Abstract
Recombination activating gene (RAG) expression in peripheral B cells increases after immunization with (4-hydroxy-3-nitrophenyl) acetyl coupled to chicken gamma globulin (NP-CGG) in alum. This increase could result from reinduction of RAG expression or, alternatively, from accumulation of RAG-expressing immature B cells in the periphery. We have used mice that carry a green fluorescent protein (GFP) RAG indicator transgene (RAG2-GFP) to characterize the RAG-expressing B cells in immunized spleens. Most of the RAG2-GFP–expressing B cells in unimmunized spleen are immature B cells. Injection with NP-CGG in alum initially suppresses lymphopoiesis in the bone marrow and decreases the number of immature RAG2-GFP–expressing B cells in the spleen. Recovery of lymphopoiesis in the bone marrow coincides with accumulation of RAG-expressing immature B cells in the spleen. Most of the RAG-expressing cells that accumulate in the spleen after immunization do not proliferate and they are not germinal center cells. Neither the initial suppression of lymphopoiesis nor the subsequent accumulation of RAG-expressing cells in the spleen is antigen dependent, since similar changes are seen with alum alone. Furthermore, such changes in the numbers of developing and circulating immature lymphoid cells are seen after injection with complete Freund's adjuvant or malaria infection. Our experiments suggest that adjuvants and infectious agents cause previously unappreciated alterations in lymphopoiesis resulting in the accumulation of RAG-expressing immature B cells in the spleen.
Keywords: recombination activating gene, adjuvant, malaria, lymphocyte development, green fluorescent protein indicator gene
Introduction
Recombination activating genes 1 and 2 (RAG1 and RAG2) encode a lymphocyte-specific enzyme that catalyzes V(D)J recombination 1 2 3. In the B lymphoid lineage, RAG expression is primarily restricted to developing B cells in the bone marrow 4 5. However, low level RAG expression is also found in B cells in peripheral lymphoid organs, and several laboratories have reported increases in RAG expression in peripheral B cells after immunization 6 7 8 9. Two hypotheses have been put forward to account for this increase: RAG expression could be reinduced in B cells undergoing antigen-activated immune responses 6 7; alternatively, RAG expression in the periphery might reflect accumulation of immature B cells containing residual RAG mRNA 6 7 10.
To examine regulation of RAG2 expression in vivo, we and others produced mice that carry RAG2 green fluorescent protein (RAG2-GFP) indicator genes 9 10 11. Although there were several differences in the techniques used to produce the indicator strains, the results of the experiments were similar: RAG2-GFP expression was found in spleen B cells, and in both cases it was the immature B cells that expressed the indicator 9 10. However, the source of the increased RAG expression observed after immunization was not determined 9 10.
Here we report experiments showing that transient increases in RAG expression in the spleens of immunized mice result from changes in the production of immature B cells and their export from the bone marrow.
Materials and Methods
Mice.
RAG2-GFP (FVB/N or [FVB/N × C57BL/6 (B6)] F1) mice 10, TNF knockout (TNF−/−) 12, type I IFN receptor knockout (IFNR−/−) 13, and signal transducer and activator of transcription (STAT)1 knockout (STAT1−/−) 14 mice and matched controls were bred and maintained under specific pathogen-free conditions. B6, RAG1 knockout (RAG1−/−), and 129S6/SvEv mice were purchased from The Jackson Laboratory or Taconic Farms. Mice were 2–5 mo old and were always age and sex matched.
Immunization.
400 μl 10% aluminum potassium sulfate with or without 100 μg of (4-hydroxy-3-nitrophenyl) acetyl (NP) coupled to chicken gamma globulin (NP-CGG; Biosearch Technologies, Inc.) was precipitated by adjusting pH to 6.2 with 1 N potassium hydroxide; the alum precipitates were washed in PBS three times and injected intraperitoneally. CFA (Sigma-Aldrich) was mixed with an equal volume of PBS, and 200 μl was injected intraperitoneally.
Malaria Infection.
Plasmodium yoelii (17X NL strain) was maintained by alternating cyclic passage of the parasites in Anopheles stephensi mosquitoes and BALB/c mice. Before infecting B6 mice, parasites were passed in this strain by injecting the mice with parasite-infected BALB/c red blood cells (PRBCs). For experimental infections in B6 mice, 1 × 103 PRBCs obtained from B6 mice were injected intraperitoneally, and parasitemia was determined by microscopic examination of Giemsa-stained thin blood smears 15.
Adoptive Transfer.
Adoptive transfer was performed as described 10. 2–4 × 107 splenocytes from wild-type B6 or RAG2-GFP ([FVB/N × B6]F1) mice were transferred by intravenous injection into B6 RAG1−/− recipients.
Flow Cytometry and Cell Sorting.
