Abstract
The lack of B cells and antibody does not prevent mice from dealing effectively with a pathogenic γ-herpesvirus. Both CD4+ and CD8+ T cells contribute to the control of virus replication in the respiratory tract, with the depletion of either lymphocyte subset leading to increased titers in the lung. However, the further neutralization of IFN-γ diminishes the effectiveness of the CD4+ T cell response and causes substantially increased mortality. Experiments with bone marrow radiation chimeras indicate that the immune CD4+ effectors operate optimally when there is the potential for direct interaction with virus-infected targets expressing MHC class II glycoproteins, suggesting that the IFN-γ produced by these lymphocytes is functioning at short range. The numbers of latently infected cells in the spleens of carrier mice are also significantly increased by the concurrent depletion of both the CD4+ population and IFN-γ. These experiments raise the possibility that the defective control of intercurrent γ-herpesvirus infections in patients with AIDS not only is due solely to the absence of helper T cells but also reflects the loss of an important set of CD4+ effectors.
The γ-herpesviruses (HVs) cause persistent infections with the potential for tumorigenesis (1–4). The most thoroughly analyzed is Epstein–Barr virus (EBV), which establishes first as a lytic infection of epithelial cells in the oropharynx, followed by lifelong latency in B lymphocytes. The acute phase is rapidly controlled by the specific host response, whereas continued immune surveillance by CD8+ T cells is considered to keep the latent infection in check (3, 5). The virus reactivates in patients with HIV-induced AIDS and in transplant recipients receiving immunosuppressive therapy (6). The possible consequences include greatly increased EBV excretion from the oropharynx, with associated oral hairy leukoplakia in AIDS patients, and the development of B cell lymphomas (7, 8). Much less is known about another human γ-HV, human HV-8, which has only been recognized since the onset of the HIV epidemic and is thought to be the cause of the AIDS-associated Kaposi’s sarcoma (4).
The principal target of HIV is, of course, the CD4+ T cell (9). The diminished control of HIV that leads to AIDS is considered to reflect a progressive decline of CD4+ T help for the CD8+ effector population (10). This idea (11–15) comes from experiments with lymphocytic choriomeningitis virus, a mouse arenavirus. Although lymphocytic choriomeningitis virus-specific CD8+ T cells continue to be present in persistently infected mice that lack CD4+ T cells, it is clear that many of these lymphocytes have lost the capacity to function (16). Decreased T help could also be thought to explain the emergence of γ-HV-induced disease in AIDS patients (4, 8) because all activated CD4+ T cells are targets for HIV infection (9).
The issue of defective immune control in the absence of the CD4+ subset (17) has also been addressed experimentally for the γ-HV-68 (MHV-68), which has considerable sequence homology to human HV-8 (18) and establishes lifelong latency in B lymphocytes and macrophages (19, 20). The conclusions differ from those reached for lymphocytic choriomeningitis virus (11, 16). Although mice that are CD4+ T cell-deficient as a consequence of homozygous disruption (−/−) of the H-2I-Ab MHC class II gene show evidence of reactivated, lytic infection in epithelial sites and eventually die from a wasting disease (17), MHV-68-specific CD8+ T cells are neither decreased in numbers nor unable to function at any stage of this process (21).
These observations with MHV-68 suggest two alternative explanations for the diminished control of the γ-HVs in AIDS. One is that CD4+ T help for B cells maintains a specific Ig response that functions to keep these viruses in check. Experiments with MHV-68 suggest that humoral immunity may, indeed, be important because depleting persistently infected, conventional mice by treatment with monoclonal antibodies (mAbs) to CD4 did not cause any reactivation of the lytic infection (22). The alternative possibility, that the CD4+ subset can exert a measure of direct control in addition to that mediated by CD8+ effector T cells, can only be addressed in the absence of a concurrent B-cell response. We have thus analyzed the consequences of CD4+ T cell depletion in MHV-68-infected Ig−/− μMT mice (23–25).
MATERIALS AND METHODS
Immunodeficient and Chimeric Mice.
The Ig−/− μMT mice (23) and MHC class II−/− C2D (26) mice were bred at St. Jude Children’s Research Hospital, whereas the congenic (H-2b) Ig+/+/MHC class II+/+ C57BL/6J (B6) controls were purchased from the Jackson Laboratories (Bar Harbor, ME). These female mice were free of specific pathogens, except for intranasal (i.n.) infection with 600 plaque-forming units (pfu) of MHV-68 under Avertin (2,2,2-tribromoethanol) anesthesia at 6–12 weeks of age (17, 27). Chimeric animals selectively lacking functional B cells and a capacity for MHC class II expression on lung epithelium (μMT → C2D) were made (28) by the i.v. reconstitution of irradiated (950 rad from a 137Cs γ source) C2D mice with 1.3 × 107 μMT bone marrow cells and 1.3 × 107 μMT lymphocytes (spleen and lymph node cells). The following day, each recipient received a further i.p. dose of 9 × 106 μMT lymphocytes. The controls were irradiated B6 mice that had been reconstituted in the same way (μMT → B6). The chimeras were then left for 24 h prior to commencing mAb treatment (27, 29) for the selective depletion of IFN-γ or T cell subsets (see below). Flow cytometric analysis (30) at the time of sampling confirmed reconstitution with CD4+ and CD8+ T cells and the lack of Ig+/+ B cells, for both the μMT → C2D and μMT → B6 chimeras.
