Abstract
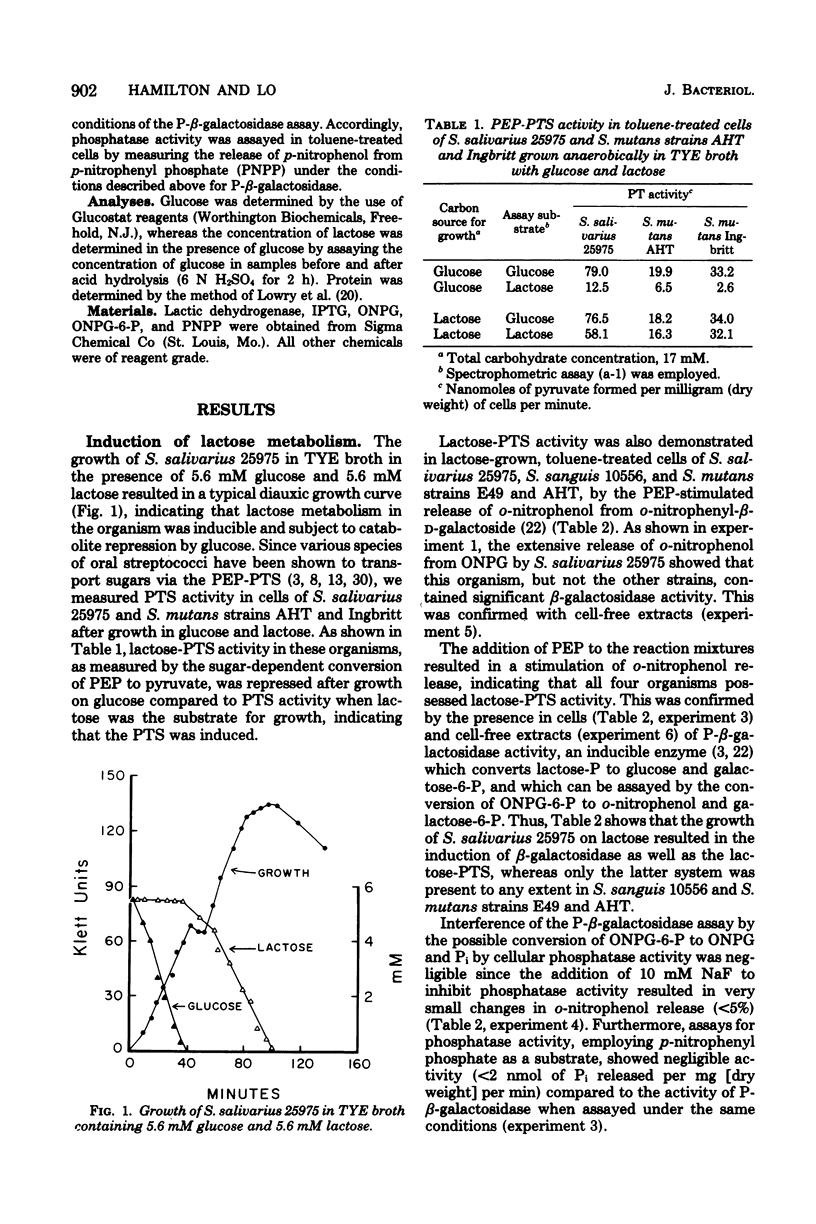
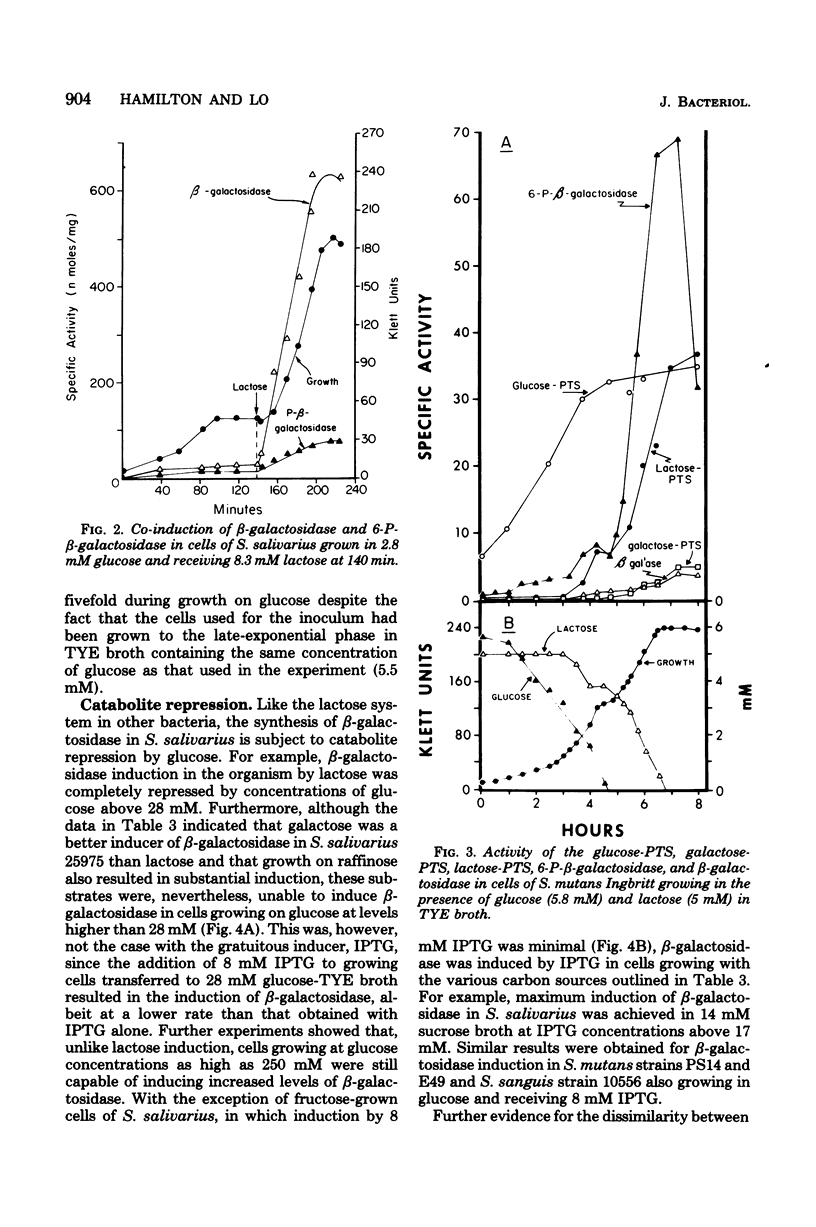
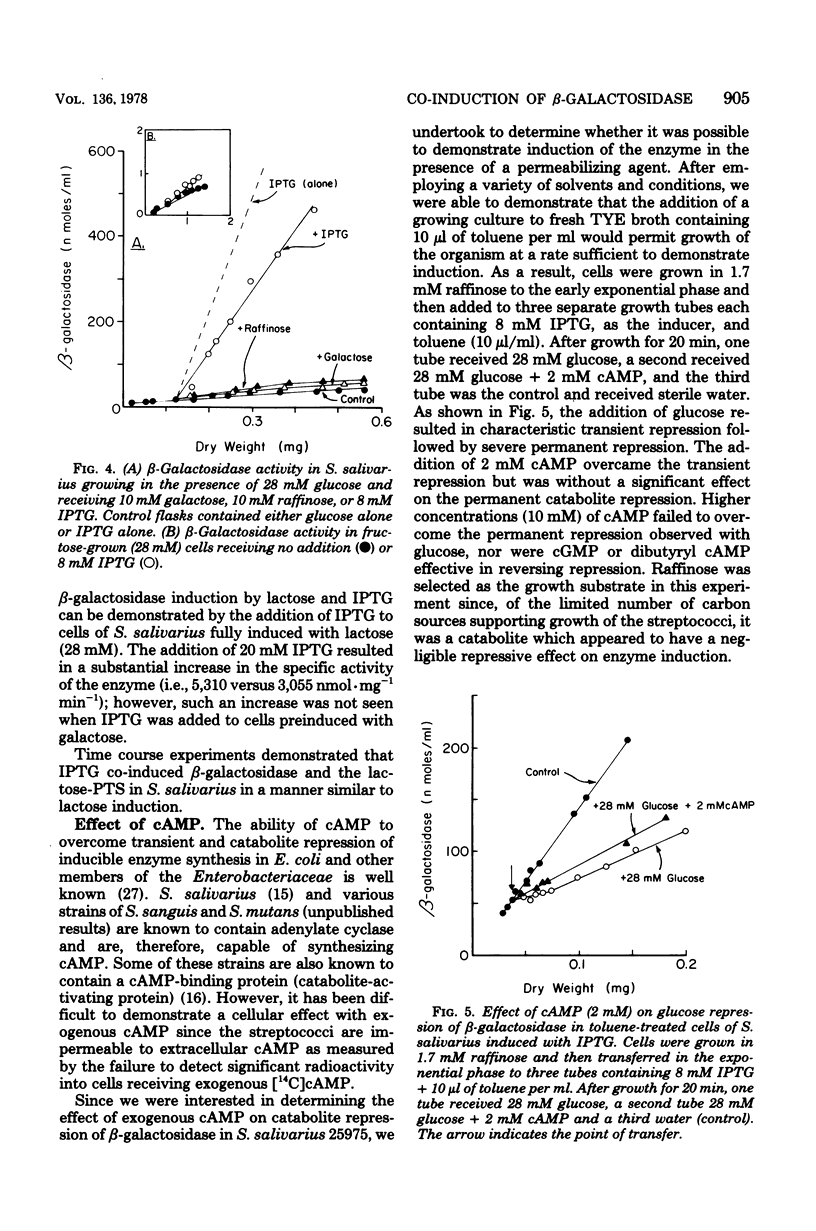
The addition of lactose, galactose, or isopropyl-beta-D-thiogalactoside (IPTG) to glucose-grown cells of Streptococcus salivarius 25975 resulted in the co-induction of both the lactose-P-enolpyruvate phosphotransferase system (lactose-PTS) and beta-galactosidase, with the latter the predominant metabolic system. With various strains of Streptococcus mutans and Streptococcus sanguis 10556, on the other hand, the lactose-PTS was the major metabolic pathway with beta-galactosidase induced either to low or negligible levels. In all cases, induction of the lactose-PTS resulted in the concomitant induction of 6-P-beta-galactosidase. The induction by lactose of both the lactose-PTS and beta-galactosidase in all strains was repressed by glucose and other catabolites, notably, fructose. Induction of beta-galactosidase in S. salivarius 25975 by IPTG was, however, relatively resistant to glucose repression. Induction experiments with IPTG and lactose suggested that a cellular metabolite of lactose metabolism was a repressor of enzyme activity. Exogenous cAMP was shown to reverse the transient repression by glucose of beta-galactosidase induction in cells of S. salivarius 25975 receiving lactose, provided the cells were grown with small amounts of toluene to overcome the permeability barrier to this nucleotide, cAMP, was however, unable to overcome the permanent repression of beta-galactosidase activity to a significant extent under these conditions.
Full text
PDF





Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Brown A. T., Christian C. P. Characterization of a novel nicotinamide adenine dinucleotide-dependent alcohol dehydrogenase from a strain of Streptococcus mutans. Arch Oral Biol. 1974 May;19(5):397–406. doi: 10.1016/0003-9969(74)90181-2. [DOI] [PubMed] [Google Scholar]
- Brown A. T., Wittenberger C. L. Mannitol and sorbitol catabolism in Streptococcus mutans. Arch Oral Biol. 1973 Jan;18(1):117–126. doi: 10.1016/0003-9969(73)90026-5. [DOI] [PubMed] [Google Scholar]