The following anti–mouse antibodies were used: biotin, PE, or allophycocyanin (APC) anti-B220 (RA3-6B2); biotin or FITC anti-CD43 (S7); biotin or PE anti–heat-stable antigen (HSA) (M1/69); biotin anti-IgM (R6-60.2); biotin anti-Igλ1λ2 (R26-46); biotin anti-Igκ (R5-250); GL7; biotin anti-CD4 (H129.19); APC anti-CD8 (Ly-2); PE anti-Fas (Jo2); biotin anti–Ly-6G (RB6-8C5); PE DX5; FITC annexin V; and FcBlock (2.4G2) (all from BD PharMingen). Biotinylated antibodies were visualized with streptavidin–Red 613 (GIBCO BRL) or streptavidin-PerCP (Becton Dickinson). GL7 was visualized using PE- or biotin-labeled mouse anti–rat IgM. For DNA staining, cells were suspended in 20 μM Hoechst 33342 (Molecular Probes)/2% fetal bovine serum/PBS and incubated at 37°C for 40 min. Data were acquired with a FACSCalibur™ or FACS Vantage™ (Becton Dickinson) and analyzed with CELLQuest™ software (Becton Dickinson). Nucleated lymphocytes were electronically gated on forward and side scatter; for DNA analysis, doublets were excluded by area and width of FL-5 signal pulses. For RNA analysis, spleen cells stained with anti-B220, anti-HSA, and anti-IgM antibodies and fractions of cells (1–2 × 105) were sorted directly into TRIzol LS (GIBCO BRL).
Reverse Transcription PCR.
RNA was extracted using TRIzol (GIBCO BRL), and cDNA was synthesized using Superscript II reverse transcriptase (GIBCO BRL). Reverse transcription (RT)-PCR reactions were performed on serial dilutions of cDNA template using HotStarTaq polymerase (Qiagen). PCR conditions and primers were as described 10. Specificity of PCR products was confirmed by Southern blotting as described 16. The oligonucleotide probe used for Igβ was 5′-GTGACCTGCCACTGAATTTCCAAG-3′.
Results
Half-Life of GFP Expression in Immature B Cells In Vivo.
GFP+ B cells in the spleens of RAG2-GFP mice display the surface features of immature B cells (B220lowHSAhigh 493+) 10, whereas GFP− B cells are mature B cells (B220high HSAlow493−) 10 17 18. Upon transfer to RAG−/− recipients, GFP+ B cells become GFP− and home to lymph nodes and spleen, suggesting that they mature and enter the long-lived B cell compartment 10. To determine the kinetics of loss of GFP expression, we transferred spleen cells from RAG2-GFP indicator mice into RAG1−/− recipients (Fig. 1 A). After adoptive transfer the number of GFPhigh B cells decreased with a half-life of 52 h (Fig. 1 A), and after 4 d these cells were no longer found in RAG−/− recipients. Similar kinetics of loss of GFP expression were found when GFP+ spleen B cells were cultured in vitro (10; and not shown). Therefore, it is unlikely that GFP+ cells preferentially migrate out from spleen. We conclude that RAG2-GFP is only expressed for a short time after B cells leave the bone marrow, and that GFP+ B cells are not generated from spleen B cell precursors.
Figure 1.

Adoptive transfer of RAG2-GFP mouse splenocytes into RAG1−/− mice. (A) Percentage of GFPhigh cells in IgM+ population recovered from RAG−/− mouse spleen, monitored at 24-h intervals after adoptive transfer (four mice per time point). The curve was fitted using Deltagraph™ software (DeltaPoint, Inc.). (B) RAG1 mRNA expression in B cells purified from the spleens of RAG1−/− mice that had received 2–4 × 107 wild-type spleen cells 7 d earlier. Serial fivefold dilutions of the cDNA are shown. PCR for Igβ is a cDNA loading control. R1, RAG1; TRSF, B cells isolated from RAG1−/− adoptive transfer recipient; WT, unmanipulated wild-type mouse B cells.
To confirm that loss of RAG2-GFP expression correlates with loss of endogenous RAG mRNA, we measured RAG1 mRNA by RT-PCR. Spleen cells were transferred into RAG1−/− mice, and B220+HSAhigh cells were purified after 7 d. RAG1 mRNA could not be detected in the transferred B cells, whereas immature spleen B cells from a wild-type control were positive for RAG1 mRNA expression (Fig. 1 B). We conclude that RAG2-GFP expression after transfer correlates with endogenous RAG mRNA expression and that RAG-expressing immature B cells are not of splenic origin.
The Number of Immature B Cells Is Regulated during Immune Responses.
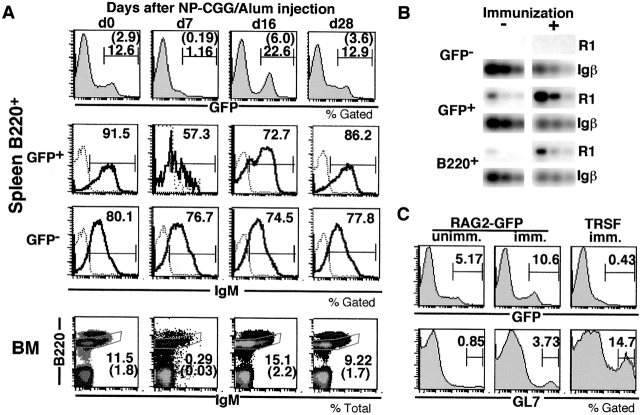
Experiments in several laboratories have shown that B cell RAG expression increases in peripheral lymphoid organs after immunization 6 7 8 9. To determine whether immunization also increases RAG2-GFP expression, we injected indicator mice with NP-CGG in alum and performed time course experiments (Fig. 2 A). Although the number of immature GFP+ B cells in the spleen initially decreased (34 ± 15% of control [n = 5]; Fig. 2 A), by day 16 their number had increased above the starting value (199 ± 38% of control [n = 6]; Fig. 2 A). Both the timing and magnitude of this transient increase in RAG expression are consistent with previous observations 6 7 9.
Figure 2.