Virus, Titration, and Statistical Analysis.
The MHV-68 stock was grown in OMK cells from an isolate originally provided by A. A. Nash (31). The extent of lytic infection in the lung was determined by using a well-established 6-day plaque assay with NIH 3T3 fibroblast monolayers under carboxymethylcellulose (17). Latent virus titers were determined for single-cell spleen suspensions by a modification of a limiting dilution assay (25) that measures the extent of virus reactivation over a 3–4-week incubation period (37°C, 5% C02). Low-passage NIH 3T3 cells were plated in DMEM (supplemented with 10% FCS, 2 mM l-glutamine, 4 mM Hepes, and gentamicin) at 1 × 104 cells/well (0.2 ml) in 96-well flat-bottomed plates and allowed to adhere for 3–4 h. Spleen cell suspensions free of erythrocytes were then added (starting with 5 × 104 cells per 0.1 ml/culture well) in eight serial 3-fold dilutions, by using a minimum of 24 replicates/dilution. A parallel series of cultures was plated with cell equivalents that had previously been subjected to mechanical disruption (four freeze–thaw cycles), to determine the level of lytic virus present before reactivation. Frequency estimates were determined by applying the Poisson formula on the number of negative wells/dilution. The measure of virus reactivation from latency (latent virus) was determined by subtracting the values obtained when disrupted spleen cells were cultured with NIH 3T3 cells (replicating virus) from the total counts recorded for viable lymphocytes. The data were analyzed by the Mann–Whitney test.
In Vivo Depletion and Measuring IFN-γ.
The mice were injected i.p. every second day with 0.5-ml aliquots of ascitic fluid containing mAbs to CD4 (GK1.5), CD8α (2.43), or IFN-γ (XMG1.2). The treatment commenced either 4 days prior to infection or, in mice with established disease, at least 14 days before sampling. These depletion protocols are used routinely in the laboratory (27, 29, 32), and they were checked for effectiveness by flow cytometry with noncrossreactive mAbs to CD4 or CD8 or for evidence of neutralization of IFN-γ in the fluid phase obtained by bronchoalveolar lavage (BAL). The IFN-γ levels were calculated by comparison with standard curves generated by using recombinant IFN-γ, with the limit of detection of the sandwich ELISA kit (Endogen, Cambridge, MA) being 15 pg/ml.
Flow Cytometry.
Single-cell suspensions were stained by incubating 1 × 106 cells in the individual wells of a 96-well round-bottomed microtiter plate with mAbs diluted in FACS medium (1% BSA and 0.1% NaN3 in PBS) (30). After a 30-min incubation on ice, the cells were washed three times with FACS medium, fixed with 1% ice-cold paraformaldehyde in PBS, and analyzed on a FACScan by using CellQuest Version 3.1 software (Becton Dickinson, San Jose, CA). The efficiency of the in vivo mAb depletions was determined by staining with anti-CD8β (53–5.8) and anti-CD4 (RM4–4). In general, <1% of the original subset remained. The extent of T cell reconstitution in the chimeric mice was checked by staining for CD4 and CD8, whereas the lack of B cells was confirmed by staining for B220. The level of CD4+ T cell activation (28, 30) was assessed by staining for CD44 (IM7) and CD62L (Mel 14). The mAbs conjugated to phycoerythrin, Cy-chrome, and FITC were purchased from PharMingen (San Diego, CA).
RESULTS
CD4+ T Cells Can Limit the Acute Phase of MHV-68 Infection.
An earlier study with BALB/c mice indicated that MHV-68 infection is lethal in the absence of CD8+ effectors (33). This was not the case in later experiments with the B6 strain (22), although there is always the concern that the long-term efficacy of CD8+ T cell depletion with mAbs may be compromised by the development of an antibody response to the rat Ig used for treatment. Neutralization of the mAb reagents is obviously not a problem with the Ig−/− μMT mice. It is clear that CD4+ T cells can indeed deal with the lytic, respiratory epithelial phase of MHV-68 replication (anti-CD8, Table 1). Any CD4+ or CD8+ T cell-independent mechanism for controlling MHV-68 is ultimately ineffective (anti-CD4/8, Table 1), although studies with other HVs suggest that natural killer cells may well play a part in limiting the early phase of this infectious process (34–37).
