Abstract
Efficient T cell activation is dependent on the intimate contact between antigen-presenting cells (APCs) and T cells. The engagement of the B7 family of molecules on APCs with CD28 and CD152 (cytotoxic T lymphocyte–associated antigen 4 [CTLA-4]) receptors on T cells delivers costimulatory signal(s) important in T cell activation. We investigated the dependence of pathologic cellular activation in psoriatic plaques on B7-mediated T cell costimulation. Patients with psoriasis vulgaris received four intravenous infusions of the soluble chimeric protein CTLA4Ig (BMS-188667) in a 26-wk, phase I, open label dose escalation study. Clinical improvement was associated with reduced cellular activation of lesional T cells, keratinocytes, dendritic cells (DCs), and vascular endothelium. Expression of CD40, CD54, and major histocompatibility complex (MHC) class II HLA-DR antigens by lesional keratinocytes was markedly reduced in serial biopsy specimens. Concurrent reductions in B7-1 (CD80), B7-2 (CD86), CD40, MHC class II, CD83, DC–lysosomal-associated membrane glycoprotein (DC-LAMP), and CD11c expression were detected on lesional DCs, which also decreased in number within lesional biopsies. Skin explant experiments suggested that these alterations in activated or mature DCs were not the result of direct toxicity of CTLA4Ig for DCs. Decreased lesional vascular ectasia and tortuosity were also observed and were accompanied by reduced presence of E-selectin, P-selectin, and CD54 on vascular endothelium. This study highlights the critical and proximal role of T cell activation through the B7-CD28/CD152 costimulatory pathway in maintaining the pathology of psoriasis, including the newly recognized accumulation of mature DCs in the epidermis.
Keywords: psoriasis vulgaris, T cell costimulation, B7, CTLA4Ig, dendritic cells
Introduction
Psoriasis vulgaris is an inflammatory skin disease afflicting ∼2% of the population. It is likely to be mediated by activated T cells, which early within the course of disease are present within the lesional skin in increased numbers 1 2. Dendritic cells (DCs), an APC population uniquely capable of efficiently stimulating resting T cells 3, are also increased in psoriatic lesions 4 and possess an activated phenotype 2 5. Cytokines released from these activated T cells and DCs are believed to contribute to the pathologic changes induced in lesional keratinocytes 6 7 and vascular endothelium 8 9.
Cytotoxic T lymphocyte–associated antigen 4 (CTLA-4)–immunoglobulin (CTLA4Ig; BMS-188667), a novel soluble chimeric protein, binds to B7-1 (CD80) and B7-2 (CD86) expressed on APCs and thereby inhibits a second signal required for optimal T cell activation 10 11 12 13. CTLA4Ig inhibits skin APC and T cell functional interactions in vitro 5 14. Administration of CTLA4Ig to patients with psoriasis vulgaris in a phase I trial produced a dose-dependent improvement in skin lesions. 9 of 11 patients in the top dosing groups achieved a 50% or greater decrease in psoriasis clinical scores 15. This use of CTLA4Ig in psoriasis patients provides a unique opportunity to ascertain the contribution of ongoing T cell costimulation to the persistence of chronic cell-mediated inflammation in a human disease 16. We will demonstrate here that the B7-targeted disruption of APC and T cell interaction effectively blocks T cell activation and reinstates a nascent phenotype to lesional tissue.
Materials and Methods
Patients.
A protocol outlining the use of CTLA4Ig (BMS-188667) in patients with moderate to severe psoriasis was approved by the Institutional Review Boards of participating clinical institutions. Patients providing informed consent for use of the investigational agent were enrolled into this study if they had a history of stable psoriasis vulgaris of at least 6-mo duration involving 10–49% of total body surface area, and had failed at least one prior antipsoriatic therapy. Before enrollment on study, systemic retinoids were discontinued for at least 2 yr; investigational drugs, methotrexate, cyclosporine, and systemic corticosteroids were discontinued for at least 16 wk; phototherapy and photochemotherapy were not administered for at least 4 wk; and topical treatments other than emollients were not administered for two or more weeks. The baseline demographics of this patient population were described in a previous publication 15.
CTLA4Ig (BMS-188667) Administration.
CTLA4Ig (BMS-188667) was administered as a 1-h intravenous infusion on day 1 (week 1), day 3, day 16 (week 3), and day 29 (week 5). Four to six patients were accrued to each of eight dose levels in this open label dose escalation study: 0.5, 1, 2, 4, 8, 16, 25, and 50 mg/kg. Patients were monitored continuously for a 4-h period after each infusion and at weekly intervals during the first 8 wk of study, then at biweekly to monthly intervals through day 176 (week 26). Safety, immunogenicity, pharmacokinetic, and biologic activity assessments were performed at each visit.
Immunohistochemical Studies.
Samples for histological analysis were obtained with a 6-mm punch biopsy from representative lesions before administration of CTLA4Ig (BMS-188667) and at study days 8, 16, 36, and 78. Each specimen was split for routine histopathology and for histochemical analysis on cryostat-cut 6-mm sections. Immunohistochemical staining procedures using 3-amino-9-ethylcarbazol as the chromagen were performed as described previously 17. Antibodies used for histochemical analysis had the following specificities: CD3, CD25, HLA-DR, CD1a, and CD80 (clones SK7, 2A3, L243, SK9, and L307, respectively; Becton Dickinson), CD8 (clone OKT8; American Type Culture Collection), CD86 and CD11c (clones FUN-1 and B-ly6, respectively; BD PharMingen), CD54 (RR 1/1; Biosource International), CD40, P-selectin, and E-selectin (clones G28.5, 5G4, and 5G11, respectively; Bristol-Myers Squibb), CD83 (clone HB15a; Immunotech), DC–lysosomal-associated membrane glycoprotein (DC-LAMP) and macrophage mannose receptor (MMR) (gifts from Ralph Steinman, The Rockefeller University), neutrophil elastase (clone 204; Biodesign), Factor XIIIa (FXIIIa; Calbiochem), or laminin (clone 2E8; a gift from Eva Engvall, La Jolla Cancer Research Foundation, La Jolla, CA). Quantitative measures of epidermal area and length (used to compute mean thickness) and cell numbers reactive with specific antibodies were obtained using a Macintosh computer using public domain IMAGE program from the National Institutes of Health (http://rsb.info.nih.gov/nih-image).
DC Migration Studies.
Split thickness biopsies of psoriatic skin lesions were obtained in The Rockefeller University General Clinical Research Center using a protocol approved by The Rockefeller University Institutional Review Board. Three psoriatic patients, who provided informed consent and did not receive infusions with CTLA4Ig (BMS-188667), underwent biopsy of psoriatic lesional skin. Split thickness samples were also prepared from the skin of four healthy donors undergoing corrective surgery of face, breast, or abdomen. The skin was stored at 4°C and used in the assays described below within 6 h of collection. Each explant was trimmed (psoriatic lesional skin: 30-mm2; normal skin: 400-mm2) and floated in 5–10 ml of culture medium. 3-d in vitro cultures of skin explants were established with the subsequent preparation of single emigrant cell suspensions as described previously 18. Skin explants were incubated in the presence of either CTLA4Ig (100 μg/ml) or media alone throughout the culture period.
Flow Cytometry.
Single cell suspensions of emigrant skin cells were incubated with saturating concentrations of the following mouse anti–human fluorochrome-conjugated antibodies, for 15 min at room temperature: PE-conjugated anti-CD3 (clone SK7; Becton Dickinson); peridinin chlorophyll protein (PerCP)-conjugated anti-CD45 (clone 2D1; Becton Dickinson); allophycocyanin-coupled HLA-DR (clone L243; Becton Dickinson), and FITC-conjugated anti-CD80 (clone L307; Becton Dickinson), anti-CD86 (clone FUN-1; BD PharMingen), anti-CD40 (clone 5C3; BD PharMingen), or anti-CD1a (clone HI149; BD PharMingen). CTLA4Ig (100 μg/ml) was added before staining in some experiments as a control for steric inhibition of binding of CD80- and CD86-specific mAbs. The controls for nonspecific Ig binding were mouse IgG1 directly conjugated to FITC (clone MOPC-21; BD PharMingen), PE, or PerCP (clone X40; Becton Dickinson), and IgG2a directly conjugated to allophycocyanin (clone X39; Becton Dickinson). 5,000 DC-gated events (CD3−, DR+, CD45+) were collected for quadruple staining on a FACSCalibur™ flow cytometer (Becton Dickinson), and data were analyzed using CELLQuest™ software (Becton Dickinson). For publication purposes, flow cytometry profiles were scanned, converted to digital files, and imported into Adobe Illustrator® v8.0 for Macintosh (Adobe Systems) where curve-fitting was performed.
