Abstract
The expression of FcγR by human skin-derived mast cells of the MCTC type was determined in the current study. Expression of mRNA was analyzed with microarray gene chips and RT-PCR; protein by Western blotting and flow cytometry; function by release of β-hexosaminidase, PGD2, leukotriene C4 (LTC4), IL-5, IL-6, IL-13, GM-CSF, and TNF-α. FcγRIIa was consistently detected along with FcεRI at the mRNA and protein levels; FcγRIIc was sometimes detected only by RT-PCR; but FcγRIIb, FcγRI, and FcγRIII mRNA and protein were not detected. FcγRIIa-specific mAb caused skin MCTC cells to degranulate secrete PGD2, LTC4, GM-CSF, IL-5, IL-6, IL-13, and TNF-α in a dose-dependent fashion. FcεRI-specific mAb caused similar amounts of each mediator to be released with the exception of LTC4, which was not released by this agonist. Simultaneous but independent cross-linking of FcεRI and FcγRIIa did not substantially alter mediator release above or below levels observed with each agent alone. Skin MCTC cells sensitized with dust-mite-specific IgE and IgG, when coaggregated by Der p2, exhibited enhanced degranulation compared with sensitization with either IgE or IgG alone. These results extend the known capabilities of human skin mast cells to respond to IgG as well as IgE-mediated signals.
Receptors for IgG mediate important regulatory and effector functions within a variety of cell types, including mast cells. Three classes have been described, FcγRI (CD64), FcγRII (CD32), and FcγRIII (CD16), each having different affinities for IgG. FcγRI, a high-affinity receptor (IgG3 Kd ~10−9 M), is composed of an α-chain that binds IgG (IgG3 >1>4 > 2), and the γ-chain dimer, each of which contains an ITAM motif (4) that transmits signals leading to cell activation and mediator release. FcγRII is a 40-kDa monomeric glycoprotein with low affinity for human IgG (IgG3 Kd ≥ 10−7 M). Three genes in humans, FCGRIIA, FCGRIIB, and FCGRIIC, and one in mice, FCGRIIB, encode the different subtypes. FCGRIIA and FCGRIIC each encode an ITAM, while FCGRIIB encodes an ITIM (1), which attenuates ITAM signaling. FcγRIIa binds IgG3 > 1 = 2 > 4, while FcγRIIb and FcγRIIc bind IgG3 > 1 = 4 > 2. FCGRIII binds IgG3 = 1 > 2 = 4 with low affinity (IgG3 Kd ≥ 10−6 M) and is derived from two genes.
Mast cells are ubiquitously located in tissues where they initiate and propagate inflammatory diseases. They are uniquely equipped to initiate type I hypersensitivity reactions through their activation by multivalent Ags that cross-link IgE-bound FcεRI on the surface of mast cells, which induces the release of preformed mediators such as histamine and newly generated mediators such as PGD2. Later release of newly formed cytokines and chemokines further contributes to this inflammatory process. In addition to their well-established role as the effector cells for allergic inflammation, murine studies indicate mast cells participate in the innate immune response against bacteria and viruses (2-4) and also against certain parasites (5, 6). Rodent mast cells play key roles in the pathogenesis of non-IgE-mediated hypersensitivity disorders (7-9) involving FcγRIIIa that is naturally expressed on their surfaces and which responds to IgG immune complexes by causing secretion of mediators. Although FcγRIII expression has not been reported on human mast cells, functional FcγRI is transiently induced by IFN-γ (10). In mice activating FcγRIIIa and FcεRI molecules are counterbalanced by inhibitory FcγRIIb (11, 12). When FcγRIIb is coaggregated with FcεRI, degranulation is attenuated (13) due to the ITIM domain (14), which recruits the phosphatase SHIP1 (15). Mice lacking FcγRIIb have augmented allergic responses (16, 17).
The attenuating function of FcγRIIb has been postulated to explain how allergen immunotherapy might work: allergen injections induce allergen-specific IgG in the presence of allergen-specific IgE, and the resultant IgG–allergen–IgE complexes co-aggregate FcεRI and FcγRIIb. CD32 expression has been reported on human basophils and skin mast cells (18) but not on lung mast cells (19). In support of this pathway, a bispecific anti-IgE/anti-FcγRII Ab was constructed that inhibited allergen–IgE-mediated activation of human basophils and cord blood-derived mast cells (20). But it was not clear whether this bispecific Ab acted through an ITIM motif, competed with IgE for binding to FcεRI, and/or interfered with the aggregation of IgE by Ag. A potentially therapeutic bispecific human Fcγ-Fcε chimera, called GE2, was constructed and shown to inhibit class switching to IgE and IgE production by human B cells (21) and mediator release from FcεRI-expressing dendritic cells (22). GE2 also attenuated allergen-dependent activation of human cord blood-derived mast cells and peripheral blood basophils, and allergen skin tests in monkeys (23, 24). However, whether tissue-derived mast cells express FcγR capable of inducing inhibitory signals, similar to rodent mast cells and human cord blood-mast cells, is not clear.
The current study examines human skin-derived mast cells (25) for expression of FcγR, and shows that they express functional FcγRIIa, but not FcγRI, FcγRIIb, FcγRIIc, and FcγRIII, and consequently can release mediators in an IgG-dependent, IgE-independent manner.
Materials and Methods
Abs and reagents
Anti-FcεRIα mAb (22E7) (26) was a gift from Dr. J. Kochan (Hoffman-La Roche, Nutley, NJ), anti-FcγRIIb and anti-FcγRIIc mAb (41H16) were from Dr. B. M. Longenecker (Edmonton, Canada) (27), and rabbit polyclonal Abs were raised against the cytoplasmic tail of FcγRIIb (Ab163) (28) and of FcγRIIa (Ab260) (29) from Dr. Clark Anderson (Ohio State University, Columbus, OH). Anti-FcγRIIa, anti-FcγRIIb, and anti-FcγRIIc mAb (2E1) (30) (Santa Cruz Biotechnology); anti-FcγRIIa mAb (IV.3) (31), anti-FcγRIIa, anti-FcγRIIb, anti-FcγRIIc mAb (AT10) (32), and anti-FcεRI mAb (32.2) (Medarex); nonspecific mouse IgG1 mAb (MOPC31C), anti-FcγRIII mAb (3G8) mAb, and soybean trypsin inhibitor (SBTI)4 (Sigma-Aldrich); anti-Kit mAb (YB5.B8) (Immunotech); recombinant Der p2 (33), mouse (Fab)-human (Fc) chimeric Der P2-specific IgE (2B12) (34) and IgG1 (αDpX) (35) mAbs (Indoor Biotechnologies); 4-hydroxy-3-nitrophenylacetyl (NP)-BSA (~24 NP moieties per BSA molecule; Biosearch Technology); and NP-specific mouse (Fab)-human (Fc) IgE (MCA333S) and IgG2 (MCA334B) (Serotec) were obtained as indicated.