Table 1.
MHV-68 titers in the lungs of μMT mice
| Treatment | Days after infection
|
||
|---|---|---|---|
| 17 | 21 | 29 | |
| Undepleted | 0.82 ± 0.17 | 1.15 ± 0.20 | 0.10 ± 0.17 |
| Anti-CD4 | ND | 3.12 ± 0.48 | 2.95 ± 0.47 |
| Anti-CD8 | ND | 2.68 ± 0.29 | 0.00 ± 0.00 |
| Anti-CD4/CD8 | 8.00 ± 0.01 | All dead | |
The μMT mice were dosed i.p. (27, 32) with the GK1.5 and/or 2.43 mAbs every second day, starting 4 days before i.n. infection with 600 pfu of MHV-68. Groups of three or four mice were sampled at different times after virus challenge, and titers are expressed as mean ± SD log10 values of lung homogenates (pfu/ml). ND, not done.
Mechanism of CD4+ T Cell Effector Function.
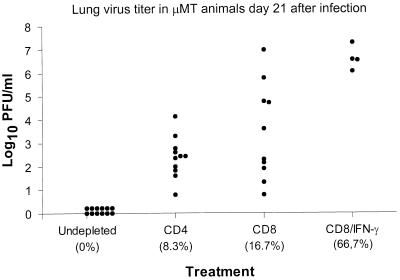
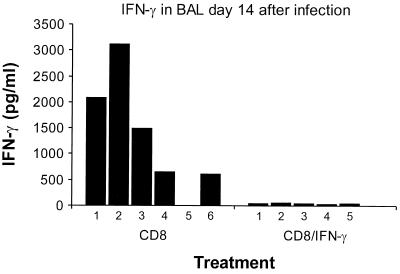
The control of MHV-68 infection in μMT mice by CD4+ T cells acting in the absence of either antibody or the CD8+ subset (anti-CD8, Table 1) rules out any process involving T cell help. A further experiment illustrated quite clearly that the CD4+ and CD8+ populations optimally operate together to limit the lytic phase of MHV-68 infection (CD4 or CD8, Fig. 1). The spread in lung virus titers for the CD8-depleted group at day 21 after infection was considerable (CD8, Fig. 1) and is reminiscent of the spectrum found previously for secondarily stimulated CD4+ effectors in μMT mice infected with an influenza A virus (28). This variability was removed and the lethality of the infectious process was greatly enhanced by the concurrent treatment of the CD8-depleted mice with a mAb to IFN-γ (CD8/IFN-γ, Fig. 1). Exposure to the XMG1.2 mAb throughout the course of a virus-specific host response does not necessarily inhibit the development of lymphocytes with the capacity to produce IFN-γ (29). However, antibody-mediated neutralizing of any IFN-γ produced by T cells that had localized to the lung was confirmed in a further experiment in which levels of this cytokine were determined in the supernatant fluid obtained by BAL (Fig. 2).
Figure 1.
Control of the respiratory phase of MHV-68 infection by T cells and IFN-γ. The Ig−/− μMT mice were dosed i.p. (27, 29, 32) with the GK1.5 (CD4), 2.43 (CD8), or XMG1.2 (IFN-γ) mAbs, starting 2 days prior to i.n. challenge with 600 pfu of MHV-68. The data are compiled from two experiments, giving final group sizes of 12 mice. The values in the parentheses show the percentage mortality prior to time of sampling on day 21 after virus challenge.
Figure 2.
Neutralization of IFN-γ in the fluid phase obtained by BAL (27) from μMT mice challenged 14 days previously with MHV-68. Treatment with the XMG1.2 mAb (29) was begun 2 days prior to i.n. infection with 600 pfu of virus and then continued every second day until time of sampling. The numbers given are for individual mice, and the sensitivity of the ELISA was 15 pg/ml.
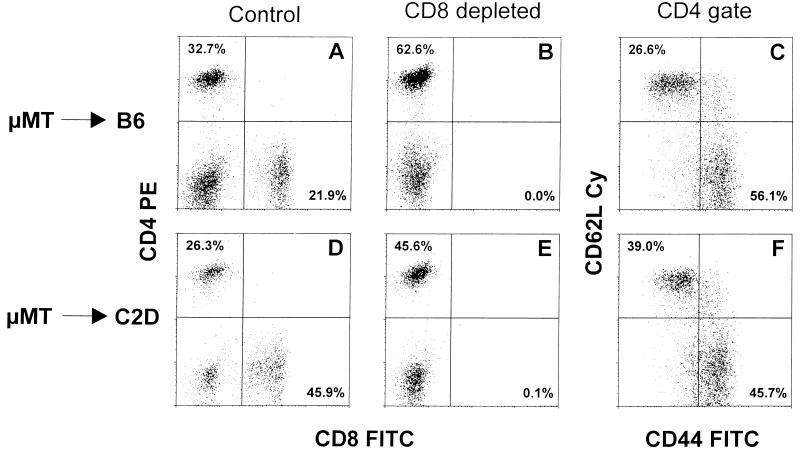
The fact that antibody to IFN-γ can modify the profile of CD4+ T cell-mediated control suggests that this cytokine is operating in the fluid phase rather than at an intimate interface between the immune lymphocytes and virus-infected lung cells. Thus, we asked whether it is important for the MHC class II glycoproteins that target the CD4+ effectors to be expressed on respiratory epithelium by making short-term bone marrow radiation chimeras (28) that were also reconstituted with mature CD4+ and CD8+ T cells (Fig. 3). The protocol used MHC class II−/− (C2D) or MHC class II+/+ (B6) recipients, Ig−/− lymphocytes (μMT), and MHC class II+/+ bone marrow (μMT).
