Abstract
A subpopulation of peripheral human CD4+CD25+ T cells that expresses CD45RO, histocompatibility leukocyte antigen DR, and intracellular cytotoxic T lymphocyte–associated antigen (CTLA) 4 does not expand after stimulation and markedly suppresses the expansion of conventional T cells in a contact-dependent manner. After activation, CD4+CD25+ T cells express CTLA-4 on the surface detectable for several weeks. These cells show a G1/G0 cell cycle arrest and no production of interleukin (IL)-2, IL-4, or interferon (IFN)-γ on either protein or mRNA levels. The anergic state of CD4+CD25+ T cells is not reversible by the addition of anti-CD28, anti–CTLA-4, anti–transforming growth factor β, or anti–IL-10 antibody. However, the refractory state of CD4+CD25+ T cells was partially reversible by the addition of IL-2 or IL-4. These data demonstrate that human blood contains a resident T cell population with potent regulatory properties.
Keywords: human regulatory T cells, CD4+CD25+ T cells, CTLA-4 expression, T cell inhibition, tolerance
Introduction
The adaptive immune response represents a highly effective and dynamic system that can protect a host from pathogens. However, the process involving the generation of pathogen-specific T effector cells intrinsically bears a high risk for the development of autoreactive T cells. Central tolerance mechanisms have been classically ascribed to clonal deletion in the thymus. Furthermore, it was found by using transgenic mouse models that in the periphery the induction of anergy prevents the development of autoreactive T cells 1. Moreover, there is accumulating evidence from various animal models for an additional active mechanism of immune suppression whereby a distinct subset of T cells inhibits the activation of “conventional” T cells in the per-iphery 2 3. The phenotype of these so-called regulatory T cells in mice has been determined as CD4+ CD25+CD45RBlow T cells 4 5 6. This population of T cells from syngeneic healthy donors can prevent the development of autoimmunity upon transfer to lymphopenic recipients 7, indicating that the normal immune system contains a population of “professional” regulatory T cells that actively suppresses autoreactivity. Functional analysis of murine CD4+CD25+ T cells showed that these cells fail to proliferate or secrete cytokines in response to polyclonal or antigen-specific stimulation but inhibit the activation of conventional responsive T cells 5. Detailed characterization in vitro revealed that their suppressive capacity is mediated by direct T cell–T cell contact, but not by cytokines like IL-10 and TGF-β 5 8 9.
Results from studies applying neonatal thymectomy indicated that regulatory T cells originate from the thymus 3. However, it is still largely unknown how regulatory T cells are activated and controlled in the periphery and what kind of antigen they recognize.
Recently, we described the induction of human regulatory T cells by repetitive stimulations of cord blood–derived naive CD4+ T cells with immature monocyte-derived dendritic cells (DCs) 10. These regulatory T cells showed several similarities to murine CD4+CD25+ T cells. They inhibit the activation of conventional T cells in a contact-dependent and cytokine-independent manner 11. In contrast to murine regulatory T cells, human regulatory T cells induced by immature DCs produced high amounts of IL-10. Since these human regulatory T cells were generated in vitro, we subsequently investigated whether regulatory T cells can also be found in vivo and isolated from human peripheral blood.
Here, we demonstrate that freshly isolated human CD4+CD25+ T cells contain T cells with regulatory properties. These T cells are nonproliferating and suppress the activation of conventional CD4+ T cells in a contact- and dose-dependent, but antigen-nonspecific, manner. The anergic state of these human regulatory T cells was partially reversed by the addition of IL-2 or IL-4, but not by anti-CD3 or anti-CD28 Ab.
Materials and Methods
Culture Medium.
X-VIVO-15 supplemented with 1% autologous plasma was used for the culture of DCs and without plasma for culture of T cells (BioWhittaker).
Cytokines.
All cytokines used in this study were recombinant human proteins. Final concentrations were the following: 800 U/ml GM-CSF (Leukomax™; Sandoz), 1,000 U/ml IL-4 and IL-6 (Strathmann Biotech GmbH), 10 ng/ml IL-1β and TNF-α (Strathmann), and 1 μg/ml prostaglandin E2 (Minprostin; Pharmacia-UpJohn). For culture and expansion of T cells, 25 or 200 U/ml IL-2 (Proleukin; Chiron Corp.) and/or 500 U/ml IL-4 were used as indicated.
Abs.
The following Abs were used. Mouse IgG: anti-CD2 (6F10.3); anti-CD14 (RMO52); anti-CD19 (J4.119); anti-CD40 (5C3); anti-CD58 (AICD58); anti-CD80 (MAB104); anti-CD86 (BU63); anti-CD83 (HB15A); and anti-CD152 (anti–cytotoxic T lymphocyte–associated antigen [CTLA]-4, BMI3). Rat IgG (from Beckman Coulter/Immunotech): anti–histocompatibility leukocyte antigen (HLA)-DR (YE2/36HLK) (Serotec/Camon); and mouse and rat subclass-specific isotypes (Beckman Coulter/Immunotech). Conjugated secondary reagents: FITC-conjugated goat anti–mouse-IgG; and PE-conjugated goat anti–rat IgG (Jackson ImmunoResearch Laboratories). Staining of MACS®-sorted T cells: FITC- or PE-conjugated anti-CD3 (UCHT1); anti-CD4 (RPAT4); anti-CD25 (M-A251); anti-CD28 (CD28.2); anti-CD45RA (HI100); and anti-CD45RO (UCHL1) anti–CTLA-4 (BNI3) (all from BD PharMingen); anti-HLA-A2 (BB7.2; American Type Culture Collection); anti–TCR-α/β (BMA031); anti–TCR-γ/δ (Immu510); and FITC- and PE-conjugated mouse IgG from Beckman Coulter/Immunotech. The anti–TGF-β (R&D Systems, used according to the manufacturer's instructions) and anti–IL-10 mAb (JES-19F1.1.1, blocking capacity tested in proliferation assays using IL-10 receptor transfected–Baf3 cells; American Type Culture Collection), anti–CTLA-4 (BNI3, blocking Ab used according to the manufacturer's instructions), and anti–CTLA-4 F(ab′)2 (ANC152.2/8H5, blocking Ab, used according to the manufacturer's instructions; Ancell) were used for blocking experiments. For T cell activation the stimulatory Abs anti-CD3 (OKT-3, purified supernatant; American Type Culture Collection) and anti-CD28 (CD28.2) were used as indicated.