Statistical Analysis.
Data were summarized as mean ± SD. Comparisons between experimental groups were performed using the two-sided Wilcoxon signed rank test for paired data. A significance level of 5% was used for all comparisons.
Results
CTLA4Ig Administration Improves Psoriasis Clinical Activity.
Psoriasis patients receiving four infusions with the soluble chimeric protein CTLA4Ig (BMS-188667) displayed a dose-dependent improvement in their global clinical parameters of psoriasis 15. Though meaningful clinical responses were observed in all but the lowest dosing cohort, the most consistent responses were observed in the top two dose groups (CTLA4Ig 25 and 50 mg/kg dose). 9 of 11 patients accrued to these 2 dosing cohorts demonstrated a 50% or greater improvement from baseline psoriasis evaluation. The duration of clinical response was sustained in many cases throughout the 147-d median observation period after the final CTLA4Ig infusion. Clinical quiescence, therefore, often persisted well after the elimination of detectable CTLA4Ig from the circulation. No discernible changes in B7-bearing target cell populations or alterations in lymphocyte subset distributions were observed in the peripheral blood.
To ascertain the mechanisms by which the inhibition of the CD28/CD152 T cell costimulatory pathway moderated the chronic inflammatory cascade and resolved psoriatic lesions, a series of immunohistochemical studies was undertaken. Histologic samples were obtained from prospectively identified lesions prestudy and at study days 8, 16, 36, and 78 after initiation of CTLA4Ig infusions.
Reduction in Intralesional T Lymphocyte Subsets and Neutrophils after Administration of CTLA4Ig.
The number of CD3, CD25, and CD8 positively staining T lymphocytes was serially reduced in specimens obtained after the initial (day 1) infusion with CTLA4Ig in 9 of 11 patients accrued to the 2 top dosing cohorts (Fig. 1A–I). The most rapid reductions were observed within the CD25+ (IL-2 receptor α subunit) T cell subset. Normalization of intralesional T cell counts correlated with meaningful clinical improvement in these patients. T (CD3+) lymphocytes, including the CD8+ and CD25+ subsets, were clustered in the papillary dermis and the suprapapillary epidermis in baseline biopsies. At day 78, the few remaining T lymphocytes were predominantly distributed within the dermis. Progressive epidermal thinning was also evident in these serial biopsies.
Figure 1.
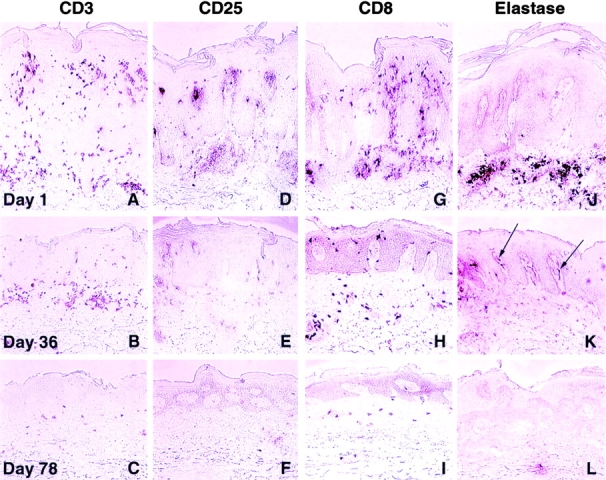
Decrease in psoriatic lesional leukocytic infiltrates after administration of CTLA4Ig. Representative immunohistochemical findings in serial biopsies at day 1 (top), day 36 (middle), and day 78 (bottom) from prospectively identified lesions in 9 of the 11 patients accrued to the CTLA4Ig 25 and 50 mg/kg dose levels. Illustrated in A–L are data from a patient who received CTLA4Ig 25 mg/kg/dose. T cells present within the lesion were detected by immunostaining with mAbs to CD3 (A–C), CD25 (D–F), or CD8 (G–I). Serial reductions in T cell infiltrates were observed after initiation of CTLA4Ig infusions (day 1) and were associated with progressive epidermal thinning. Neutrophilic infiltrates detected by immunostaining with mAbs to elastase (J–L) were also decreased in serial biopsies. At day 36 (K), neutrophils were largely confined to capillaries in the papillary dermis (arrows). Original magnifications: ×200.
Elastase-positive neutrophilic infiltrates were also decreased after the initiation of therapy (Fig. 1J–L). At day 1, neutrophils were detected in the papillary dermis and in the stratum corneum. At subsequent biopsy time points, neutrophils appeared to be concentrated within the papillary capillaries (Fig. 1 K; arrows). In general, the elimination of neutrophils from psoriatic lesions occurred more rapidly than that of T lymphocytes, and this population of cells was no longer present in extravascular spaces on day 78.
Reduction in Intralesional T Lymphocytes Is Associated with Alterations in Keratinocyte Proliferation and the Diminished Expression of Keratinocyte Accessory Molecules.
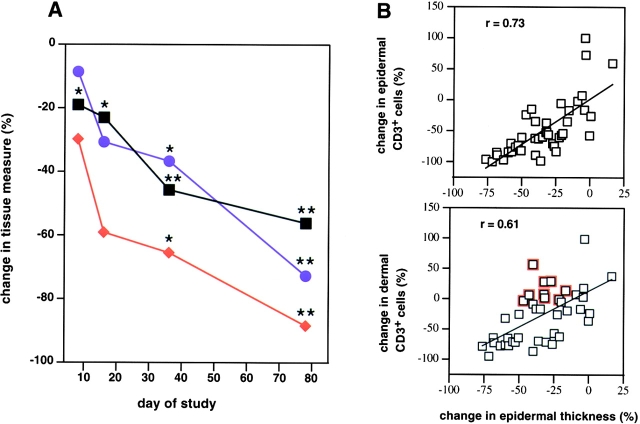
Soluble factors released from activated T lymphocytes are believed to contribute to the hyperproliferation of keratinocytes within psoriatic lesions 6 7. Therefore, intralesional T lymphocyte counts were correlated with epidermal thickness in serial lesional biopsies obtained from patients accrued to the 25 and 50 mg/kg dose groups. Peak reductions in intralesional T lymphocytes were observed at day 78 with an 88% mean decrease in the epidermal compartment (P < 0.001) and a 73% mean reduction in the dermal compartment compared with baseline examination (P < 0.001) (Fig. 2 A). A statistically meaningful decline in epidermal thickness was observed as early as day 8 (19% reduction; P = 0.016); at day 78, a 56% mean percent reduction from baseline epidermal thickness was observed (P < 0.001). The decrease in lesional epidermal T lymphocytes correlated most closely with the observed reductions in epidermal thickness (r = 0.73). Individual patient correlation plots for each of the four biopsies obtained after the day 1 infusion are illustrated in Fig. 2 B.
Figure 2.
Changes in psoriatic lesional T cell numbers and keratinocyte hyperplasia across the 11 patients accrued to the CTLA4Ig 25 and 50 mg/kg dose groups. (A) Mean percent decrease in values for epidermal thickness (black squares) and numbers of T cells within the epidermis (red diamonds) or dermis (blue circles) compared with baseline (day 1) are illustrated over the first 78 d of the study period. Asterisks indicate statistical significance (*P < 0.05; **P < 0.001). P values were based on a two-sided t test for no change at the indicated study day compared with day 1. (B) Correlation plots of the percent change in infiltrating epidermal or dermal T cells versus percent change in epidermal thickness for each patient at all sampling time points. Individual data points are an average derived from triplicate analyses. Epidermal thickness was calculated by quantitating the cross-sectional surface area beneath a 1-mm linear region of a representative histological section using computer-assisted image analysis. Positive correlations between the change in T (CD3+) cell numbers and epidermal thickness were evident (epidermal CD3+: r = 0.73; dermal CD3+: r = 0.61). Discordant responses characterized by reductions in epidermal thickness that were not accompanied by similar reductions in T cell numbers within a specific compartment are indicated (open red squares).
Keratinocytes, upon activation, are able to express a variety of cell surface proteins that are thought to play an important role in amplifying the cutaneous inflammatory reaction in psoriasis, including the clonal expansion of intralesional T cells 19 20 21 22 23 24. Membrane-bound and soluble T cell factors induce these keratinocyte phenotypic alterations 6 19 20. Therefore, a paracrine loop for intralesional T cell activation may be provided by activated lesional keratinocytes. Accordingly, immunohistochemical studies of select keratinocyte accessory molecules were undertaken. At day 1, strong immunostaining with mAbs reactive with CD40 was detected on lesional keratinocytes, particularly in the suprapapillary areas. The CD40 expression on keratinocytes, colocalized with the marked expression of CD54 (intercellular adhesion molecule [ICAM]-1), MHC class II HLA-DR antigens (Fig. 3A, Fig. D, and Fig. G), and the influx of juxtaposed T lymphocytes (Fig. 1A, Fig. D, and Fig. G). At day 78, clinically responding patients displayed weak CD40 reactivity on the basal layer of keratinocytes. CD54 and HLA-DR expression were similarly markedly diminished within the day 78 biopsy specimens (Fig. 3C, Fig. F, and Fig. I). Clinically nonresponding patients did not demonstrate discernible changes in their immunohistology from baseline examination with regard to all serial immunohistochemical studies incorporated in this study.