Culture of human skin mast cells
All study protocols involving human tissues were approved by the Human Studies Internal Review Board at Virginia Commonwealth University (Richmond, VA). Surgical skin samples were obtained from Virginia Commonwealth University Medical Center, the Cooperative Human Tissue Network of the National Cancer Institute, or the National Disease Research Interchange. Skin-derived mast cells were prepared as described (25). After removing s.c. fat by blunt dissection, residual tissue is cut into 1- to 2-mm fragments and digested with type 2 collagenase (1.5 mg/ml), hyaluronidase (0.7 mg/ml), and type 1 DNase (0.3 mg/ml) in HBSS for 2 h at 37°C. The dispersed cells were collected by filtering through a no. 80 mesh sieve and resuspended in HBSS containing 1% FCS and 10 mM HEPES. Cells were resuspended in HBSS, layered over a Percoll cushion, and centrifuged at 700 × g at room temperature for 20 min. Nucleated cells were collected from the buffer/Percoll interface, while erythrocytes sediment to the bottom of the tube. Cells enriched by Percoll density-dependent sedimentation were resuspended at a concentration of 1 × 106 cells/ml in serum-free AIM-V medium (Life Technologies) containing 100 ng/ml of recombinant human stem cell factor (SCF) (a gift from Amgen). Skin mast cells were split into separate wells every 4–5 days. Total cell numbers and viabilities were assessed by trypan blue staining. Cultures of skin-derived mast cells were maintained for up to 3 mo and were ~100% mast cells. Alternatively, freshly dispersed, Percoll-enriched mast cells were labeled with anti-FcεRI-α and anti-CD117 mAbs (5 μg/ml), and then with FITC-labeled anti-mouse F(ab′)2 at 4°C. Labeled cells were purified to ≥95% by sorting in a MoFlo high-performance cell sorter (Cytomation) and subjected to Western blotting for detection of different forms of CD32.
Gene expression
RNA extracted from skin-derived mast cells in culture with SCF was reverse-transcribed, labeled, and hybridized to Affymetrix HG-U133A and HG-U133B GeneChips by the DNA Microarray Section of the Nucleic Acids Core Facility at Virginia Commonwealth University as recommended by the manufacturer. The average signal intensity of genes determined to be expressed by the detection of cDNA was normalized to 500.
For RT-PCR total cellular RNA was extracted using the MicroFast-Track Method (Invitrogen Life Technologies) from 1 × 106 mast cells or from the peripheral blood leukocytes shown previously to express FcγRIIa, FcγRIIb, and FcγRIIc isoforms. RNA was concentrated by ethanol precipitation, resuspended in diethylpyrocarbonate-treated water, and stored at −70°C. cDNA was synthesized from 20 μl of RNA using a first strand cDNA kit (Pharmacia Biotech). FcγRII isoforms were specifically amplified using primers and conditions described previously (36). Southern blot analysis of PCR products was performed using a digoxygenin-UTP (Boehringer Mannheim) labeled RS91-46 probe as described previously (37).
Flow cytometry
Cells were recovered by centrifugation at 800 × g at 4°C, washed with PBS/1%BSA, and blocked for 30 min at 4°C with a 1/500 dilution of normal human serum. The cells were washed and incubated with the indicated Ab (10 μg/ml) for 1 h at 4°C. After Ab labeling, the cells were washed and incubated with a 1/100 dilution of F(ab′)2-FITC-goat anti-mouse Ab (BD Pharmingen) for 30 min at 4°C. After three washes, cells were resuspended in 400 μl of PBS. The mean intensity of fluorescence was determined for at least 10,000 cells using a FACScan flow cytometer (BD Biosciences). MOPC31C, nonspecific IgG, was used as a negative control. All experiments were performed at least three times.
Cell activation
Cultured skin mast cells (0.5 × 105 cells) were washed and activated in AIM-V with Abs at the concentrations indicated. In some experiments, freshly dispersed Percoll-enriched skin mast cells were activated with these Abs. For degranulation and lipid mediator measurements, activation was stopped after 30 min by adding three volumes of ice-cold PBS. The cells were centrifuged at 1000 rpm for 10 min at 4°C. Supernatants were transferred into a separate tube. Cell pellets were resuspend in PBS, sonicated in a Branson sonifier (model 350; power 5, 50% pulse cycle × 4 pulses) and microfuged. For cultured skin mast cells where purities >95%, β-hexosaminidase was assayed by measuring release of p-nitrophenol from the substrate p-nitrophenyl N-acetyl-β-d-glucosaminide as described (38). Absorbance values were read at 405 nm. For the freshly isolated skin mast cells where purities were typically ~5%, tryptase levels were measured in the releasates and retentates by ELISA using G4-biotin and B12 antitryptase mAbs as described (39). In each case, degranulation was calculated as a percentage of release values using the following formula:
To determine cytokine production, human skin mast cells were washed, suspended at 106 cells/ml in AIM-V medium containing SBTI (100 μg/ml) and activated in 24-well plates by incubation with anti-FcεRI (22E7) mAb at 1 μg/ml for 24 h as described (40).
FcεRI–FcγRII coaggregation
To examine FcεRI–FcγRII coaggregation, mast cells were sensitized with Der p2-specific IgE (10 μg/ml), Der p2-specific IgG1 (10 μg/ml) (each recognizing a noncompeting epitope (41) or both for at least 2 h at 37°C. This human IgG1 mAb bound to the mast cell surface under our experimental conditions as determined by flow cytometry (data not shown). Cells were washed and activated with 5 μg/ml Der p2 that had been aggregated using a (BS3)-coupling procedure according to the manufacturer (Pierce), or with buffer or mAb controls.
Immune complexes were generated with IgE and IgG anti-NP mAbs and NP-BSA. Stock solutions of NP-BSA (0.13 μg/ml) and Ig (8.7 μg/ml) were prepared and used to challenge skin-derived mast cells. Reactions were stopped after 30 min and analyzed for degranulation as above.
Lipid and cytokine measurement
Cysteinyl-leukotriene levels in releasates were measured with an enzyme immunoassay (EIA) for leukotriene C4 (LTC4)/D4 (LTD4)/E4 (LTE4) (Amersham Biosciences). For this EIA, the cross-reactivity between LTC4 and LTD4 is 100%; LTC4 and LTE4 is 70%; and LTC4 and LTB4 is 0.3%. Concentrations were determined according to the manufacturer’s instructions with a lower limit of detection of 15 pg/ml.
A PGD2-MOX EIA kit (Cayman Chemical) was used to measure PGD2 levels according to manufacturer’s instructions. PGD2 from fresh samples was first converted into PGD2 methoxime to prevent degradation. The lower limit of detection was 10 pg/ml.
Cytokines were measured using sandwich ELISAs in 384-well plates as described (40). Purified and biotinylated mouse or rat mAbs specific for each cytokine and standard recombinant cytokines were purchased from BD Biosciences as follows: IL-5, rat JES1-39D10/JES1-5A10; IL-6, rat MQ2–13A5/MQ2–39C3; IL-13, rat JES10–5A2/mouse B69–2; GM-CSF, rat BVD2-23B6/BVD2-21C11; and TNF-α, mouse MAB1/MAB11. Lower limits of sensitivity were 16 pg/ml.
Statistical analysis
One-way ANOVA was used to compare data among different treatment groups. If significant differences were detected, a Dunnett test was then used to compare all treatment groups vs the control group. Values of p ≤ 0.05 were considered to be significant.