Cytokine Assays.
106 T cells were stimulated with allogeneic DCs (105) in 24-well plates in 1 ml X-VIVO-15. Cytokine synthesis was determined by analysis of supernatants 48 h after stimulation. The commercially available ELISA kits for the human cytokines IFN-γ, IL-2, IL-4, IL-5, and IL-10 (BD PharMingen) were used as indicated by the manufacturer. For intracellular analysis of cytokine production, anti–IL-2–PE, anti–IL-4–PE, anti–IL-5–PE, anti–IL-10–PE, anti–IFN-γ–PE mAb, and FITC- and PE-conjugated isotypic mAb (BD PharMingen) were used according to manufacturer's instructions. In brief, 106 T cells were activated with 2.4 μg/ml PHA plus 1 ng/ml PMA for 6 h. 1.3 μM/ml monensin was added for the last 4 h, cells were collected, washed, fixed/saponin-permeabilized (perm/fix solution; BD PharMingen), and stained with 0.5 μg/test of cytokine-specific Abs.
Generation of DCs.
DCs were generated from buffy coats as described previously 12. In brief, PBMCs were isolated by Ficoll density gradient centrifugation. Monocytes were isolated by plastic adherence and cultured in X-VIVO-15 plus 1% heat-inactivated autologous plasma including 800 U/ml GM-CSF and 1,000 U/ml IL-4. At day 7, nonadherent cells were rinsed off, washed once in PBS, and transferred to fresh six-well plates at 7 × 105 cells in 3 milliliter per well. For differentiation into mature DCs, cells were additionally stimulated with 10 ng/ml IL-1β, 10 ng/ml TNF-α, 1,000 U/ml IL-6, and 1 μg/ml prostaglandin E2. At day 9 mature DCs were used for T cell stimulation.
Flow Cytometric Analysis.
Immunofluorescence staining was performed after washing the cells twice with PBS plus 0.5% human serum albumin (HSA). Cells were incubated for 20 min at 4°C with each mAb (5 μg/ml, 105 cells per test). After washing with cold PBS/HSA the indirectly labeled cells were incubated with FITC- and PE-conjugated second-step mAb for 20 min at 4°C, washed three times, and analyzed by flow cytometry (FACScalibur™, CELLQuest™ software; Becton Dickinson). Necrosis versus apoptosis was determined by propidium iodide and annexin V staining according to the manufacturer's instructions (BD PharMingen).
Isolation of T Cell Subpopulations.
CD4+ T cells were purified from buffy coats using CD4 MACS® MultiSort beads (Miltenyi Biotec) as described previously 10 12. After detaching, CD4+ T cells were washed once in PBS plus 0.5% HSA plus 3 mM EDTA, and stained with anti-CD25 beads (3 μl per 107 cells; Miltenyi Biotec). Alternatively, cells were stained with FITC-conjugated anti-CD25 mAb (10 μg per 100 × 106 CD4+ T cells) for 15 min on ice, washed two times, and incubated for an additional 15 min with anti-FITC MultiSort beads (3 μl per 107 cells; Miltenyi Biotec) and positively selected according to the manufacturer's instructions. After detaching, CD4+CD25+ T cells were washed once and stained with anti-CD45RO beads (3 μl per 107 cells; Miltenyi Biotec) for separation of CD4+ CD25+CD45RO+ T cells and CD4+CD25+CD45RO− T cells. CD4+, CD25-depleted T cells (named as CD4+ T cells in the following), CD4+CD25+ T cells, and the CD45RO+ and CD45RO− T cell subpopulations of CD4+CD25+ T cells were used immediately after isolation.
T Cell Stimulation Assays.
Stimulation with allogeneic DCs. For an optimal stimulation of freshly isolated T cell populations in an antigen-specific (alloantigen) manner, mature DCs from allogeneic donors were used for stimulation. Freshly isolated T cells (2 × 105 cells per well) were used for primary proliferation assays in the presence of different numbers of allogeneic DCs in 96-well plates (200 μl per well) as indicated in the figure legends. T cell proliferation of primary cultures was measured after 4 d of incubation and an additional 16-h pulse with [3H]Tdr (37 kBq per well) using a liquid scintillation counter. For secondary stimulation assays, T cells (106 cells per well) were primarily stimulated with 105 allogeneic DCs (105 cells per well) in 24-well plates. 25 U/ml IL-2 was added once at day 5 of culture.
10 d after the onset of the primary culture, T cells were harvested and used for secondary proliferation assays.
All secondary proliferation assays were performed with 5 × 104 T cells per well in the presence of different ratios of allogeneic DCs. T cell proliferation of secondary proliferation assays was measured after 2 d of incubation and an additional 16-h pulse with 3H[Tdr].