Figure 3.
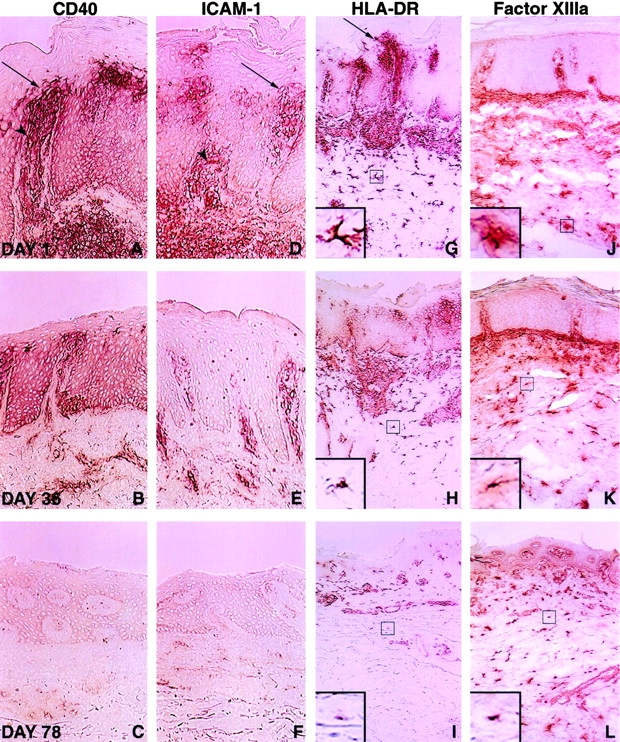
Diminished immunohistochemical detection of CD40, ICAM-1, HLA-DR, and FXIIIa in psoriatic lesional skin after administration of CTLA4Ig. Keratinocytes within the suprapapillary regions (arrows) and DCs displayed marked upregulation of expression of CD40, ICAM-1 (CD54), and HLA-DR at day 1 (top), which serially decreased at study days 36 (middle) and 78 (bottom). Superficial capillaries in untreated psoriatic lesions also exhibited increased ICAM-1 and CD40 staining (arrowheads; A and D), which was reduced at days 36 (B and E) and 78 (C and F). Phenotypic alterations of lesional DCs stained with mAbs to HLA-DR and FXIIIa are observed at days 36 and 78 after the initial infusion (G–L). Higher magnification views of the boxed regions are shown in the inset areas in G–L, and demonstrate a progressive decrease in the number and length of branching processes radiating from the cell body (dendricity) of the stained cells. Results shown are from serial biopsies of a single lesion in a patient administered CTLA4Ig 25 mg/kg and are representative of the nine patients achieving a 50% or greater improvement in clinical scores. Original magnifications: (A–F) ×400; (G–L) ×200; (G–L, insets) ×800.
Presence of Mature DCs in Psoriatic Epidermis and Their Diminished Density after Administration of CTLA4Ig.
Keratinocytes do not express B7 molecules, the ligands for CTLA4Ig 25. Therefore, we studied the effects of CTLA4Ig on epidermal Langerhans cells (LCs) and dermal DCs, the predominant B7-bearing cells within the skin, providing costimulatory signals for intralesional T cell activation 3 5 14 26 27 28 29 30 31. We assessed whether the decreased number of intralesional T lymphocytes observed on study could be partially attributed to a change in the number of skin DCs or their complement of accessory/costimulatory molecules that have an upregulated expression on mature DC.
The density of CD40, CD54, and HLA-DR staining on intralesional DCs decreased progressively in clinically responding patients. Weak residual CD40, CD54, and HLA-DR reactivity was evident on infiltrating dermal mononuclear cells at day 78 (Fig. 3A–I). A diminished number of FXIIIa-staining dermal DCs was also observed in clinical responders 4, particularly in the upper reticular and papillary dermis (Fig. 3J–L).
The morphology of the lesional DC population also changed over time. DCs possessing long, branching processes evident at the time of the initial skin biopsy (Fig. 3G and Fig. J, inset) were replaced with a population of smaller cells that had less numerous, clipped processes (Fig. 3H, Fig. I, Fig. K, and Fig. L, inset). The morphological features of this latter population resembled immature DCs. mAbs to CD1a, expressed on immature but not mature LCs 30 32, displayed serial increases in reactivity predominantly within the suprabasal layer of the epidermis, in the majority of patients accrued to the top two dosing cohorts (Fig. 4A–C). Thus, CD1a positivity originally “displaced” to the upper zone of the thickened epidermis at day 1 was replaced by a more equitable CD1a staining distribution at day 78.
Figure 4.
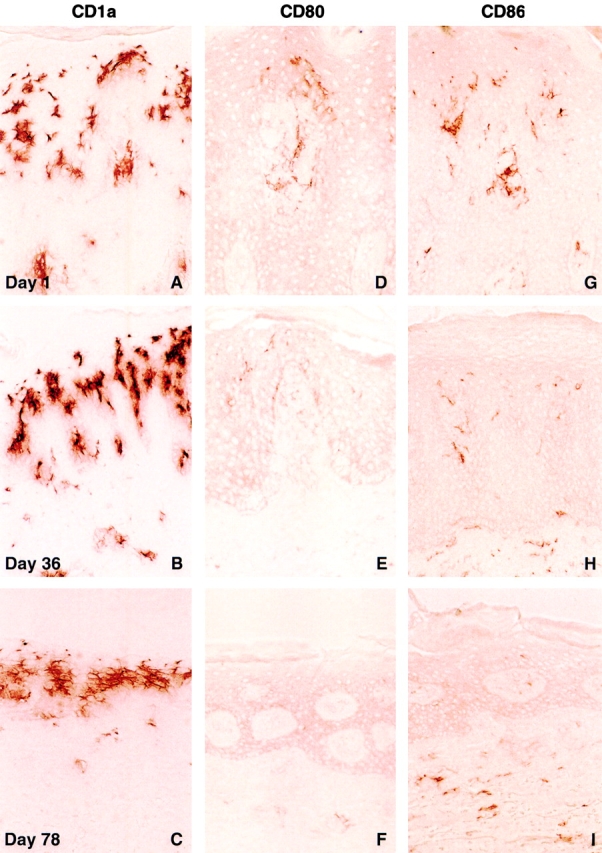
Increased CD1a reactivity and reduced CD80 and CD86 expression in psoriatic lesional biopsies after administration of CTLA4Ig. Serial biopsies were obtained from the perimeter of a single lesion in a patient accrued to the CTLA4Ig 25 mg/kg dose level, and are representative of the histological findings in the clinical responders within the 25 and 50 mg/kg dosing cohorts. An increasing density of CD1a+ epidermal LC staining was observed after initiation of treatment (A–C). Positive CD80 and CD86 immunoreactivity was seen in both the epidermis and dermis at day 1 (D and G). Diminished staining for CD80 and CD86 is seen at day 36 (E and H). At day 78, CD80 expression was no longer detectable within the epidermis, and minimal CD80+ staining was present in the dermis (F). A low level of staining for CD86, predominantly within the dermis, was present at day 78 (I). Original magnifications: ×400.
Conversely, there was a progressive decrease in the intensity of staining with mAbs reactive with CD80 (B7-1) and CD86 (B7-2) in serial biopsies (Fig. 4D–I). Notably, reagents selected for immunohistochemical studies of CD80 and CD86 expression did not bind to the same B7 epitopes as CTLA4Ig (data not shown; reference 33). At day 1, CD80 and CD86 staining was present in both the epidermis and dermis (Fig. 4D and Fig. G). Day 78 biopsy specimens, however, revealed low level staining of DCs only within the dermal compartment, with slightly higher density staining for CD86 than CD80 (Fig. 4F and Fig. I).