Results
Skin and lung mast cells express FcγRIIa and not FcγRIIb
Microarray analysis with Affymetrix gene chips were used to examine gene expression in cultured human skin mast cells. As shown in Table I, expression of FcγRIIa was detected along with strong expression of FcεRIα, FcεRI, and FcεRI, but expression was absent for FcγRIIb and FcγRIIc along with FcγRI, FcγRIII, and FcεRII. To confirm the microarray analysis, RT-PCR with FcγRII-specific primers (36, 37) was performed. RNA from cultured skin mast cells yielded RT-PCR products representative of FcγRIIa (Fig. 1A, upper panel), while no FcγRIIb products were detected in three separate cultures. An FcγRIIc product was detected in one of three cultures (data not shown). The identities of the FcγRII PCR products from positive controls and from cultured skin mast cells were confirmed by Southern blotting with labeled probes that recognize gene-specific internal sequences (Fig. 1A, lower panel).
Table I.
Gene expression of IgG and IgE receptors on skin-derived mast cells using Affymetrix gene chips
| Gene Product | Expression Signal |
|---|---|
| FcεRIα | 3260 |
| FcεRIβ | 2979 |
| FcεRIγ | 6453 |
| FcγRI (CD64) | Aa |
| FcγRIIa (CD32a) | 133 |
| FcγRIIb (CD32b) | A |
| FcγRIIc (CD32c) | A |
| FcγRIII (CD16) | A |
| FcεRII (CD23) | A |
A, absent.
FIGURE 1.

FcγRII mRNA and protein expression in human skin mast cells. A, RT-PCR. RNA preparations from skin mast cells and peripheral blood leukocytes (+, positive control) were reverse-transcribed and FcγRIIa, FcγRIIb, and FcγRIIc transcripts were selectively amplified. PCR products were separated by electrophoresis on 2% agarose gels and detected by ethidium bromide staining (upper panel). Reaction mixtures with primers only (no RNA/DNA template) served as negative controls (data not shown). These results were representative of three independent experiments, meaning that cells were obtained from three separate donors. Southern blotting performed with a digoxygenin-UTP-labeled probe recognizing all FcγRII isoforms was used to confirm amplification of this gene. The results are representative of studies performed on two separate skin mast cell cultures. The two bands in FcγRIIC blots represent donor-dependent allelic polymorphisms (57). B, Western blot. Lysates from two separate mast cell cultures (SMC1 and SMC2) of 6–8 wk were separated by SDS-PAGE under reducing conditions, and analyzed by immunoblotting with rabbit anti-FcγRIIa (Ab260) (lower panel) or rabbit anti-FcγRIIb (Ab163) (upper panel). As a control, the FcγRIIa-expressing cell line U937 and FcγRIIb-expressing peripheral blood leukocytes (PBL) were included. Each lane contains 2 × 105 cell equivalents. Molecular mass markers are indicated to the left of these gels. C, Western blot of freshly dispersed mast cells. Lysates from freshly dipersed mast cells were tested as in B. These mast cells had been purified by sorting using FITC-labeled anti-Kit and anti-FcεRI mAbs within 2 days after their dispersal from skin. Peripheral blood basophils (PBB) and cord blood-derived mast cells (CBMC), obtained as described (24, 36), served as positive controls for CD32b, while Raji cells served as a positive control for CD32a. Each lane contains 5 × 105 cell equivalents.
Western blotting was used to analyze gene expression at the protein level. As shown in Fig. 1B, FcγRIIb protein was detected in extracts of peripheral blood leukocytes, but not in extracts of two preparations of cultured skin mast cells using Ab163. In contrast Ab260 against the cytoplasmic tail of FcγRIIa revealed a band migrating at ~43 kDa from U937 cells as well as mast cells, as expected for the glycosylated form of this receptor. To determine whether this expression pattern reflected the conditions of culture, Western blots also were performed with skin mast cells that had been purified by cell sorting using anti-FcεRI and anti-CD117 mAbs within 36 h after their dispersal and enrichment by Percoll density-dependent sedimentation. As shown in Fig. 1C, FcγRIIa, but not FcγRIIb, was detected in such mast cells. In contrast, FcγRIIb, but not FcγRIIa, was detected in cord blood-derived mast cells and peripheral blood basophils as noted previously (24, 36). Thus, freshly dispersed and cultured human skin mast cells do not express the FcγRIIb isoform but do express FcγRIIa.
Flow cytometry was used to reveal the surface expression of FcγR, FcεRI, and Kit on skin-derived human mast cells as shown in Fig. 2. As expected, the vast majority of cells expressed FcεRI and Kit. Surface expression of FcγRII was detected with IV.3 (FcγRIIa), AT10 (FcγRIIa, FcγRIIb, and FcγRIIc), and 2E1 (FcγRIIa, FcγRIIb, and FcγRIIc) (37, 42, 43), but not with 41H16 (FcγRIIb and FcγRIIc). This pattern of labeling indicates that membrane expression of FcγRII isoforms is limited to FcγRIIa. Analogous to earlier studies in which cultured human mast cells were examined, no FcγRI (CD64) or FcγRIII (CD16) expression was detected in resting cells (10).
FIGURE 2.

Surface expression of mast cell FcγRII. Mast cells were incubated at 4°C with mAbs against FcγR, FcεRI, and Kit as described, followed by FITC-labeled goat IgG anti-mouse IgG. Each primary mAb was used at 10 μg/ml. The isotype-matched control mAb (MOPC31C) is shown as the dotted line. Results are representative of a total of five separate experiments.
Activation of mast cells through direct FcγRIIa cross-linking
The functionality of FcγRIIa on skin mast cells was examined by challenging them with the FcγRIIa-specific mAb IV.3. A dose-dependent degranulation response was observed as measured by release of 17–64% of β-hexosaminidase in response to 0.01 μg/ml to 5 μg/ml of IV.3, with maximal release observed by 0.1 μg/ml of IV.3 (Fig. 3A). Activation through the FcεRI with mAb 22E7 released β-hexosaminidase to a similar degree, ranging from 15 to 33% in response to 0.01 μg/ml to 1 μg/ml of 22E7. Spontaneous release of β-hexosaminidase was <5%. When FcγRIIa and FcγεRI were challenged simultaneously, degranulation levels were comparable with those with IV.3 stimulation (0.1–5.0 μg/ml) alone; at the lowest dose of IV.3 (0.01 μg/ml) degranulation increased with simultaneous FcεRI cross-linking, but only to the degree expected with FcεRI cross-linking alone. To ensure that IV.3-mediated activation had occurred with the unaggregated mAb, mast cell activation was performed with IV.3 that had been subjected to gel filtration (Fig. 3, B and C). Portions of the peak fractions and those on either side along with prechromatography IV.3 and 22E7 Ab (each adjusted to a concentration of 1 μg/ml) were used to stimulate skin mast cells. IV.3 from the peak fraction (#22) as well as those from the ascending (#20) and descending (#24) portions of this peak induced a similar magnitude of mast cell activation when compared with the unfractionated IV.3, indicating that free (nonaggregated) IV.3 was responsible for stimulating these mast cells to degranulate. To compare the time courses for degranulation after FcγRIIa and FcεRI cross-linking, mast cells were stimulated with IV.3 and/or 22E7 at 1 μg/ml concentration for 0, 5, and 30 s, and for 3 and 15 min. As shown in Fig. 3D, the time courses of β-hexosaminidase release were similar in each case, with significant degranulation being detected by 30 s. Maximal release values also were similar.