Polyclonal anti-CD3 stimulation. Alternatively to allogeneic stimulation, CD4+ and CD4+CD25+ T cells (106 cells per well) were primarily activated with anti-CD3 mAb (2 μg/ml, OKT-3) in the presence of 4 × 106 irradiated (4,000 rads) syngeneic PBMCs in 24-well plates. IL-2 was added at days 2, 5, and 7 after primary stimulation. At day 10 the CD4+ and CD4+CD25+ T cells were harvested and used in a secondary anti-CD3 stimulation with identical conditions, but in the presence of syngeneic irradiated PBMCs in 96-well plates as indicated in the figure legends. T cell proliferation in anti-CD3 assays was measured after 2 d of incubation and an additional 16-h pulse with [3H]Tdr (37 kBq per well) using a liquid scintillation counter.
Cocultures of CD4+ and CD4+CD25+ T cells (primary stimulation as well as secondary stimulation) activated with allogeneic DCs or anti-CD3 plus syngeneic PBMCs were also performed to analyze the influence of CD4+CD25+ T cells on the proliferation and cytokine production of conventional CD4+ T cells. A constant number of CD4+ T cells was cocultured with different numbers of CD4+CD25+ T cells as indicated in the figures, and both T cell populations were activated with either allogeneic DCs or anti-CD3 plus syngeneic PBMCs.
Transwell Experiments.
Transwell experiments were performed in 24-well plates as described previously 13. In brief, CD4+ T cells (106 cells per milliliter) and CD4+CD25+ T cells (106 cells per milliliter) were separately stimulated with allogeneic DCs (105 cells per milliliter). Additionally, CD4+CD25+ T cells stimulated with allogeneic DCs were cocultured and activated with CD4+ T cells in the same well or were placed in transwell chambers (Millicell, 0.4 μm; Millipore) in the same well. For some coculture experiments, additional inhibitory Abs were added as indicated in the figure legend. After 2 d of culture, 3 × 200 μl per well (containing 2 × 105 T cells) of each culture were transferred to three individual wells of 96-well plates. Proliferation was measured after an additional 16-h pulse with [3H]Tdr using a liquid scintillation counter.
Cell Cycle Analysis.
Cell cycle analysis of each T cell population was performed as described by determination of the DNA content using propidium iodide staining 14. 24–96 h after restimulation T cells were washed in PBS and fixed in 70% ethanol for 2 h at −20°C. After incubation, cells were treated with PBS containing 1% glucose, 2 mg/ml RNAse, and 0.05 mg/ml propidium iodide (Sigma Aldrich) for 30 min at room temperature. DNA content of stained cells was analyzed by flow cytometry.
mRNA Detection.
RNA was isolated as described previously 15 and used for reverse transcription (RT) with Superscript RnaseH− following the recommendations of the supplier (Life Technologies). RT-PCR was performed using the following oligonucleotides: β-actin forward: GAGCGGGAAATCGTGCGTGACATT; β-actin reverse: GAAGGTAGTTTCGTGGATGCC (225 bp); IL-10 forward: ATGCCCCAAGCTGAGAACCAAGAC; IL-10 reverse: CCCAGAGCCCCAGATCCGATTTTG (227 bp); IL-4 forward: CTGCTTCCCCCTCTGTTCTTCC; IL-4 reverse: TCTGGTTGGCTTCCTTCACAGG (379 bp); IL-2 forward: ATGTACAGGATGCAACTCCTGTC; IL-2 reverse: GTCAGTGTTGAGATGATGCTTTGAC (458 bp); TGF-β1 forward: GACATCAACGGGTTCACTACCGG; TGF-β1 reverse: GAGGCAGAAGTTGGCATGGTAGC (267 bp); IFN-γ forward: CTGTTACTGCCAGGACCCATATG; and IFN-γ reverse: GAACCATTACTGGGATGCTCTTCG (448 bp). Oligonucleotides were chosen to span at least one intron or to include sequences of two adjacent exons at the level of genomic DNA.
Results
Freshly Isolated Human CD4+CD25+ T Cells Are Anergic and Do Not Proliferate after Allogeneic or Polyclonal Activation.
To analyze the functional properties of human CD4+ T cell subpopulations, we isolated CD4+ T cells from buffy coats of randomly selected healthy volunteers. In the next step, CD4+CD25+ T cells were positively selected from isolated CD4+ T cells. Both populations showed a purity of >95% (CD4+CD25− or CD4+CD25+; Fig. 1 A). Analyzing 17 different buffy coats from healthy donors, we observed a resident population of CD4+ CD25+ T cells that represented ∼0.7–5.5% of total PBMCs. CD4+CD25− (in the following, abbreviated as CD4+ T cells) and CD4+CD25+ T cells expressed CD4, TCR-α/β, and CD3 molecules in comparable amounts. However, the CD4+CD25+ T cell population contained significantly more cells, which expressed CD45RO and HLA-DR, a phenotype characteristic for differentiated or activated T cells (Fig. 1 A). After activation with allogeneic DCs, the CD4+CD25+ T cells showed an upregulation of CD25 and HLA-DR (Fig. 1 B) comparable to conventional CD4+ T cells (data not shown).
Figure 1.

Phenotype of freshly isolated and stimulated CD4+ and CD4+CD25+ T cells. CD4+ and CD4+CD25+ T cells were isolated from buffy coats of healthy volunteers by positive selection using paramagnetic beads as described in Materials and Methods. (A) The figure shows the surface expression of freshly isolated T cells, representative of 10 independent experiments. (B) Phenotype of CD4+CD25+ T cells 10 d after stimulation with allogeneic DCs.
