1997).
The process of extravasation in the CNS is frequently described as passage through the blood–brain barrier (BBB; Noseworthy 1999), a structure characterized by interendothelial cell tight junctions and induced and maintained by the close association between microvascular endothelial cells and astrocyte endfeet processes (Janzer and Raff 1987; Wolburg and Risau 1995). However, our data clearly showed that T cell transmigration never occurred at microvascular sites, but rather at postcapillary venules where endothelial cells were separated from the astrocyte endfeet by two basement membranes and at least one cell layer (leptomeningeal cells). Therefore, this site does not fulfill the above described characteristics of the BBB. The association of epithelial leptomeningeal cells with veins and postcapillary venules has not been reported previously, but may be related to the nature of the studies performed to date: ultrastructural studies on human tissues have shown that cerebral arteries and arterioles are ensheeted by leptomeningeal cells (Alcolado et al. 1988; Zhang et al. 1990), but such studies cannot easily distinguish between vessel types. Only through the use of laminin chain–specific antibodies and our knowledge of their cellular origins, in combination with examination of inflamed brain tissue, was it possible to specifically define the cell layers and basement membranes associated with sites of cellular infiltration. It now remains to be determined how the leptomeningeal sheet appears at the ultrastructural level in postcapillary venules and veins in the mouse brain, and the nature of the BBB at such sites.
In conclusion, our data enforce the concept that T cell emigration of the endothelial cell basement membrane is distinct from transmigration of the parenchymal basement membrane. For the first time, two biochemically distinct basement membranes encountered by emigrating T cells in EAE have been defined, and not only laminin α4 in the endothelial cell basement membrane and integrin α6 on emigrating T cells, but also the endothelium of inflamed vessels, have been identified as key players in transmigration of the endothelial basement membrane.
Acknowledgments
The authors thank Stefanie Karosi for careful and critical reading of the manuscript and Monika Bruckner for expert technical assistance. We are particularly grateful to Winfried Neuhuber for help with confocal microscopy and interpretation of the data.
This work was supported by Deutsche Forschungsgemeinschaft grants So285/1-3 and So285/1-4 to L.M. Sorokin.
Footnotes
Abbreviations used in this paper: BBB, blood–brain barrier; CNS, central nervous system; EAE, experimental autoimmune encephalomyelitis; GADPH, glyceraldehyde-3-phosphate dehydrogenase; GFAP, glial fibrillar acidic protein; IL, interleukin; MMP, matrix metalloproteinase; PECAM, platelet endothelial cell adhesion molecule; PLP, protein–lipid–protein; TNF, tumor necrosis factor.
References
- Alcolado R., Weller R.O., Parrish E.P., Garrod D. The cranial arachnoid and piamater in mananatomical and ultrastructural observations. Neuropathol. Appl. Neurobiol. 1988;14:1–17. doi: 10.1111/j.1365-2990.1988.tb00862.x. [DOI] [PubMed] [Google Scholar]
- Aumailley M., Smyth N. The role of laminins in basement membranes. J. Anat. 1998;193:1–21. doi: 10.1046/j.1469-7580.1998.19310001.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boado R.J., Pardridge W.M. Differential expression of alpha-actin mRNA and immunoreactive protein in brain microvascular pericytes and smooth muscle cells. J. Neurosci. Res. 1994;39:430–435. doi: 10.1002/jnr.490390410. [DOI] [PubMed] [Google Scholar]