FIGURE 3.
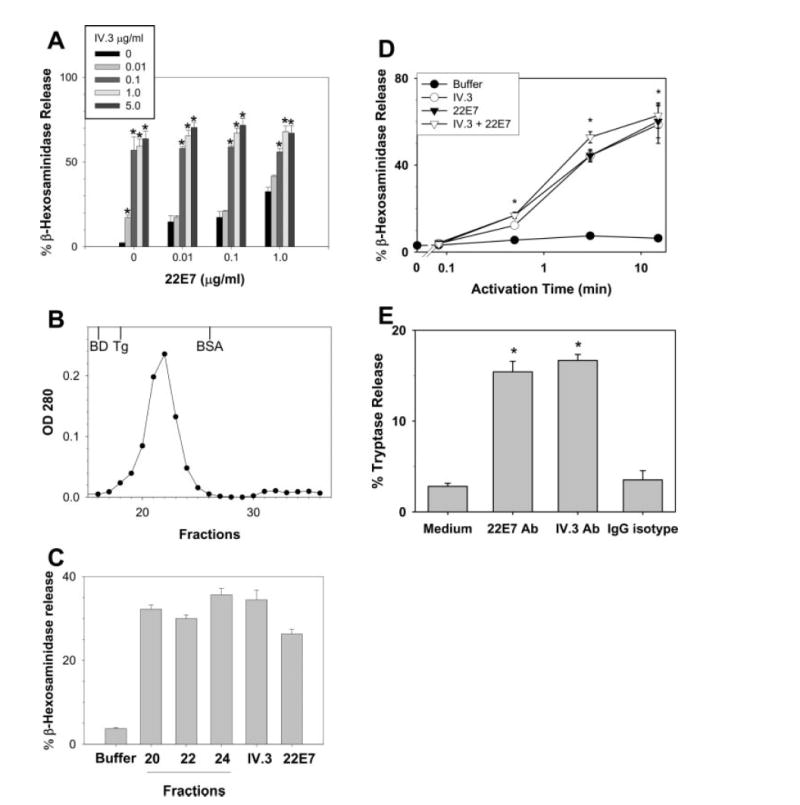
Degranulation by skin-derived mast cells after FcγRIIa and/or FcεRI cross-linking. A, Degranulation dose response. β-Hexosaminidase release was determined after 30 min of incubation with the mAbs as indicated. Release values were greater than the buffer control (p < 0.05) at all mAb concentrations. B, Gel filtration of IV.3 mAb. IV.3 mAb (0.5 mg/180 μl) was loaded onto a Superose 12 HR 10/30 column (Pharmacia) using a Shimadzu LC-10Avp HPLC system (Shimadzu) at a flow rate of 1 ml/min. The column was equilibrated and run with PBS. Elution fractions were collected at 0.5 ml/tube at 1 ml/min. The OD of each fraction was monitored at 280 nm and plotted. Gel filtration standards are shown at their elution positions for Blue Dextran (BD, ~2 × 106 Da), thyroglobulin (Tg, 669,000 Da), and BSA (66,000 Da). C, Activation of skin mast cells with unaggregated IV.3 mAb. IV.3 in fractions from the ascending (#20), peak (#22), and descending (#24) portions of the elution profile along with prechromatography IV.3 and 22E7 were each used at 1 μg/ml to stimulate skin mast cells. β-Hexosaminidase was measured as a marker of degranulation. D, Time-course. β-Hexosaminidase release was determined after mast cells were incubated with 22E7 (1 μg/ml) and/or IV.3 (0.1 μg/ml) mAbs for 0, 5 and 30 s, and for 3 and 15 min. E, Degranulation of freshly dispersed skin mast cells. Mast cells were dispersed from skin, enriched to ~5% purity by Percoll density-dependent sedimentation and then stimulated with 22E7 (1 μg/ml) or IV.3 (1 μg/ml) mAbs for 30 min. Degranulation was assessed by measuring release of tryptase. Spontaneous release and release with a nonimmune IgG-isotype control were each <4%. Mean ± SE values from three independent experiments are shown. *, p < 0.05 by ANOVA when experimental values are compared with 0 μg/ml of IV.3 within each 22E7 group in A, and to the buffer control in D and E.
The functionality of FcγRIIa on freshly dispersed skin mast cells was examined by measuring tryptase release after stimulation with optimal amounts of anti-FcεRI and FcγRIIa mAbs. By measuring tryptase release, in contrast with β-hexosaminidase, the mast cell source of both the releasate and the retentate could be assured. As shown in Fig. 3E, cross-linking these receptors led to comparable tryptase release values that were significantly higher than those observed with the buffer or IgG controls, indicating that both of these receptors were functionally present on freshly dispersed skin mast cells.
The release of PGD2 (Fig. 4A) and LTC4 (Fig. 4B) were assessed next. Similar to 22E7 stimulation, IV.3-stimulated skin mast cells produced comparable levels of PGD2 in a dose-dependent pattern from 0.001 to 1 μg/ml. However, no synergistic or additive effect was observed when FcγRIIa and FcεRI were simultaneously but independently cross-linked with FcεRI. LTC4 production was not observed at any of the concentrations of 22E7 tested. In contrast, FcγRIIa cross-linking induced release of LTC4 at 0.1 and 1 μg/ml; this release was not significantly altered when FcγRIIa was simultaneously challenged with FcεRI.
FIGURE 4.
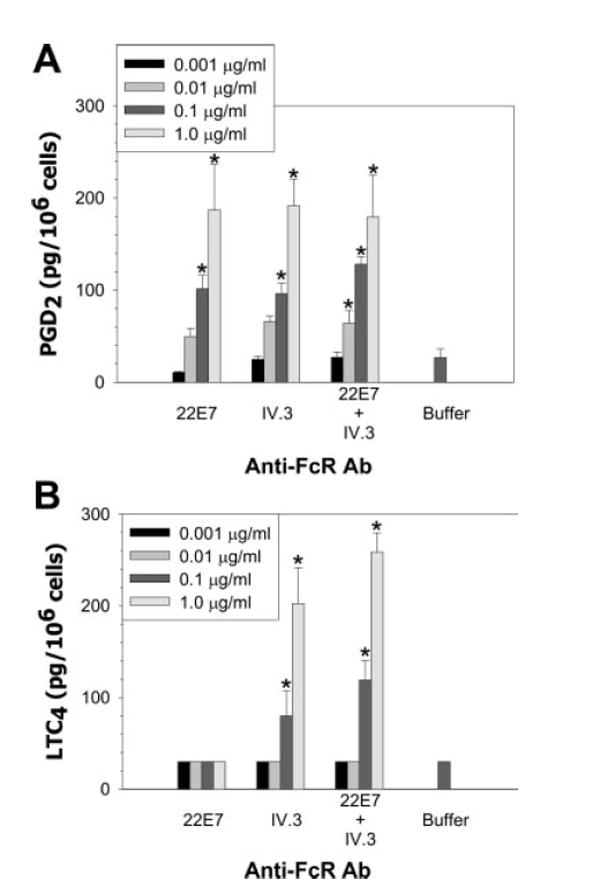
Release of PGD2 (A) and LTC4 (B) by skin mast cells stimulated by cross-linking FcεRI (22E7), FcγRIIa (IV.3) or both receptors simultaneously. PGD2 and LTC4 release were determined after skin mast cells (106 cells/ml) had been incubated with the indicated mAbs for 45 min in AIM-V medium. Results from three independent experiments, each single experiment performed in triplicate, are presented. *, p < 0.05 by ANOVA when experimental values within each group are compared with the buffer control.