Figure 3.
The magnitude of the CD4+ and CD8+ T cell responses in spleen (A and D) and the extent of depletion following treatment with the 2.43 mAb to CD8 (B and E) is shown for μMT → B6 (A and B) and μMT → C2D (D and E) chimeras at 21 days after i.n. infection with MHV-68. In both cases, the predominant CD4+ T cell population in mice lacking the CD8+ subset showed the activated CD44high CD62Llow phenotype (C and F, Lower Right). The μMT → B6 mice have the potential to express the MHC class II glycoproteins that target the CD4+ subset in the radiation sensitive (μMT) and radiation-resistant (B6) compartments, whereas only cells of recent hemopoietic origin (μMT) could be MHC class II+ stimulators in the μMT → C2D chimeras.
As would be expected, the virus-specific CD8+ T cells (Fig. 3, A and D) were operating in the μMT → B6 and μMT → C2D mice, with their elimination by mAb treatment (Fig. 3, B and E) significantly increasing virus titers in both cases (CD8, Table 2). However, the higher levels of both lytic (lung) and latent (spleen) virus and the greater mortality in the undepleted μMT → C2D group indicates that the CD4+ T cells did not function optimally (if at all) in these mice (undepleted, Table 2), although there is the potential for MHC class II glycoprotein expression on monocyte/macrophages and dendritic cells originating from the μMT bone marrow. Apparently, the control of MHV-68 by activated CD4+ T cells (Fig. 3, C and F) is optimal when MHC class II glycoproteins can be expressed on the target cells supporting virus replication and/or latency. Obviously, the lung epithelial cells of the μMT → B6 chimeras have the potential to express the MHC class II phenotype of the radiation-resistant host, but the minimal effectiveness of the CD4+ T cells in the μMT → C2D chimeras also raises the possibility (compare CD8 groups, Table 2) that at least a proportion of the latently infected cells in the spleen may be from the radiation-resistant background (C2D) rather than of immediate hemopoietic origin (μMT). What is again apparent (Fig. 1; CD8/IFN-γ, Table 2) is that any control mechanism involving the CD4+ T cells (Fig. 3) is ineffectual in the absence of IFN-γ (Fig. 2).
Table 2.
Effect of differential MHC class II glycoprotein expression in the radiation-resistant compartment on the control of MHV-68 infection in chimeric mice
| Group | Depletion | Mortality | Virus in lung
|
Virus in spleen
|
||
|---|---|---|---|---|---|---|
| No. | Titer | Latent virus | Replicating virus | |||
| Nil | 2/9 | 2 | 0.30, 0.30 | 0.74 ± 0.81* | 1.19 ± 0.49 | |
| μMT → B6 | CD8 | 0/7 | 7 | 2.46 ± 1.03†‡ | 2.78 ± 0.44†§ | 1.28 ± 0.26‡ |
| CD8/IFN-γ | 5/7 | 2 | 4.19 ± 0.58 | 4.45 ± 0.84 | 2.29 ± 0.57 | |
| Nil | 6/10 | 4 | 2.24 ± 0.23 | 3.10 ± 0.39* | 1.06 ± 0.70 | |
| μMT → C2D | CD8 | 2/10 | 8 | 5.27 ± 1.29‡§ | 4.45 ± 0.54†‡ | 2.64 ± 0.40†‡ |
| CD8/IFN-γ | 6/6 | |||||
The chimeric mice were dosed i.p. with mAbs to CD4, CD8, and IFN-γ every second day, commencing 2 days before i.n. challenge with 600 pfu of MHV-68. Some of the mice died (mortality) before the time of sampling on day 21 after infection. No virus was detected in the lungs of 5 of 7 undepleted μMT → B6 mice, but all other lung samples were positive and evidence of both virus replication and latency was found uniformly for the spleens. The titers shown are the mean ± SD log10 values for the lungs (pfu/ml) and the mean ± SD log10 values for cell equivalents containing virus for the spleens.