- Calmes R. Involvement of phosphoenolpyruvate in the catabolism of caries-conducive disaccharides by Streptococcus mutans: lactose transport. Infect Immun. 1978 Mar;19(3):934–942. doi: 10.1128/iai.19.3.934-942.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carlsson J., Elander B. Regulation of dextransucrase formation by Streptococcus sanguis. Caries Res. 1973;7(2):89–101. doi: 10.1159/000259834. [DOI] [PubMed] [Google Scholar]
- Douglas W. H. The metal oxide/eugenol cements. I. The chelating power of the eugenol type molecule. J Dent Res. 1978 Jul-Aug;57(7-8):800–804. doi: 10.1177/00220345780570070801. [DOI] [PubMed] [Google Scholar]
- Ferenci T., Kornberg H. L. Pathway of fructose utilization by Escherichia coli. FEBS Lett. 1971 Feb 19;13(2):127–130. doi: 10.1016/0014-5793(71)80216-8. [DOI] [PubMed] [Google Scholar]
- Gibbons R. J. Presence of an invertase-like enzyme and a sucrose permeation system in strains of Streptococcus mutans. Caries Res. 1972;6(2):122–131. doi: 10.1159/000259784. [DOI] [PubMed] [Google Scholar]
- Hamilton I. R. Effects of fluoride on enzymatic regulation of bacterial carbohydrate metabolism. Caries Res. 1977;11 (Suppl 1):262–291. doi: 10.1159/000260304. [DOI] [PubMed] [Google Scholar]
- Hamilton I. R., Ellwood D. C. Effects of fluoride on carbohydrate metabolism by washed cells of Streptococcus mutans grown at various pH values in a chemostat. Infect Immun. 1978 Feb;19(2):434–442. doi: 10.1128/iai.19.2.434-442.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamilton I. R. Synthesis and degradiation of intracellular polyglucose in Streptococcus salivarius. Can J Microbiol. 1968 Jan;14(1):65–77. doi: 10.1139/m68-011. [DOI] [PubMed] [Google Scholar]
