Abstract
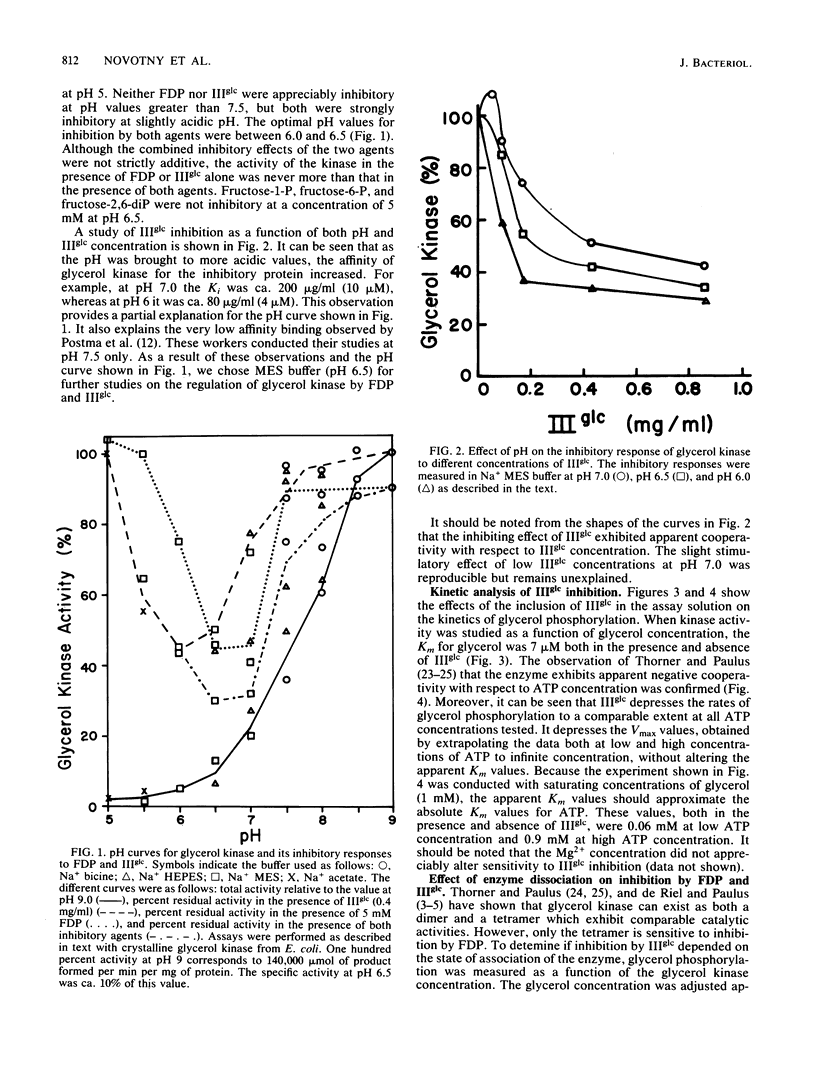
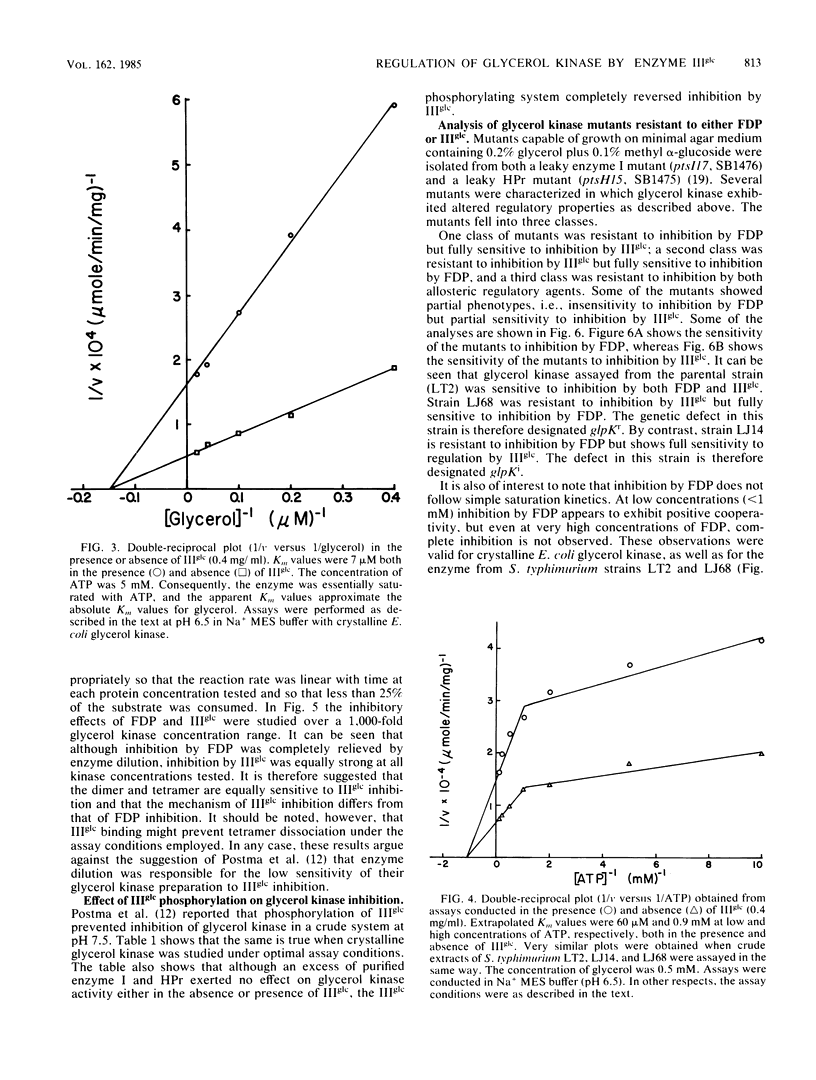
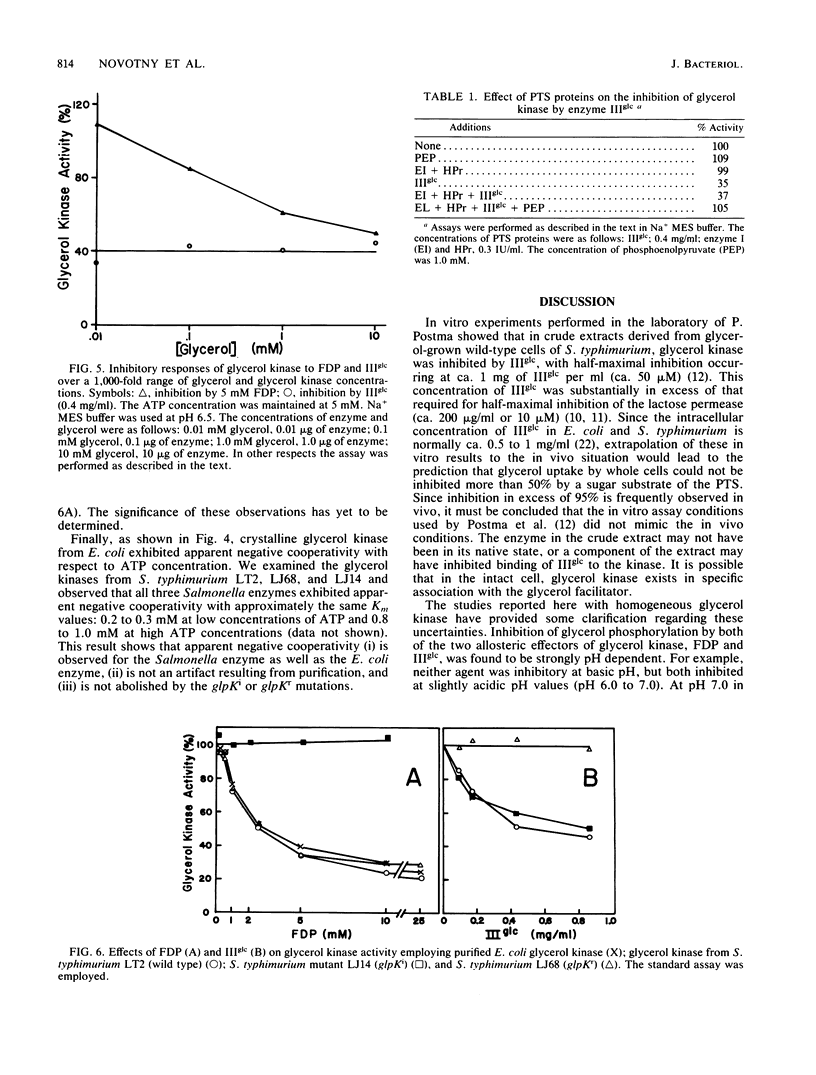
The mechanism by which enzyme IIIglc of the bacterial phosphotransferase system regulates the activity of crystalline glycerol kinase from Escherichia coli has been studied, and the inhibitory effects have been compared with those produced by fructose-1,6-diphosphate. It was shown that the free, but not the phosphorylated, form of enzyme IIIglc inhibits the kinase. Mutants of Salmonella typhimurium were isolated which were resistant to inhibition by either enzyme IIIglc (glpKr mutants) or fructose-1,6-diphosphate (glpKi mutants), and each mutant type was shown to retain full sensitivity to inhibition by the other regulatory agent. Other mutants were fully or partially resistant to regulation by both agents. The two regulatory sites on the kinase are evidently distinct but must overlap or interact functionally. Kinetic analyses have revealed several mechanistic features of the regulatory interactions. (i) Inhibition by both allosteric regulatory agents is strongly pH dependent, with maximal inhibition occurring at ca. pH 6.5 under the assay conditions employed. (ii) Binding of enzyme IIIglc to glycerol kinase is also pH dependent, the Ki being near 4 microM at pH 6.0 but near 10 microM at pH 7.0. (iii) Whereas fructose-1,6-diphosphate inhibition apparently requires that the enzyme exist in a tetrameric state, both the dimer and the tetramer appear to be fully sensitive to enzyme IIIglc inhibition. (iv) Inhibition by enzyme IIIglc (like that by fructose-1,6-diphosphate) is noncompetitive with respect to both substrates. (v) The inhibitory responses of glycerol kinase to fructose-1, 6-diphosphate and enzyme IIIglc show features characteristic of positive cooperativity at low inhibitor concentration. (vi) Neither agent inhibits completely at high inhibitor concentration. (vii) Apparent negative cooperativity with respect to ATP binding is observed