Recently, it was shown that resting murine regulatory CD4+CD25+ T cells intracellularly express CTLA-4 9 16. To investigate the CTLA-4 expression of human T cells, we analyzed intracellular and surface expression of this marker on freshly isolated and activated CD4+ and CD4+CD25+ T cells. As shown in Fig. 2, no surface expression of CTLA-4 was observed on freshly isolated T cells of both populations. However, a subpopulation of CD4+CD25+ T cells, the CD45RO+ population, constantly expressed CTLA-4 intracellularly (Fig. 2 A). Furthermore, CTLA-4 expression was detectable early after activation by allogeneic DCs (16 h) solely on the surface of CD4+CD25+ T cells but not on the surface of conventional CD4+ T cells (Fig. 2 B). This strong upregulation of CTLA-4 on a subpopulation of CD4+ CD25+ T cells was also observed 6 h after primary activation with anti-CD3 or PHA and then detectable for at least 21 d after priming (data not shown).
Figure 2.


A subpopulation of CD4+CD25+ human anergic T cells shows constitutive expression of CTLA-4. (A) Surface and intracellular CTLA-4 expression of freshly isolated CD4+ and CD4+CD25+ T cells. (B) Surface expression of CTLA-4 16 h after stimulation with allogeneic DCs. The analysis is representative of seven independent experiments. (C) Proliferation assays of CD4+ and CD4+CD25+ T cells stimulated with varying numbers of allogeneic mature DCs. Proliferation of T cells was determined by the addition of [3H]Tdr after 4 (primary culture) or 2 d (restimulation) of culture for 16 h. Similar results were obtained in five independent experiments. (D) Cell cycle analysis of alloreactive CD4+ and CD4+CD25+ T cells 4 d after the first restimulation with allogeneic DCs. Similar results were obtained in five independent experiments.
Both T cell populations were primed and restimulated with potent immunostimulatory allogeneic DCs in different DC/T cell ratios to induce maximal activation of alloantigen-specific T cells. As shown in Fig. 2 C, the primary stimulation of CD4+ T cells resulted in a strong and dose-dependent proliferation, whereas only low rates of proliferation were observed after primary stimulation of CD4+CD25+ T cells. Furthermore, this weak proliferative capacity of CD4+CD25+ T cells further decreased after restimulation and could not be enhanced by the addition of allogeneic PBMCs, anti-CD3, anti-CD28 Abs, or PHA (data not shown). By contrast, the respective CD4+ T cell population showed a considerably high alloreactive proliferation after restimulation. As shown in Fig. 2 D, alloantigen-activated CD4+CD25+ T cells showed a cell cycle arrest in the G1/G0 phase.
Anergic Human CD4+CD25+ T Cells Do Not Produce IL-2, IL-4, IFN-γ, or IL-10 after Activation.
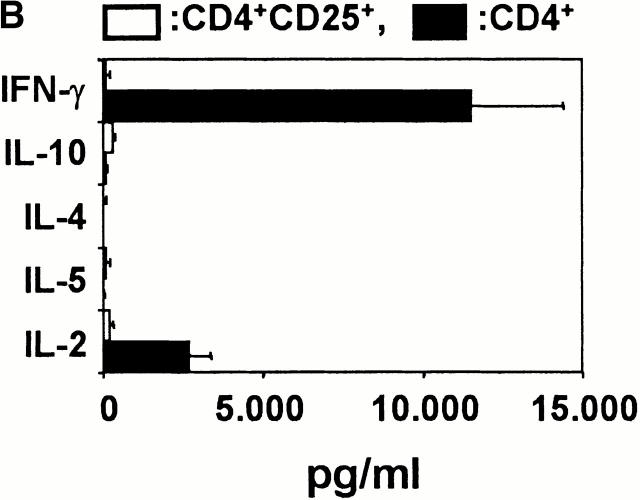
Next we analyzed the cytokine profiles of alloreactive CD4+ and CD4+CD25+ T cells stimulated twice with allogeneic mature DCs. It has been shown that mature DCs induce the polarization of naive CD4+ T cells towards Th1 cells with a high production of IFN-γ and IL-2 in the absence of IL-4 or IL-10 10 12. This cytokine profile could also be observed for alloreactive peripheral CD4+ T cells after two stimulations (Fig. 3). In contrast, CD4+CD25+ T cells showed only marginal synthesis of IL-2, IL-4, IL-10, and IFN-γ after two stimulations with allogeneic DCs or polyclonal restimulation.
Figure 3.


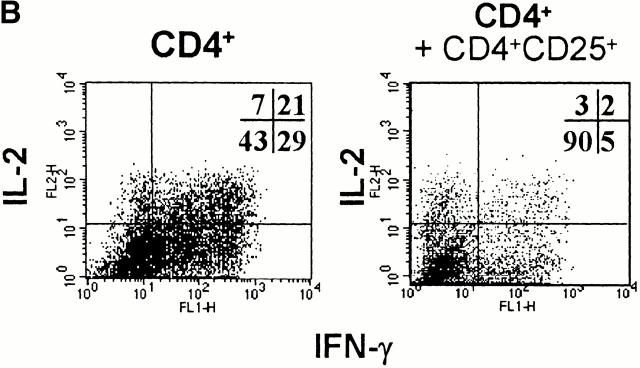
Cytokine profiles of alloreactive CD4+ and CD4+CD25+ T cells after the first restimulation. CD4+ and CD4+CD25+ T cells were primed and restimulated with allogeneic DCs from the same donor. The figure shows the cytokine profiles of T cells after the first restimulation. (A) 6 d after the first restimulation, T cells were activated with PHA/PMA in the presence of monensin. The cytokine profiles were detected by intracellular FACS® staining 6 h later. (B) Cytokine profiles of T cells determined by ELISA 48 h after the first restimulation with allogeneic DCs. Black bars, CD4+ T cells; white bars, CD4+CD25+ T cells. (C) The cytokine mRNA profile of T cells 2 h after restimulation with allogeneic DCs as detected by RT-PCR. A representative result of four independent experiments is shown.