These two values were significantly different (P < 0.02).
P < 0.01, compared to the Nil group.
These two values were significantly different (P < 0.02).
P < 0.05, compared to the Nil group.
Increased Virus Load in Persistently Infected μMT Mice.
Eliminating both T cell subsets from mice infected at least 4.5 months previously leads to the consistent (albeit low-level) reactivation of the lytic phase in the lung, an increase in the numbers of latently infected cells, and the death of some mice within 21 days of initiating the lymphocyte depletions (CD4/CD8, Table 3). Further neutralization of IFN-γ, which may be produced by natural killer cells, was associated with increased mortality, although we did not measure a significant effect on virus titers in the lungs (experiment 2, Table 3). As with the MHC class II−/− mice (17), the amounts of virus detected do not explain the fatal outcome.
Table 3.
Consequences of depleting T cells subsets and IFN-γ in persistently infected μMT mice
| Experiment | Depletion | Mortality | Virus in lung
|
Virus in spleen
|
||
|---|---|---|---|---|---|---|
| No. | Titer virus | Latent virus | Replicating virus | |||
| 1 | Nil | 0/15 | 0/10 | 0.62 ± 0.57 | 0.64 ± 0.08 | |
| Day 14 | CD4 | 0/10 | 3/5 | 0.50 ± 0.17 | 1.71 ± 0.43* | 0.72 ± 0.27 |
| CD8 | 0/14 | 5/9 | 1.32 ± 0.36 | 1.26 ± 0.70† | 0.74 ± 0.40 | |
| CD8/IFN-γ | 0/14 | 8/9 | 0.94 ± 0.28 | 2.73 ± 0.73*† | 0.92 ± 0.25* | |
| CD4/CD8 | 0/13 | 4/5 | 1.90 ± 0.21 | 2.62 ± 0.52* | 0.81 ± 0.38 | |
| Day 21 | Nil | 0/5 | 0/5 | 0.58 ± 0.63 | 0.60 ± 0.14 | |
| CD4 | 0/5 | 2/5 | 0.30, 0.18 | 1.10 ± 1.17 | 0.63 ± 0.12 | |
| CD8 | 0/5 | 0/5 | 0.63 ± 0.64 | 0.54 ± 0.05 | ||
| CD8/IFN-γ | 2/5 | 1/3 | 0.30 | 2.37 ± 0.54‡ | 0.77 ± 0.32 | |
| CD4/CD8§ | 3/8 | 5/5 | 2.41 ± 0.23 | 2.15 ± 0.32* | 0.70 ± 0.22 | |
| 2 | CD4/CD8 | 0/9 | 8/9 | 1.55 ± 0.52 | 2.76 ± 0.41 | 0.67 ± 0.73 |
| Day 16 | CD4/CD8/IFN-γ | 3/9 | 6/6 | 2.22 ± 0.64 | 3.05 ± 0.11 | 1.42 ± 0.61 |
Mice that had been infected i.n. with 600 pfu of virus 4.5 (experiment 1) or 5.5 (experiment 2) months previously were treated every second day with mAbs to CD4, CD8 or IFN-γ and sampled after a further 14–21 days. The mean ± SD log10 values for lung samples (pfu/ml) are shown where more than two were positive. All spleen samples were positive, and titers are expressed as the mean ± SD log10 values for cell equivalents containing virus.
P < 0.02, compared to Nil group.
These two values were significantly different (P < 0.05).
P < 0.05, compared to Nil group.
These mice were sampled when moribund on day 19 after the mAb depletions were started.
The titers of replicating virus in lung and spleen samples from these persistently infected mice were generally too low to allow any useful conclusions about the nature of the differential immune control of reactivation to lytic phase (Table 3). Otherwise, a significant increase in the extent of latency in the spleen was only seen for CD8-depleted mice that were also treated with the mAb to IFN-γ (CD8/IFN-γ, Table 3). In fact, the numbers of infected spleen cells in the CD8/IFN-γ and CD4/CD8 depletion groups were equivalent (Table 3). As with the acute infection (Figs. 1 and 2, Table 2), the results indicate that any CD4+ T cell-mediated control of latent MHV-68 in lymphoid tissue is IFN-γ-dependent (Table 3).
DISCUSSION
Experiments with IFN-γ−/− and IFN-γ receptor−/− mice indicate that this cytokine is not essential for controlling the initial stages of MHV-68 infection (38, 39), although it is produced at high levels during the acute phase of the host response in conventional mice (40). Under different regimes of virus challenge, however, the IFN-γ receptor−/− mice may develop either acute splenic fibrosis (39) or a chronic vascular disease characterized by persistent, lytic infection of smooth muscle cells in the large arteries (41). The reactive pathology in the blood vessel wall leads to progressive thickening and eventual occlusion, with death resulting from vascular insufficiency. Neither the acute splenic atrophy nor the chronic arterial disease was observed here, although the IFN-γ-depletion experiments were probably too short to allow time for the emergence of the latter.