We next examined the effects of FcγRIIa stimulation on the release of cytokines (Fig. 5). Stimulation of skin mast cells by cross-linking FcεRI and FcγRIIa by themselves or simultaneously resulted in dose-dependent increases in production of IL-5, IL-6, IL-13, GM-CSF, and TNF-α over a 24-h interval. At 0.01 μg/ml of mAb, IV.3 resulted in significantly less production of IL-5, GM-CSF, and TNF-α than did 22E7. However, in no case did stimulation with both mAbs together result in a significantly higher level of cytokine secretion than the sum of cytokine released by the two mAbs alone. Although no synergistic or additive effects were detected when cells were simultaneously stimulated through both FcεRI and FcγRIIa pathways, there also was no inhibition of FcεRI-mediated release of these cytokines when FcγRIIa was simultaneously cross-linked.
FIGURE 5.

Cytokine production by skin-derived mast cells challenged with anti-FcγRIIa and/or anti-FcεRI mAbs. A, IL-5. B, IL-6. C, IL-13. D, GM-CSF. E, TNF-α. Mast cells (106 cells/ml) were incubated with the indicated mAbs for 24 h in AIM-V medium containing SBTI and SCF. Results from three independent experiments, each single experiment performed in triplicate, are presented as box plots (because not all data followed a normal distribution) where the hatch mark is the median and the upper and lower ends of the box are the 25th and 75th percentiles. *, p < 0.05 by ANOVA when values for each cytokine were compared with those for the buffer control; †, p < 0.05 against IV.3 within the same mAb dose group.
Activation of skin-derived mast cells by Ag
Immune complexes
To test whether IgE or IgG immune complexes would activate skin-derived mast cells, cells were challenged with either IgG anti-NP–NP-BSA or IgE anti-NP–NP-BSA immune complexes. Degranulation was assessed by measuring β-hexosaminidase release. The result, as shown in Fig. 6A, clearly demonstrated immune complex-mediated mast cell activation for both IgE and IgG immune complexes. In each case, the magnitude of degranulation diminished with lesser amounts of immune complex.
FIGURE 6.

Activation of skin-derived human mast cells by Ag through FcγRIIa and FcεRI pathways. A, Activation by preformed IgG anti-NP–NP-BSA and IgE anti-NP–NP-BSA immune complexes. Immune complexes containing 8.7 μg/ml Ab with 0.13 μg/ml NP-BSA, 4.4 μg/ml Ab with 0.7 μg/ml NP-BSA, and 2.2 μg/ml Ab with 0.3 μg/ml NP-BSA were incubated with skin mast cells for 30 min. Supernatants and cell lysates were prepared for β-hexosaminidase measurements. Data are expressed as mean ± SE from three independent experiments, each performed in triplicate. B, Co-crosslinking FcγRIIa and FcεRI by Ag. Skin-derived mast cells that had been sensitized with Der p2-specific IgE, Der p2-specific IgG, or both mAbs were challenged with aggregated Der p2. Releasates were collected after 30 min to measure mediator release as described in materials and methods. Data expressed as mean ± SE from three individual experiments. *, p < 0.05 by ANOVA when experimental values are compared with the Ab-only control in A.
Coaggregation of FcεRI and FcγRIIa
The effects of coaggregating FcεRI and FcγRII using IgE and IgG anti-Der p2 mAbs were next examined. For these experiments, mast cells were sensitized with IgE to Der p2, IgG to Der p2, or both. Cells were then challenged with different concentrations of aggregated Der p2. As seen in Fig. 6B, degranulation occurred when IgE- or IgG-armed mast cells were exposed to Der p2. When mast cells were armed with both isotypes, FcεRI coaggregation with FcγRIIa led to significantly higher amounts of degranulation than to either mAb by itself (p < 0.05), even though this increase was additive at best. Importantly, no inhibition of IgE-mediated activation was observed when IgG and IgE were coaggregated by Ag.
Discussion
The novel finding that human skin-derived mast cells normally express functional FcγRIIa, but not FcγRI, FcγRIIb, or FcγRIII, implies a potential role for mast cells in IgG immune complex-mediated processes in vivo. This may be analogous to the role that FcγRIII plays in rodent mast cells whereby mast cells become involved in IgG-mediated hypersensitivity disorders such as the Arthus reaction (7), or in IgG-mediated anaphylaxis (44). Mice, unlike humans, express only the inhibitory FcγRIIb form of this receptor (45). Transient expression of FcγRI on human mast cells (10) also might contribute to IgG-mediated activation of these cells.
A current theory predicts that the allergen–IgE-mediated response of mast cells will be attenuated by allergen-specific IgG induced by allergen immunotherapy and acting by coaggregating ITIM-containing FcγRIIb with ITAM-containing FcεRI receptors (45-47). The expression of FcγRIIb on rodent mast cells, human basophils, and human cord blood-derived mast cells and the ability of this receptor to inhibit FcεRI-dependent activation (36, 45) supports this paradigm. Indeed, the therapeutic potential for regulating FcεRI-mediated activation by coaggregation of ITIM-containing receptors led to the development of GE2, human IgG1 Hinge-CHγ2-CHγ3 region linked to the human IgE CHε2-CHε3-CHε4 region. GE2 showed dose- and time-dependent inhibition of Ag-driven IgE-mediated histamine release from human basophils and cord blood-derived mast cells and increased inhibition of IgE-mediated passive cutaneous anaphylaxis in human FcεRI-α transgenic mice (23, 24, 48). Whether the inhibitory capabilities of GE2 act solely through the ITIM domain of FcγRIIb or also by competing with Ag-specific IgE and/or IgG for binding to their receptors remains to be fully understood.
However, the absence of FcγRIIb and the presence of FcγRIIa and possibly FcγRIIc on human skin-derived mast cells argues against this mechanistic explanation for the efficacy of immunotherapy, at least for the MCTC type of mast cell that predominates in skin (49). In fact, production of IgG against allergens might lead to activation of MCTC cells through FcγRIIa. The current study using skin-derived mast cells found no evidence for inhibition of degranulation when FcγRIIa and FcεRI were simultaneously but independently cross-linked with Ags or anti-receptor mAbs or when co-cross-linked with Ag. In fact, co-cross-linking led to a higher level of degranulation than with either FcγRIIa or FcεRI cross-linking alone. Whether these observations can be extended to MCTC cells from other tissues, or to the MCT type of mast cell that predominates in lung and small bowel mucosa remains to be studied. The finding of FcγRIIb, but neither FcγRIIa or FcγRIIc, in cord blood-derived mast cells (24) distinguishes this in vitro-derived mast cell from skin-derived MCTC cells, which may reflect differences in the progenitors, the conditions for development, or the stage of maturation of these mast cells.