- Hengstenberg W., Penberthy W. K., Hill K. L., Morse M. L. Phosphotransferase system of Staphylococcus aureus: its requirement for the accumulation and metabolism of galactosides. J Bacteriol. 1969 Aug;99(2):383–388. doi: 10.1128/jb.99.2.383-388.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanapka J. A., Hamilton I. R. Fluoride inhibition of enolase activity in vivo and its relationship to the inhibition of glucose-6-P formation in Streptococcus salivarius. Arch Biochem Biophys. 1971 Sep;146(1):167–174. doi: 10.1016/s0003-9861(71)80053-x. [DOI] [PubMed] [Google Scholar]
- Khandelwal R. L., Hamilton I. R. Effectors of purified adenyl cyclase from Streptococcus salivarius. Arch Biochem Biophys. 1972 Jul;151(1):75–84. doi: 10.1016/0003-9861(72)90475-4. [DOI] [PubMed] [Google Scholar]
- Khandelwal R. L., Hamilton I. R. Purification and properties of adenyl cyclase from Streptococcus salivarius. J Biol Chem. 1971 May 25;246(10):3297–3304. [PubMed] [Google Scholar]
- Khandelwal R. L., Spearman T. N., Hamilton I. R. Protein kinase activity in cariogenic and non-cariogenic oral streptococci: Activation and inhibition by cyclic AMP. FEBS Lett. 1973 Apr 15;31(2):246–250. doi: 10.1016/0014-5793(73)80114-0. [DOI] [PubMed] [Google Scholar]
- Kornberg H. L. Genetics in the study of carbohydrate transport by bacteria. Sixth Griffith Memorial Lecture. J Gen Microbiol. 1976 Sep;96(1):1–16. doi: 10.1099/00221287-96-1-1. [DOI] [PubMed] [Google Scholar]
- Kornberg H. L., Reeves R. E. Inducible phosphoenolpyruvate-dependent hexose phosphotransferase activities in Escherichia coli. Biochem J. 1972 Aug;128(5):1339–1344. doi: 10.1042/bj1281339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krasse B., Jordan H. V., Edwardsson S., Svensson I., Trell L. The occurrence of certain "caries-inducing" streptococci in human dental plaque material with special reference to frequency and activity of caries. Arch Oral Biol. 1968 Aug;13(8):911–918. doi: 10.1016/0003-9969(68)90006-x. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Maryanski J. H., Wittenberger C. L. Mannitol transport in Streptococcus mutans. J Bacteriol. 1975 Dec;124(3):1475–1481. doi: 10.1128/jb.124.3.1475-1481.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McKay L. L., Walter L. A., Sandine W. E., Elliker P. R. Involvement of phosphoenolpyruvate in lactose utilization by group N streptococci. J Bacteriol. 