It has been demonstrated that murine CD4+CD25+ T cells do not produce detectable amounts of cytokines on the protein level but show enhanced cytokine mRNA synthesis after activation 5. Here we show, using RT-PCR for cytokine mRNA analysis of human CD4+CD25+ T cells, that only mRNA signals for TGF-β and IL-10 were detectable in resting and activated CD4+CD25+ T cells. Neither IL-2 and IFN-γ nor IL-4 could be detected at the level of mRNA expression under such conditions (Fig. 3 C). In contrast, CD4+ T cells showed a strong upregulation of T cell–associated cytokines IL-2, IL-4, and IFN-γ after activation. It is important to note that no significant increase of apoptosis or necrosis measured by annexin V and propidium iodide staining could be observed in activated CD4+ or CD4+CD25+ T cells (data not shown).
The Anergic State of Human CD4+CD25+ T Cells Can Be Partially Reversed by the Addition of IL-2 and/or IL-4.
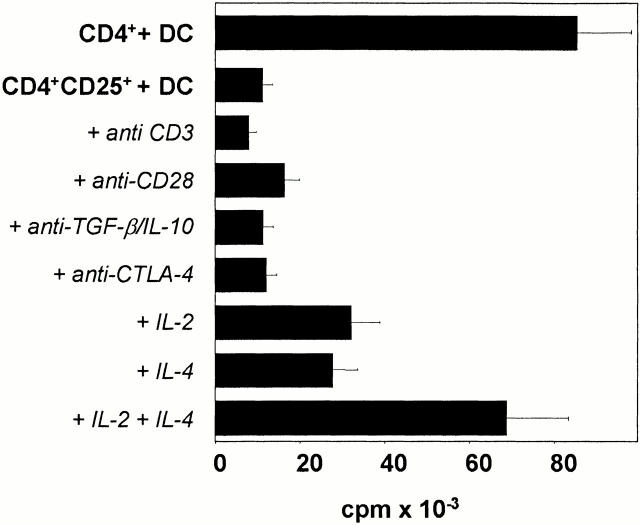
To analyze the anergic state of human CD4+CD25+ T cells in more detail we activated freshly isolated CD4+CD25+ T cells in the presence or absence of blocking/stimulatory Abs or T cell growth factors. The suppressed proliferative capacity could not be reversed by the addition of blocking Abs directed against TGF-β, IL-10, or CTLA-4 (Fig. 4). Furthermore, addition of stimulatory anti-CD3 or anti-CD28 Abs could not reverse the anergic state of human CD4+CD25+ T cells. However, this refractory state of CD4+CD25+ T cells could be partially reversed by the addition of high amounts of IL-2 (200 U/ml) or IL-4 (500 U/ml) and, even more pronounced, by a combination of both.
Figure 4.
The anergic state of CD4+CD25+ T cells is partially reversible by exogenous IL-2 or IL-4. CD4+CD25+ T cells were primed and restimulated with allogeneic DCs. T cell proliferation (5 × 104 T cells per well plus 5 × 103 DCs per well, first restimulation) in the presence or absence of blocking Abs (2 μg/ml anti–TGF-β mAb, 10 μg/ml anti–IL-10 mAb, 20 μg/ml anti–CTLA-4 mAb) or additional stimulation with 2 μg/ml anti-CD3, 2 μg/ml anti-CD28, or cytokines (200 U/ml IL-2, 500 U/ml IL-4). [3H]Tdr was added after 2 d of culture for the final 16 h. Proliferation of restimulated CD4+ T cells served as a control. Similar results were obtained in seven independent experiments.
Proliferation and Cytokine Production of CD4+ T Cells Is Inhibited by Coculture with CD4+CD25+ T Cells.
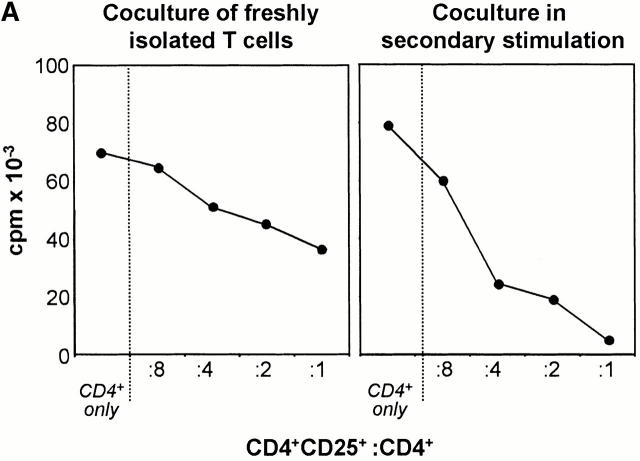
Next, we analyzed the regulatory properties of human CD4+CD25+ T cells. Freshly isolated CD4+ T cells were either immediately cocultured with CD4+CD25+ T cells and stimulated with allogeneic DCs or at first separately activated, and such prestimulated CD4+ and CD4+CD25+ T cells were subsequently used for cocultures. As shown in Fig. 5 A, in both types of coculture experiments using freshly isolated or prestimulated T cells, activated CD4+CD25+ T cells suppressed the proliferation of alloreactive CD4+ T cells. Analogous to the decreased proliferation of CD4+CD25+ T cells after restimulation (Fig. 2 C), the inhibitory capacity of such cells is even more pronounced in secondary cocultures. Furthermore, the inhibitory capacity of CD4+ CD25+ T cells was dose dependent (Fig. 5 A). Finally, CD4+CD25+ T cells also markedly inhibited the cytokine synthesis of CD4+ T cells (Fig. 5 B).
