Abstract
The energy derived from pyrophosphate (PPi) hydrolysis is used to pump protons across the tonoplast membrane, thus forming a proton gradient. In a plant's cytosol, the concentration of PPi varies between 10 and 800 μm, and the PPi concentration needed for one-half maximal activity of the maize (Zea mays) root tonoplast H+-pyrophosphatase is 30 μm. In this report, we show that the H+-pyrophosphatase of maize root vacuoles is able to hydrolyze PPi (Reaction 2) formed by Reaction 1, which is catalyzed by PPi-dependent phosphofructokinase (PFP):
Table 1.
| Fructose-1,6-bisphosphate (F1,6BP) + Pi ↔ PPi +Fructose-6-phosphate (F6 P) | (reaction 1) |
| PPi → 2 Pi | (reaction 2) |
| H+cyt → H+vac | (reaction 3) |
| F1,6BP + H+cyt ↔ H+vac + F6P + Pi | (reaction 4) |
During the steady state, one-half of the inorganic phosphate released (Reaction 4) is ultimately derived from F1,6BP, whereas PFP continuously regenerates the pyrophosphate (PPi) hydrolyzed. A proton gradient (ΔpH) can be built up in tonoplast vesicles using PFP as a PPi-regenerating system. The Δ pH formed by the H+-pyrophosphatase can be dissipated by addition of 20 mm F6P, which drives Reaction 1 to the left and decreases the PPi available for the H+-pyrophosphatase. The maximal Δ pH attained by the pyrophosphatase coupled to the PFP reaction can be maintained by PFP activities far below those found in higher plants tissues.
Plants and some protozoans of clinical importance possess a PPi-dependent phosphofructokinase (PFP; EC 2.7.1.90) that converts Fru-6-phosphate (F6P) to Fru-1,6-bisphosphate (F1,6BP) and inorganic phosphate (Pi) using PPi as phosphoryl donor. In contrast to the reaction catalyzed by the isoform that uses ATP as substrate (ATP-dependent phosphofructokinase, EC 2.7.1.11), the equilibrium constant (Keq) of the reaction catalyzed by PFP is close to 1 and is, therefore, readily reversible (O'Brien et al., 1975; Kombrink et al., 1984). PFP is activated by Fru-2,6-bisphosphate (F2,6BP), its allosteric modulator. The cytosolic concentration of F2,6BP can fluctuate depending on the environmental conditions or the level of PFP expression in plant tissues (Hajirezaei et al., 1994; Nielsen and Stitt, 2001). Attempts have been made to ascertain whether PFP plays a role in glycolysis by converting F6P to F1,6BP; in a number of cases, this has been shown convincingly (Smyth and Chevalier, 1984; Duff et al., 1989). However, other studies demonstrated that PFP is unable to support glycolysis in vivo because it operates as a gluconeogenic enzyme (ap Rees et al., 1985; Black et al., 1987; Paul et al., 1995). Therefore, there is no consensus about the metabolic function of this enzyme.
Plants, protozoans, and certain bacteria have a membrane-bound H+-translocating inorganic pyrophosphatase (H+-PPase) that works as a proton pump. This enzyme couples the hydrolysis of PPi with electrogenic translocation of protons across the membrane (Baykov et al., 1999). In plants, this enzyme is located in the tonoplast membrane, where it supports vacuolar functions, such as preventing the harmful accumulation of Ca2+ and Na+ in the cytosol and allowing storage of organic metabolites that can be recovered by the cytosol when needed by the H+-energized tonoplast membranes. The cytosolic PPi concentration in plant cells is approximately 200 μm (Weiner et al., 1987; Maeshima, 2000). This concentration is sufficient to promote the maximal activity of vacuolar H+-PPase in plant cells (Maeshima, 2000). The PPi concentration is determined by the balance between the rates of PPi hydrolysis and synthesis. The hydrolysis of PPi is catalyzed by the H+-PPase (Davies et al., 1993). On the other hand, PPi is synthesized as a by-product of various biosynthetic reactions such as the synthesis of nucleic acids, polysaccharides, coenzymes, and proteins; activation of fatty acids; and isoprenoid synthesis. Under stress conditions, such as anoxia, the biosynthetic reactions are slowed down, The aim of this study was to evaluate whether the maize (Zea mays) root H+-PPase is able to bind and hydrolyze the PPi formed by Reaction 1 catalyzed by PFP in vitro under conditions that mimic the concentrations of F1,6BP, Pi, Mg2+, F2,6BP, and F6P found in the plant cell cytosol (Rebeille et al., 1983; Ukaji and Ashihara, 1987; Weiner et al., 1987; Kubota and Ashihara, 1990; Davies et al., 1993; Stitt, 1998; Table I). Here, we show that the affinity of maize root H+-PPase for PPi under these conditions is sufficient to shift Reaction 1 toward PPi formation, making it possible to build up a proton gradient in the tonoplast even in the presence of very low amounts of PFP.
Table I.