- Breier G., Albrecht U., Sterrer S., Risau W. Expression of vascular endothelial growth factor during embryonic angiogenesis and endothelial cell differentiation. Development. 1992;114:521–532. doi: 10.1242/dev.114.2.521. [DOI] [PubMed] [Google Scholar]
- Chandler S., Miller K.M., Clements J.M., Lury J., Corkill D., Anthony D.C.C., Adams S.E., Gearing A.J.H. Matrix metalloproteinases, tumor necrosis factor and multiple sclerosisan overview. J. Neuroimmunol. 1997;72:155–161. doi: 10.1016/s0165-5728(96)00179-8. [DOI] [PubMed] [Google Scholar]
- Chang A.C., Salomon D.R., Wadsworth S., Hong M.J., Mojcik C.F., Otto S., Shevach E.M., Coligan J.E. Alpha 3 beta 1 and alpha 6 beta 1 integrins mediate laminin/merosin binding and function as costimulatory molecules for human thymocyte proliferation. J. Immunol. 1995;154:500–510. [PubMed] [Google Scholar]
- Clements J.M., Cossins J.A., Wells G.M.A., Corkill D.J., Helfrich K., Wood L.M., Pigott R., Stabler G., Ward G.A., Gearing A.J.H., Miller K.M. Matrix metalloproteinase expression during experimental autoimmune encephalomyelitis and effects of a combined matrix metalloproteinase and tumour necrosis factor-α inhibitor. J. Neuroimmunol. 1997;74:85–94. doi: 10.1016/s0165-5728(96)00210-x. [DOI] [PubMed] [Google Scholar]
- Cross A.H., O'Mara T., Raine C.S. Chronologic localization of myelin-reactive cells in the lesions of relapsing EAEimplications for the study of multiple sclerosis. Neurology. 1993;43:1028–1033. doi: 10.1212/wnl.43.5.1028. [DOI] [PubMed] [Google Scholar]
- Defilippi P., Silengo L., Tarone G. α6 β1 integrin (laminin receptor) is down-regulated by tumor necrosis factor alpha and interleukin-1 beta in human endothelial cells. J. Cell Biol. 1992;267:18303–18307. [PubMed] [Google Scholar]
- Delwel G.O., Sonnenberg A. Laminin isoforms and their integrin receptors. In: Horton M.A., editor. Adhesion Receptors as Therapeutic Targets. CRC Press; London: 1996. pp. 9–36. [Google Scholar]
- Dubois B., Masure S., Hurtenbach U., Paemen L., Heremans H., van der Oord J., Sciot R., Meinhardt T., Hämmerling G., Opdenakker G., Arnold B. Resistance of young gelatinase B-deficient mice to experimental autoimmune encephalomyelitis and necrotizing tail lesions. J. Clin. Invest. 1999;104:1507–1515. doi: 10.1172/JCI6886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Durbeej M., Fecker L., Hjalt T., Zhang H.Y., Salmivirta K., Klein G., Timpl R., Sorokin L., Ebendal T., Ekblom P., Ekblom M. Expression of laminin alpha 1, alpha 5 and beta 2 chains during embryogenesis of the kidney and vasculature. Matrix Biol. 1996;15:397–413. doi: 10.1016/s0945-053x(96)90159-6. [DOI] [PubMed] [Google Scholar]
- Echtermeyer F., Schöber S., Pöschl E., von der Mark H., von der Mark K. Specific induction of cell motility on laminin by α7 integrin. J. Biol. Chem. 1996;271:2071–2075. doi: 10.1074/jbc.271.4.2071. [DOI] [PubMed] [Google Scholar]
- Ehrig K., Leivo I., Argraves W.S., Ruoslahti E., Engvall E. Merosina tissue-specific basement membrane protein is a laminin-like protein. Proc. Natl. Acad. Sci. USA. 1990;87:3264–3268. doi: 10.1073/pnas.87.9.3264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ekblom, P., and R. Timpl. 1996. The Laminins. P. Ekblom and R. Timpl, editors. Harwood Academic Publishers, Amsterdam. 321 pp.