To confirm that the diminished density of DC activation markers and the modified DC morphology within lesional biopsies were associated with decreased numbers of mature DCs, immunohistochemical studies of serial lesional biopsies were performed using DC-restricted, maturation-associated markers (CD83, DC-LAMP, and MMR) and the leukocyte integrin CD11c. CD83 and DC-LAMP, expressed at high levels on mature DCs 34 35, displayed markedly increased staining in day 1 lesional biopsy specimens in a pattern similar to CD80 and CD86 expression (Fig. 5A and Fig. C). Little residual staining was evident at day 78 in clinical responders (Fig. 5B and Fig. D). In contrast, unaffected skin from psoriatic patients showed no significant staining in the epidermis or dermis with these antibodies (data not shown). CD11c, expressed at high levels on some DCs 36, demonstrated increased staining on DCs in both the dermis and epidermis at day 1 (Fig. 5 E). Marked reductions in CD11c immunoreactivity were observed at day 78 in clinical responders, with residual staining exclusively in the dermal compartment (Fig. 5 F). A pattern of CD11c immunoreactivity similar to the day 78 biopsy findings was observed in nonlesional biopsies from psoriatic patients (data not shown). MMR, a carbohydrate-recognizing receptor important in the uptake and delivery of antigens to MHC class II compartments for antigen processing and presentation, is expressed on immature DCs. MMR expression on DCs is downregulated in vitro by inflammatory stimuli 37. Immunohistochemical studies in human tissue have not detected expression of MMR on LCs 38. Day 1 and 78 biopsy specimens demonstrated MMR staining within the dermis with little apparent alteration in the density of staining (Fig. 5G and Fig. H). Therefore, psoriatic epidermis, and to a lesser extent psoriatic dermis, were heavily infiltrated with activated/mature DCs. Lesional biopsies at day 78 possessed a lower density of the requisite accessory/costimulatory molecules for T cell activation compared with pretreatment biopsies. These findings may also be contributory to the observed reduction in lesional T cells.
Figure 5.
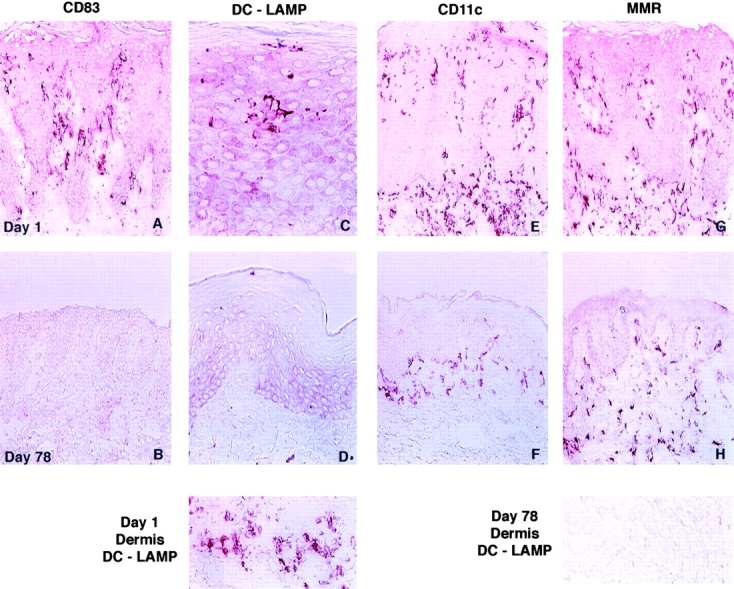
Alterations in immunohistochemical markers of DC maturational status in psoriatic lesional biopsies after CTLA4Ig administration. Biopsy material obtained at day 1 (top) and day 78 (bottom) from a patient accrued to the CTLA4Ig 25 mg/kg dose level are illustrated and were reflective of the changes observed in all clinical responders. The DC-restricted markers CD83 and DC-LAMP, expressed at high levels on mature DCs, demonstrated an increased magnitude of staining in both the epidermis and dermis at day 1 (A, C, and Day 1 Dermis DC-LAMP). Marked reductions in the density of staining for both CD83 and DC-LAMP were observed; little residual staining was evident at day 78 (B, D, and Day 78 Dermis DC-LAMP). Immunoreactivity for the leukocyte integrin CD11c, abundantly expressed on DCs, was also decreased within the epidermal and dermal compartments at day 78 compared with day 1 (E and F). Residual CD11c reactivity was restricted to the dermis at day 78. The population of immature DCs characterized by MMR positivity was distributed exclusively within the dermal compartment and displayed small decreases in immunoreactivity at day 78 compared with baseline examination (G and H). Original magnifications: (A, B, E–H) ×400; (C and D) ×800.
Addition of CTLA4Ig to Normal and Psoriatic Skin Explant Cultures Does Not Affect DC Maturation, Migration, or Survival.
The diminished incidence of mature DCs in serial biopsies could be attributed to either (a) direct toxicity of CTLA4Ig for DCs or (b) indirect effects of CTLA4Ig on DCs resulting from impaired T lymphocyte activation, with secondary effects on DC maturation, survival, and/or recruitment 39 40 41. To distinguish between these possibilities, we performed several experiments in which the migration/maturation in vitro of DCs from split thickness skin was conducted in the presence or absence of saturating concentrations of CTLA4Ig 10 42 43 44 45 46 47 48 49.
The harvest of DCs from explant cultures was unchanged in the presence of CTLA4Ig. The mean recovery of emigrating cells across the seven paired experiments (lesional and normal skin) was 1.37 × 105 in the presence of CTLA4Ig or 1.41 × 105 in control cultures (P = 0.447). Approximately 60% of exfiltrators were CD3−HLA-DR+ CD45+ regardless of the culture conditions (control mean percent: 58.2 ± 13.2; CTLA4Ig mean percent: 62.8 ± 18.4; P = 0.375). Cell viability in all experiments was >90% as assessed by trypan blue exclusion. Therefore, the presence of CTLA4Ig in the culture medium had no major effect on the migration or survival of DCs from skin explants after 3 d of incubation.
Flow cytometric analyses of DCs emigrating either under control conditions or in the presence of CTLA4Ig failed to demonstrate significant differences in the levels of CD86, CD80, CD40, or CD1a expression (Table , and Fig. 6). The proportion of CD3−HLA-DR+CD45+ cells expressing each of these cell surface proteins was equivalent across the paired experimental conditions. Further fractionation of the CD3−CD45+ emigrating cell population into high and medium HLA-DR expression indicated that the CD80/CD86 mean fluorescence intensities (MFIs) correlated with the level of HLA-DR expression after culture of skin explants but not with the presence of CTLA4Ig during culture (data not shown). DCs, upon activation or emigration from explants, are known to increase in size 43. Forward scatter flow cytometric analyses failed to discern a difference in the mean cell size when comparisons were made between those explants cultured in the presence of CTLA4Ig versus media alone (data not shown). These data indicated that the maturation of DCs and their acquisition of costimulatory/accessory molecules were not inhibited by the presence of CTLA4Ig in short term culture. The diminished density of DC activation/maturation markers and the modified DC morphology observed on study are therefore likely attributable to an altered lesional milieu resulting from decreased T cell activation, with secondary effects on DC extravasation into skin and/or DC maturation.
Table 1.
Characterization of Skin DC Exfiltrators after 3 d of Culture
| −CTLA4Ig | +CTLA4Ig | ||||
|---|---|---|---|---|---|
| Skin explant source | Cell surface protein | Mean percentage of CD3−HLA-DR+ CD45+ cells ± SD | MFI ± SD | Mean percentage of CD3−HLA-DR+ CD45+ cells ± SD | MFI ± SD |
| Normal | CD86 | 93.7 ± 2.6 | 109.2 ± 54.4 | 91.1 ± 8.0 | 92.0 ± 38.6 |
| (n = 4) | CD80 | 60.7 ± 11.8 | 35.6 ± 21.8 | 44.0 ± 15.1 | 22.4 ± 10.9 |
| CD40 | 72.0 ± 7.6 | 33.3 ± 16.0 | 71.2 ± 10.8 | 31.6 ± 14.2 | |
| CD1a | 38.2 ± 13.8 | 40.3 ± 24.1 | 35.8 ± 18.3 | 33.0 ± 18.8 | |
| Psoriatic lesion | CD86 | 75.9 ± 10.2 | 135.8 ± 51.6 | 83.2 ± 11.9 | 158.0 ± 61.0 |
| (n = 3) | CD80 | 33.0 ± 2.2 | 61.4 ± 38.4 | 38.6 ± 18.9 | 57.2 ± 63.3 |
| CD40 | 53.6 ± 9.7 | 54.0 ± 4.2 | 62.8 ± 10.2 | 53.6 ± 35.2 | |
| CD1a | 43.3 ± 3.3 | 75.2 ± 40.0 | 34.6 ± 21.6 | 52.1 ± 40.9 | |
Skin explants were cultured for 3 d either in media alone or with CTLA4Ig (100 μg/ml). Exfiltrators were quadruple-stained with mAbs reactive against CD3, HLA-DR, CD45, and a panel of mAbs to CD86, CD80, CD40, or CD1a. Values represent mean ± SD from independent experiments of either the percentage of the total DC population possessing a given cell surface protein or the MFI for each of these proteins (n = 4 normal skin; n = 3 psoriatic lesional skin). No significant difference between paired data sets was apparent.
