Abstract
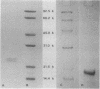
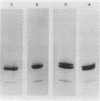
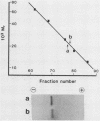
The bacterioferritin from Azotobacter vinelandii exhibits properties which in ferritins from other sources are attributed to the heteropolymeric nature of the holoprotein. The native bacterioferritin displayed multiple bands on isoelectric focusing gels. On discontinuous sodium dodecyl sulfate-polyacrylamide gels, there were two subunit polypeptides of approximate Mr 21,000 and 23,000. These molecular weights were corroborated by gel filtration experiments. Peptide maps produced by partial trypsin digestion and electrophoresis showed no detectable differences between the subunits. Similarities to well-characterized mammalian ferritins and apparent anomalies in two commonly applied electrophoretic procedures are discussed.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Adelman T. G., Arosio P., Drysdale J. W. Multiple subunits in human ferritins: evidence for hybrid molecules. Biochem Biophys Res Commun. 1975 Apr 21;63(4):1056–1062. doi: 10.1016/0006-291x(75)90676-2. [DOI] [PubMed] [Google Scholar]
- Arosio P., Adelman T. G., Drysdale J. W. On ferritin heterogeneity. Further evidence for heteropolymers. J Biol Chem. 1978 Jun 25;253(12):4451–4458. [PubMed] [Google Scholar]
- Bjork I., Fish W. W. Native and subunit molecular weights of apoferritin. Biochemistry. 1971 Jul 20;10(15):2844–2848. doi: 10.1021/bi00791a007. [DOI] [PubMed] [Google Scholar]
- Bomford A., Conlon-Hollingshead C., Munro H. N. Adaptive responses of rat tissue isoferritins to iron administration. Changes in subunit synthesis, isoferritin abundance, and capacity for iron storage. J Biol Chem. 1981 Jan 25;256(2):948–955. [PubMed] [Google Scholar]
- Bryce C. F., Crichton R. R. The subunit structure of horse spleen apoferritin. I. The molecular weight of the subunit. J Biol Chem. 1971 Jul 10;246(13):4198–4205. [PubMed] [Google Scholar]
- Bulen W. A., LeComte J. R., Lough S. A hemoprotein from azotobacter containing non-heme iron: isolation and crystallization. Biochem Biophys Res Commun. 1973 Oct 15;54(4):1274–1281. doi: 10.1016/0006-291x(73)91125-x. [DOI] [PubMed] [Google Scholar]
- Burgess B. K., Jacobs D. B., Stiefel E. I. Large-scale purification of high activity Azotobacter vinelandII nitrogenase. Biochim Biophys Acta. 1980 Jul 10;614(1):196–209. doi: 10.1016/0005-2744(80)90180-1. [DOI] [PubMed] [Google Scholar]
- Cleveland D. W., Fischer S. G., Kirschner M. W., Laemmli U. K. Peptide mapping by limited proteolysis in sodium dodecyl sulfate and analysis by gel electrophoresis. J Biol Chem. 1977 Feb 10;252(3):1102–1106. [PubMed] [Google Scholar]
- Harker A. R., Wullstein L. H. Resolution of two subunits from the molybdenum-iron protein of Azotobacter vinelandii nitrogenase. J Biol Chem. 1981 Dec 10;256(23):11981–11983. [PubMed] [Google Scholar]
- Hoare R. J., Harrison P. M., Hoy T. G. Structure of horse-spleen apoferritin at 6 angstom resolution. Nature. 1975 Jun 19;255(5510):653–654. doi: 10.1038/255653a0. [DOI] [PubMed] [Google Scholar]
- Kennedy C., Eady R. R., Kondorosi E., Rekosh D. K. The molybdenum--iron protein of Klebsiella pneumoniae nitrogenase. Evidence for non-identical subunits from peptide 'mapping'. Biochem J. 1976 May 1;155(2):383–389. doi: 10.1042/bj1550383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Leibold E. A., Aziz N., Brown A. J., Munro H. N. Conservation in rat liver of light and heavy subunit sequences of mammalian ferritin. Presence of unique octopeptide in the light subunit. J Biol Chem. 1984 Apr 10;259(7):4327–4334. [PubMed] [Google Scholar]
- Li J. D., Wang J. W., Zhong Z. P., Tu Y., Dong B. Presence of a cytochrome b-containing ferritin in Azotobacter vinelandii. Sci Sin. 1980 Jul;23(7):897–904. [PubMed] [Google Scholar]
- Lundell D. J., Howard J. B. Isolation and partial characterization of two different subunits from the molybdenum-iron protein of Azotobacter vinelandii nitrogenase. J Biol Chem. 1978 May 25;253(10):3422–3426. [PubMed] [Google Scholar]
- NEWTON J. W., WILSON P. W., BURRIS R. H. Direct demonstration of ammonia as an intermediate in nitrogen fixation by Azotobacter. J Biol Chem. 1953 Sep;204(1):445–451. [PubMed] [Google Scholar]
- O'Farrell P. H. High resolution two-dimensional electrophoresis of proteins. J Biol Chem. 1975 May 25;250(10):4007–4021. [PMC free article] [PubMed] [Google Scholar]
- Powell L. W., Alpert E., Isselbacher K. J., Drysdale J. W. Abnormality in tissue isoferritin distribution in idiopathic haemochromatosis. Nature. 1974 Jul 26;250(464):333–335. doi: 10.1038/250333a0. [DOI] [PubMed] [Google Scholar]
- Stiefel E. I., Watt G. D. Azotobacter cytochrome b557.5 is a bacterioferritin. Nature. 1979 May 3;279(5708):81–83. doi: 10.1038/279081a0. [DOI] [PubMed] [Google Scholar]
- Van de Bogart M., Beinert H. Micro methods for the quantitative determination of iron and copper in biological material. Anal Biochem. 1967 Aug;20(2):325–334. doi: 10.1016/0003-2697(67)90038-3. [DOI] [PubMed] [Google Scholar]
- Weber K., Pringle J. R., Osborn M. Measurement of molecular weights by electrophoresis on SDS-acrylamide gel. Methods Enzymol. 1972;26:3–27. doi: 10.1016/s0076-6879(72)26003-7. [DOI] [PubMed] [Google Scholar]
- Yariv J., Kalb A. J., Sperling R., Bauminger E. R., Cohen S. G., Ofer S. The composition and the structure of bacterioferritin of Escherichia coli. Biochem J. 1981 Jul 1;197(1):171–175. doi: 10.1042/bj1970171. [DOI] [PMC free article] [PubMed] [Google Scholar]