RAG2 expression in peripheral B cells in immune responses. (A) RAG2-GFP expression in mice injected with NP-CGG in alum. The first row shows GFP intensity on B220+ spleen cells. The second and third rows show IgM intensity (bold lines) on GFP+B220+ and GFP−B220+ spleen cells; dotted lines indicate B220− cell controls. The fourth row shows B220/IgM density plots of bone marrow (BM) cells. Numbers are the percentages of gated cells or absolute cell numbers ×106 (shown in parentheses). (B) Increased endogenous RAG1 mRNA expression in immunized mouse spleen. RAG1 mRNA levels were estimated by RT-PCR. Sorted GFP−B220+ (GFP−), GFP+B220+ (GFP+), and B220+ splenocytes from mice immunized (+) 16 d earlier or unimmunized (−) were analyzed. Serial fivefold dilutions of the cDNA are shown. PCR for Igβ is a cDNA loading control. (C) RAG2-GFP and GL7 expression in adoptively transferred RAG1−/− mice 16 d after immunization with NP-CGG in alum. Histograms show GFP and GL7 expression on B220+ spleen cells. Percentage of cells found in each category is indicated. R1, RAG1; RAG2-GFP, RAG2-GFP mouse control; unimm., unimmunized control; imm., immunized mice; TRSF imm., RAG1−/− mouse that received 3 × 107 RAG2-GFP spleen cells 1 d before immunization.
The GFP+ B cells in the spleen on day 16 after immunization were heterogeneous and showed an increased proportion of less mature IgMlow B cells (Fig. 2 A). In contrast, there was no shift in the surface IgM expression in GFP− B cells with immunization. To confirm that changes in RAG2-GFP expression after immunization reflect changes in endogenous RAG mRNA expression, we compared RAG1 mRNA levels in GFP+ and GFP− B cells purified from spleens of immunized and control mice. Endogenous RAG1 mRNA was found in GFP+ but not in GFP− cells, and the level of RAG1 expression per cell increased after immunization (Fig. 2 B). The relative increase in RAG1 expression in GFP+ B cells may reflect a relative increase in less mature IgMlow cells, since these are known to express higher levels of RAGs than IgMhigh immature B cells (Fig. 2 A, middle panels [9, 10]). We conclude that the number of RAG-expressing immature B cells in the spleen increases after immunization with NP-CGG in alum. Finally, the increase in RAG2-GFP expression in spleen at day 16 was transient and the number of GFP+ B cells returned to normal by day 28 after immunization (Fig. 2 A). Interestingly, the number of immature B lineage cell in the bone marrow also varied during the response, but GFP intensity was not significantly altered by immunization (Fig. 2 A, and data not shown).
As noted above, the majority of the GFP+ B cells in unimmunized spleens are not derived from splenic precursors. To determine whether immunization induces spleen B cells to express RAG2-GFP, we performed adoptive transfer experiments. Spleen cells from RAG2-GFP indicator mice were transferred into RAG1−/− hosts that were immunized on day 1 after transfer. Although the immunized recipients showed an increase in spleen GL7+ B cells consistent with an ongoing immune response, we found no increase in GFP+ B cells at day 16 after adoptive transfer of spleen cells (Fig. 2 C). We conclude that the GFP+ B cells found in the spleen at day 16 after immunization are not generated from splenic precursors.
To determine whether GFP+ B cells in the spleen at day 16 after immunization were dividing, we measured their DNA content with Hoechst dye. In seven independent experiments, only 1.18 ± 0.52% of the GFP+ B cells had S or G2/M phase DNA content. Most of these cycling cells were found in a GL7lowB220low but Fas− subpopulation of GFP+ cells (Fig. 3A and Fig. B; see gate G2). Therefore, most of the GFP+ B cells in the spleens of immunized RAG2-GFP indicator mice are not germinal center (GC) cells, which are predominantly GL7high and Fas+ (Fig. 3A and Fig. B; see gate G1) 6 19 20.
Figure 3.

Cell cycle analysis of spleen B cells from immunized mice. RAG2-GFP mice were immunized with NP-CGG in alum and analyzed at day 16. (A) Histograms show DNA content of GFP−GL7high (G1), GFP+ GL7low (G2), and GFP+GL7− (G3) B cells. The gates are shown on the density plot. (B) Expression of Fas in B220+GL7+ GFP+ (G1 in A) and B220+ GL7+GFP− (G2 in A) spleen cells at day 16 of NP-CGG/alum immunization. Numbers are percentages of cells within the marked area.
Altered B Lymphopoiesis Induced by Adjuvants or Infectious Agents.
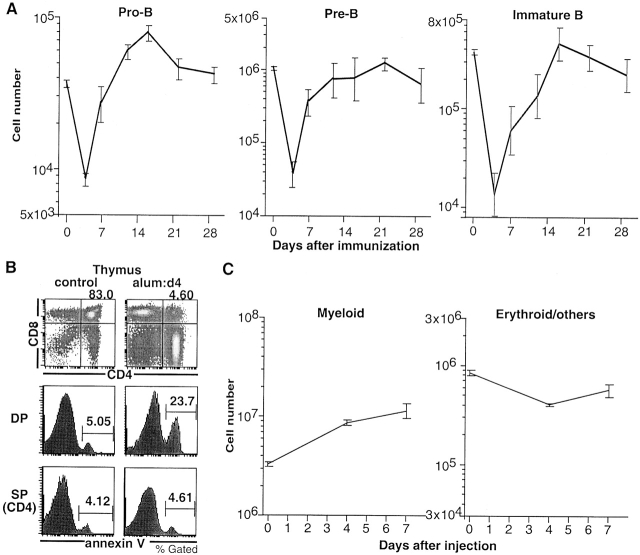
As noted above, B lymphopoiesis is initially suppressed by intraperitoneal injection with NP-CGG in alum. To determine which stages of B cell development are affected, we immunized B6 mice and counted B cell progenitors in the bone marrow (Fig. 4 A). We found that pro-B cell, pre-B cell, and immature B cell numbers are all reduced by 5–20-fold by day 4 after immunization but all of these precursors recover by day 16. In contrast, mature B cell numbers in the spleen change little with immunization (not shown). We conclude that immunization with NP-CGG in alum initially suppresses B lymphopoiesis.