Figure 6.
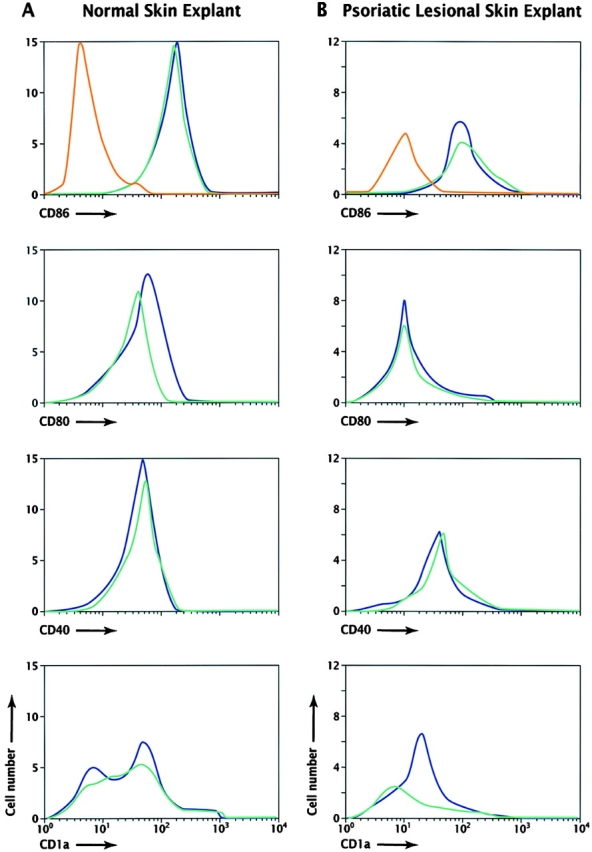
Four-color flow cytometry analysis of emigrating leukocytes from normal and psoriatic lesional skin explants. DCs (CD3−DR+CD45+) emigrating from normal skin (A) or psoriatic lesional skin explants (B) after 3 d of culture were analyzed for expression of CD80, CD86, CD40, or CD1a, and the results were plotted as histograms of cell number versus log10 fluorescence. Isotype controls for the DC population gates (CD3−DR+CD45+) used in the two series of analyses are illustrated in A and B (red lines). Cultures were performed with media alone (blue lines) or in the presence of CTLA4Ig (100 μg/ml; green lines). The data in A are representative of four independent experiments with normal skin explants; data in B are comparable to three independent experiments with psoriatic lesional skin. The results from all experiments are summarized in Table .
T Cell Costimulatory Blockade Reduces Lesional Vascular Endothelial Selectin and Integrin Expression.
Due to the inability of the skin explant model to examine the possible effects of CTLA4Ig administration on dermal vasculature and leukocyte recruitment, immunostaining with antibodies reactive to laminin (present in the basement membrane of blood vessels), selectins, and integrins was undertaken. E- and P-selectin (CD62E, CD62P) mediate the initial tethering to, and rolling on, endothelium by neutrophils, T cells, and DCs. These selectins are key contributors to the recruitment of skin-homing T cells and DCs 40 41 50 51. Their expression is upregulated after endothelial exposure to a variety of inflammatory cytokines, including TNF-α, IFN-γ, IL-1β, and IL-4 8 9. CD54 mediates the firm adhesion of tethered leukocytes to endothelium, and its expression is upregulated by many of the same cytokines 52 53.
Lesional vessels displayed progressively reduced ectasia and tortuosity after day 1 infusions with CTLA4Ig (Fig. 7A–C). At day 1, CD62E and CD62P were strongly expressed in the superficial and deep vascular plexus. Expression of both selectins was diminished in serial biopsy specimens (Fig. 7D–I). Though modest decreases in CD62P immunoreactivity were selectively observed in the superficial capillaries at days 36 and 78, these changes were detected consistently across all clinically responding patients. CD54 also displayed progressively decreased vascular immunoreactivity (Fig. 3D–F). Downmodulation of these vascular adhesion molecules likely impaired the ability of endothelial cells to activate and recruit circulating leukocytes to the psoriatic lesions, as evidenced by the diminished inflammatory infiltrate present at day 78. Human endothelial cells do not express CD80 or CD86 54. Thus, it appears that the observed immunohistochemical changes were indirectly mediated through the binding of CTLA4Ig to other local cellular B7-bearing targets.
Figure 7.
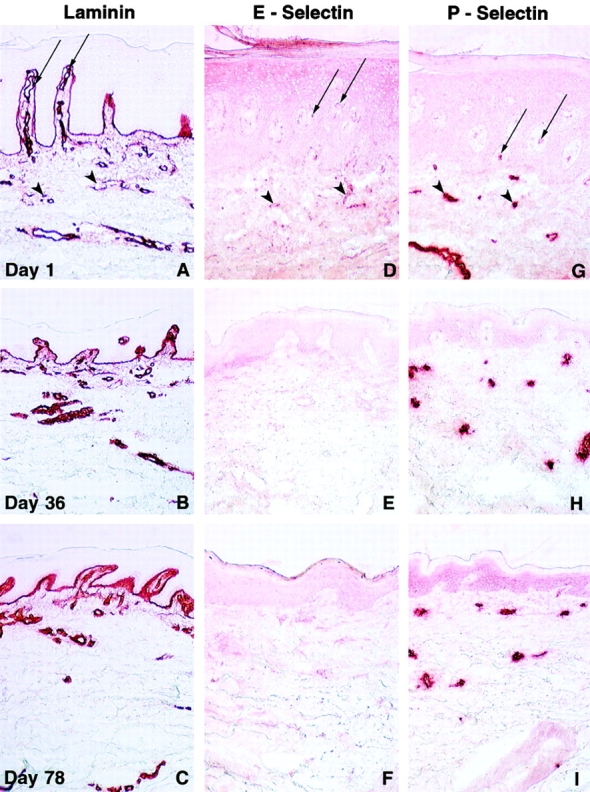
Alterations in psoriatic lesional vascular immunohistology in serial cryostat sections after administration of CTLA4Ig. Illustrated in A–I are representative immunohistochemical findings in biopsies obtained at day 1 (top), day 36 (middle), and day 78 (bottom) from the perimeter of a single lesion in clinically responding subjects accrued to the CTLA4Ig 25 and 50 mg/kg dose levels; data shown are from one subject accrued to the 50 mg/kg dose level. Immunostaining with mAbs to laminin (A–C) present in the basement membrane of blood vessels (and epidermis) illustrates the progressive decrease in lesional vascular ectasia and tortuosity after CTLA4Ig administration. Increased E-selectin (CD62E) and P-selectin (CD62P) reactivity was noted in superficial (arrows) and deep dermal vessels (arrowheads) within chronically inflamed psoriatic skin at day 1 (D and G). E-selectin staining decreases serially in both the superficial and deep dermal vascular beds after administration of CTLA4Ig (E and F). Expression of P-selectin is selectively decreased in expression in the superficial capillaries at days 36 and 78. Original magnifications: ×200.
Discussion
Our findings support the hypothesis that T cell activation is a critical and proximal event in the complex chronic inflammatory cascade that culminates in the generation of psoriatic plaques. To our knowledge, this is the first report documenting the accumulation of mature DCs in a human autoimmune target organ, the skin. Additionally, the resolution of activated phenotypes on lesional keratinocytes, DCs, and vascular endothelium after T cell costimulatory blockade has not previously been reported. Thus, these data expand upon our prior observations 15 and provide possible mechanisms for the observed durable reduction in intralesional T cells after administration of CTLA4Ig. We have demonstrated that a reduced capacity for lesional T cell clonal expansion (due to reversion of the lesional skin APC population to a less mature/immunocompetent state) and decreased vascular recruitment may both have been contributory factors. These observations suggest that clinical activity in this chronic autoimmune disease is dependent on the continued activation of T cells.
Many models have been proposed to explain the interplay of keratinocytes with lesional T cells in the immunopathogenesis of psoriasis. The upregulated expression of MHC class II HLA-DR antigens on activated keratinocytes could be regarded as an in vivo bioassay for the presence of IFN-γ released from activated T cells 6 22. The adequacy of keratinocytes to provide T cell activation signals, however, has been a matter of some debate 55 56. The clinical resolution of psoriatic lesions after the specific disruption of B7 engagement with CD28/CD152 on T cells has demonstrated that keratinocytes, which do not express detectable B7, are not sufficient for perpetuating T cell activation in psoriatic lesions.
