Abstract
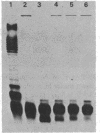
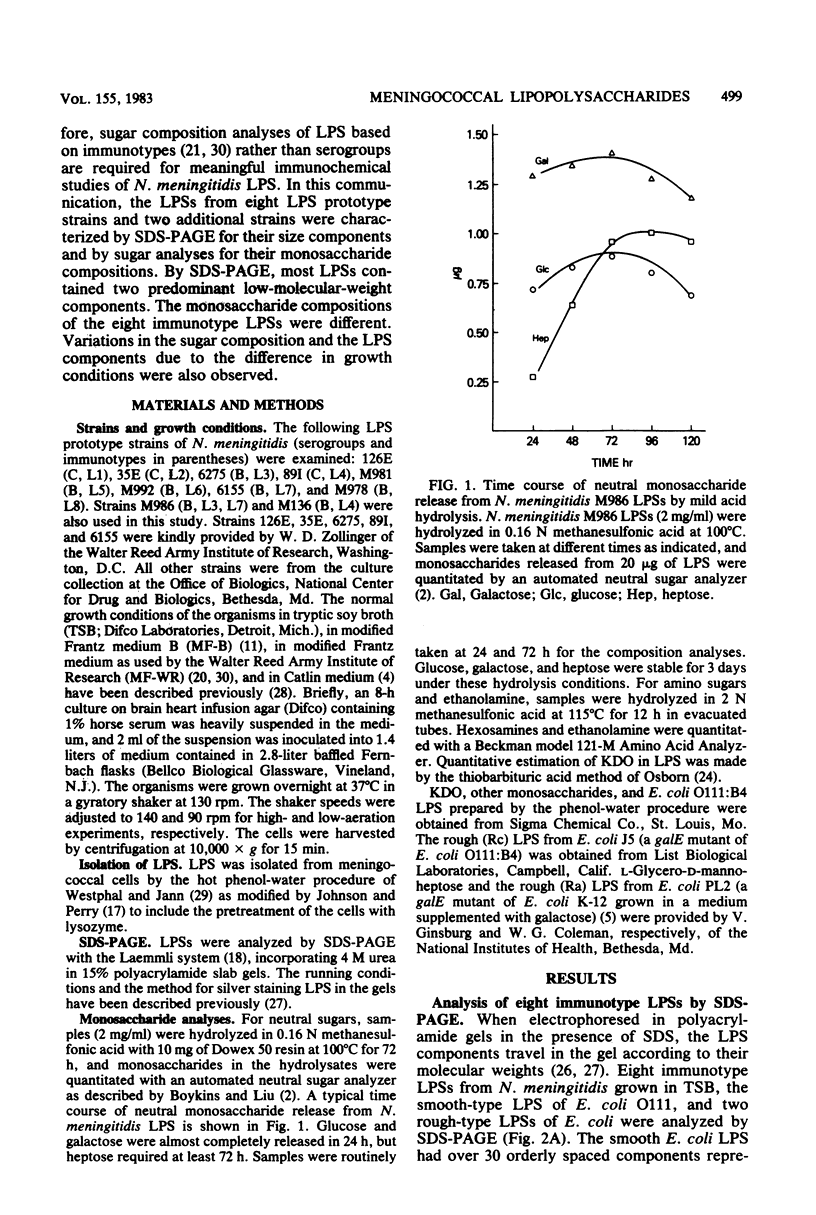
Eight immunotype lipopolysaccharides (LPSs) of Neisseria meningitidis were prepared by the phenol-water procedure and characterized by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and sugar analyses. By SDS-PAGE and a highly sensitive silver strain. N. meningitidis LPSs from cells grown in tryptic soy broth were shown to contain one or two predominant components and a few minor, somewhat higher-molecular-weight components. The molecular sizes of the two predominant components were approximately the same as those of two E. coli rough-type LPSs, one with a complete core and the other with an incomplete core. The molecular weight of the major LPS component varied somewhat among different immunotypes but was estimated to be in the range of 4,200 to 5,000. By sugar analyses, the eight immunotype LPSs were different in their monosaccharide compositions. All contained glucose, galactose, heptose, glucosamine, and 2-keto-3-deoxyoctonate, but in different molar ratios. The growth of N. meningitidis in tryptic soy broth under different levels of aeration resulted in a change in the two major LPS components seen on the SDS-PAGE gel. High aeration increased the amount of the smaller component, whereas low aeration increased the amount of the larger component. Sugar analyses of LPSs from high and low aeration indicated that the larger LPS component contained more galactose residues per molecule. Use of different media for cell growth may also result in small, but noticeable, variations in the LPS components and in the galactose content of the LPS. The observed heterogeneity of N. meningitidis LPS may explain why many strains of N. meningitidis appear to possess more than one immunotype.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Apicella M. A. Lipopolysaccharide-derived serotype polysaccharides from Neisseria meningitidis group B. J Infect Dis. 1979 Jul;140(1):62–72. doi: 10.1093/infdis/140.1.62. [DOI] [PubMed] [Google Scholar]
- BRANHAM S. E. Serological relationships among meningococci. Bacteriol Rev. 1953 Sep;17(3):175–188. doi: 10.1128/br.17.3.175-188.1953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boykins R. A., Liu T. Y. Automatic analysis of neutral sugar components in glycoproteins and complex carbohydrates. J Biochem Biophys Methods. 1980 Jan-Feb;2(1):71–78. doi: 10.1016/0165-022x(80)90075-5. [DOI] [PubMed] [Google Scholar]
- COLLINS F. M. THE EFFECT OF THE GROWTH RATE ON THE COMPOSITION OF S. ENTERITIDIS CELL WALLS. Aust J Exp Biol Med Sci. 1964 Apr;42:255–262. doi: 10.1038/icb.1964.27. [DOI] [PubMed] [Google Scholar]