Levels of metabolites in plant cytosol; PFP and H+-PPase activities from different plants and experimental conditions used in this study
| Plants and Conditions
|
Metabolites in Plant Cytosola
|
Enzyme Activity Level
|
||||
|---|---|---|---|---|---|---|
| F1,6BP | Pi | Fru-6P | PPi | PFPb | H+-PPasec | |
| mm | milliunits g fresh wt-1 | |||||
| Wild-type plants | 0.6 | 5 | 1 | 0.33 | 293-1,813 | 15-25 |
| Antisense transformant plants (lower levels) | - | - | - | - | 20-58 | - |
| Experimental conditions (Figs. 1, 2, and 4) | - | - | - | - | 0.5-60d | 18-25d |
a The concentrations of F1,6BP, Pi, and F6P are those reported in the literature for plant cell cytosol (Rebeille et al., 1983; Ukaji and Ashihara, 1987; Weiner et al., 1987; Kubota and Ashihara, 1990; Davies et al., 1993; Stitt, 1998). b The PFP activities for wild-type and antisense transformant tobacco (Nicotiana tabacum) plants were taken from Nielsen and Stitt (2001) for antisense transformant potato (Solanum tuberosum) plants. Values are from Hajirezaei et al. (1994). c The H+-PPase activities for wild-type mung bean, tomato (Lycopersicon esculentum) fruit, and maize leaves were taken from Maeshima and Yoshida (1989), Milner et al. (1995), and Clayton et al. (1993), respectively. d The PFP and H+-PPase activities shown in this report were converted to milliunits per gram fresh wt-1 computing the enzyme activity level (milliunits) present in 1 mL of reaction medium. This volume was converted to grams to obtain the activities shown.
RESULTS
Cleavage of F1,6BP, pH, and Mg2+ Dependence
PFP and maize root H+-PPase catalyzed the release of Pi from F1,6BP (Fig. 1). There was no measurable thereby limiting the rate of PPi formation. Thus, the use of the reaction catalyzed by PFP in the direction of PPi formation may be an auxiliary mechanism to prevent a drop in the cytosolic levels of PPi under the metabolic conditions of the living cell (Davies et al., 1993). As far as we know, this proposal has never been demonstrated experimentally.
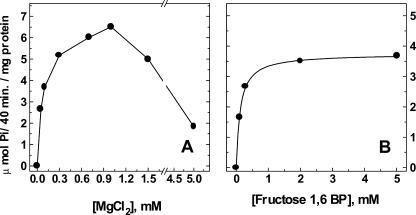
Figure 1.
Cleavage of F1,6BP and Mg2+ dependence. The F1,6BP cleavage was dependent on MgCl2 (A) and F1,6BP (B) concentrations. The MgCl2 dependence was measured in a medium containing: 50 mm MOPS-Tris buffer (pH 7.0), 60 milliunits mL–1 mung bean (Vigna radiata) PFP, 0.1 mg mL–1 maize vacuolar microsomal protein, 100 mm KCl, 2 μm F2,6BP, and 10 mm F1,6BP (A). When the F1,6BP dependence was measured, the MgCl2 concentration was fixed at 0.6 mm(B). The reaction was performed at 30°C and started by addition of 0.1 mm Pi. Aliquots of 0.8 mL were removed, and the reaction was stopped by the addition of 0.2 mL of 50% (w/v) trichloroacetic acid. The figure shows a representative experiment. Similar results were obtained with three different vesicle preparations.
The mass to action ratio measured for the substrates and products of PFP in plants is close to its Keq value (Kubota and Ashihara, 1990). This implies that in the plant cytosol, the direction of the reaction may be determined by the [PPi] to [Pi] ratio and that the PPi formed at the expense of F1,6BP could ultimately be used to pump H+ across the tonoplast membrane as follows:
Table 2.
| Pi+F1,6BP ↔ PPi+F6P | (reaction 1) | ΔG° = +0.7 kcal mol-1 |
| PPi → 2Pi | (reaction 2) | ΔG° = -5.1 kcal mol-1 |
| H+cyt ↔ H+vac (ΔpH = 2) | (reaction 3) | ΔG = +1.5 kcal mol-1 |
| F1,6BP + H+cyt → F6P + Pi+H+vac | (reaction 4) | ΔG° = -2.9 kcal mol-1 |
release of Pi when the two enzymes needed to regenerate PPi or the substrates F1,6BP and Pi were omitted from the assay medium. The rate of cleavage varied with the concentration of F1,6BP (Fig. 1B), and the half-maximal rate was observed with 123 μm F1,6BP. PFP and H+-PPase both require Mg2+ for their activities (Maeshima and Yoshida, 1989; Givan, 1999; Maeshima, 2000). Magnesium was needed for the coupled reaction in Figure 1; the maximal rate of F1,6BP cleavage was obtained in the presence of 0.6 to 1 mm MgCl2. Higher MgCl2 concentrations were found to be inhibitory (Fig. 1A). The findings of Figure 1 indicate that the true substrate for the maize root H+-PPase was the very low PPi concentration derived from Reaction 1 and that the Pi released into the medium originated from F1,6BP by the action of PFP, which continuously regenerated the PPi hydrolyzed (Reactions 1 and 2).
The concentration of PPi that is attained when the PPi-regenerating reaction reaches a steady state varied depending on the pH of the medium. The rate of F1,6BP cleavage was maximal in the pH range of 6.0 to 7.0 and decreased by 50% when the pH was raised from 7.0 to 8.0 (data not shown).