FcγRIIa cross-linking on the surface of skin MCTC cells leads to degranulation and secretion of newly generated lipids and cytokines. Although mostly comparable to what is observed with FcεRI cross-linking, the one difference pertains to LTC4 secretion, which follows cross-linking of FcγRIIa but not FcεRI. The absence of LTC4 production by FcεRI-cross-linked MCTC cells from skin has been reported (49-51). Of note is that MCTC cells from lung do produce LTC4 after FcεRI cross-linking (49). The observations that MCTC cells from a noncutaneous site produce FcεRI-mediated LTC4, and that skin MCTC cells produce FcγRIIa-mediated LTC4, raise the possibility that skin MCTC cells, if properly primed, also might produce FcεRI-mediated LTC4.
Several factors may regulate FcγRII isoform expression. Cytokines influence whether monocytes express the FcγRIIa or FcγRIIb isoform. Specifically, FcγRIIb expression is up-regulated by IL-4 (28), and FcγRIIa by IL-10 (52). But IL-10 is up-regulated after allergen immunotherapy (53). How cytokines affect FcγRII expression on mast cells needs to be understood. Another factor is the culture condition, e.g., human monocytes cultured at higher cell densities express higher levels of FcγRIIb (28). Finally, recent studies show that a promoter haplotype in FCGRIIB results in diminished expression and may predispose to autoimmune disorders such as systemic lupus erythematosus (54-56). The presence of FcγRIIa and absence of FcγRIIb was a consistent phenotype among the skin mast cell preparations obtained from different subjects, making genetic polymorphisms an untenable explanation for our observations.
In summary, the expression of functional FcγRIIa receptors on human skin-derived mast cells of the MCTC type extends the response capabilities of human mast cells beyond those associated with IgE to include those associated with IgG.
Acknowledgments
We thank Henry Bateman and the Nucleic Acids Core Facility at Virginia Commonwealth University for their excellent technical assistance.
Footnotes
This work was supported in part by National Institutes of Health Grants R01-AI27517 (to L.B.S.) and K08-AI057357 (to W.Z.), by a grant from Philip Morris USA and Philip Morris International (to L.B.S.), and by the Food Allergy and Anaphylaxis Network (to C.L.K.)
Disclosures L. B. Schwartz has received royalties from commercial tryptase assay. His spouse, A.-M. Irani, has received royalties from Merck & Co., Glaxo-SmithKline, Novartis, and AstraZeneca Pharmaceuticals.
Abbreviations used in this paper: SBTI, soybean trypsin inhibitor; LT, leukotriene; NP, 4-hydroxy-3-nitrophenylacetyl; EIA, enzyme immunoassay.
References
- 1.Unkeless JC, Jin J. Inhibitory receptors, ITIM sequences and phosphatases. Curr Opin Immunol. 1997;9:338–343. doi: 10.1016/s0952-7915(97)80079-9. [DOI] [PubMed] [Google Scholar]
- 2.Malaviya R, Georges A. Regulation of mast cell-mediated innate immunity during early response to bacterial infection. Clin Rev Allergy Immunol. 2002;22:189–204. doi: 10.1385/CRIAI:22:2:189. [DOI] [PubMed] [Google Scholar]
- 3.Marone G, Florio G, Petraroli A, Triggiani M, De Paulis A. Role of human Fcε RI+ cells in HIV-1 infection. Immunological Reviews. 2001;179:128–138. doi: 10.1034/j.1600-065x.2001.790113.x. [DOI] [PubMed] [Google Scholar]
- 4.Marshall JS, King CA, McCurdy JD. Mast cell cytokine and chemokine responses to bacterial and viral infection. Curr Pharm Des. 2003;9:11–24. doi: 10.2174/1381612033392413. [DOI] [PubMed] [Google Scholar]
- 5.Urban JF, Schopf L, Morris SC, Orekhova T, Madden KB, Betts CJ, Gamble HR, Byrd C, Donaldson D, Else K, Finkelman FD. Stat6 signaling promotes protective immunity against Trichinella spiralis through a mast cell- and T cell-dependent mechanism. J Immunol. 2000;164:2046–2052. doi: 10.4049/jimmunol.164.4.2046. [DOI] [PubMed] [Google Scholar]
- 6.Knight PA, Wright SH, Lawrence CE, Paterson YY, Miller HR. Delayed expulsion of the nematode Trichinella spiralis in mice lacking the mucosal mast cell-specific granule chymase, mouse mast cell protease-1. J Exp Med. 2000;192:1849–1856. doi: 10.1084/jem.192.12.1849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Sylvestre DL, Ravetch JV. A dominant role for mast cell Fc receptors in the Arthus reaction. Immunity. 1996;5:387–390. doi: 10.1016/s1074-7613(00)80264-2. [DOI] [PubMed] [Google Scholar]
- 8.Lee DM, Friend DS, Gurish MF, Benoist C, Mathis D, Brenner MB. Mast cells: a cellular link between autoantibodies and inflammatory arthritis. Science. 2002;297:1689–1692. doi: 10.1126/science.1073176. [DOI] [PubMed] [Google Scholar]
- 9.Robbie-Ryan M, Tanzola MB, Secor VH, Brown MA. Cutting edge: both activating and inhibitory Fc receptors expressed on mast cells regulate experimental allergic encephalomyelitis disease severity. J Immunol. 2003;170:1630–1634. doi: 10.4049/jimmunol.170.4.1630. [DOI] [PubMed] [Google Scholar]
- 10.Okayama Y, Kirshenbaum AS, Metcalfe DD. Expression of a functional high-affinity IgG receptor, Fcγ RI, on human mast cells: up-regulation by IFN-γ. J Immunol. 2000;164:4332–4339. doi: 10.4049/jimmunol.164.8.4332. [DOI] [PubMed] [Google Scholar]
- 11.Chong HJ, Andrew BL, Bailey DP, Wright H, Ramirez C, Gharse A, Oskeritzian C, Xia HZ, Zhu J, Paul WE, et al. IL-4 selectively enhances FcγRIII expression and signaling on mouse mast cells. Cell Immunol. 2003;224:65–73. doi: 10.1016/j.cellimm.2003.08.004. [DOI] [PubMed] [Google Scholar]
- 12.Daeron M. Fc receptors and allergies. Rev Fr Allergol. 2000;40:445–465. [Google Scholar]
- 13.Daeron M, Malbec O, Latour S, Arock M, Fridman WH. Regulation of high-affinity IgE receptor-mediated mast cell activation by murine low-affinity IgG receptors. J Clin Invest. 1995;95:577–585. doi: 10.1172/JCI117701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Daeron M, Latour S, Malbec O, Espinosa E, Pina P, Pasmans S, Fridman WH. The same tyrosine-based inhibition motif, in the intracytoplasmic domain of Fcγ RIIB, regulates negatively BCR-, TCR-, and FcR-dependent cell activation. Immunity. 1995;3:635–646. doi: 10.1016/1074-7613(95)90134-5. [DOI] [PubMed] [Google Scholar]