- Engelhardt B. Lymphocyte trafficking through the central nervous system. In: Hamann A., editor. Adhesion Molecules and Chemokines in Lymphocyte Trafficking. Harwood Academic Publishers; Amsterdam: 1997. pp. 173–200. [Google Scholar]
- Engelhardt B., Vestweber D., Hallmann R., Schulz M. E- and P-selectin are not involved in the recruitment of inflammatory cells across the blood-brain barrier in experimental autoimmune encephalomyelitis. Blood. 1997;90:4459–4472. [PubMed] [Google Scholar]
- Engelhardt B., Laschinger M., Schulz M., Samulowitz U., Vestweber D., Hoch G. The development of experimental autoimmune encephalomyelitis in the mouse requires α4-integrin but not α4β7-integrin. J. Clin. Invest. 1998;102:2096–2105. doi: 10.1172/JCI4271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frieser M., Hallmann R., Johansson S., Vestweber D., Goodman S.L., Sorokin L. Mouse polymorphonuclear granulocyte binding to extracellular matrix molecules involves beta 1 integrins. Eur. J. Immunol. 1996;26:3127–3136. doi: 10.1002/eji.1830261245. [DOI] [PubMed] [Google Scholar]
- Frieser M., Nöckel H., Pausch F., Röder C., Hahn A., Deutzmann R., Sorokin L.M. Cloning of the mouse laminin alpha 4 geneexpression in a subset of endothelium. Eur. J. Biochem. 1997;246:727–735. doi: 10.1111/j.1432-1033.1997.t01-1-00727.x. [DOI] [PubMed] [Google Scholar]
- Galliano M.-F., Aberdam D., Aguzzi A., Ortonne J.-P., Meneguzzi G. Cloning and complete primary structure of the mouse laminin α3 chain. J. Biol. Chem. 1995;270:21820–21826. doi: 10.1074/jbc.270.37.21820. [DOI] [PubMed] [Google Scholar]
- Gardinier M.V., Amiguet P., Lininton C., Matthieu J.M. Myelin/oligodendrocyte glycoprotein is a unique member of the immunoglobin superfamily. J. Neurosci. Res. 1992;33:177–187. doi: 10.1002/jnr.490330123. [DOI] [PubMed] [Google Scholar]
- Goodman S.L., Deutzmann R., von der Mark K. Two distinct cell-binding domains in laminin can independently promote non-neuronal cell adhesion and spreading. J. Cell Biol. 1987;105:589–598. doi: 10.1083/jcb.105.1.589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goodman S.L., Deutzmann R., Nurcombe V. Locomotory competence and laminin-specific cell surface binding sites are lost during myoblast differentiation. Development. 1989;105:795–802. doi: 10.1242/dev.105.4.795. [DOI] [PubMed] [Google Scholar]
- Graesser D., Mahooti S., Haas T., Davis S., Clark R.B., Madri J.A. The interrelationship of α4 integrin and matrix metalloproteinase-2 in the pathogenesis of experimental autoimmune encephalomyelitis. Lab. Invest. 1998;78:1445–1458. [PubMed] [Google Scholar]
- Graesser D., Mahooti S., Madri J. Distinct roles for matrix metaloproteinase-2 and α4 integrin in autoimmune T cell extravasation and residency in brain parenchyma during experimental autoimmune encepholmyelitis. J. Neuroimmunol. 2000;109:121–131. doi: 10.1016/s0165-5728(00)00275-7. [DOI] [PubMed] [Google Scholar]
- Hartung H.-P., Kieseier B.C. The role of matrix metalloproteinases in autoimmune damage to the central and peripheral nervous system. J. Neuroimmunol. 2000;107:140–147. doi: 10.1016/s0165-5728(00)00225-3. [DOI] [PubMed] [Google Scholar]
- Henry M.D., Campbell K.P. A role for dystroglycan in basement membrane assembly. Cell. 1998;95:859–870. doi: 10.1016/s0092-8674(00)81708-0. [DOI] [PubMed] [Google Scholar]
- Hunter D.D., Shah V., Merlie J.P., Sanes J.R. A laminin-like adhesive protein concentrated in the synaptic cleft of the neuromuscular junction. Nature. 1989;338:229–234. doi: 10.1038/338229a0. [DOI] [PubMed] [Google Scholar]
- Janzer R.C., Raff M.C. Astrocytes induce blood-brain barrier properties in endothelial cells. Nature. 1987;325:253–257. doi: 10.1038/325253a0. [DOI] [PubMed] [Google Scholar]