Transmembrane Proton Gradient
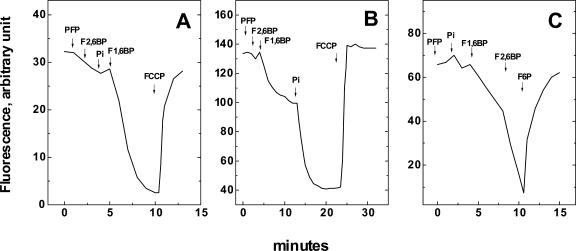
The assay in Figure 2 shows that the energy derived from the steady-state cleavage of F1,6BP was sufficient to form and maintain a transmembrane proton gradient in tonoplast vesicles. In the presence of PFP, the addition of F2,6BP, Pi, and F1,6BP promoted a quenching of ACMA fluorescence, indicating that a proton gradient was formed across the tonoplast membrane (Fig. 2A). The gradient was abolished when 1 μm FCCP, a proton ionophore, was added to the medium (Fig. 2, A and B).
Figure 2.
Membrane proton gradient supported by PFP, F1,6BP, and Pi. The assay conditions were identical to those described in Figure 1B, except that the 9-amino-6-chloro-2-methoxyacridine (ACMA; 3 μm) was present and F1,6BP, F2,6BP, and Pi concentrations are listed below for each panel. At the end of gradient formation, 1 μm carbonyl cyanide p-(trifluoromethoxy)phenylhydrazone (FCCP) was added to dissipate the H+ gradient in A and B. In C, the addition of PFP activator, F2,6BP, enhanced the proton gradient formation, and the addition of 20 mm F6P dissipated it. The arrows indicate the sequential addition of: A, 25 milliunits of PFP, 2 μm F2,6BP, 2 mm Pi, and 0.6 mm F1,6BP; B, 30 μm Pi was included in the reaction medium before the sequential addition of: 25 milliunits of PFP; 4 μm F2,6BP; 0.6 mm F1,6BP, and 2 mm Pi; C, 10 milliunits of PFP, 2 mm Pi, 0.1 mm F1,6BP, 4 μm F2,6BP, and 20 mm F6P. The figure shows a representative experiment. Similar results were obtained in three different vesicle preparations.
The proton gradient formed depended on the concentration of Pi present in the reaction medium (Fig. 2B). In the presence of 30 μm Pi, a small proton gradient was observed after the addition of 0.1 mm F1,6BP (Fig. 2B). Further addition of Pi (2 mm), promoted both an increase in the initial rate of proton pumping and an enhancement of the proton gradient. This result suggests that the addition of Pi to the medium displaces Reaction 1 toward PPi formation, increasing its steady-state concentration.
The capacity of the system to form the proton gradient was highly dependent on the presence of the PFP activator F2,6BP (Fig. 2C). A slow rate of proton pumping was measured in presence of 2 mm Pi, 0.1 mm F1,6BP, and 10 milliunits of PFP. However, when 2 μm F2,6BP was added to the medium, the rate of proton pumping was accelerated by more than 2-fold. This result confirms that the rate of the PPi formation by PFP is stimulated by F2,6BP and that the proton gradient is also dependent on the rate of PPi formation (Fig. 2C). Finally, the transmembrane proton gradient was abolished when 20 mm F6P was added to the medium, indicating that when the equilibrium is driven toward F1,6BP formation, the steady-state concentration of PPi available to the maize root H+-PPase is greatly reduced, a condition that led to a decrease of the H+ gradient.
Equilibrium Concentrations of PPi Formed from F1,6BP and Pi and PPi Dependence of Maize Root H+-PPase
The equilibrium concentration of PPi formed in the reaction catalyzed by PFP varied depending on the initial concentrations of F1,6BP, Pi, and F6P. To evaluate if the range of PPi concentrations formed in our experiments was close to that needed for one-half maximal activation of the maize root H+-PPase, the PPi concentration was calculated in two ways: by using the concentrations of substrates and products found in plant cytosol (Kubota and Ashihara, 1990; Davies et al., 1993) or by using the results obtained under the conditions of the experiments of Figure 2. In addition, we measured the apparent affinity of maize root H+-PPase for PPi (Fig. 3). Table II shows the PPi concentrations that would be formed under these different conditions. These values were calculated using the Keq of 0.31 (Kubota and Ashihara, 1990) for the reaction catalyzed by PFP.
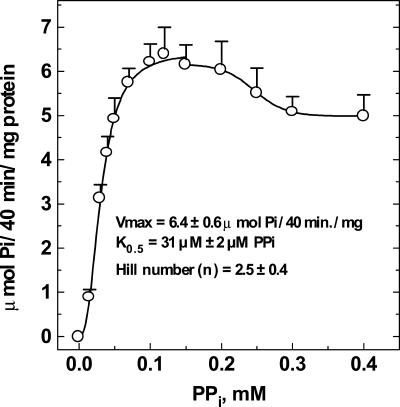
Figure 3.