Though the role of CD28/CD152 costimulation in T cell activation and cytokine production is well understood 11 12 13, comparatively little is known regarding the possible direct effects of this pathway on DC activation. In this study, we have demonstrated that the presence of saturating CTLA4Ig concentrations in short term skin explant cultures did not impair DC maturation, migration, or survival. These ex vivo data contrast with the immunohistochemical observations in serial biopsies harvested over a 78-d period after systemic administration of CTLA4Ig. The clinical samples displayed a progressively less mature complement of lesional DCs. The apparently conflicting observations suggest that CTLA4Ig did not block DC activation through direct inhibition of the B7 axis on DCs. Instead, these data support a model in which the primary blockade of T cell costimulatory signals leads to progressive diminution in the number, activation status, and cytokine elaboration of lesional T cells, with secondary effects on keratinocyte activation and cytokine release. A gradual transformation of the skin microenvironment ensued. Under this scenario, immature DCs recruited to lesional skin after initiation of CTLA4Ig infusions presumably could no longer acquire sufficient stimuli required to be optimally activated, further compromising T cell activation signals. This model is in accord with the view of DCs as the consummate immunologic “rheostats,” integrating a variety of activating signals, which ultimately “tune” the capacity to stimulate T cells 29. The cadence of the clinical response in this study was slower than that observed with antipsoriatic agents that directly inhibit T cell activation and may be explained by these observations 15 57 58.
Our immunohistochemical data indicate that T cell costimulatory signals delivered through B7/CD28 are pivotal in creating a microenvironment, which facilitates the activation of tissue DCs and the perpetuation of autoimmune inflammation. However, these B7/CD28 signals in isolation are not likely sufficient to initiate a chronic inflammatory cascade. The key requirements in constructing a microenvironment conducive to T cell–mediated, organ-specific autoimmune inflammation have been characterized in transgenic models of pancreatic islet β cell inflammation. Autoimmune inflammation of the pancreas in these animals requires not only ectopic and/or increased expression of CD80 but also either a genetically predisposed host 59, increased levels of autoantigen and autoreactive T cells 60, elevated MHC class II antigens 61, or enhanced levels of an inflammatory cytokine 62. Mice expressing any of these transgenes alone maintain normal pancreatic islet architecture 60 63 64. A repertoire of inflammatory stimuli analogous to this array of transgenes was identified within the day 1 psoriatic lesional tissue. At day 78, the intrinsic T cell activating capacity was likely vastly different, with lesional mononuclear cells possessing fewer of the requisite class II antigens, accessory and costimulatory molecules for T cell activation. The downregulation via B7-CD28/CTLA-4 blockade of a variety of proinflammatory stimuli required for disruption of immunologic homeostasis may explain the profound and durable clinical response, well beyond the active treatment period, when contrasted with other T cell–based therapies 15 17. Additional explanations for the durable response could include the possible induction of tolerance or the emergence of regulatory T cells. Currently, no data exist in support of either of these possible explanations.
In addition to the functional modulation of the T cell activating capacity of lesional APCs, the alteration in endothelial expression of CD54, E-selectin, and P-selectin likely impaired the ability to recruit neutrophils, lymphocytes, and DCs to inflamed skin 40 41 50 51. These selectins and integrins have been demonstrated to be important in the large scale influx of mononuclear cells to a variety of other inflamed tissues including synovium and respiratory tract 51 65. Their presence on vascular endothelium at heightened levels is important in the reseeding of peripheral tissues with DCs to replenish the pool of tissue DCs lost through DC migration–invoking stimuli 46 48. Thus, vascular recruitment of effector cells, an additional common pathway important in sustaining chronic inflammation, is also potentially impacted by B7/CD28 costimulatory blockade.
In summary, we have shown that CTLA4Ig blockade of the B7-CD28/CD152 pathway of T cell costimulation in patients with psoriasis modulates the state of chronic inflammatory activation of T cells, DCs, and endothelial cells in psoriatic plaques. These observations could have important implications for the treatment of other acute or chronic T cell–mediated diseases, where these effector cells may play an important role in the pathogenesis of the disease.
Acknowledgments
The authors wish to thank Drs. Ralph Steinman (The Rockefeller University, New York, NY) and Hans Ochs (University of Washington, Seattle, WA) for critical discussions during the preparation of the manuscript, and Robert Bruschini (Bristol-Myers Squibb, Princeton, NJ) for help in preparing the figures.
Footnotes
Abbreviations used in this paper: CTLA-4, cytotoxic T lymphocyte–associated antigen 4; CTLA4Ig, CTLA-4–immunoglobulin; DC, dendritic cell; DC-LAMP, DC–lysosomal-associated membrane glycoprotein; ICAM, intercellular adhesion molecule; LC, Langerhans cell; MFI, mean fluorescence intensity; MMR, macrophage mannose receptor; PerCP, peridinin chlorophyll protein.
Portions of this work have been published in abstract form (1997. J. Invest. Dermatol. 108:555).
References
- Paukkonen K., Naukkarinen A., Horsmanheimo M. The development of manifest psoriatic lesions is linked with the invasion of CD8+ T cells and CD11c+ macrophages into the epidermis. Arch. Dermatol. Res. 1992;284:375–379. doi: 10.1007/BF00372065. [DOI] [PubMed] [Google Scholar]
- Demiden A., Taylor J.R., Grammer S.F., Streilein J.W. T-lymphocyte-activating properties of epidermal antigen-presenting cells from normal and psoriatic skinevidence that psoriatic epidermal antigen-presenting cells resemble cultured normal Langerhans cells. J. Invest. Dermatol. 1991;97:454–460. doi: 10.1111/1523-1747.ep12481465. [DOI] [PubMed] [Google Scholar]
- Banchereau J., Steinman R.M. Dendritic cells and the control of immunity. Nature. 1998;392:245–252. doi: 10.1038/32588. [DOI] [PubMed] [Google Scholar]
- Cerio R., Griffiths C.E.M., Cooper K.D., Nickoloff B.J., Headington J.T. Characterization of factor XIIIa positive dermal dendritic cells in normal and inflamed skin. Br. J. Dermatol. 1989;121:421–431. doi: 10.1111/j.1365-2133.1989.tb15509.x. [DOI] [PubMed] [Google Scholar]
- Nestle F.O., Turka L.A., Nickoloff B.J. Characterization of dermal dendritic cells in psoriasisautostimulation of T lymphocytes and induction of Th1 type cytokines. J. Clin. Invest. 1994;94:202–209. doi: 10.1172/JCI117308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baadsgaard O., Tong P., Elder J.T., Hansen E.R., Ho V., Hammerberg C., Lange-Vejlsgaard G., Fox D.A., Fisher G., Chan L.S. UM4D4+ (CDw60) T cells are compartmentalized into psoriatic skin and release lymphokines that induce a keratinocyte phenotype expressed in psoriatic lesions. J. Invest. Dermatol. 1990;95:275–282. doi: 10.1111/1523-1747.ep12484908. [DOI] [PubMed] [Google Scholar]
- Wrone-Smith T., Nickoloff B.J. Dermal injection of immunocytes induces psoriasis. J. Clin. Invest. 1996;98:1878–1887. doi: 10.1172/JCI118989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bevilacqua M.P., Stengelin S., Gimbrone M.A., Jr., Seed B. Endothelial leukocyte adhesion molecule 1an inducible receptor for neutrophils related to complement regulatory proteins and lectins. Science. 1989;243:1160–1164. doi: 10.1126/science.2466335. [DOI] [PubMed] [Google Scholar]