with purified E. coli glycerol kinase, with glycerol kinase in crude extracts of wild-type S. typhimurium cells, and with glpKr and glpKi mutant forms of glycerol kinase from S. typhimurium. These results serve to characterize the regulatory interactions which control the activity of glycerol kinase by fructose-1,6-diphosphate and by enzyme IIIglc of the phosphotransferase system.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Castro L., Feucht B. U., Morse M. L., Saier M. H., Jr Regulation of carbohydrate permeases and adenylate cyclase in Escherichia coli. Studies with mutant strains in which enzyme I of the phosphoenolpyruvate:sugar phosphotransferase system is thermolabile. J Biol Chem. 1976 Sep 25;251(18):5522–5527. [PubMed] [Google Scholar]
- Conrad C. A., Stearns G. W., 3rd, Prater W. E., Rheiner J. A., Johnson J. R. Characterization of a glpK transducing phage. Mol Gen Genet. 1984;193(2):376–378. doi: 10.1007/BF00330696. [DOI] [PubMed] [Google Scholar]
- Dills S. S., Apperson A., Schmidt M. R., Saier M. H., Jr Carbohydrate transport in bacteria. Microbiol Rev. 1980 Sep;44(3):385–418. doi: 10.1128/mr.44.3.385-418.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nelson S. O., Postma P. W. Interactions in vivo between IIIGlc of the phosphoenolpyruvate:sugar phosphotransferase system and the glycerol and maltose uptake systems of Salmonella typhimurium. Eur J Biochem. 1984 Feb 15;139(1):29–34. doi: 10.1111/j.1432-1033.1984.tb07971.x. [DOI] [PubMed] [Google Scholar]
- Osumi T., Saier M. H., Jr Mechanism of regulation of the lactose permease by the phosphotransferase system in Escherichia coli: evidence for protein-protein interaction. Ann Microbiol (Paris) 1982 Mar-Apr;133(2):269–273. [PubMed] [Google Scholar]