PPi saturation kinetics of maize root H+-PPase. The assay medium composition was 50 mm MOPS-Tris (pH 7.0), 100 mm KCl, 0.6 mm MgCl2, 3 μm FCCP, and 0.030 mg mL–1 maize vesicle protein. The PPi concentrations are shown on the abscissa. The reaction was started by addition of vesicles. The amount of PPi hydrolyzed never exceeded 5% of the total PPi added to the medium. The kinetic parameters shown in the figure were calculated from a nonlinear regression analysis applied to the Hill equation using the program ENZFITTER. The specific activities shown are means ± se of at least four independent preparations.
Table II.
PPi formation in presence of F1,6BP, Pi, and F6P
After PPi formation catalyzed by PFP, PPi is hydrolysed by H+ PPase, forming a pH gradient. The respective free energy change (ΔG1 and ΔG2) are calculated. In the table, the concentrations of PPi found after the reaction F1,6BP + Pi → PPi + F-6P (reaction 1, ΔG1) reaches equilibrium were calculated using the value of 0.31 for the apparent equilibrium constant (Keq) as previously described (Kubota and Ashihara, 1990). The equilibrium PPi concentrations were calculated using the following equation: [PPi] = [F1,6BP] × [Pi] × Keq/[F6P]. The PPi formed in the presence of 0.6 mm MgCl2 is further hydrolyzed by the H+-PPase in the following reaction: H+cyt + PPi → 2Pi + H+vac (reaction 2, ΔG2). The ΔG° = -5.1 kcal mol-1 was taken from the values measured by de Meis (1984) in the presence of 0.6 mm MgCl2 to calculate the ΔG2.
| Conditions
|
Concentrations
|
ΔG
|
|||||
|---|---|---|---|---|---|---|---|
| F1,6BP | PI | F6P | PPi formed | ΔG1a | ΔG2a | ΔG1 + 2a | |
| m | kcal mol-1 | ||||||
| Ab | 6×10-4 | 5×10-3 | 1×10-3 | 3.3×10-4 | -0.6 | -3.9 | -4.5 |
| Bc | 6×10-4 | 2×10-3 | 3×10-5 | 3.5×10-4 | -2.1 | -5.0 | -7.2 |
| Cc | 6×10-4 | 3×10-5 | 3×10-5 | 2.3×10-5 | -1.3 | -8.4 | -9.7 |
| Dc | 1×10-4 | 2×10-3 | 2×10-2 | 3.0×10-6 | -0.02 | -2.2 | -2.2 |
a The ΔG1 values (kilocalories per mole) for different conditions were calculated from the equation ΔG1 = ΔG° + RT In [PPi] × [F6P]/[F1,6BP] × [Pi] using the concentrations shown in the table, and ΔG2 values were calculated from the equation ΔG2 = ΔG° + RT In [Pi]2 × [H+vac]/[PPi] × [H+cyt], assuming a ΔpH of 2 units. The final concentration of F6P derived from contamination of F1,6BP and its hydrolysis was estimated to be 5% (3 × 10-5 m). This concentration of F6P was used to calculate the concentration of PPi formed in experiments in which extra amounts of F6P were not added to the assay medium (conditions B and C). bThe molar concentrations of F1,6BP, Pi, and F6P are those reported in the literature for plant cell cytosol (Rebeille et al., 1983; Ukaji and Ashihara, 1987; Weiner et al., 1987; Kubota and Ashihara, 1990; Davies et al., 1993; Stitt, 1998). cThe molar concentrations of F1,6BP, Pi, and F6P are those used in Figure 2.
The PPi concentration needed for one-half maximal rate activity of maize root H+-PPase was 3.2 × 10–5 m (Fig. 3). This value is similar to that previously reported by Maeshima (2000), is 10-fold lower than the PPi equilibrium concentrations calculated in Table II (conditions A and B), and is close to the equilibrium values calculated for condition C. This means that the rate of proton pumping (Fig. 2) responds readily to the equilibrium of the reaction catalyzed by PFP. The addition of 20 mm F6P (Fig. 2C) reduces the PPi concentration to such a low level (Table II, condition D) that can no longer be used by the maize root H+-PPase to accumulate H+ inside the tonoplast vesicles.
PFP Dependence for Formation of the Transmembrane Proton Gradient
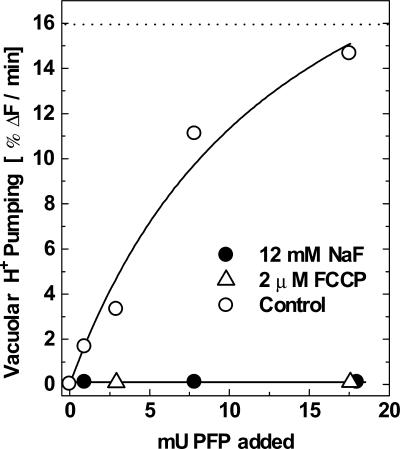
The rate of vacuolar proton pumping varied depending on the amount of PFP added to the reaction medium (Fig. 4). The proton pumping was blocked by 12 mm fluoride, an anion that inhibits the vacuolar maize root H+-PPase (Fig. 4, black circles). The assay condition of 2 mm Pi, 0.5 mm F1,6BP, 2 μm F2,6BP, and 18 milliunits of PFP was sufficient to stimulate the proton pumping to a level similar to that observed using 0.3 mm PPi, a saturating concentration (Fig. 4, white circles and dotted line). The amount of PFP used in Figure 4 is very small and is close to the levels observed in antisense transformant plants in which more than 95% of the total PFP activity has been abolished (Hajirezaei et al., 1994; Nielsen and Stitt, 2001).