The MHV-68-specific CD4+ T cell response in B6 mice is substantial and is maintained at a constant level in the face of viral persistence (42). Production of IFN-γ at levels detectable by a single cell ELISpot assay (43) is largely a property of the CD4+ subset following in vitro culture with irradiated, virus-infected stimulator cells, although CD8+ T cells synthesize this cytokine when high doses of cognate peptide are substituted for live virus (21, 42). The finding that there is an effective CD4+ T cell response to MHV-68 in the Ig−/− μMT mice is not surprising because previous studies with the influenza A viruses have shown that both the initial clonal expansion and the maintenance of this lymphocyte subset follows essentially normal kinetics. Even so, the influenza-specific response overall was of lower total magnitude than that observed in the Ig+/+ controls (44). The reason for the difference is that the Ig−/− mice have very small spleens, an effect that may be associated with the absence of germinal centers and follicular dendritic cells (44).
The theme that IFN-γ is important for limiting the extent of HV infections has also emerged from mouse experiments with other HVs (45–50). The CD4+ T cell-mediated control of cutaneous zosteriform lesions induced by herpes simplex virus is significantly diminished in IFN-γ−/− BALB/c mice (47, 48). Similarly, herpes simplex virus clearance following local infection of the mouse eye depends on IFN-γ, although the stromal keratitis associated with the CD4+ T cell response is not obviously less severe in the absence of this cytokine (49). Treatment of cultured human keratinocytes with IFN-γ has, however, been shown to promote MHC class II glycoprotein expression and to increase the extent of CD4+ T cell-mediated cytotoxicity (51). Similarly, the control of persistent murine cytomegalovirus infection in the mouse salivary gland depends on both IFN-γ and CD4+ T cells (37, 45).
A variety of molecular mechanisms for diminishing the expression of peptide-loaded MHC class I glycoproteins have now been identified for both the α(herpes simplex virus) and β(cytomegalovirus) HVs (reviewed in ref. 52). At least some of these inhibitory processes can be modified by exposure to IFN-γ. Such effects have yet to be identified for the γ-HVs. The mechanism that EBV uses to escape immune surveillance during latency is to shut down the expression of various viral genes in particular cell types (5). Similarly, current experiments are identifying the limited spectrum of MHV-68 genes expressed in the persistent, latent phase (53, 54).
It is abundantly clear from the human experience with EBV in the immunosuppressed and from mouse studies with murine cytomegalovirus, herpes simplex virus, and MHV-68 that the immune system functions to maintain viral latency. Although the focus with EBV has largely been on the CD8+ T cell response, the experiments with both murine cytomegalovirus and MHV-68 highlight the fact that CD4+ T cells are also important effectors that operate, at least in part, via mechanisms involving IFN-γ. Lack of CD4+ T cell help for CD8+ T cells may not be the sole or even the primary mechanism underlying the induction of the disease processes classically associated with EBV and human HV-8 in AIDS patients. Like the intracellular bacteria (55), the γ-HVs seem, at least in part, to be controlled by mechanisms involving IFN-γ production by CD4+ effector T cells.
Acknowledgments
We thank Vicki Henderson for help with the manuscript and Mehdi Mehrpooya for assistance with virus titrations. J.P.C. is the recipient of a fellowship from the Alfred Benson Foundation (Copenhagen, Denmark). This work was supported by United States Public Health Service Grants CA 21765 and AI38359 and by the American Lebanese-Syrian Associated Charities.
ABBREVIATIONS
- HV
herpesvirus
- EBV
Epstein–Barr virus
- MHV-68
murine γ-HV-68
- mAb
monoclonal antibody
- i.n.