Figure 4.
Changes in hemato-lymphopoiesis after injection with NP-CGG in alum. (A) Bone marrow pro-B, pre-B, and immature B cells. B6 mice were immunized with NP-CGG in alum, and bone marrow cells obtained from a single femur were analyzed. Cells with the B220lowCD43+ HSAhighIgM−, B220lowCD43−HSAhighIgM−, and B220lowIgM+ phenotypes were counted as pro-, pre-, and immature B cells, respectively. Mean ± SE (n = 4–5) is shown. (B) Thymocytes: increased cell death in the thymus 4 d after alum injection. Density plots show B6 thymocytes stained with anti-CD4 and anti-CD8. Percentage of CD4+ CD8+ cells is indicated. Histograms show annexin V staining on gated populations of CD4+CD8+ (DP) and CD4+CD8− (SP) thymocytes. Control mice were not injected with alum. (C) Myeloid and erythroid cells obtained from single femurs. Ly-6G+B220−DX5− and Ly-6G−B220−DX5− cells were counted as myeloid or erythroid/others respectively. Mean ± SE (n = 3–4) is shown.
To determine whether the effect of NP-CGG plus alum is antigen dependent, we injected RAG2-GFP indicator mice and controls with alum alone. Such mice were indistinguishable from mice injected with NP-CGG in alum. The number of GFP+ B cells in the spleen dropped to 36 ± 12% of control on day 7 (n = 5) and then increased to 189 ± 26% of control on day 16 after alum injection (n = 5). These changes coincided with initial suppression of B lymphopoiesis followed by recovery at day 16. Furthermore, CFA had similar effects on B lymphopoiesis (Table ). Thus, altered lymphopoiesis in NP-CGG and alum–injected mice is antigen independent and can be induced by either alum or CFA.
Table 1.
Effect of Adjuvant Injection on T and B Lineage Cells
| Number of cells (×105) | |||
|---|---|---|---|
| Cell | PBS | Alum | CFA |
| Bone marrow | |||
| Total | 106.38 ± 9.93 | 126.67 ± 3.33 | 112.67 ± 7.22 |
| Pro-B | 1.02 ± 0.17 | 0.12 ± 0.01 | 0.12 ± 0.03 |
| Pre-B | 5.52 ± 0.69 | 0.62 ± 0.11 | 1.05 ± 0.20 |
| Immature B | 2.31 ± 0.41 | 0.45 ± 0.07 | 0.36 ± 0.10 |
| Recirculating B | 7.67 ± 1.48 | 0.35 ± 0.12 | 0.20 ± 0.08 |
| Myeloid | 37.50 ± 2.97 | 85.28 ± 4.97 | 65.92 ± 3.16 |
| Thymus | |||
| Total | 772.50 ± 52.82 | 64.67 ± 21.36 | 136.67 ± 50.44 |
| DP | 440.87 ± 41.69 | 9.06 ± 4.08 | 61.46 ± 26.37 |
| Spleen | |||
| B | 26.01 ± 1.78 | 26.37 ± 0.34 | 28.56 ± 3.02 |
| T | 13.37 ± 1.37 | 10.06 ± 0.55 | 10.14 ± 0.68 |
Reagents were intraperitoneally injected 4 d before analysis. Numbers are mean ± SE (n = 3 or 4). Pro-B, B220lowCD43+HSAhighIgM−; Pre-B, B220lowCD43−HSAhighIgM−; Immature B, B220lowIgM+; Recirculating B, B220high; Myeloid, Ly-6G+B220−DX5−; DP, CD4+CD8+; T, CD3+.
In addition to suppressing B lymphopoiesis, adjuvant injection also suppressed thymopoiesis. 4 d after administration of alum or CFA, the number of CD4+CD8+ thymocytes decreased 7–50-fold (Fig. 4 B, and Table ). The loss of CD4+CD8+ thymocytes was associated with an increase in annexin V staining indicative of increased programmed cell death (Fig. 4 B). In contrast, the number of myeloid cells in the bone marrow increased while the number of erythroid cells was not significantly altered (Fig. 4 C). We conclude that administration of alum or CFA initially suppresses both T and B lymphopoiesis, that this effect is cell type and developmental stage specific, and that it correlated with an increase in thymocyte apoptosis 21.