- Jucker M., Tian M., Norton D.D., Sherman C., Kusiak J.W. Laminin α2 is a component of brain capillary basement membranereduced expression in dystrophic dy mice. Neuroscience. 1996;71:1153–1161. doi: 10.1016/0306-4522(95)00496-3. [DOI] [PubMed] [Google Scholar]
- Kikkawa Y., Sanzen N., Fujiwara H., Sonneberg A., Sekiguchi K. Integrin binding specificty of laminin 10/11laminin 10/11 are recognized by α3β1, α6β1 and α6β4 integrins. J. Cell Sci. 2000;113:869–876. doi: 10.1242/jcs.113.5.869. [DOI] [PubMed] [Google Scholar]
- Koch M., Olson P., Albus A., Jin W., Hunter D.D., Brunken W., Burgeson R.E., Champliaud M.-F. Characterization and expression of the laminin gamma 3 chaina novel non-basement membrane-associated, laminin chain. J. Cell Biol. 1999;145:605–617. doi: 10.1083/jcb.145.3.605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Körner H., Riminton D.S., Strickland D.H., Lemckert F.A., Pollard J.D., Sedgwick J.D. Critical points of tumor necrosis factor action in central nervous system autoimmune inflammation defined by gene targeting. J. Exp. Med. 1997;186:1585–1590. doi: 10.1084/jem.186.9.1585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kortesmaa J., Yurchenco P., Tryggvason K. Recombinant laminin-8 (alpha4, beta1, gamma1). Production, purification, and interactions with integrins. J. Biol. Chem. 2000;275:14853–14859. doi: 10.1074/jbc.275.20.14853. [DOI] [PubMed] [Google Scholar]
- Landegren U. Measurement of cell numbers by means of the endogenous enzyme hexosaminidase. Applications to detection of lymphokines and cell surface antigens. J. Immunol. Methods. 1984;67:379–388. doi: 10.1016/0022-1759(84)90477-0. [DOI] [PubMed] [Google Scholar]
- Laschinger M., Engelhardt B. Interaction of alpha4-integrin with VCAM-1 is involved in adhesion of encephalitogenic T cell blasts to brain endothelium but not in their transendothelial migration in vitro. J. Neuroimmunol. 2000;102:32–43. doi: 10.1016/s0165-5728(99)00156-3. [DOI] [PubMed] [Google Scholar]
- Leivo I., Engvall E. Merosin, a protein specific for basement membranes of Schwann cells, striated muscle, and trophoblast, is exressed late in nerve and muscle development. Proc. Natl. Acad. Sci. USA. 1988;85:1544–1548. doi: 10.1073/pnas.85.5.1544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lenter M., Vestweber D. The integrin chain beta 1 and alpha 6 associate with the chaperone calnexin prior to integrin assembly. J. Biol. Chem. 1994;269:12263–12268. [PubMed] [Google Scholar]
- Lichtinghagen R., Seifert T., Kracke A., Marckmann S., Wurster U., Heidenreich F. Expression of matrix metalloproteinase-9 and its inhibitos in mononuclear blood cells of patients with multiple sclerosis. J. Neuroimmunol. 1999;99:19–26. doi: 10.1016/s0165-5728(99)00094-6. [DOI] [PubMed] [Google Scholar]
- Liedtke W., Edelmann W., Chui F.-C., Kucherlapati R., Raine C.S. Experimental autoimmune encephalomyelitis in mice lacking glial fibrillar acidic protein is characterized by a more severe clinical course and an infiltrative central nervous system lesion. Am. J. Pathol. 1998;152:251–259. [PMC free article] [PubMed] [Google Scholar]
- Lindblom A., Engel J., Paulsson M. Characterization of native laminin from bovine kidney and comparisons with other laminin variants. Eur. J. Biochem. 1994;219:383–392. doi: 10.1111/j.1432-1033.1994.tb19950.x. [DOI] [PubMed] [Google Scholar]
- Losasso C., Di Tomaso F., Sgambato A., Ardito R., Cittadini A., Giardina B., Petrucci T.C., Brancaccio A. Anomalous dystroglycan in carcinoma cell lines. FEBS Lett. 2000;484:194–198. doi: 10.1016/s0014-5793(00)02157-8. [DOI] [PubMed] [Google Scholar]