intranasal(ly)
- pfu
plaque-forming unit(s)
- BAL
bronchalveolar lavage
References
- 1.Borysiewicz L K, Sissons J G. Curr Top Microbiol Immunol. 1994;189:123–150. doi: 10.1007/978-3-642-78530-6_8. [DOI] [PubMed] [Google Scholar]
- 2.Klein G. Cell. 1994;77:791–793. doi: 10.1016/0092-8674(94)90125-2. [DOI] [PubMed] [Google Scholar]
- 3.Rickinson A B, Moss D J. Annu Rev Immunol. 1997;15:405–431. doi: 10.1146/annurev.immunol.15.1.405. [DOI] [PubMed] [Google Scholar]
- 4.Ganem D. Curr Biol. 1995;5:469–471. doi: 10.1016/s0960-9822(95)00093-5. [DOI] [PubMed] [Google Scholar]
- 5.Heslop H E, Brenner M K, Rooney C. Hum Gene Ther. 1994;5:381–397. doi: 10.1089/hum.1994.5.3-381. [DOI] [PubMed] [Google Scholar]
- 6.Yao Q Y, Rickinson A B, Gaston J S, Epstein M A. Int J Cancer. 1985;35:43–49. doi: 10.1002/ijc.2910350108. [DOI] [PubMed] [Google Scholar]
- 7.Walling D M, Raab-Traub N. J Virol. 1994;68:7909–7917. doi: 10.1128/jvi.68.12.7909-7917.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Brink A A, Dukers D F, van den Brule A J, Oudejans J J, Middeldorp J M, Meijer C J, Jiwa M. J Clin Pathol. 1997;50:911–918. doi: 10.1136/jcp.50.11.911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Pantaleo G, Fauci A S. Annu Rev Microbiol. 1996;50:825–854. doi: 10.1146/annurev.micro.50.1.825. [DOI] [PubMed] [Google Scholar]
- 10.Kalams S A, Walker B D. J Exp Med. 1998;188:2199–2204. doi: 10.1084/jem.188.12.2199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Moskophidis D, Lechner F, Pircher H, Zinkernagel R M. Nature (London) 1993;362:758–761. doi: 10.1038/362758a0. [DOI] [PubMed] [Google Scholar]
- 12.Matloubian M, Concepcion R J, Ahmed R. J Virol. 1994;68:8056–8063. doi: 10.1128/jvi.68.12.8056-8063.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Battegay M, Moskophidis D, Rahemtulla A, Hengartner H, Mak T W, Zinkernagel R M. J Virol. 1994;68:4700–4704. doi: 10.1128/jvi.68.7.4700-4704.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Thomsen A R, Johnansen J, Marker O, Christensen J P. J Immunol. 1996;157:3074–3080. [PubMed] [Google Scholar]
- 15.Christensen J P, Marker O, Thomsen A R. Scand J Immunol. 1994;40:373–382. doi: 10.1111/j.1365-3083.1994.tb03477.x. [DOI] [PubMed] [Google Scholar]
- 16.Zajac A J, Blattman J N, Murali-Krishna K, Sourdive D J D, Suresh M, Altman J D, Ahmed R. J Exp Med. 1998;188:2205–2213. doi: 10.1084/jem.188.12.2205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Cardin R D, Brooks J W, Sarawar S R, Doherty P C. J Exp Med. 1996;184:863–871. doi: 10.1084/jem.184.3.863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Virgin H W, Latreille P, Wamsley P, Hallsworth K, Weck K E, Dal C A, Speck S H. J Virol. 1997;71:5894–5904. doi: 10.1128/jvi.71.8.5894-5904.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Sunil-Chandra N P, Efstathiou S, Nash A A. J Gen Virol. 1992;73:3275–3279. doi: 10.1099/0022-1317-73-12-3275. [DOI] [PubMed] [Google Scholar]
- 20.Weck, K. E., Kim, S. S., Virgin, H. W. & Speck, S. H. (1999) J. Virol., in press.
- 21.Stevenson P G, Belz G T, Altman J D, Doherty P C. Proc Natl Acad Sci USA. 1998;95:15565–15570. doi: 10.1073/pnas.95.26.15565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Stevenson P G, Cardin R D, Christensen J P, Doherty P C. J Gen Virol. 1999;80:477–483. doi: 10.1099/0022-1317-80-2-477. [DOI] [PubMed] [Google Scholar]
- 23.Kitamura D, Roes J, Kuhn R, Rajewsky K. Nature (London) 1991;350:423–426. doi: 10.1038/350423a0. [DOI] [PubMed] [Google Scholar]
- 24.Usherwood E J, Stewart J P, Robertson K, Allen D J, Nash A A. J Gen Virol. 1996;77:2819–2825. doi: 10.1099/0022-1317-77-11-2819. [DOI] [PubMed] [Google Scholar]
- 25.Weck K E, Barkon M L, Yoo L I, Speck S H, Virgin H W. J Virol. 1996;70:6775–6780. doi: 10.1128/jvi.70.10.6775-6780.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Grusby M J, Johnson R S, Papaioannou V E, Glimcher L H. Science. 1991;253:1417–1420. doi: 10.1126/science.1910207. [DOI] [PubMed] [Google Scholar]