IFNs and TNF are known to suppress hematopoiesis during certain viral infections 22 23. For example, type I IFNs are essential for suppression of hemato-lymphopoiesis after lymphocytic choriomeningitis virus (LCMV) infection 22. To determine whether suppression of lymphopoiesis by adjuvants is mediated by IFNs or TNF, we injected type I IFNR−/−, TNF−/−, and STAT1−/− mice with alum and examined B and T cell development on day 4 12 13 14 (Fig. 5). Although we found significant differences in the magnitude of the adjuvant-induced suppression of lymphopoiesis in the different control mouse strains, the absence of type I IFNR, STAT1, or TNF had no effect on adjuvant-mediated suppression of B or T lymphopoiesis. We conclude that neither type I IFNR, nor STAT1, nor TNF is essential for the suppression of lymphopoiesis by adjuvant.
Figure 5.
STAT1−/−, type I IFNR−/−, and TNF−/− mice are susceptible to alum-mediated suppression of lymphopoiesis. Mice with null mutations of Stat1, Ifnar, or Tnf were injected with alum 4 d before analysis. B220low cells from a single femur and CD4+CD8+ (DP) thymocytes were counted and compared with background-matched controls. □, uninjected wild-type mouse; ▪, injected wild-type mouse; ○, uninjected knockout mouse; •, injected knockout mouse; W, wild-type; −/−, knockout.
Adjuvants are thought to impact the immune system by some of the same mechanisms as natural infections. To determine whether lymphopoiesis is also suppressed during acute infection, we exposed mice to P. yoelii. At the peak of parasitemia, day 14 after infection, the number of B220low immature B lineage cells in the bone marrow was severely reduced (Fig. 6), but the number of developing B cells recovered and even increased by day 28 when the malaria parasites had been cleared 24. We conclude that the development of immature B lineage cells is suppressed during malaria infection, as it is after administration of adjuvants.
Figure 6.
B220/IgM staining of bone marrow cells from malaria-infected mice. Date after infection is shown above the density plots and parasitemia below. Percentages of B220low cells and absolute cell numbers ×106 (shown in parentheses) are indicated.
Discussion
RAG2-GFP indicator mouse strains produced by two different technologies have been used to examine RAG expression in vivo 9 10 11. In one case, GFP was targeted into RAG2 to produce a locus that directs the synthesis of a RAG2-GFP fusion protein 9. The dynamic range of GFP fluorescence in this strain is about one log over background and to enhance detection analyses must be performed in homozygous mutant mice 9. The second indicator strain carries a 165-kb bacterial artificial chromosome transgene in which GFP was inserted into the RAG2 gene 10. In this transgenic indicator strain, GFP is not produced as a fusion protein and fluorescence levels are much higher, with a dynamic range of three to four logs. The half-life and precise specific activity of the targeted RAG2-GFP fusion protein are not known, but a clear disadvantage of the transgenic system is that the half-life of the GFP indicator in this strain is 2–3 d, probably much longer than that of the endogenous RAG2 protein. However, despite the differences between the two indicator strains, and the long half-life of the transgenic GFP, the results reported for the two indicator strains are very similar 9 10. In both cases, there were two peaks of RAG2-GFP indicator gene expression in developing B cells corresponding to two waves of RAG transcription during heavy and light chain gene recombination 4 5 9 10. In addition, both indicator strains showed GFP expression in immature B cells in the bone marrow and spleen 9 10. The amount of endogenous RAG mRNA found in immature B cells was inversely correlated with the levels of surface IgM expression and up to two orders of magnitude lower than that found in pro- or pre-B cells 9 10. Immature B cells with the highest levels of surface IgM had exceedingly low levels of RAG mRNA, below the dynamic range of detection in the targeted indicator strain, and the significance of this low level of RAG expression has yet to be determined.
Several groups have shown that immunization increases RAG expression in B cells in peripheral lymphoid organs, raising the possibility that RAG could be reinduced in mature B cells 6 7 8 9. In our initial experiments, we found that RAG expression was not reinduced in mature B cells after immunization and we were unable to induce RAG expression in vitro with LPS plus IL-4 10. Nevertheless, there is an increase in the number of RAG2-GFP–expressing B cells in spleens of mice injected with alum. Most of these cells are not of splenic origin, they are nondividing cells that carry the cell surface markers of immature B cells. Only a subset of the GFP+ B cells in the spleen is rapidly proliferating, and a small subfraction of these rapidly dividing cells expresses high levels of both GL7 and Fas, which are markers of GC B cells 19 20. Therefore, most GFP+ B cells found in the spleen on day 16 after injection with alum are immature, and only a very small number of these cells have the characteristics of GC cells. It has been proposed that RAG expression in peripheral B cells might contribute to further diversification of the antibody repertoire in GCs during immune reactions 6 7 8 25 26. Our results suggest that antibody gene replacement in GCs is a rare event. However, inflammatory or pathological processes that lead to accumulation of immature B cells might increase the frequency of such events 16 27.
What is the origin of the RAG2-GFP–expressing cells that accumulate in the spleen after immunization? Immature B cells are thought to be produced by the bone marrow at a steady rate of 1–2 × 107/d, and homeostasis maintained by selecting a variable fraction of these cells into the long-lived B cell compartment 28 29 30 31 32 33. However, our experiments suggest that the increase in RAG2-GFP–expressing B cells found in the spleen after immunization is due to an increase in immature B cell production and export 12 d after initial suppression of B lymphopoiesis by adjuvant. This rebound effect resembles the increase in immature B cell production and emigration from the bone marrow found 12–14 d after sublethal irradiation 17 and differs from the augmentation of pre-B cell production observed 4 d after injection with sheep RBCs 34. We conclude that, like irradiation, adjuvants and infection affect lymphopoiesis and alter the rates of immature B cell production and export from the bone marrow.