Figure 5.
CD4+CD25+ T cells inhibit dose dependently proliferation and cytokine production of conventional CD4+ T cells. (A, left) Freshly isolated CD4+ T cells (2 × 105 cells per well) were stimulated with allogeneic DCs (2 × 104 cells per well) in the presence of different numbers of freshly isolated CD4+CD25+ T cells from the same donor. Proliferation was determined after 4 d of culture by the addition of [3H]Tdr for the final 16 h. (Right) Coculture of separately primed CD4+ and CD25+ T cells in a secondary stimulation assay. Proliferation was determined after 2 d of culture by addition of [3H]Tdr for the final 16 h. (B) On the left, the cytokine profile of CD4+ T cells primed and restimulated with allogeneic DCs alone and on the right, the cytokine profile of CD4+ T cells restimulated with allogeneic DCs in the presence of CD4+CD25+ T cells are illustrated. A representative result of four independent experiments is shown. (C) Anti-CD3 assay. CD4+ and CD4+CD25+ T cells were stimulated with 2 μg/ml anti-CD3 in the presence of irradiated syngeneic PBMCs and were used for secondary anti-CD3 mAb proliferation assays 10 d after as described in Materials and Methods. The proliferative response of CD4+ and CD4+CD25+ T cells restimulated separately or in coculture is shown. A representative result of three independent experiments is shown.
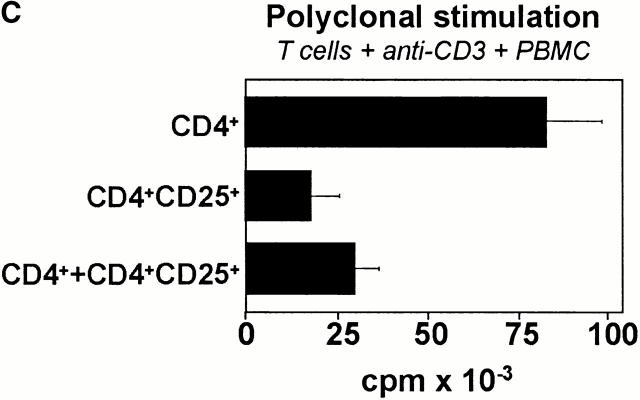
The usage of allogeneic DCs as T cell stimulators may result in the preferential selection of an alloreactive subpopulation of CD4+CD25+ T cells. To prevent a potential selection of a nonrepresentative CD4+CD25+ T cell population, freshly isolated CD4+ and CD4+CD25+ T cells were polyclonally activated by anti-CD3 mAb. Such T cells were subsequently restimulated alone or in cocultures in the presence of anti-CD3 mAb and syngeneic PBMCs. Fig. 5 C demonstrates that the polyclonally activated CD4+ T cells showed a profound anti-CD3–induced proliferation, whereas the proliferation of CD4+CD25+ T cells was rather low and the coculture of both populations led to a considerable suppression of the CD4+ T cell–based proliferation. Thus, the fact that alloreactive CD4+CD25+ T cells and the polyclonally activated CD4+CD25+ T cells have equivalent suppressive properties strongly argues for the assumption that the alloreactive CD4+CD25+ T cell population is representative for the whole regulatory CD4+CD25+ T cell population.
Human CD4+CD25+ T Cells Inhibit the Activation of Conventional CD4+ T Helper Cells by Contact-dependent, but Cytokine-independent Mechanisms.
Transwell experiments were performed to investigate whether cell–cell contact or soluble mediators mediate the suppressive effect of human CD4+CD25+ T cells. The inhibition of soluble factors such as IL-10 or TGF-β, or blocking Abs against CTLA-4, or anti–CTLA-4 F(ab′)2-fragments, or the additional stimulation by anti-CD28 mAb were not able to restore the proliferation of CD4+ T cells in coculture with CD4+ CD25+ T cells. In contrast, separation of both T cell populations by a semipermeable membrane in transwell chambers completely abolished the suppressive capacity of CD4+CD25+ T cells for the proliferation of conventional CD4+ T cells (Fig. 6 A).
Figure 6.
The inhibitory effect of human regulatory CD4+CD25+ T cells requires cell–cell contact and is antigen nonspecific. (A) CD4+ and CD4+CD25+ T cells from the same donor were primed with allogeneic DCs. After 10 d, the alloreactive T cells were restimulated with mature DCs from the same allogeneic donor. CD4+CD25+ T cells (106 cells per well) and CD4+ T cells (106 cells per well) were placed and activated separately in transwell chambers in the same well. Alternatively, CD4+CD25+ T cells were added directly to the cocultures of CD4+ T cells plus allogeneic DCs in the presence or absence of inhibitory Abs as indicated (2 μg/ml anti–TGF-β, 10 μg/ml anti–IL-10, 20 μg/ml anti–CTLA-4/CTLA-4 F(ab′)2 fragments or 2 μg/ml anti-CD28). Proliferation of restimulated CD4+ T cells served as a control. (B) Syngeneic CD4+ and CD4+CD25+ T cells were primed with allogeneic DCs from different donors (A or B). 10 d after, donor A–specific CD4+ T cells (ACD4+) were restimulated in the presence of donor B–specific CD4+CD25+ T cells (BCD4+CD25+) stimulated with allogeneic DCs (donor A and/or B) as indicated. Additionally, BCD4+CD25+ T cells were preactivated with 0.5 μg/ml anti-CD3 at 37°C for 30 min, washed in PBS, and added to cultures of activated ACD4+ T cells. After 2 d of culture, activated T cells were transferred in 96-well plates to measure incorporation of [3H]Tdr for the final 16 h. Results representative of five independent experiments are presented as mean cpm of triplicate determinations.