- Catlin B. W. Nutritional profiles of Neisseria gonorrhoeae, Neisseria meningitidis, and Neisseria lactamica in chemically defined media and the use of growth requirements for gonococcal typing. J Infect Dis. 1973 Aug;128(2):178–194. doi: 10.1093/infdis/128.2.178. [DOI] [PubMed] [Google Scholar]
- Coleman W. G., Jr, Leive L. Two mutations which affect the barrier function of the Escherichia coli K-12 outer membrane. J Bacteriol. 1979 Sep;139(3):899–910. doi: 10.1128/jb.139.3.899-910.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- ELBEIN A. D., HEATH E. C. THE BIOSYNTHESIS OF CELL WALL LIPOPOLYSACCHARIDE IN ESCHERICHIA COLI. I. THE BIOCHEMICAL PROPERTIES OF A URIDINE DIPHOSPHATE GALACTOSE 4-EPIMERASELESS MUTANT. J Biol Chem. 1965 May;240:1919–1925. [PubMed] [Google Scholar]
- Frasch C. E., Chapman S. S. Classification of Neisseria meningitidis group B into distinct serotypes. I. Serological typing by a microbactericidal method. Infect Immun. 1972 Jan;5(1):98–102. doi: 10.1128/iai.5.1.98-102.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frasch C. E., Gotschlich E. C. An outer membrane protein of Neisseria meningitidis group B responsible for serotype specificity. J Exp Med. 1974 Jul 1;140(1):87–104. doi: 10.1084/jem.140.1.87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frasch C. E., McNelis R. M., Gotschlich E. C. Strain-specific variation in the protein and lipopolysaccharide composition of the group B meningococcal outer membrane. J Bacteriol. 1976 Aug;127(2):973–981. doi: 10.1128/jb.127.2.973-981.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldman R. C., Leive L. Heterogeneity of antigenic-side-chain length in lipopolysaccharide from Escherichia coli 0111 and Salmonella typhimurium LT2. Eur J Biochem. 1980;107(1):145–153. doi: 10.1111/j.1432-1033.1980.tb04635.x. [DOI] [PubMed] [Google Scholar]
- Hitchcock P. J., Brown T. M. Morphological heterogeneity among Salmonella lipopolysaccharide chemotypes in silver-stained polyacrylamide gels. J Bacteriol. 1983 Apr;154(1):269–277. doi: 10.1128/jb.154.1.269-277.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jennings H. J., Bhattacharjee A. K., Kenne L., Kenny C. P., Calver G. The R-type lipopolysaccharides of Neisseria meningitidis. Can J Biochem. 1980 Feb;58(2):128–136. doi: 10.1139/o80-018. [DOI] [PubMed] [Google Scholar]
- Jennings H. J., Hawes G. B., Adams G. A., Kenny C. P. The chemical composition and serological reactions of lipopolysaccharides from serogroups A,B,X, and Y Neisseria meningitidis. Can J Biochem. 1973 Oct;51(10):1347–1354. doi: 10.1139/o73-178. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Limjuco G. A., Karkhanis Y. D., Zeltner J. Y., Maigetter R. Z., King J. J., Carlo D. J. Studies on the chemical composition of lipopolysaccharide from Neisseria meningitidis group B. J Gen Microbiol. 1978 Feb;104(2):187–191. doi: 10.1099/00221287-104-2-187. [DOI] [PubMed] [Google Scholar]