- 15.Lesourne R, Fridman WH, Daeron M. Dynamic interactions of Fcγ receptor IIB with filamin-bound SHIP1 amplify filamentous actin-dependent negative regulation of Fcε receptor I signaling. J Immunol. 2005;174:1365–1373. doi: 10.4049/jimmunol.174.3.1365. [DOI] [PubMed] [Google Scholar]
- 16.Takai T, Ono M, Hikida M, Ohmori H, Ravetch JV. Augmented humoral and anaphylactic responses in FcγRII- deficient mice. Nature. 1996;379:346–349. doi: 10.1038/379346a0. [DOI] [PubMed] [Google Scholar]
- 17.Ujike A, Ishikawa Y, Ono M, Yuasa T, Yoshino T, Fukumoto M, Ravetch JV, Takai T. Modulation of immunoglobulin (Ig)E-mediated systemic anaphylaxis by low-affinity Fc receptors for IgG. J Exp Med. 1999;189:1573–1579. doi: 10.1084/jem.189.10.1573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ghannadan M, Baghestanian M, Wimazal F, Eisenmenger M, Latal D, Kargül G, Walchshofer S, Sillaber C, Lechner K, Valent P. Phenotypic characterization of human skin mast cells by combined staining with toluidine blue and CD antibodies. J Invest Dermatol. 1998;111:689–695. doi: 10.1046/j.1523-1747.1998.00359.x. [DOI] [PubMed] [Google Scholar]
- 19.Valent P, Ashman LK, Hinterberger W, Eckersberger F, Majdic O, Lechner K, Bettelheim P. Mast cell typing: demonstration of a distinct hematopoietic cell type and evidence for immunophenotypic relationship to mononuclear phagocytes. Blood. 1989;7:1778–1785. [PubMed] [Google Scholar]
- 20.Tam SW, Demissie S, Thomas D, Daeron M. A bispecific antibody against human IgE and human FcγRII that inhibits antigen-induced histamine release by human mast cells and basophils. Allergy. 2004;59:772–780. doi: 10.1111/j.1398-9995.2004.00332.x. [DOI] [PubMed] [Google Scholar]
- 21.Yamada T, Zhu D, Zhang K, Saxon A. Inhibition of interleukin-4-induced class switch recombination by a human immunoglobulin Fcγ-Fc epsilon chimeric protein. J Biol Chem. 2003;278:32818–32824. doi: 10.1074/jbc.M304590200. [DOI] [PubMed] [Google Scholar]
- 22.Kepley CL, Zhang K, Zhu D, Saxon A. FcεRI-FcγRII coaggregation inhibits IL-16 production from human Langerhans-like dendritic cells. Clin Immunol. 2003;108:89–94. doi: 10.1016/s1521-6616(03)00155-4. [DOI] [PubMed] [Google Scholar]
- 23.Zhang K, Kepley CL, Terada T, Zhu D, Perez H, Saxon A. Inhibition of allergen-specific IgE reactivity by a human Ig Fcγ-Fcε bifunctional fusion protein. J Allergy Clin Immunol. 2004;114:321–327. doi: 10.1016/j.jaci.2004.03.058. [DOI] [PubMed] [Google Scholar]
- 24.Kepley CL, Taghavi S, Mackay GA, Zhu D, Morel PA, Zhang K, Ryan JJ, Satin LS, Zhang M, Pandolfi PP, Saxon A. Coaggregation of Fcγ RII With Fcε RI on human mast cells inhibits antigen-induced secretion and involves SHIP-Grb2-Dok complexes. J Biol Chem. 2004 doi: 10.1074/jbc.M404318200. [DOI] [PubMed] [Google Scholar]
- 25.Kambe N, Kambe M, Kochan JP, Schwartz LB. Human skin-derived mast cells can proliferate while retaining their characteristic functional and protease phenotypes. Blood. 2001;97:2045–2052. doi: 10.1182/blood.v97.7.2045. [DOI] [PubMed] [Google Scholar]
- 26.Riske F, Hakim J, Mallamaci M, Griffin M, Pilson B, Tobkes N, Lin P, Danho W, Kochan J, Chizzonite R. High affinity human IgE receptor (FcεRI). Analysis of functional domains of the α-subunit with monoclonal antibodies. J Biol Chem. 1991;266:11245–11251. [PubMed] [Google Scholar]
- 27.Antoun GR, Longenecker BM, Zipf TF. Comparison of the 40 kDa hematopoietic cell antigens bound by monoclonal antibodies IV. 3, 41H. 16 and KB61. Mol Immunol. 1989;26:333–338. doi: 10.1016/0161-5890(89)90088-6. [DOI] [PubMed] [Google Scholar]
- 28.Tridandapani S, Siefker K, Teillaud JL, Carter JE, Wewers MD, Anderson CL. Regulated expression and inhibitory function of Fcγ RIIb in human monocytic cells. J Biol Chem. 2002;277:5082–5089. doi: 10.1074/jbc.M110277200. [DOI] [PubMed] [Google Scholar]
- 29.Vely F, Gruel N, Moncuit J, Cochet O, Rouard H, Dare S, Galon J, Sautes C, Fridman WH, Teillaud JL. A new set of monoclonal antibodies against human Fcγ RII (CD32) and Fcγ RIII (CD16): characterization and use in various assays. Hybridoma. 1997;16:519–528. doi: 10.1089/hyb.1997.16.519. [DOI] [PubMed] [Google Scholar]
- 30.Micklem KJ, Stross WP, Willis AC, Cordell JL, Jones M, Mason DY. Different isoforms of human FcRII distinguished by CDw32 antibodies. J Immunol. 1990;144:2295–2303. [PubMed] [Google Scholar]
- 31.Looney RJ, Abraham GN, Anderson CL. Human monocytes and U937 cells bear two distinct Fc receptors for IgG. J Immunol. 1986;136:1641–1647. [PubMed] [Google Scholar]
- 32.Greenman J, Tutt AL, George AJ, Pulford KA, Stevenson GT, Glennie MJ. Characterization of a new monoclonal anti-Fc gamma RII antibody, AT10, and its incorporation into a bispecific F(ab′)2 derivative for recruitment of cytotoxic effectors. Mol Immunol. 1991;28:1243–1254. doi: 10.1016/0161-5890(91)90011-8. [DOI] [PubMed] [Google Scholar]
- 33.Smith AM, Benjamin DC, Hozic N, Derewenda U, Smith WA, Thomas WR, Gafvelin G, Hage-Hamsten M, Chapman MD. The molecular basis of antigenic cross-reactivity between the group 2 mite allergens. J Allergy Clin Immunol. 2001;107:977–984. doi: 10.1067/mai.2001.115629. [DOI] [PubMed] [Google Scholar]
- 34.Schuurman J, Perdok GJ, Lourens TE, Parren PW, Chapman MD, Aalberse RC. Production of a mouse/human chimeric IgE monoclonal antibody to the house dust mite allergen Der p 2 and its use for the absolute quantification of allergen-specific IgE. J Allergy Clin Immunol. 1997;99:545–550. doi: 10.1016/s0091-6749(97)70083-6. [DOI] [PubMed] [Google Scholar]
- 35.Van der Zee JS, van Swieten P, Jansen HM, Aalberse RC. Skin tests and histamine release with P1-depleted Dermatophagoides pteronyssinus body extracts and purified P1. J Allergy Clin Immunol. 1988;81:884–896. doi: 10.1016/0091-6749(88)90946-3. [DOI] [PubMed] [Google Scholar]