- Yao L., Pan J., Setiadi H., Patel K.D., McEver R.P. Interleukin 4 or oncostatin M induces a prolonged increase in P-selectin mRNA and protein in human endothelial cells. J. Exp. Med. 1996;184:81–92. doi: 10.1084/jem.184.1.81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Linsley P.S., Brady W., Urnes M., Grosmaire L.S., Damle N.K., Ledbetter J.A. CTLA-4 is a second receptor for the B cell activation antigen B7. J. Exp. Med. 1991;174:561–569. doi: 10.1084/jem.174.3.561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Linsley P.S., Ledbetter J.A. The role of CD28 receptor during T cell response to antigen. Annu. Rev. Immunol. 1993;11:191–212. doi: 10.1146/annurev.iy.11.040193.001203. [DOI] [PubMed] [Google Scholar]
- Viola A., Schroeder S., Sakakibara Y., Lanzavecchia A. T lymphocyte costimulation mediated by reorganization of membrane microdomains. Science. 1999;283:680–682. doi: 10.1126/science.283.5402.680. [DOI] [PubMed] [Google Scholar]
- Sayegh M.H., Turka L.A. The role of T-cell costimulatory activation pathways in transplant rejection. N. Engl. J. Med. 1998;338:1813–1821. doi: 10.1056/NEJM199806183382506. [DOI] [PubMed] [Google Scholar]
- Symington F.W., Brady W., Linsley P.S. Expression and function of B7 on human epidermal Langerhans cells. J. Immunol. 1993;150:1286–1295. [PubMed] [Google Scholar]
- Abrams J.R., Lebwohl M.G., Guzzo C.A., Jegasothy B.V., Goldfarb M.T., Goffe B.S., Menter A., Lowe N.J., Krueger G., Brown M.J. CTLA4Ig-mediated blockade of T-cell costimulation in patients with psoriasis vulgaris. J. Clin. Invest. 1999;103:1243–1252. doi: 10.1172/JCI5857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwartz R.S. The new immunology—the end of immunosuppressive drug therapy? N. Engl. J. Med. 1999;340:1754–1756. doi: 10.1056/NEJM199906033402209. [DOI] [PubMed] [Google Scholar]
- Vallat V.P., Gilleaudeau P., Battat L., Wolfe J., Nabeya R., Heftler N., Hodak E., Gottlieb A.B., Krueger J.G. PUVA bath therapy strongly suppresses immunological and epidermal activation in psoriasisa possible cellular basis for remittive therapy. J. Exp. Med. 1994;180:283–296. doi: 10.1084/jem.180.1.283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pope M., Betjes M.G.H., Hirmand H., Hoffman L., Steinman R.M. Both dendritic cells and memory T lymphocytes emigrate from organ cultures of human skin and form distinctive dendritic-T-cell conjugates. J. Invest. Dermatol. 1995;104:11–17. doi: 10.1111/1523-1747.ep12613452. [DOI] [PubMed] [Google Scholar]
- Denfeld R.W., Hollenbaugh D., Fehrenbach A., Weiss J.M., von Leoprechting A., Mai B., Voith U., Schöpf E., Aruffo A., Simon J.C. CD40 is functionally expressed on human keratinocytes. Eur. J. Immunol. 1996;26:2329–2334. doi: 10.1002/eji.1830261009. [DOI] [PubMed] [Google Scholar]
- Gaspari A.A., Sempowski G.D., Chess P., Gish J., Phipps R.P. Human epidermal keratinocytes are induced to secrete interleukin-6 and co-stimulate T lymphocyte proliferation by a CD40-dependent mechanism. Eur. J. Immunol. 1996;26:1371–1377. doi: 10.1002/eji.1830260629. [DOI] [PubMed] [Google Scholar]
- Singer K.H., Tuck D.T., Sampson H.A., Hall R.P. Epidermal keratinocytes express the intercellular adhesion molecule-1 in inflammatory dermatoses. J. Invest. Dermatol. 1989;92:746–750. doi: 10.1111/1523-1747.ep12722441. [DOI] [PubMed] [Google Scholar]
- Auböck J., Romani N., Grubauer G., Fritsch P. HLA-DR expression on keratinocytes is a common feature of diseased skin. Br. J. Dermatol. 1986;114:465–472. doi: 10.1111/j.1365-2133.1986.tb02851.x. [DOI] [PubMed] [Google Scholar]
- Dunn D., Gadenne A.S., Simha S., Lerner E.A., Bigby M., Bleicher P.A. T cell receptor Vβ expression in normal human skin. Proc. Natl. Acad. Sci. USA. 1993;90:1267–1271. doi: 10.1073/pnas.90.4.1267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang J.C.C., Smith L.R., Froning K.J., Schwabe B.J., Laxer J.A., Caralli L.L., Kurland H.H., Karasek M.A., Wilkinson D.I., Carlo D.J., Brostoff S.W. CD8+ T cells in psoriatic lesions preferentially use T-cell receptor Vβ3 and/or Vβ13.1 genes. Proc. Natl. Acad. Sci. USA. 1994;91:9282–9286. doi: 10.1073/pnas.91.20.9282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Freeman G.J., Cardoso A.A., Boussiotis V.A., Anumanthan A., Groves R.W., Kupper T.S., Clark E.A., Nadler L.M. The BB1 monoclonal antibody recognizes both cell surface CD74 (MHC class II-associated invariant chain) as well as B7-1 (CD80), resolving the question regarding a third CD28/CTLA-4 counterreceptor. J. Immunol. 1998;161:2708–2715. [PubMed] [Google Scholar]
- Yokozeki H., Katayama I., Ohki O., Matsunaga T., Watanabe K., Satoh T., Azuma M., Okumura K., Nishioka K. Functional CD86 (B7-2/B70) on cultured human Langerhans cells. J. Invest. Dermatol. 1996;106:147–153. doi: 10.1111/1523-1747.ep12329735. [DOI] [PubMed] [Google Scholar]
- Nickoloff B.J., Nestle F.O., Zheng X.-G., Turka L.A. T lymphocytes in skin lesions of psoriasis and mycosis fungoides express B7-1a ligand for CD28. Blood. 1994;83:2580–2586. [PubMed] [Google Scholar]
- Caux C., Vanbervliet B., Massacrier C., Azuma M., Okumura K., Lanier L.L., Banchereau J. B70/B7-2 is identical to CD86 and is the major functional ligand for CD28 expressed on human dendritic cells. J. Exp. Med. 1994;180:1841–1847. doi: 10.1084/jem.180.5.1841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lanzavecchia A. Licence to kill. Nature. 1998;393:413–414. doi: 10.1038/30845. [DOI] [PubMed] [Google Scholar]
- Caux C., Massacrier C., Vanbervliet B., Dubois B., Van Kooten C., Durand I., Banchereau J. Activation of human dendritic cells through CD40 cross-linking. J. Exp. Med. 1994;180:1263–1272. doi: 10.1084/jem.180.4.1263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ridge J.P., Di Rosa F., Matzinger P. A conditioned dendritic cell bridge between a CD4+ T-helper cell and a T-killer cell. Nature. 1998;393:474–478. doi: 10.1038/30989. [DOI] [PubMed] [Google Scholar]
- Romani N., Lenz A., Glassel H., Stössel H., Stanzl U., Majdic O., Fritsch P., Schuler G. Cultured human Langerhans cells resemble lymphoid dendritic cells in phenotype and function. J. Invest. Dermatol. 1989;93:600–609. doi: 10.1111/1523-1747.ep12319727. [DOI] [PubMed] [Google Scholar]
- Engel P., Gribben J.G., Freeman G.J., Zhou L.-J., Nozawa Y., Abe M., Nadler L.M., Wakasa H., Tedder T.F. The B7-2 (B70) costimulatory molecule expressed by monocytes and activated B lymphocytes is the CD86 differentiation antigen. Blood. 1994;84:1402–1407. [PubMed] [Google Scholar]
- Zhou L.-J., Tedder T.F. Human blood dendritic cells selectively express CD83, a member of the immunoglobulin superfamily. J. Immunol. 1995;154:3821–3835. [PubMed] [Google Scholar]
- de Saint-Vis B., Vincent J., Vandenabeele S., Vanbervliet B., Pin J.-J., Aït-Yahia S., Patel S., Mattei M.-G., Banchereau J., Zurawski S. A novel lysosome-associated membrane glycoprotein, DC-LAMP, induced upon DC maturation, is transiently expressed in MHC class II compartment. Immunity. 1998;9:325–336. doi: 10.1016/s1074-7613(00)80615-9. [DOI] [PubMed] [Google Scholar]
- Metlay J.P., Witmer-Pack M.D., Agger R., Crowley M.T., Lawless D., Steinman R.M. The distinct leukocyte integrins of mouse spleen dendritic cells as identified with new hamster monoclonal antibodies. J. Exp. Med. 1990;171:1753–1771. doi: 10.1084/jem.171.5.1753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sallusto F., Cella M., Danieli C., Lanzavecchia A. Dendritic cells use macropinocytosis and the mannose receptor to concentrate macromolecules in the major histocompatibility complex class II compartmentdownregulation by cytokines and bacterial products. J. Exp. Med. 1995;182:389–400. doi: 10.1084/jem.182.2.389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uccini S., Sirianni M.C., Vincenzi L., Topino S., Stoppacciaro A., Lesnoni La Parola I., Capuano M., Masini C., Cerimele D., Cella M. Kaposi's sarcoma cells express the macrophage-associated antigen mannose receptor and develop in peripheral blood cultures of Kaposi's sarcoma patients. Am. J. Pathol. 1997;150:929–938. [PMC free article] [PubMed] [Google Scholar]