Figure 4.
PFP dependence for the formation of the membrane proton gradient. The reaction was carried out in medium containing 10 mm MOPS-Tris (pH 7.0), 3 μm ACMA, 1 mm MgCl2, 100 mm KCl, 2 μm F2,6BP, 2 mm Pi, 0.1 mg mL–1 maize vacuolar microsomal protein, and 0.6 mm F1,6BP. The initial rate of H+ pumping was taken in the linear phase of fluorescence quenching of ACMA and was expressed as percentage ΔF per minute after the addition of 1, 3, 8, and 18 milliunits of PFP to the cuvette (2 mL) in the absence (○) or in the presence (•) of 12 mm NaF or 2 μm FCCP (Δ). The figure shows a representative experiment. Similar results were obtained in three different vesicle preparations. The dotted line represents the maximal rate of H+ pumping measured when 0.3 mm PPi was added to the reaction mixture in the absence of the PPi-PFP regenerating system.
DISCUSSION
In this report, the capacity of maize root vacuolar H+-PPase to use the PPi formed at equilibrium by PFP was evaluated. It was found that in the presence of Mg2+, Pi, F1,6BP and F2,6BP concentrations similar to those available in plant cytosol (Rebeille et al., 1983; Ukaji and Ashihara, 1987; Weiner et al., 1987; Kubota and Ashihara, 1990; Stitt, 1998), the PPi formed by even a small amount of PFP was sufficient to activate the vacuolar H+-PPase and to form a proton gradient across the tonoplast membrane. This reaction is coupled to a decrease in the F1,6BP concentration, a compound that has a lower energy of hydrolysis than PPi (de Meis, 1989, 1993). Table II shows that the free energy change (associated with Reaction 4) is markedly negative (Table II, ΣΔG1 + 2 varying from –2.19 to –9.68 kcal mol–1), despite different levels of PPi formed under the various conditions used to calculate the equilibrium PPi concentration. Even when the PPi concentration was as low as 3 μm (Table II, condition D), the calculated ΣΔG1 + 2 was –2.19 kcal mol–1, a value that indicates a favorable shift to PPi hydrolysis. In maize roots tips, the contribution to the steady-state level to PPi in the cytosol of only one of the reactions that produce PPi, the formation of UDP-Glc from Glc-1 P and UTP, was estimated to be 10 μm (Roberts, 1990) a value that is more than 3 times higher than that used to obtain a value of ΣΔG1 + 2 of –2.19 kcal mol–1 (Table II, condition D). This means that there are no thermodynamic constraints for the pumping of protons into the vacuole lumen using F1,6BP, Pi and 20 mm F6P to achieve the equilibrium concentration of PPi of 3 μm. The observation that the protons leak out of the vesicles when 20 mm F6P is added to the medium (Fig. 2C) is related to the decrease of the steady-state PPi concentration to a level far below that needed by the tonoplast H+-PPase (Fig. 3).
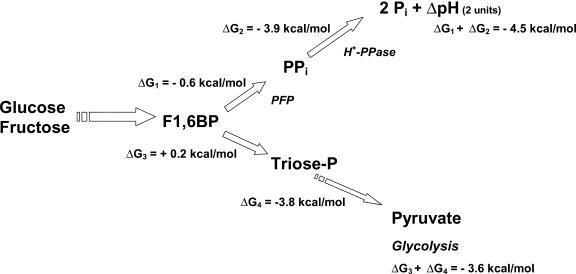
Based on reported concentrations of glycolytic intermediates of plant cells (Kubota and Ashihara, 1990), the ΔG for the formation of PPi (Reaction 1; Kubota and Ashihara, 1990) and the ΔG of PPi hydrolysis (Reactions 2 and 3; de Meis, 1984; Davies et al., 1993), it was possible to calculate the overall ΔG for the glycolytic conversion of F1,6BP to either pyruvate or PPi and the subsequent hydrolysis of PPi by the H+-PPase needed to maintain a ΔpH of 2 units across the tonoplast membrane. From these estimates, it becomes apparent that thermodynamic pull for the utilization of F1,6BP toward H+ pumping (ΔG1 + ΔG2 in Fig. 5) is slightly more favorable than the formation of pyruvate (ΔG3 + ΔG4 in Fig. 5). The readily reversible PFP reaction may provide an adaptive pathway for glycolysis and gluconeogenesis in higher plant cells (Black et al., 1987). The result shown in Figure 2 suggests a new role of PFP, the formation of PPi to be used for the formation of a proton gradient in tonoplast vesicles.
Figure 5.