- 27.Allan W, Tabi Z, Cleary A, Doherty P C. J Immunol. 1990;144:3980–3986. [PubMed] [Google Scholar]
- 28.Topham D J, Tripp R A, Sarawar S R, Sangster M Y, Doherty P C. J Virol. 1996;70:1288–1291. doi: 10.1128/jvi.70.2.1288-1291.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Sarawar S R, Sangster M, Coffman R L, Doherty P C. J Immunol. 1994;153:1246–1253. [PubMed] [Google Scholar]
- 30.Tripp R A, Hou S, Doherty P C. J Immunol. 1995;154:5870–5875. [PubMed] [Google Scholar]
- 31.Efstathiou S, Ho Y M, Minson A C. J Gen Virol. 1990;71:1355–1364. doi: 10.1099/0022-1317-71-6-1355. [DOI] [PubMed] [Google Scholar]
- 32.Hou S, Doherty P C, Zijlstra M, Jaenisch R, Katz J M. J Immunol. 1992;149:1319–1325. [PubMed] [Google Scholar]
- 33.Ehtisham S, Sunil-Chandra N P, Nash A A. J Virol. 1993;67:5247–5252. doi: 10.1128/jvi.67.9.5247-5252.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Biron C A, Byron K S, Sullivan J L. N Engl J Med. 1989;320:1731–1735. doi: 10.1056/NEJM198906293202605. [DOI] [PubMed] [Google Scholar]
- 35.Salazar-Mather T P, Orange J S, Biron C A. J Exp Med. 1998;187:1–14. doi: 10.1084/jem.187.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Welsh R M, O’Donnell C L, Shultz L D. Natl Immun. 1994;13:239–245. [PubMed] [Google Scholar]
- 37.Polic B, Hengel H, Krmpotic A, Trgovcich J, Pavic I, Luccaronin P, Jonjic S, Koszinowski U H. J Exp Med. 1998;188:1047–1054. doi: 10.1084/jem.188.6.1047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Sarawar S R, Cardin R D, Brooks J W, Mehrpooya M, Hamilton-Easton A M, Mo X Y, Doherty P C. J Virol. 1997;71:3916–3921. doi: 10.1128/jvi.71.5.3916-3921.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Dutia B M, Clarke C J, Allen D J, Nash A A. J Virol. 1997;71:4278–4283. doi: 10.1128/jvi.71.6.4278-4283.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Sarawar S R, Cardin R D, Brooks J W, Mehrpooya M, Tripp R A, Doherty P C. J Virol. 1996;70:3264–3268. doi: 10.1128/jvi.70.5.3264-3268.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Weck K E, Dal Canto A J, Gould J D, O’Guin A K, Roth K A, Saffitz J E, Speck S H, Virgin H W. Nat Med. 1997;3:1346–1353. doi: 10.1038/nm1297-1346. [DOI] [PubMed] [Google Scholar]
- 42.Christensen, J. P. & Doherty, P. C. (1999) J. Virol., in press.
- 43.Sarawar S R, Carding S R, Allan W, McMickle A, Fujihashi K, Kiyono H, McGhee J R, Doherty P C. Reg Immunol. 1993;5:142–150. [PubMed] [Google Scholar]
- 44.Topham D J, Tripp R A, Hamilton-Easton A M, Sarawar S R, Doherty P C. J Immunol. 1996;157:2947–2952. [PubMed] [Google Scholar]
- 45.Lucin P, Pavic I, Polic B, Jonjic S, Koszinowski U H. J Virol. 1992;66:1977–1984. doi: 10.1128/jvi.66.4.1977-1984.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Cunningham A L, Merigan T C. J Immunol. 1983;130:2397–2400. [PubMed] [Google Scholar]
- 47.Smith P M, Wolcott R M, Chervenak R, Jennings S R. Virology. 1994;202:76–88. doi: 10.1006/viro.1994.1324. [DOI] [PubMed] [Google Scholar]
- 48.Bouley D M, Kanangat S, Wire W, Rouse B T. J Immunol. 1995;155:3964–3971. [PubMed] [Google Scholar]
- 49.Yu Z Y, Manickan E, Rouse B T. J Leukocyte Biol. 1996;60:528–532. doi: 10.1002/jlb.60.4.528. [DOI] [PubMed] [Google Scholar]
- 50.Presti R M, Pollock J L, Dal C A, O’Guin A K, Virgin H W. J Exp Med. 1998;188:577–588. doi: 10.1084/jem.188.3.577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Mikloska Z, Kesson A M, Penfold M E, Cunningham A L. J Infect Dis. 1996;173:7–17. doi: 10.1093/infdis/173.1.7. [DOI] [PubMed] [Google Scholar]
- 52.Johnson D C, Hill B. Curr Top Microbiol Immunol. 1998;232:150–177. doi: 10.1007/978-3-642-72045-1_8. [DOI] [PubMed] [Google Scholar]
- 53.Bowden R J, Simas J P, Davis A J, Efstathiou S. J Gen Virol. 1997;78:1675–1687. doi: 10.1099/0022-1317-78-7-1675. [DOI] [PubMed] [Google Scholar]
- 54.Virgin, H. W., Presti, R. M., Li, X-Y, Liu, C. & Speck, S. H. (1999) J. Virol., in press. [DOI] [PMC free article] [PubMed]
- 55.Kaufmann S H. Curr Opin Immunol. 1994;6:518–525. doi: 10.1016/0952-7915(94)90135-x. [DOI] [PubMed] [Google Scholar]