- Madri J.A., Graesser D. Cell migration in the immune systemthe evolving inter-related roles of adhesion molecules and proteinases. Dev. Immunol. 2000;7:103–116. doi: 10.1155/2000/79045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mayer U., Nischt R., Pöschl E., Mann K., Fukuda K., Gerl M., Yamada Y., Timpl R. A single EGF-like motif of laminin is responsible for high affinity nidogen binding. EMBO (Eur. Mol. Biol. Organ.) J. 1993;12:1879–1885. doi: 10.1002/j.1460-2075.1993.tb05836.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mayer U., Saher G., Fässler R., Bornemann A., Echtermeyer F., von der Mark H., Miosge N., Pöschl E., von der Mark K. Absence of integrin alpha 7 causes a novel form of muscular dystrophy. Nat. Genet. 1997;17:318–323. doi: 10.1038/ng1197-318. [DOI] [PubMed] [Google Scholar]
- Miner J.H., Lewis R.M., Sanes J.R. Molecular cloning of a novel laminin chain, alpha 5, and widespread expression in adult mouse tissues. J. Biol. Chem. 1995;270:28523–28526. doi: 10.1074/jbc.270.48.28523. [DOI] [PubMed] [Google Scholar]
- Nehls V., Drenckhahn D. Heterogeneity of microvascular pericytes for smooth muscle. J. Cell Biol. 1991;113:147–154. doi: 10.1083/jcb.113.1.147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nehls V., Denzer K., Drenckhahn D. Pericyte involvement in capillary sprouting during angiogenesis in situ. Cell Tissue Res. 1992;270:469–474. doi: 10.1007/BF00645048. [DOI] [PubMed] [Google Scholar]
- Noseworthy J.H. Progress in determining the causes and treatment of multiple sclerosis. Nature. 1999;399:A40–A47. doi: 10.1038/399a040. [DOI] [PubMed] [Google Scholar]
- Patton B.L., Miner J.H., Chiu A.Y., Sanes J.R. Distribution and function of laminins in the neuromuscular system of developing, adult and mutant mice. J. Cell Biol. 1997;139:1507–1521. doi: 10.1083/jcb.139.6.1507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paulsson M., Saladin K. Mouse heart laminin. Purification of the native protein and structural comparison with Engelbreth-Holm-Swarm tumour laminin. J. Biol. Chem. 1989;264:18726–18732. [PubMed] [Google Scholar]
- Paulsson M., Aumailley M., Deutzmann R., Timpl R., Beck K., Engel J. Laminin-nidogen complexextraction with chelating agents and structural characterization. Eur. J. Biochem. 1987;166:11–19. doi: 10.1111/j.1432-1033.1987.tb13476.x. [DOI] [PubMed] [Google Scholar]
- Pedraza C., Geberhiwot T., Ingerpuu S., Assefa D., Wondimu Z., Kortesmaa J., Tryggvason K., Virtanen I., Patarroyo M. Monocytic cells synthesize, adhere to, and migrate on laminin 8. J. Immunol. 2000;165:5831–5838. doi: 10.4049/jimmunol.165.10.5831. [DOI] [PubMed] [Google Scholar]
- Pinkstaff J.K., Detterich J., Lynch G., Gall C. Integrin subunit gene expression is regionally differentiated in adult brain. J. Neurosci. 1999;19:1541–1556. doi: 10.1523/JNEUROSCI.19-05-01541.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Powell S.K., Kleinman H.K. Neuronal laminins and their cellular receptors. Int. J. Biochem. Cell Biol. 1997;29:401–414. doi: 10.1016/s1357-2725(96)00110-0. [DOI] [PubMed] [Google Scholar]
- Riminton D.S., Körner H., Strickland D.H., Lemckert F.A., Pollard J.D., Sedgwick J.D. Challenging cytokine redundancyinflammatory cell movement and clinical course of experimental autoimmune encephalomyelitis are normal in lymphotoxin-deficient, but not tumor necrosis factor–deficient, mice. J. Exp. Med. 1998;187:1517–1528. doi: 10.1084/jem.187.9.1517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ringelmann B., Röder C., Hallmann R., Maley M., Davies M., Grounds M., Sorokin L.M. Expression of laminin α1, α2, α4 and α5 chains, fibronectin and tenascin-C in skeletal muscle of dystrophic 129ReJ dy/dy mice. Exp. Cell Res. 2000;246:165–182. doi: 10.1006/excr.1998.4244. [DOI] [PubMed] [Google Scholar]
- Saito F., Masaki T., Kamakura K., Anderson L.V.B., Fujita S., Fukuta-Ohi H., Sunada Y., Shimizu T., Matsumura K. Characterization of the transmembrane molecular architecture of the dystroglycan complex in Schwann cells. J. Biol. Chem. 1999;274:8240–8246. doi: 10.1074/jbc.274.12.8240. [DOI] [PubMed] [Google Scholar]