- 36.Kepley CL, Cambier JC, Morel PA, Lujan D, Ortega E, Wilson BS, Oliver JM. Negative regulation of Fc epsilon RI signaling by Fc gamma RII costimulation in human blood basophils. J Allergy Clin Immunol. 2000;106:337–348. doi: 10.1067/mai.2000.107931. [DOI] [PubMed] [Google Scholar]
- 37.Metes D, Ernst LK, Chambers WH, Sulica A, Herberman RB, Morel PA. Expression of functional CD32 molecules on human NK cells is determined by an allelic polymorphism of the FcγRIIC gene. Blood. 1998;91:2369–2380. [PubMed] [Google Scholar]
- 38.Schwartz LB, Lewis RA, Seldin D, Austen KF. Acid hydrolases and tryptase from secretory granules of dispersed human lung mast cells. J Immunol. 1981;126:1290–1294. [PubMed] [Google Scholar]
- 39.Schwartz LB, Bradford TR, Rouse C, Irani A-M, Rasp G, Van der Zwan JK, Van Der Linden P-WG. Development of a new, more sensitive immunoassay for human tryptase: use in systemic anaphylaxis. J Clin Immunol. 1994;14:190–204. doi: 10.1007/BF01533368. [DOI] [PubMed] [Google Scholar]
- 40.Zhao W, Oskeritzian CA, Pozez AL, Schwartz LB. Cytokine production by skin-derived mast cells: endogenous proteases are responsible for degradation of cytokines. J Immunol. 2005;175:2635–2642. doi: 10.4049/jimmunol.175.4.2635. [DOI] [PubMed] [Google Scholar]
- 41.Ovsyannikova IG, Vailes LD, Li Y, Heymann PW, Chapman MD. Monoclonal antibodies to group II Dermatophagoides spp. allergens: murine immune response, epitope analysis, and development of a two-site ELISA. J Allergy Clin Immunol. 1994;94:537–546. doi: 10.1016/0091-6749(94)90211-9. [DOI] [PubMed] [Google Scholar]
- 42.Warmerdam PA, van den Herik-Oudijk IE, Parren PW, Westerdaal NA, van de Winkel JG, Capel PJ. Interaction of a human FcγRIIb1 (CD32) isoform with murine and human IgG subclasses. Int Immunol. 1993;5:239–247. doi: 10.1093/intimm/5.3.239. [DOI] [PubMed] [Google Scholar]
- 43.Ierino FL, Hulett MD, McKenzie IF, Hogarth PM. Mapping epitopes of human FcγRII (CDw32) with monoclonal antibodies and recombinant receptors. J Immunol. 1993;150:1794–1803. [PubMed] [Google Scholar]
- 44.Miyajima I, Dombrowicz D, Martin TR, Ravetch JV, Kinet JP, Galli SJ. Systemic anaphylaxis in the mouse can be mediated largely through IgG, and FcγRIII: assessment of the cardiopulmonary changes, mast cell degranulation, and death associated with active or IgE- or IgG1-dependent passive anaphylaxis. J Clin Invest. 1997;99:901–914. doi: 10.1172/JCI119255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Daeron M. Fc receptor biology. Annu Rev Immunol. 1997;15:203–234. doi: 10.1146/annurev.immunol.15.1.203. [DOI] [PubMed] [Google Scholar]
- 46.Ott VL, Cambier JC. Activating and inhibitory signaling in mast cells: new opportunities for therapeutic intervention? J Allergy Clin Immunol. 2000;106:429–440. doi: 10.1067/mai.2000.109428. [DOI] [PubMed] [Google Scholar]
- 47.Katz HR. Inhibitory receptors and allergy. Curr Opin Immunol. 2002;14:698–704. doi: 10.1016/s0952-7915(02)00400-4. [DOI] [PubMed] [Google Scholar]
- 48.Zhu D, Kepley CL, Zhang M, Zhang K, Saxon A. A novel human immunoglobulin Fcγ Fcε bifunctional fusion protein inhibits Fcε RI-mediated degranulation. Nat Med. 2002;8:518–521. doi: 10.1038/nm0502-518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Oskeritzian CA, Zhao W, Min HK, Xia HZ, Pozez A, Kiev J, Schwartz LB. Surface CD88 functionally distinguishes the MCTC from the MCT type of human lung mast cell. J Allergy Clin Immunol. 2005;115:1162–1168. doi: 10.1016/j.jaci.2005.02.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Lawrence ID, Warner JA, Cohan VL, Hubbard WC, Kagey-Sobotka A, Lichtenstein LM. Purification and characterization of human skin mast cells. J Immunol. 1987;139:3062–3069. [PubMed] [Google Scholar]
- 51.Columbo M, Horowitz EM, Botana LM, MacGlashan DW, Jr, Bochner BS, Gillis S, Zsebo KM, Galli SJ, Lichtenstein LM. The human recombinant c-kit receptor ligand, rhSCF, induces mediator release from human cutaneous mast cells and enhances IgE-dependent mediator release from both skin mast cells and peripheral blood basophils. J Immunol. 1992;149:599–608. [PubMed] [Google Scholar]
- 52.Coopman PJ, Do MT, Barth M, Bowden ET, Hayes AJ, Basyuk E, Blancato JK, Vezza PR, McLeskey SW, Mangeat PH, Mueller SC. The Syk tyrosine kinase suppresses malignant growth of human breast cancer cells. Nature. 2000;406:742–747. doi: 10.1038/35021086. [DOI] [PubMed] [Google Scholar]
- 53.Burks W, Helm R, Stanley S, Bannon GA. Food allergens. Curr Opin Allergy Clin Immunol. 2001;1:243–248. doi: 10.1097/01.all.0000011021.73682.01. [DOI] [PubMed] [Google Scholar]
- 54.Jiang Y, Hirose S, Abe M, Sanokawa-Akakura R, Ohtsuji M, Mi X, Li N, Xiu Y, Zhang D, Shirai J, et al. Polymorphisms in IgG Fc receptor IIB regulatory regions associated with autoimmune susceptibility. Immunogenetics. 2000;51:429–435. doi: 10.1007/s002510050641. [DOI] [PubMed] [Google Scholar]
- 55.Su K, Wu J, Edberg JC, Li X, Ferguson P, Cooper GS, Langefeld CD, Kimberly RP. A promoter haplotype of the immunoreceptor tyrosine-based inhibitory motif-bearing FcγRIIb alters receptor expression and associates with autoimmunity. I. Regulatory FCGR2B polymorphisms and their association with systemic lupus erythematosus. J Immunol. 2004;172:7186–7191. doi: 10.4049/jimmunol.172.11.7186. [DOI] [PubMed] [Google Scholar]
- 56.Blank MC, Stefanescu RN, Masuda E, Marti F, King PD, Redecha PB, Wurzburger RJ, Peterson MG, Tanaka S, Pricop L. Decreased transcription of the human FCGR2B gene mediated by the -343 G/C promoter polymorphism and association with systemic lupus erythematosus. Hum Genet. 2005;117:220–227. doi: 10.1007/s00439-005-1302-3. [DOI] [PubMed] [Google Scholar]
- 57.Ernst LK, Metes D, Herberman RB, Morel PA. Allelic polymorphisms in the FcγRIIC gene can influence its function on normal human natural killer cells. J Mol Med. 2002;80:248–257. doi: 10.1007/s00109-001-0294-2. [DOI] [PubMed] [Google Scholar]