Transient suppression of B and T lymphopoiesis has been reported during infection with LCMV and after IFN injection, but these agents produced a more general suppression of hematopoiesis not found with adjuvant injection 22 35. In LMCV infection, bone marrow suppression is mediated by type I IFNs 22, but neither type I IFNs, nor STAT1, nor TNF were essential for the effect produced by alum injection.
What is the significance of altering lymphopoiesis after administration of adjuvant or during infection? Alterations in lymphopoiesis could be limited to malaria and LCMV, and to administration of CFA or alum, and therefore have little physiological significance. However, the documented suppressive effects of inflammatory cytokines, IFNs, and TNF on lymphopoiesis 36 37 38 39 suggest that any infectious or inflammatory agent that significantly increases the production of these cytokines and possibly other such effectors may alter lymphopoiesis. Therefore, subsets of T and B cells recently exported from the bone marrow or thymus may resemble myeloid cells, which display well-characterized shifts during infection and inflammation. Such shifts in the composition of the lymphocyte populations might change the outcome of immune responses. In particular, immature B cells recently emigrating from the bone marrow are highly susceptible to tolerance and less likely to enter the memory pool 40 41 42 43 44 45. Thus, decreasing the number of such cells exported in acute infection may protect against tolerance and indirectly enhance B cell memory.
Acknowledgments
We thank Dr. Ruth S. Nussenzweig and members of the M.C. Nussenzweig laboratory for comments on the manuscript and helpful discussions, Dr. David E. Levy for providing type I IFNR−/− and STAT1−/− mice, and Dr. Michael W. Marino for TNF−/− mice.
This work was supported by grants from the National Institutes of Health to M.C. Nussenzweig and to M. Tsuji (AI40656 and AI33890). M.C. Nussenzweig is an Investigator in the Howard Hughes Medical Institute.
Footnotes
Abbreviations used in this paper: APC, allophycocyanin; B6, C57BL/6; CGG, chicken gamma globulin; GC, germinal center; GFP, green fluorescent protein; HSA, heat-stable antigen; LCMV, lymphocytic choriomeningitis virus; NP, (4-hydroxy-3-nitrophenyl) acetyl; PRBC, parasite-infected BALB/c red blood cell; RAG, recombination activating gene; RT, reverse transcription; STAT, signal transducer and activator of transcription.
References
- Schatz D.G., Oettinger M.A., Baltimore D. The V(D)J recombination activating gene, RAG-1. Cell. 1989;59:1035–1048. doi: 10.1016/0092-8674(89)90760-5. [DOI] [PubMed] [Google Scholar]
- Oettinger M.A., Schatz D.G., Gorka C., Baltimore D. RAG-1 and RAG-2, adjacent genes that synergistically activate V(D)J recombination. Science. 1990;248:1517–1523. doi: 10.1126/science.2360047. [DOI] [PubMed] [Google Scholar]
- McBlane J.F., van Gent D.C., Ramsden D.A., Romeo C., Cuomo C.A., Gellert M., Oettinger M.A. Cleavage at a V(D)J recombination signal requires only RAG1 and RAG2 proteins and occurs in two steps. Cell. 1995;83:387–395. doi: 10.1016/0092-8674(95)90116-7. [DOI] [PubMed] [Google Scholar]
- Grawunder U., Leu T.M., Schatz D.G., Werner A., Rolink A.G., Melchers F., Winkler T.H. Down-regulation of RAG1 and RAG2 gene expression in preB cells after functional immunoglobulin heavy chain rearrangement. Immunity. 1995;3:601–608. doi: 10.1016/1074-7613(95)90131-0. [DOI] [PubMed] [Google Scholar]
- Li Y.S., Hayakawa K., Hardy R.R. The regulated expression of B lineage–associated genes during B cell differentiation in bone marrow and fetal liver. J. Exp. Med. 1993;178:951–960. doi: 10.1084/jem.178.3.951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han S., Zheng B., Schatz D.G., Spanopoulou E., Kelsoe G. Neoteny in lymphocytesRag1 and Rag2 expression in germinal center B cells. Science. 1996;274:2094–2097. doi: 10.1126/science.274.5295.2094. [DOI] [PubMed] [Google Scholar]
- Hikida M., Mori M., Takai T., Tomochika K., Hamatani K., Ohmori H. Reexpression of RAG-1 and RAG-2 genes in activated mature mouse B cells. Science. 1996;274:2092–2094. doi: 10.1126/science.274.5295.2092. [DOI] [PubMed] [Google Scholar]
- Hertz M., Kouskoff V., Nakamura T., Nemazee D. V(D)J recombinase induction in splenic B lymphocytes is inhibited by antigen-receptor signaling. Nature. 1998;394:292–295. doi: 10.1038/28419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Monroe R.J., Seidl K.J., Gaertner F., Han S., Chen F., Sekiguchi J., Wang J., Ferrini R., Davidson L., Kelsoe G., Alt F.W. RAG2:GFP knockin mice reveal novel aspects of RAG2 expression in primary and peripheral lymphoid tissues. Immunity. 1999;11:201–212. doi: 10.1016/s1074-7613(00)80095-3. [DOI] [PubMed] [Google Scholar]