- Sanes J.R., Engvall E., Butkowski R., Hunter D.D. Molecular heterogeneity of basal laminaeisoforms of laminin and collagen IV at the neuromuscular junction and elsewhere. J. Cell Biol. 1990;111:1685–1699. doi: 10.1083/jcb.111.4.1685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schuler F., Sorokin L.M. Expression of laminin isoforms in mouse myogenic cells in vitro and in vivo. J. Cell Sci. 1995;108:3795–3805. doi: 10.1242/jcs.108.12.3795. [DOI] [PubMed] [Google Scholar]
- Sedgwick J.D., Riminton D.S., Cyster J.G., Körner H. Tumor necrosis factora master-regulator of leukocyte movement. Immunol. Today. 2000;21:110–113. doi: 10.1016/s0167-5699(99)01573-x. [DOI] [PubMed] [Google Scholar]
- Shimizu Y., van Seventer G.A., Horgan K.J., Shaw S. Regulated expression and binding of three VLA (β1) integrin receptors on T-cells. Nature. 1990;345:250–253. doi: 10.1038/345250a0. [DOI] [PubMed] [Google Scholar]
- Sixt M., Hallmann R., Wendler O., Scharffetter-Kochanek K., Sorokin L.M. Cell adhesion and migration properties of β2-integrin negative, polymorphonuclear granulocytes (PMN) on defined extracellular matrix moleculesrelevance for leukocyte extravasation. (March 14, 2001) J. Biol. Chem. 2001;In press doi: 10.1074/jbc.M010898200. [DOI] [PubMed] [Google Scholar]
- Sobel R.A., Hinojoza J.R., Maeda A., Chen M. Endothelial cell integrin laminin receptor expression in multiple sclerosis lesions. Am. J. Pathol. 1998;135:405–415. doi: 10.1016/S0002-9440(10)65584-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sonnenberg A., Modderman P.W., Hogervorst F. Laminin receptor on platelets is the integrin VLA-6. Nature. 1988;336:487–489. doi: 10.1038/336487a0. [DOI] [PubMed] [Google Scholar]
- Sorokin L., Sonnenberg A., Aumailley M., Timpl R., Ekblom P. Recognition of the laminin E8 cell-binding site by an integrin processing the α6 subunit is essential for epithelial polarization in developing kidney tubules. J. Cell Biol. 1990;111:1265–1273. doi: 10.1083/jcb.111.3.1265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sorokin L.M., Conzelmann S., Ekblom P., Aumailley M., Battaglia C., Timpl R. Monoclonal antibodies against laminin A chain fragment E3 and their effects on cell- and heparin-binding, and on kidney development. Exp. Cell Res. 1992;201:137–144. doi: 10.1016/0014-4827(92)90357-e. [DOI] [PubMed] [Google Scholar]
- Sorokin L.M., Girg W., Gopfert T., Hallmann R., Deutzmann R. Expression of novel 400 kD laminin chains by mouse and bovine endothelial cells. Eur. J. Biochem. 1994;223:603–610. doi: 10.1111/j.1432-1033.1994.tb19031.x. [DOI] [PubMed] [Google Scholar]
- Sorokin L.M., Frieser M., Pausch F., Kröger S., Ohage E., Deutzmann R. Developmental regulation of laminin alpha 5 suggests a role in epithelial and endothelial cell maturation Dev. Biol. 189 1997. 285 300a [DOI] [PubMed] [Google Scholar]
- Sorokin L.M., Pausch F., Durbeej M., Ekblom P. Differential expression of five laminin α (1–5) chains in developing and adult mouse kidney Dev. Dyn. 210 1997. 446 462b [DOI] [PubMed] [Google Scholar]
- Sorokin L.M., Maley M., Moch H., von der Mark H., von der Mark K., Karosi S., Davies M.J., McGeachie J.K., Grounds M.D. Laminin α4 and integrin α6 are upregulated in regenerating dy/dy skeletal musclecomparative expression of laminin and integrin isoforms in muscles regenerating after crush injury. Exp. Cell Res. 2000;256:500–514. doi: 10.1006/excr.2000.4842. [DOI] [PubMed] [Google Scholar]