- Postma P. W., Epstein W., Schuitema A. R., Nelson S. O. Interaction between IIIGlc of the phosphoenolpyruvate:sugar phosphotransferase system and glycerol kinase of Salmonella typhimurium. J Bacteriol. 1984 Apr;158(1):351–353. doi: 10.1128/jb.158.1.351-353.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saier M. H., Jr Bacterial phosphoenolpyruvate: sugar phosphotransferase systems: structural, functional, and evolutionary interrelationships. Bacteriol Rev. 1977 Dec;41(4):856–871. doi: 10.1128/br.41.4.856-871.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saier M. H., Jr, Feucht B. U. Coordinate regulation of adenylate cyclase and carbohydrate permeases by the phosphoenolpyruvate:sugar phosphotransferase system in Salmonella typhimurium. J Biol Chem. 1975 Sep 10;250(17):7078–7080. [PubMed] [Google Scholar]
- Saier M. H., Jr, Novotny M. J., Comeau-Fuhrman D., Osumi T., Desai J. D. Cooperative binding of the sugar substrates and allosteric regulatory protein (enzyme IIIGlc of the phosphotransferase system) to the lactose and melibiose permeases in Escherichia coli and Salmonella typhimurium. J Bacteriol. 1983 Sep;155(3):1351–1357. doi: 10.1128/jb.155.3.1351-1357.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saier M. H., Jr, Roseman S. Sugar transport. The crr mutation: its effect on repression of enzyme synthesis. J Biol Chem. 1976 Nov 10;251(21):6598–6605. [PubMed] [Google Scholar]
- Saier M. H., Jr, Straud H., Massman L. S., Judice J. J., Newman M. J., Feucht B. U. Permease-specific mutations in Salmonella typhimurium and Escherichia coli that release the glycerol, maltose, melibiose, and lactose transport systems from regulation by the phosphoenolpyruvate:sugar phosphotransferase system. J Bacteriol. 1978 Mar;133(3):1358–1367. doi: 10.1128/jb.133.3.1358-1367.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scholte B. J., Schuitema A. R., Postma P. W. Isolation of IIIGlc of the phosphoenolpyruvate-dependent glucose phosphotransferase system of Salmonella typhimurium. J Bacteriol. 1981 Oct;148(1):257–264. doi: 10.1128/jb.148.1.257-264.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simoni R. D., Roseman S., Saier M. H., Jr Sugar transport. Properties of mutant bacteria defective in proteins of the phosphoenolpyruvate: sugar phosphotransferase system. J Biol Chem. 1976 Nov 10;251(21):6584–6597. [PubMed] [Google Scholar]