- Yu W., Nagaoka H., Jankovic M., Misulovin Z., Suh H., Rolink A., Melchers F., Meffre E., Nussenzweig M.C. Continued RAG expression in late stages of B cell development and no apparent re-induction after immunization. Nature. 1999;400:682–687. doi: 10.1038/23287. [DOI] [PubMed] [Google Scholar]
- Yu W., Misulovin Z., Suh H., Hardy R.R., Jankovic M., Yannoutsos N., Nussenzweig M.C. Coordinate regulation of RAG1 and RAG2 by cell type-specific DNA elements 5′ of RAG2. Science. 1999;285:1080–1084. doi: 10.1126/science.285.5430.1080. [DOI] [PubMed] [Google Scholar]
- Marino M.W., Dunn A., Grail D., Inglese M., Noguchi Y., Richards E., Jungbluth A., Wada H., Moore M., Williamson B. Characterization of tumor necrosis factor-deficient mice. Proc. Natl. Acad. Sci. USA. 1997;94:8093–8098. doi: 10.1073/pnas.94.15.8093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muller U., Steinhoff U., Reis L.F., Hemmi S., Pavlovic J., Zinkernagel R.M., Aguet M. Functional role of type I and type II interferons in antiviral defense. Science. 1994;264:1918–1921. doi: 10.1126/science.8009221. [DOI] [PubMed] [Google Scholar]
- Durbin J.E., Hackenmiller R., Simon M.C., Levy D.E. Targeted disruption of the mouse Stat1 gene results in compromised innate immunity to viral disease. Cell. 1996;84:443–450. doi: 10.1016/s0092-8674(00)81289-1. [DOI] [PubMed] [Google Scholar]
- Tsuji M., Miyahira Y., Nussenzweig R.S., Aguet M., Reichel M., Zavala F. Development of antimalaria immunity in mice lacking IFN-gamma receptor. J. Immunol. 1995;154:5338–5344. [PubMed] [Google Scholar]
- Qin X.F., Schwers S., Yu W., Papavasiliou F., Suh H., Nussenzweig A., Rajewsky K., Nussenzweig M.C. Secondary V(D)J recombination in B-1 cells. Nature. 1999;397:355–359. doi: 10.1038/16933. [DOI] [PubMed] [Google Scholar]
- Allman D.M., Ferguson S.E., Cancro M.P. Peripheral B cell maturation. I. Immature peripheral B cells in adults are heat-stable antigen high and exhibit unique signaling characteristics. J. Immunol. 1992;149:2533–2540. [PubMed] [Google Scholar]
- Rolink A.G., Andersson J., Melchers F. Characterization of immature B cells by a novel monoclonal antibody, by turnover and by mitogen reactivity. Eur. J. Immunol. 1998;28:3738–3748. doi: 10.1002/(SICI)1521-4141(199811)28:11<3738::AID-IMMU3738>3.0.CO;2-Q. [DOI] [PubMed] [Google Scholar]
- Smith K.G., Nossal G.J., Tarlinton D.M. FAS is highly expressed in the germinal center but is not required for regulation of the B-cell response to antigen. Proc. Natl. Acad. Sci. USA. 1995;92:11628–11632. doi: 10.1073/pnas.92.25.11628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watanabe D., Suda T., Nagata S. Expression of Fas in B cells of the mouse germinal center and Fas-dependent killing of activated B cells. Int. Immunol. 1995;7:1949–1956. doi: 10.1093/intimm/7.12.1949. [DOI] [PubMed] [Google Scholar]
- Vermes I., Haanen C., Steffens-Nakken H., Reutelingsperger C. A novel assay for apoptosis. Flow cytometric detection of phosphatidylserine expression on early apoptotic cells using fluorescein labeled Annexin V. J. Immunol. Methods. 1995;184:39–51. doi: 10.1016/0022-1759(95)00072-i. [DOI] [PubMed] [Google Scholar]
- Binder D., Fehr J., Hengartner H., Zinkernagel R.M. Virus-induced transient bone marrow aplasiamajor role of interferon-α/β during acute infection with the noncytopathic lymphocytic choriomeningitis virus. J. Exp. Med. 1997;185:517–530. doi: 10.1084/jem.185.3.517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maciejewski J.P., Weichold F.F., Young N.S. HIV-1 suppression of hematopoiesis in vitro mediated by envelope glycoprotein and TNF-alpha. J. Immunol. 1994;153:4303–4310. [PubMed] [Google Scholar]
- Osmond D.G., Priddle S., Rico-Vargas S. Proliferation of B cell precursors in bone marrow of pristane-conditioned and malaria-infected miceimplications for B cell oncogenesis. Curr. Top. Microbiol. Immunol. 1990;166:149–157. doi: 10.1007/978-3-642-75889-8_19. [DOI] [PubMed] [Google Scholar]
- Papavasiliou F., Casellas R., Suh H., Qin X.F., Besmer E., Pelanda R., Nemazee D., Rajewsky K., Nussenzweig M.C. V(D)J recombination in mature B cellsa mechanism for altering antibody responses. Science. 1997;278:298–301. doi: 10.1126/science.278.5336.298. [DOI] [PubMed] [Google Scholar]
- Han S., Dillon S.R., Zheng B., Shimoda M., Schlissel M.S., Kelsoe G. V(D)J recombinase activity in a subset of germinal center B lymphocytes. Science. 1997;278:301–305. doi: 10.1126/science.278.5336.301. [DOI] [PubMed] [Google Scholar]