To analyze the antigen specificity of this inhibitory effect of CD4+CD25+ T cells, we primed syngeneic CD4+ and CD4+CD25+ T cells with allogeneic DCs from different donors (donor A or B). As shown in Fig. 6 B, donor B–specific CD4+CD25+ T cells suppressed the proliferation of donor A–specific CD4+ T cells only after activation with donor B DCs. Alternatively, the suppressive capacity of CD4+CD25+ T cells could also be induced by anti-CD3–mediated stimulation. These results suggest that the inhibitory properties of human CD4+CD25+ T cells are activation-dependent, but antigen-nonspecific, and do not require the simultaneous presentation of the target antigen on the same APC.
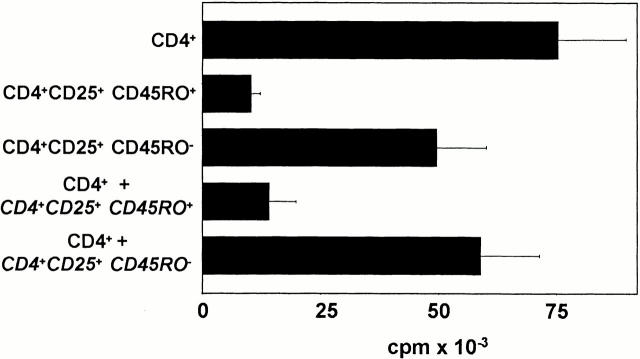
Freshly isolated CD4+CD25+ T cells contain two subpopulations, CD45RO+ and CD45RO− T cells (Fig. 1 A). To analyze the suppressive capacities of both subpopulations independently, CD45RO beads were used for the separation of both populations. As shown in Fig. 7, the CD45RO− T cell population showed a normal proliferation after activation with allogeneic DCs and in coculture with CD4+ T cells, only a weak inhibitory capacity for the proliferation of conventional CD4+ T cells. In contrast, the CD45RO+ subpopulation was anergic and suppressed the proliferation of conventional CD4+ T cells. These data suggest that peripheral human CD4+CD25+ T cells contain two subpopulations, CD4+CD25+CD45RO+ regulatory T cells and CD4+CD25+CD45RO− conventional T cells.
Figure 7.
Human CD4+CD25+ T cell populations contain regulatory and conventional T cells. Freshly isolated CD4+CD25+ T cells were separated into CD45RO+ and CD45RO− cells using anti-CD45RO beads as described in Materials and Methods and separately primed with allogeneic DCs. 10 d after, the T cell populations were restimulated under the same conditions or in coculture with primed CD4+ T cells. The proliferation of restimulated T cell populations is shown (2-d culture plus an additional 16-h [3H]Tdr). The figure is representative for three independent experiments.
Discussion
The delicate balance between pathogen-induced effector functions and endogenous tolerance-mediating mechanisms is of vital importance for the integrity of a host in the course of an immune response. In particular, with regard to peripheral tolerance induction, there is increasing evidence for a distinct population of regulatory T cells that controls autoimmunity and transplant rejection in mice 3. Our results indicate that human peripheral blood also contains a population of regulatory CD4+CD25+ T cells that inhibits the activation of CD4+CD25− T effector cells significantly. This resident human T cell population represents ∼0.7–5.5% of total PBMCs of randomly selected healthy donors. Freshly isolated, these cells showed no significant proliferation or cytokine secretion after allogeneic activation with mature DCs, or polyclonal stimulation with PHA or anti-CD3 Abs. Nevertheless, in coculture these anergic T cells after activation efficiently suppressed the proliferation and cytokine production of CD4+CD25− T cells in a dose- and contact-dependent, but cytokine-independent manner.
These inhibitory properties of human regulatory CD4+ T cells as outlined in this study suggest that these T cells are the human analogue of a previously described population of regulatory CD4+CD25+ T cells in mice. Such cells arise from the thymus 3, express CD4, CD25 17, and intracellular CTLA-4 9 16, and their activation via the B7-CD28 pathway is essential for their induction and survival in the periphery 18. They comprise 5–10% of peripheral T cells and can not be induced to proliferate in vitro upon polyclonal activation using anti-CD3 Abs 5. Analogous to murine CD4+CD25+ T cells, the anergic state of human regulatory T cells could not be reversed by the addition of anti-CD28 Abs. However, besides striking similarities there are also some differences between murine and human regulatory T cells. The suppressive activity of murine CD4+CD25+ T cells was overcome by the addition of anti-CD28 mAb 5, and the unresponsiveness and suppressive activity of murine CD4+CD25+ thymocytes was abrogated by a combination of anti-CD3 and anti-CD28 mAb 19. In contrast, coactivation of human regulatory T cells via CD28 could not reduce their inhibitory capacity (Fig. 6 A). In addition, human regulatory T cells showed no significant proliferation after stimulation with mature DCs that expressed high amounts of costimulatory molecules such as CD80, CD86, CD58, CD40 12, and cytokines IL-12 20, IL-15 21, and IL-18 22. These findings are in accord with an earlier observation that the anergic state of human regulatory T cells, induced by repetitive stimulations of naive cord blood–derived CD4+ T cells with immature DCs, also could not be reversed by mature DCs, or the addition of anti-CD3 or anti-CD28 Abs 10. Thus, the deficient proliferative response of human regulatory T cells is not the result of a lack of costimulation.