- Thorner J. W. Glycerol kinase. Methods Enzymol. 1975;42:148–156. doi: 10.1016/0076-6879(75)42109-7. [DOI] [PubMed] [Google Scholar]
- Thorner J. W., Paulus H. Catalytic and allosteric properties of glycerol kinase from Escherichia coli. J Biol Chem. 1973 Jun 10;248(11):3922–3932. [PubMed] [Google Scholar]
- Thorner J. W., Paulus H. Composition and subunit structure of glycerol kinase from Escherichia coli. J Biol Chem. 1971 Jun 25;246(12):3885–3894. [PubMed] [Google Scholar]
- Waygood E. B., Mattoo R. L., Peri K. G. Phosphoproteins and the phosphoenolpyruvate: sugar phosphotransferase system in Salmonella typhimurium and Escherichia coli: evidence for IIImannose, IIIfructose, IIIglucitol, and the phosphorylation of enzyme IImannitol and enzyme IIN-acetylglucosamine. J Cell Biochem. 1984;25(3):139–159. doi: 10.1002/jcb.240250304. [DOI] [PubMed] [Google Scholar]
- Waygood E. B., Steeves T. Enzyme I of the phosphoenolpyruvate: sugar phosphotransferase system of Escherichia coli. Purification to homogeneity and some properties. Can J Biochem. 1980 Jan;58(1):40–48. doi: 10.1139/o80-006. [DOI] [PubMed] [Google Scholar]
- de Riel J. K., Paulus H. Subunit dissociation in the allosteric regulation of Glycerol kinase from Escherichia coli. 3. Role in desensitization. Biochemistry. 1978 Nov 28;17(24):5146–5150. doi: 10.1021/bi00617a012. [DOI] [PubMed] [Google Scholar]
- de Riel J. K., Paulus H. Subunit dissociation in the allosteric regulation of glycerol kinase from Escherichia coli. 1. Kinetic evidence. Biochemistry. 1978 Nov 28;17(24):5134–5140. doi: 10.1021/bi00617a010. [DOI] [PubMed] [Google Scholar]
- de Riel J. K., Paulus H. Subunit dissociation in the allosteric regulation of glycerol kinase from Escherichia coli. 2. Physical evidence. Biochemistry. 1978 Nov 28;17(24):5141–5146. doi: 10.1021/bi00617a011. [DOI] [PubMed] [Google Scholar]