- Rissoan M.-C., Soumelis V., Kadowaki N., Grouard G., Briere F., de Waal Malefyt R., Liu Y.-J. Reciprocal control of T helper cell and dendritic cell differentiation. Science. 1999;283:1183–1186. doi: 10.1126/science.283.5405.1183. [DOI] [PubMed] [Google Scholar]
- Robert C., Fuhlbrigge R.C., Kieffer J.D., Ayehunie S., Hynes R.O., Cheng G., Grabbe S., von Andrian U.H., Kupper T.S. Interaction of dendritic cells with skin endotheliuma new perspective on immunosurveillance. J. Exp. Med. 1999;189:627–635. doi: 10.1084/jem.189.4.627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strunk D., Egger C., Leitner G., Hanau D., Stingl G. A skin homing molecule defines the Langerhans cell progenitor in human peripheral blood. J. Exp. Med. 1997;185:1131–1136. doi: 10.1084/jem.185.6.1131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lukas M., Stössel H., Hefel L., Imamura S., Fritsch P., Sepp N.T., Schuler G., Romani N. Human cutaneous dendritic cells migrate through dermal lymphatic vessels in a skin organ culture model. J. Invest. Dermatol. 1996;106:1293–1299. doi: 10.1111/1523-1747.ep12349010. [DOI] [PubMed] [Google Scholar]
- Larsen C.P., Steinman R.M., Witmer-Pack M., Hankins D.F., Morris P.J., Austyn J.M. Migration and maturation of Langerhans cells in skin transplants and explants. J. Exp. Med. 1990;172:1483–1493. doi: 10.1084/jem.172.5.1483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinman R., Hoffman L., Pope M. Maturation and migration of cutaneous dendritic cells. J. Invest. Dermatol. 1995;105:2S–7S. doi: 10.1111/1523-1747.ep12315162. [DOI] [PubMed] [Google Scholar]
- Larsen C.P., Ritchie S.C., Hendrix R., Linsley P.S., Hathcock K.S., Hodes R.J., Lowry R.P., Pearson T.C. Regulation of immunostimulatory function and costimulatory molecule (B7-1 and B7- 2) expression on murine dendritic cells. J. Immunol. 1994;152:5208–5219. [PubMed] [Google Scholar]
- Rambukkana A., Pistoor F.H.M., Bos J.D., Kapsenberg M.L., Das P.K. Effects of contact allergens on human Langerhans cells in skin organ culturemigration, modulation of cell surface molecules, and early expression of interleukin-1β protein. Lab. Invest. 1996;74:422–436. [PubMed] [Google Scholar]
- Randolph G.J., Beaulieu S., Pope M., Sugawara I., Hoffman L., Steinman R.M., Muller W.A. A physiologic function for p-glycoprotein (MDR-1) during the migration of dendritic cells from skin via afferent lymphatic vessels. Proc. Natl. Acad. Sci. USA. 1998;95:6924–6929. doi: 10.1073/pnas.95.12.6924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roake J.A., Rao A.S., Morris P.J., Larsen C.P., Hankins D.F., Austyn J.M. Dendritic cell loss from nonlymphoid tissues after systemic administration of lipopolysaccharide, tumor necrosis factor, and interleukin 1. J. Exp. Med. 1995;181:2237–2247. doi: 10.1084/jem.181.6.2237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Price A.A., Cumberbatch M., Kimber I., Ager A. α6 integrins are required for Langerhans cell migration from the epidermis. J. Exp. Med. 1997;186:1725–1735. doi: 10.1084/jem.186.10.1725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Picker L.J., Kishimoto T.K., Smith C.W., Warnock R.A., Butcher E.C. ELAM-1 is an adhesion molecule for skin-homing T cells. Nature. 1991;349:796–799. doi: 10.1038/349796a0. [DOI] [PubMed] [Google Scholar]
- Austrup F., Vestweber D., Borges E., Löhning M., Bräuer R., Herz U., Renz H., Hallmann R., Scheffold A., Radbruch A., Hamann A. P- and E-selectin mediate recruitment of T-helper-1 but not T-helper-2 cells into inflamed tissues. Nature. 1997;385:81–83. doi: 10.1038/385081a0. [DOI] [PubMed] [Google Scholar]
- Dustin M.L., Rothlein R., Bhan A.K., Dinarello C.A., Springer T.A. Induction of IL-1 and interferon-γtissue distribution, biochemistry, and function of a natural adherence molecule (ICAM-1) J. Immunol. 1986;137:245–254. [PubMed] [Google Scholar]
- Smith C.W., Rothlein R., Hughes B.J., Mariscalco M.M., Rudloff H.E., Schmalsteig F.C., Anderson D.C. Recognition of an endothelial determinant for CD18-dependent human neutrophil adherence and transendothelial migration. J. Clin. Invest. 1988;82:1746–1756. doi: 10.1172/JCI113788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marelli-Berg F.M., Hargreaves R.E.G., Carmichael P., Dorling A., Lombardi G., Lechler R.I. Major histocompatibility complex class II–expressing endothelial cells induce allospecific nonresponsiveness in naive T cells. J. Exp. Med. 1996;183:1603–1612. doi: 10.1084/jem.183.4.1603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bos J.D., de Rie M.A. The pathogenesis of psoriasisimmunological facts and speculations. Immunol. Today. 1999;20:40–46. doi: 10.1016/s0167-5699(98)01381-4. [DOI] [PubMed] [Google Scholar]
- Nickoloff B.J., Turka L.A. Keratinocyteskey immunocytes of the integument. Am. J. Pathol. 1993;143:325–331. [PMC free article] [PubMed] [Google Scholar]
- Ellis C.N., Fradin M.S., Messana J.M., Brown M.D., Siegel M.T., Hartley A.H., Rocher L.L., Wheeler S., Hamilton T.A., Parish T.G. Cyclosporine for plaque-type psoriasisresults of a multidose, double-blind trial. N. Engl. J. Med. 1991;324:277–284. doi: 10.1056/NEJM199101313240501. [DOI] [PubMed] [Google Scholar]
- The European FK 506 Multicentre Psoriasis Group. 1996. Systemic tacrolimus (FK 506) is effective for the treatment of psoriasis in a double-blind, placebo-controlled study. Arch. Dermatol. 132:419–423. [PubMed]
- Wong S., Guerder S., Visintin I., Reich E.-P., Swenson K.E., Flavell R.A., Janeway C.A., Jr. Expression of the co-stimulator molecule B7-1 in pancreatic β-cells accelerates diabetes in the NOD mouse. Diabetes. 1995;44:326–329. doi: 10.2337/diab.44.3.326. [DOI] [PubMed] [Google Scholar]
- Harlan D.M., Hengartner H., Huang M.L., Kang Y.-H., Abe R., Moreadith R.W., Pircher H., Gray G.S., Ohashi P.S., Freeman G.J. Mice expressing both B7-1 and viral glycoprotein on pancreatic beta cells along with glycoprotein-specific transgenic T cells develop diabetes due to a breakdown of T-lymphocyte unresponsiveness. Proc. Natl. Acad. Sci. USA. 1994;91:3137–3141. doi: 10.1073/pnas.91.8.3137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guerder S., Meyerhoff J., Flavell R. The role of the T cell costimulator B7-1 in autoimmunity and the induction and maintenance of tolerance to peripheral antigen. Immunity. 1994;1:155–166. doi: 10.1016/1074-7613(94)90109-0. [DOI] [PubMed] [Google Scholar]
- Guerder S., Picarella D.E., Linsley P.S., Flavell R.A. Costimulator B7-1 confers antigen-presenting-cell function to parenchymal tissue and in conjunction with tumor necrosis factor α leads to autoimmunity in transgenic mice. Proc. Natl. Acad. Sci. USA. 1994;91:5138–5142. doi: 10.1073/pnas.91.11.5138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lo D., Burkly L.C., Widera G., Cowing C., Flavell R.A., Palmiter R.D., Brinster R.L. Diabetes and tolerance in transgenic mice expressing class II MHC molecules in pancreatic beta cells. Cell. 1988;53:150–168. doi: 10.1016/0092-8674(88)90497-7. [DOI] [PubMed] [Google Scholar]
- Higuchi Y., Herrera P., Muniesa P., Huarte J., Belin D., Ohashi P., Aichele P., Orci L., Vassalli J.-D., Vassalli P. Expression of a tumor necrosis factor α transgene in murine pancreatic β cells results in severe and permanent insulitis without evolution towards diabetes. J. Exp. Med. 1992;176:1719–1731. doi: 10.1084/jem.176.6.1719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McWilliam A.S., Napoli S., Marsh A.M., Pemper F.L., Nelson D.J., Pimm C.L., Stumbles P.A., Wells T.N.C., Holt P.G. Dendritic cells are recruited into the airway epithelium during the inflammatory response to a broad spectrum of stimuli. J. Exp. Med. 1996;184:2429–2432. doi: 10.1084/jem.184.6.2429. [DOI] [PMC free article] [PubMed] [Google Scholar]