- Talts J.F., Andac Z., Göhring W., Brancaccio A., Timpl R. Binding of G domains of laminin alpha 1 and alpha 2 chains and perlecan to heparin, sulfatides, alpha-dystroglycan and several extracellular matrix proteins. EMBO (Eur. Mol. Biol. Organ.) J. 1999;18:863–870. doi: 10.1093/emboj/18.4.863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tian M., Jacobson C., Gee S.H., Campbell K.P., Carbonetto S., Jucker M. Dystroglycan in the cerebellum is a laminin a2-chain binding protein at the glial-vascular interface and is expressed in purkinje cells. Eur. J. Neurosci. 1997;8:2739–2747. doi: 10.1111/j.1460-9568.1996.tb01568.x. [DOI] [PubMed] [Google Scholar]
- Tiger C., Champliaud M., Pedrosa-Domellof F., Thornell L., Ekblom P., Gullberg D. Presence of laminin alpha 5 chain and lack of laminin alpha 1 chain during human muscle development and in muscular dystrophies. J. Biol. Chem. 1997;272:28590–28595. doi: 10.1074/jbc.272.45.28590. [DOI] [PubMed] [Google Scholar]
- Timpl R., Brown J. The laminins. Matrix Biol. 1994;14:275–281. doi: 10.1016/0945-053x(94)90192-9. [DOI] [PubMed] [Google Scholar]
- Timpl R., Rohde H., Gehron-Robey P., Rennard S.I., Foidart J.M., Martin G. Laminin—a glycoprotein from basement membranes. J. Biol. Chem. 1979;254:9933–9937. [PubMed] [Google Scholar]
- Tran E.H., Hoekstra K., Rooijen N.V., Dijkstra C.D., Owens T. Immune evasion of the central nervous system parenchyma and experimental allergic encephalomyelitis, but not leukocyte extravasation from blood, are prevented in macrophage-depleted mice. J. Immunol. 1998;161:3767–3775. [PubMed] [Google Scholar]
- Valles A.M., Boyer B., Tarone G., Thiery J.-P. Alpha 2 beta 1 integrin is required for the collagen and FGF-1 induced cell dispersion in a rat bladder carcinoma cell line. Cell Adhes. Commun. 1996;4:187–199. doi: 10.3109/15419069609014222. [DOI] [PubMed] [Google Scholar]
- van de Wiel-van Kemenade E., van Kooyk Y., de Boer A.J., Huijbens R.J., Weder P., van deKasteele W., Melief C.J., Figdor C.G. Adhesion of T and B lymphocytes to extracellular matrix and endothelial cells can be regulated through the beta subunit of VLA. J. Cell Biol. 1992;117:461–470. doi: 10.1083/jcb.117.2.461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vecchi A., Garlanda C., Lampugnani M.G., Resnati M., Matteucci C., Stoppacciaro A., Schnürch H., Risau W., Ruco L., Mantovani A., Dejana E. Monoclonal antibodies specific for endothelial cells of mouse blood vessels. Their application in the identification of adult and embryonic endothelium. Eur. J. Cell Biol. 1994;63:247–254. [PubMed] [Google Scholar]
- Virtanen I., Laitinen L., Korhonen M. Differential expression of laminin polypeptides in developing and adult human kidney. J. Histochem. Cytochem. 1995;43:621–628. doi: 10.1177/43.6.7769233. [DOI] [PubMed] [Google Scholar]
- Virtanen I., Gullberg D., Rissanen J., Kivilaakso E., Kiviluoto T., Laitinen L.A., Lehto V.P., Ekblom P. Laminin alpha1-chain shows a restricted distribution in epithelial basement membranes of fetal and adult human tissues. Exp Cell Res. 2000;257:298–309. doi: 10.1006/excr.2000.4883. [DOI] [PubMed] [Google Scholar]
- von der Mark K., Schober S., Goodman S. Integrins in cell migration. Methods Mol. Biol. 1999;129:219–230. doi: 10.1385/1-59259-249-X:219. [DOI] [PubMed] [Google Scholar]
- Wolburg H., Risau W. Development of the Hood-barrier. Trends Neurosci. 1990;13:174–178. doi: 10.1016/0166-2236(90)90043-a. [DOI] [PubMed] [Google Scholar]
- Wolburg H., Risau W. Formation of the blood-brain barrier. In: Kettenmann H., Ransom B.R., editors. Neuroglia. Oxford University Press; Oxford, UK: 1995. pp. 763–776. [Google Scholar]
- Wolburg K., Gerhardt H., Schulz M., Wolburg H., Engelhardt B. Ultrastructural localization of adhesion molecules in the healthy and inflamed choroid plexus of the mouse. Cell Tissue Res. 1999;296:259–269. doi: 10.1007/s004410051287. [DOI] [PubMed] [Google Scholar]
- Zhang E.T., Inman C.B.E., Weller R.O. Interrelationships of the pia mater and the perivascular (Virchow-Robin) spaces in the human cerebrum. J. Anat. 1990;170:111–123. [PMC free article] [PubMed] [Google Scholar]