Free-energy changes (ΔG) associated with the conversion of F1,6BP to build up a H+ gradient in tonoplast vesicles or with conversion to pyruvate by the glycolytic pathway. After Glc or Fru phosphorylation, the F1,6BP formed can be diverted to PPi synthesis by PPi-PFP (ΔG1), and the PPi is hydrolyzed by H+-PPase to build up a proton gradient of 2 units of pH in the tonoplast vesicles (ΔG2). Alternatively, the F1,6BP is converted into triose-P by aldolase (ΔG3). The triose-P is converted by glycolytic enzymes to pyruvate (ΔG4). ΔG1 and ΔG2 were calculated as shown in Table II (condition A) using concentrations in the plant cell cytosol. The ΔG3 and ΔG4 were calculated from glycolytic reactions described by Kubota and Ashihara (1990).
One of the possible limiting factors for the utilization of F1,6BP and Pi to form PPi would be the amount of PFP and/or H+-PPase present in the cells. A simultaneous up-regulation in the levels of H+-PPase and PFP have been detected during anoxia in rice (Oryza sativa) seedlings (Mertens et al., 1990; Carystinos et al., 1995; Drew, 1997). Table I shows that the levels of PFP and H+-PPase activities detected in different plants (Hajirezaei et al., 1994; Nielsen and Stitt, 2001) were sufficient in our assays to promote the formation of a H+ gradient. We found that it was possible to detect a proton gradient in tonoplast vesicles even in the presence of a PFP activity similar to that observed in antisense transformant plants (Hajirezaei et al., 1994; Nielsen and Stitt, 2001). The H+-PPase levels used in our experiments are similar to those detected in maize roots and other plants (Table I). These data suggest that, given the low ΔG values of Reactions 1 and 2 (Fig. 5), the low ratio in the activity levels of PFP to H+-PPase would limit the vacuolar proton pumping when the concentration of PFP decreases to levels below 4 milliunits g fresh weight–1. In recent reports (Hajirezaei et al., 1994; Nielsen and Stitt, 2001) using tobacco and potato plant transformants that lead up to a 95% reduction in the PFP activity (see also Table I), there were no major changes in carbon fluxes or leaf or plant growth. Nevertheless, there was a compensatory increment in the F2,6BP levels both in tobacco leaves and potato tubers (Hajirezaei et al., 1994; Nielsen and Stitt, 2001). In our study, the levels of PFP activity are similar to those detected in tobacco and potato plant transformants (Fig. 4; Table I). This raises the possibility that under steady-state conditions in the plant cytosol, different enzymes involved in energy transduction, such as maize root vacuolar H+-PPase, may use phosphate compounds with a low energy of hydrolysis (de Meis, 1989, 1993; de Meis et al., 1992a, 1992b; Montero-Lomeli and de Meis, 1992; Galina et al., 1995) to perform the work that is usually associated with the consumption of higher energy phosphate compounds such as PPi (Romero and de Meis, 1989).
MATERIALS AND METHODS
Materials and Chemicals
All chemicals were obtained from Sigma (St. Louis). PPi:Fru-6-phosphate 1-phosphotransferase was from mung bean (Vigna radiata; Sigma F-8757). FCCP was dissolved in ethanol. The amounts used were such that the ethanol concentration in the reaction mixture was never higher than 0.5% (v/v). At this concentration, ethanol had no effect on either the H+ gradient or PPi hydrolysis.
Preparation of Plant Material and Isolation of Microsomes from Maize Root Cells
Maize (Zea mays) seeds were surface sterilized with sodium hypochlorite (approximately 10 min in a 3% [v/v] solution), then washed with sterile water and soaked in water for 24 h. Radicles were harvested for preparation of vesicles from seeds allowed to germinate for 5 d on wet filter paper in the dark at 28°C.
Vacuolar membrane vesicles were isolated from whole roots using differential centrifugation. Roots (approximately 100 g wet weight) were homogenized in an IKA-Euroturrax T25 basic (speed 3 for 15 s using an S25N-18G probe, Wilmington, NC) with 200 mL of an ice-cold extraction buffer containing 10% (v/v) glycerol, 0.5% (v/v) polyvinylpyrrolidone-40, 0.13% (w/v) bovine serum albumin, 5 mm EDTA, and 0.1 m Tris-HCl (pH 8.0). Just before use, 3.3 mm dithiothreitol, 150 mm KCl, and 1 mm phenylmethylsulfonyl fluoride (final concentrations) were added to the buffer. The homogenate was strained through four layers of cheesecloth and centrifuged at 8,000g for 20 min. The supernatant was centrifuged at 100,000g for 40 min. The pellet was resuspended in 80 mL of the extraction buffer and centrifuged once more at 100,000g for 40 min. The pellet was resuspended in a small volume of ice-cold buffer containing 10 mm Tris-HCl (pH 7.6), 10% (v/v) glycerol, 1 mm EDTA, and 1 mm dithiothreitol. The vesicles were frozen under liquid N2. Protein concentrations were determined by the method of Lowry et al. (1951).