1969 Aug;99(2):603–610. doi: 10.1128/jb.99.2.603-610.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McKay L., Miller A., 3rd, Sandine W. E., Elliker P. R. Mechanisms of lactose utilization by lactic acid streptococci: enzymatic and genetic analyses. J Bacteriol. 1970 Jun;102(3):804–809. doi: 10.1128/jb.102.3.804-809.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nord C. E., Linder L., Wadström T., Lindberg A. A. Formation of glycoside-hydrolases by oral streptococci. Arch Oral Biol. 1973 Mar;18(3):391–402. doi: 10.1016/0003-9969(73)90163-5. [DOI] [PubMed] [Google Scholar]
- Perlman R. L., De Crombrugghe B., Pastan I. Cyclic AMP regulates catabolite and transient repression in E. coli. Nature. 1969 Aug 23;223(5208):810–812. doi: 10.1038/223810a0. [DOI] [PubMed] [Google Scholar]
- Rickenberg H. V. Cyclic AMP in prokaryotes. Annu Rev Microbiol. 1974;28(0):353–369. doi: 10.1146/annurev.mi.28.100174.002033. [DOI] [PubMed] [Google Scholar]
- Saier M. H., Jr, Feucht B. U., McCaman M. T. Regulation of intracellular adenosine cyclic 3':5'-monophosphate levels in Escherichia coli and Salmonella typhimurium. Evidence for energy-dependent excretion of the cyclic nucleotide. J Biol Chem. 1975 Oct 10;250(19):7593–7601. [PubMed] [Google Scholar]
- Schachtele C. F. Glucose transport in Streptococcus mutans: preparation of cytoplasmic membranes and characteristics of phosphotransferase activity. J Dent Res. 1975 Mar-Apr;54(2):330–338. [PubMed] [Google Scholar]
- Schachtele C. F., Mayo J. A. Phosphoenolpyruvate-dependent glucose transport in oral streptococci. J Dent Res. 1973 Nov-Dec;52(6):1209–1215. doi: 10.1177/00220345730520060801. [DOI] [PubMed] [Google Scholar]
- Tanzer J. M., Brown A. T., McInerney M. F. Identification, preliminary characterization, and evidence for regulation of invertase in Streptococcus mutans. J Bacteriol. 1973 Oct;116(1):192–202. doi: 10.1128/jb.116.1.192-202.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]