- Maloney P. C., Schneider H., Brandt B. L. Production and degradation of serogroup B Neisseria meningitidis polysaccharide. Infect Immun. 1972 Nov;6(5):657–658. doi: 10.1128/iai.6.5.657-661.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mandrell R. E., Zollinger W. D. Lipopolysaccharide serotyping of Neisseria meningitidis by hemagglutination inhibition. Infect Immun. 1977 May;16(2):471–475. doi: 10.1128/iai.16.2.471-475.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Munford R. S., Hall C. L., Rick P. D. Size heterogeneity of Salmonella typhimurium lipopolysaccharides in outer membranes and culture supernatant membrane fragments. J Bacteriol. 1980 Nov;144(2):630–640. doi: 10.1128/jb.144.2.630-640.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- OSBORN M. J. STUDIES ON THE GRAM-NEGATIVE CELL WALL. I. EVIDENCE FOR THE ROLE OF 2-KETO- 3-DEOXYOCTONATE IN THE LIPOPOLYSACCHARIDE OF SALMONELLA TYPHIMURIUM. Proc Natl Acad Sci U S A. 1963 Sep;50:499–506. doi: 10.1073/pnas.50.3.499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Orskov I., Orskov F., Jann B., Jann K. Serology, chemistry, and genetics of O and K antigens of Escherichia coli. Bacteriol Rev. 1977 Sep;41(3):667–710. doi: 10.1128/br.41.3.667-710.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palva E. T., Mäkelä P. H. Lipopolysaccharide heterogeneity in Salmonella typhimurium analyzed by sodium dodecyl sulfate polyacrylamide gel electrophoresis. Eur J Biochem. 1980;107(1):137–143. doi: 10.1111/j.1432-1033.1980.tb04634.x. [DOI] [PubMed] [Google Scholar]
- Tsai C. M., Frasch C. E. A sensitive silver stain for detecting lipopolysaccharides in polyacrylamide gels. Anal Biochem. 1982 Jan 1;119(1):115–119. doi: 10.1016/0003-2697(82)90673-x. [DOI] [PubMed] [Google Scholar]
- Tsai C. M., Frasch C. E. Chemical analysis of major outer membrane proteins of Neisseria meningitidis: comparison of serotypes 2 and 11. J Bacteriol. 1980 Jan;141(1):169–176. doi: 10.1128/jb.141.1.169-176.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zollinger W. D., Mandrell R. E. Outer-membrane protein and lipopolysaccharide serotyping of Neisseria meningitidis by inhibition of a solid-phase radioimmunoassay. Infect Immun. 1977 Nov;18(2):424–433. doi: 10.1128/iai.18.2.424-433.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zollinger W. D., Mandrell R. E. Type-specific antigens of group A Neisseria meningitidis: lipopolysaccharide and heat-modifiable outer membrane proteins. Infect Immun. 1980 May;28(2):451–458. doi: 10.1128/iai.28.2.451-458.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]