Measurement of F1,6BP Cleavage
The cleavage of F1,6BP was measured colorimetrically at 30°C by determining the rate of liberation of Pi (Fiske and Subbarow, 1925) in reaction media containing: 50 mm MOPS-Tris buffer (pH 7.0), 60 milliunits mL–1 mung bean PFP, 0.1 mg mL–1 maize vacuolar microsomal protein, 100 mm KCl, 2 μm F2,6BP, 0.6 mm MgCl2, and different F1,6BP concentrations. When the MgCl2 dependence was measured, the F1,6BP was fixed at 10 mm. When the pH dependence was measured, 50 mm MOPS-Tris and 50 mm glycyl-Gly buffer were added (pH from 6.0–8.5), and the concentrations of MgCl2 and F1,6BP were 1 and 5 mm, respectively. The reaction was started by the addition of 0.1 mm Pi. Aliquots of 0.8 mL were removed and the reaction was stopped by addition of 0.2 mL of 50% (w/v) trichloroacetic acid. Reaction times were adjusted so as to limit the cleavage of F 1,6 BP to <10%. Cleavage was linear with time under these conditions.
Measurement of PPi-Dependent Proton Transport into Maize Tonoplast Vesicles
The accumulation of H+ by the vesicles was determined by measuring the fluorescence quenching of ACMA (Molecular Probes, Eugene, OR) using a spectrofluorimeter (model F-3010, Hitachi, Tokyo). The excitation wavelength was set at 415 nm, and the emission wavelength was set at 485 nm. The reaction was carried out in 2 mL of medium containing 10 mm MOPS-Tris (pH 7.0), 3 μm ACMA, 1 mm MgCl2, and 100 mm KCl.
Acknowledgments
We are grateful to Dr. Martha Sorenson for critical reading of the manuscript and valuable suggestions.
Article, publication date, and citation information can be found at www.plantphysiol.org/cgi/doi/10.1104/pp.103.026633.
This work was supported by Programa de Apoio a Núcleos de Excelência-PRONEX-Financiadora de Estudos e Projetos (grant), by Conselho Nacional de Desenvolvimento Científico e Tecnológico (grant and fellowship to A.C.d.S.), and by Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro (grant and fellowship to W.S.d.-S.).
References
- ap Rees T, Green JH, Wilson PM (1985) Pyrophosphate-fructose 6-phosphate 1-phosphotransferase and glycolysis in non-photosynthetic tissues of higher plants. Biochem J 227: 299–304 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baykov AA, Cooperman BS, Goldman A, Lahti R (1999) Cytoplasmic inorganic pyrophosphatase. Prog Mol Subcell Biol 23: 127–150 [DOI] [PubMed] [Google Scholar]
- Black CC, Mustardy L, Suna SS, Kormanik PP, Xu D-P, Paz N (1987) Regulation and roles for alternative pathways of hexose metabolism in plants. Physiol Plant 69: 387–394 [Google Scholar]
- Carystinos GD, MacDonald HR, Monroy AF, Dhindsa RS, Poole RJ (1995) Vacuolar H+-translocating pyrophosphatase is induced by anoxia or chilling in seedlings of rice. Plant Physiol 108: 641–649 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clayton H, Ranson J, ap Rees T (1993) Pyrophosphate-fructose-6-phosphate 1-phosphotransferase and fructose 2,6-bisphosphate in the bundle sheath of maize leaves. Arch Biochem Biophys 301: 151–157 [DOI] [PubMed] [Google Scholar]
- Davies JM, Poole RJ, Sanders D (1993) The computed free-energy change of hydrolysis of inorganic pyrophosphate and ATP. Apparent significance for inorganic-pyrophosphate-driven reactions of intermediary metabolism. Biochim Biophys Acta 1141: 29–36 [Google Scholar]
- de Meis L (1984) Pyrophosphate of high and low energy: contribution of pH, Ca2+, Mg2+, and water to free energy of hydrolysis. J Biol Chem 259: 6090–6097 [PubMed] [Google Scholar]
- de Meis L (1989) Role of water in the energy of hydrolysis of phosphate compounds-energy transduction in biological membranes. Biochim Biophys Acta 973: 333–349 [DOI] [PubMed] [Google Scholar]
- de Meis L (1993) The concept of energy rich phosphate compounds: water, transport ATPases and entropic energy. Arch Biochem Biophys 366: 287–296 [DOI] [PubMed] [Google Scholar]
- de Meis L, Grieco MA, Galina A (1992a) Reversal of oxidative phosphorylation in submitochondrial particles using glucose 6-phosphate and hexokinase as an ATP regenerating system. FEBS Lett 308: 197–201 [DOI] [PubMed] [Google Scholar]
- de Meis L, Montero-Lomelí M, Grieco MAB, Galina A (1992b) The Maxwell Demon in biological systems: use of glucose 6-phosphate and hexokinase as an ATP regenerating system by the Ca2+-ATPase of sarcoplasmic reticulum and submitochondrial particles. Ann N Y Acad Sci 671: 19–31 [DOI] [PubMed] [Google Scholar]
- Drew MC (1997) Oxygen deficiency and root metabolism: injury and acclimation under hypoxia and anoxia. Annu Rev Plant Physiol Plant Mol Biol 48: 223–250 [DOI] [PubMed] [Google Scholar]