- Brard F., Shannon M., Prak E.L., Litwin S., Weigert M. Somatic mutation and light chain rearrangement generate autoimmunity in anti–single-stranded DNA transgenic MRL/lpr mice. J. Exp. Med. 1999;190:691–704. doi: 10.1084/jem.190.5.691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Opstelten D., Osmond D.G. Pre-B cells in mouse bone marrowimmunofluorescence stathmokinetic studies of the proliferation of cytoplasmic mu-chain-bearing cells in normal mice. J. Immunol. 1983;131:2635–2640. [PubMed] [Google Scholar]
- Forster I., Rajewsky K. The bulk of the peripheral B-cell pool in mice is stable and not rapidly renewed from the bone marrow. Proc. Natl. Acad. Sci. USA. 1990;87:4781–4784. doi: 10.1073/pnas.87.12.4781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allman D.M., Ferguson S.E., Lentz V.M., Cancro M.P. Peripheral B cell maturation. II. Heat-stable antigenhi splenic B cells are an immature developmental intermediate in the production of long-lived marrow-derived B cells. J. Immunol. 1993;151:4431–4444. [PubMed] [Google Scholar]
- Agenes F., Rosado M.M., Freitas A.A. Independent homeostatic regulation of B cell compartments. Eur. J. Immunol. 1997;27:1801–1807. doi: 10.1002/eji.1830270731. [DOI] [PubMed] [Google Scholar]
- Agenes F., Freitas A.A. Transfer of small resting B cells into immunodeficient hosts results in the selection of a self-renewing activated B cell population. J. Exp. Med. 1999;189:319–330. doi: 10.1084/jem.189.2.319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cyster J.G., Hartley S.B., Goodnow C.C. Competition for follicular niches excludes self-reactive cells from the recirculating B-cell repertoire. Nature. 1994;371:389–395. doi: 10.1038/371389a0. [DOI] [PubMed] [Google Scholar]
- Fulop G.M., Osmond D.G. Regulation of bone marrow lymphocyte production. III. Increased production of B and non-B lymphocytes after administering systemic antigens. Cell. Immunol. 1983;75:80–90. doi: 10.1016/0008-8749(83)90307-6. [DOI] [PubMed] [Google Scholar]
- Lin Q., Dong C., Cooper M.D. Impairment of T and B cell development by treatment with a type I interferon. J. Exp. Med. 1998;187:79–87. doi: 10.1084/jem.187.1.79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grawunder U., Melchers F., Rolink A. Interferon-gamma arrests proliferation and causes apoptosis in stromal cell/interleukin-7-dependent normal murine pre-B cell lines and clones in vitro, but does not induce differentiation to surface immunoglobulin-positive B cells. Eur. J. Immunol. 1993;23:544–551. doi: 10.1002/eji.1830230237. [DOI] [PubMed] [Google Scholar]
- Veiby O.P., Jacobsen F.W., Cui L., Lyman S.D., Jacobsen S.E. The flt3 ligand promotes the survival of primitive hemopoietic progenitor cells with myeloid as well as B lymphoid potential. Suppression of apoptosis and counteraction by TNF-alpha and TGF-beta. J. Immunol. 1996;157:2953–2960. [PubMed] [Google Scholar]
- Wang J., Lin Q., Langston H., Cooper M.D. Resident bone marrow macrophages produce type 1 interferons that can selectively inhibit interleukin-7-driven growth of B lineage cells. Immunity. 1995;3:475–484. doi: 10.1016/1074-7613(95)90176-0. [DOI] [PubMed] [Google Scholar]
- Su D.M., Wang J., Lin Q., Cooper M.D., Watanabe T. Interferons alpha/beta inhibit IL-7-induced proliferation of CD4− CD8− CD3− CD44+ CD25+ thymocytes, but do not inhibit that of CD4− CD8− CD3− CD44− CD25− thymocytes. Immunology. 1997;90:543–549. doi: 10.1046/j.1365-2567.1997.00205.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nossal G.J. Cellular mechanisms of immunologic tolerance. Annu. Rev. Immunol. 1983;1:33–62. doi: 10.1146/annurev.iy.01.040183.000341. [DOI] [PubMed] [Google Scholar]
- Melamed D., Benschop R.J., Cambier J.C., Nemazee D. Developmental regulation of B lymphocyte immune tolerance compartmentalizes clonal selection from receptor selection. Cell. 1998;92:173–182. doi: 10.1016/s0092-8674(00)80912-5. [DOI] [PubMed] [Google Scholar]
- Benschop R.J., Melamed D., Nemazee D., Cambier J.C. Distinct signal thresholds for the unique antigen receptor–linked gene expression programs in mature and immature B cells. J. Exp. Med. 1999;190:749–756. doi: 10.1084/jem.190.6.749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yellen A.J., Glenn W., Sukhatme V.P., Cao X.M., Monroe J.G. Signaling through surface IgM in tolerance-susceptible immature murine B lymphocytes. Developmentally regulated differences in transmembrane signaling in splenic B cells from adult and neonatal mice. J. Immunol. 1991;146:1446–1454. [PubMed] [Google Scholar]
- Sandel P.C., Monroe J.G. Negative selection of immature B cells by receptor editing or deletion is determined by site of antigen encounter. Immunity. 1999;10:289–299. doi: 10.1016/s1074-7613(00)80029-1. [DOI] [PubMed] [Google Scholar]
- Klinman N.R. The “clonal selection hypothesis” and current concepts of B cell tolerance. Immunity. 1996;5:189–195. doi: 10.1016/s1074-7613(00)80314-3. [DOI] [PubMed] [Google Scholar]