- Duff SMG, Moorhead GBG, Lefebvre DD, Plaxton WC (1989) Phosphate starvation inducible bypasses of adenylate and phosphate-dependent glycolytic enzymes in Brassica nigra suspension cells. Plant Physiol 90: 1275–1278 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fiske CH, Subbarow Y (1925) The colorimetric determination of phosphorus. J Biol Chem 66: 375–400 [Google Scholar]
- Galina A, Reis M, Albuquerque MC, Gomez-Puyou A, Gomez-Puyou MT, de Meis L (1995) Different properties of the mitochondrial and cytosolic hexokinases in maize roots. Biochem J 309: 105–112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Givan CV (1999) Evolving concepts in plant glycolysis: two centuries of progress. Biol Rev 74: 277–309 [Google Scholar]
- Hajirezaei M, Sonnewald U, Viola R, Carlisle S, Dennis DT, Stitt M (1994) Transgenic potato plants with strongly decreased expression of pyrophosphate: fructose-6 phosphate phosphotransferase show no visible phenotype and only minor changes in metabolic fluxes in their tubers. Planta 192: 16–30 [Google Scholar]
- Kombrink E, Kruger NJ, Beevers H (1984) Kinetic properties of pyrophosphate-fructose-6-phosphate phosphotransferase from germinating castor bean endosperm. Plant Physiol 74: 395–401 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kubota K, Ashihara H (1990) Identification of non-equilibrium glycolytic reactions in suspension-cultured plant cells. Biochim Biophys Acta 1036: 138–142 [DOI] [PubMed] [Google Scholar]
- Lowry OH, Rosebrough JN, Farr AL, Randall RJ (1951) Protein measurements with the Folin phenol reagent. J Biol Chem 193: 265–275 [PubMed] [Google Scholar]
- Maeshima M (2000) Vacuolar H+-pyrophosphatase. Biochim Biophys Acta 1465: 37–51 [DOI] [PubMed] [Google Scholar]
- Maeshima M, Yoshida S (1989) Purification and properties of vacuolar membrane proton-translocating inorganic pyrophosphatase from mung bean. J Biol Chem 264: 20068–20073 [PubMed] [Google Scholar]
- Mertens E, Larondelle Y, Hers H-G (1990) Induction of pyrophosphate: fructose 6-phosphate 1 phosphotransferase by anoxia in rice seedlings. Plant Physiol 93: 584–587 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milner ID, Ho LC, Hall JL (1995) Properties of proton and sugar transport at the tonoplast of tomato (Lycopersicon esculentum) fruit. Physiol Plant 94: 399–410 [Google Scholar]
- Montero-Lomeli M, de Meis L (1992) Glucose 6-phosphate and hexokinase can be used as an ATP-regenerating system by the Ca2+-ATPase of sarcoplasmic reticulum. J Biol Chem 267: 1829–1833 [PubMed] [Google Scholar]
- Nielsen TH, Stitt M (2001) Tobacco transformants with strongly decreased expression of pyrophosphate: fructose-6-phosphate 1-phosphotransferase expression in the base of their young growing leaves contain much higher levels of fructose-2,6-bisphosphate but no major changes in fluxes. Planta 214: 106–116 [DOI] [PubMed] [Google Scholar]
- O'Brien WE, Bowien S, Wood HG (1975) Isolation and characterization of a pyrophosphate-dependent phosphofructokinase from Propionibacterium shermanii. J Biol Chem 250: 8690–8695 [PubMed] [Google Scholar]
- Paul M, Sonnewald U, Hajirezaei M, Dennis D, Stitt M (1995) Transgenic tobacco plants with strongly decreased expression of pyrophosphatefructose-6-phosphate 1-phosphotransferase do not differ significantly from wild-type in photosynthate partitioning, plant growth or their ability to cope with limiting phosphate. Planta 196: 277–283 [Google Scholar]
- Rebeille F, Bligny R, Martin J-B, Douce R (1983) Relationship between the cytoplasm and the vacuole phosphate pool in Acer pseudoplatanus cells. Arch Biochem Biophys 225: 143–148 [DOI] [PubMed] [Google Scholar]
- Roberts JKM (1990) Observation of uridine triphosphate:glucose-1-phosphate uridylyltransferase activity in maize roots tips by saturation transfer 31P-NMR: estimation of cytoplasmic PPi. Biochim Biophys Acta 1051: 29–36 [DOI] [PubMed] [Google Scholar]
- Romero P, de Meis L (1989) Role of water in the energy of hydrolysis of phosphoanhydride and phosphoesters bonds. J Biol Chem 264: 7869–7873 [PubMed] [Google Scholar]
- Smyth DA, Chevalier P (1984) Phosphate accumulation in leaves of wheat seedlings measured in leaf cell protoplasts isolated after root or foliar treatment with ortho-phosphate. Physiol Plant 61: 135–140 [Google Scholar]
- Stitt M (1998) Pyrophosphate as an energy donor in the cytosol of plant cells: an enigmatic alternative to ATP. Bot Acta 111: 167–175 [Google Scholar]
- Ukaji T, Ashihara H (1987) Metabolic regulation in plant-cell culture: XXI. Effect of inorganic phosphate on synthesis of 5-phosphoribosyl-1-pyrophosphate in cultured plant cells. Int J Biochem 11: 1127–1131 [Google Scholar]
- Weiner H, Stitt M, Heldt HW (1987) Subcellular compartmentation of pyrophosphate and alkaline pyrophosphatase in leaves. Biochim Biophys Acta 893: 13–21.0.0 [Google Scholar]