It should be noted at this point that in contrast to freshly isolated human CD4+CD25+ T cells, cord blood–derived regulatory T cells induced by immature DCs secreted high amounts of IL-10. The proliferation of these cells remained refractory even in the presence of IL-2, whereas their suppressive activities were abrogated by IL-2 10. These functional differences imply the existence of different subpopulations of human CD4+CD25+ regulatory T cells.
A characteristic property of human and murine regulatory T cells is the constitutive and long-lasting membrane expression of CTLA-4 after activation 9 10 16. However, the relevance of this marker for the function of this regulatory T cell population is not defined in detail. Whereas overwhelming evidence exists that CTLA-4 acts as a negative regulator of T cell activation in vivo 23, there have been contradictory results regarding the function of CTLA-4 for the suppressive activity of murine CD4+CD25+ T cells in vitro. Thornton and Shevach 5 have reported that anti–CTLA-4 mAb did not abrogate suppression, whereas Takahashi et al. 9 demonstrated recently that the application of Fab anti–CTLA-4 mAb completely removed the inhibitory capacity of murine CD4+CD25+ T cells. In agreement with Thornton and Shevach 5, blocking of CTLA-4 by mAb or F(ab′)2 fragments did not reverse the anergic state and the suppressive properties of CD4+CD25+ human regulatory cells (Fig. 4 and Fig. 6) and did not significantly affect the responses of human IL-10–producing regulatory T cells 10. These discrepancies may be partially at least the result of the fact that the anti–human CTLA-4 mAb and F(ab′)2 fragments have a comparatively low neutralizing activity.
It has been described that murine CD4+CD25+ regulatory T cells contain mRNA for IL-10, IL-4, and TGF-β, and that the level of IL-10 mRNA increases upon polyclonal activation 5. In contrast, human CD4+CD25+ regulatory T cells only expressed mRNA for IL-10 and TGF-β, but not for IL-4. Additionally, it has been shown that the anergic state of murine regulatory T cells was completely reversed by the addition of IL-2 5, whereas we could demonstrate in this study that exogenous IL-2 and/or IL-4 only partially reversed the anergic state of human regulatory T cells. However, the combination of IL-2 and IL-4 completely restored the proliferation of these cells.
Our data report on the first description of regulatory CD4+CD25+ T cells isolated ex vivo from human peripheral blood. As demonstrated for their murine analogues, these cells may have profound influence on the control of human autoimmune diseases. The continuous and constitutive presence of this novel human T cell subset may regulate autoaggressive T and B cells. Furthermore, physiological immune responses may be terminated by the activation of these regulatory T cells thereby preventing tissue damage and harm to the human organism. Future studies will have to define the molecular pathways that regulatory T cells use to control immune responses. The better understanding of the underlying mechanisms of regulation will then potentially allow the application of these cells as therapeutic vehicles for the control of human autoimmune diseases.
Acknowledgments
The authors are grateful to Drs. E. Rude, K. Steinbrink, and T. Tuting for critical reading of this manuscript and helpful discussions. We also thank L. Paragnik, A. Kandemir, and P. Holter for their expert technical assistance.
This work was supported by the Deutsche Forschungsgemeinschaft grant A6SFB548 (to E. Schmitt) and grant A3SFB548 (to A.H. Enk).
Footnotes
Abbreviations used in this paper: CTLA, cytotoxic T lymphocyte–associated antigen; DC, dendritic cell; HLA, histocompatibility leukocyte antigen; HSA, human serum albumin; RT, reverse transcription.
References
- Hammerling G.J., Schonrich G., Momburg F., Auphan N., Malissen M., Malissen B., Schmitt-Verhulst A.M., Arnold B. Non-deletional mechanisms of peripheral and central tolerancestudies with transgenic mice with tissue-specific expression of a foreign MHC class I antigen. Immunol. Rev. 1991;122:47–67. doi: 10.1111/j.1600-065x.1991.tb00596.x. [DOI] [PubMed] [Google Scholar]
- Seddon B., Mason D. Peripheral autoantigen induces regulatory T cells that prevent autoimmunity. J. Exp. Med. 1999;189:877–882. doi: 10.1084/jem.189.5.877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seddon B., Mason D. The third function of the thymus. Immunol. Today. 2000;21:95–99. doi: 10.1016/s0167-5699(99)01559-5. [DOI] [PubMed] [Google Scholar]
- Takahashi T., Kuniyasu Y., Toda M., Sakaguchi N., Itoh M., Iwata M., Shimizu J., Sakaguchi S. Immunologic self-tolerance maintained by CD25+CD4+ naturally anergic and suppressive T cellsinduction of autoimmune disease by breaking their anergic/suppressive state. Int. Immunol. 1998;10:1969–1980. doi: 10.1093/intimm/10.12.1969. [DOI] [PubMed] [Google Scholar]
- Thornton A.M., Shevach E.M. CD4+CD25+ immunoregulatory T cells suppress polyclonal T cell activation in vitro by inhibiting interleukin 2 production. J. Exp. Med. 1998;188:287–296. doi: 10.1084/jem.188.2.287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Papiernik M., de Moraes M.L., Pontoux C., Vasseur F., Penit C. Regulatory CD4 T cellsexpression of IL-2R α chain, resistance to clonal deletion and IL-2 dependency. Int. Immunol. 1998;10:371–378. doi: 10.1093/intimm/10.4.371. [DOI] [PubMed] [Google Scholar]
- Boitard C., Yasunami R., Dardenne M., Bach J.F. T cell-mediated inhibition of the transfer of autoimmune diabetes in NOD mice. J. Exp. Med. 1989;169:1669–1680. doi: 10.1084/jem.169.5.1669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thornton A.M., Shevach E.M. Suppressor effector function of CD4+CD25+ immunoregulatory T cells is antigen nonspecific. J. Immunol. 2000;164:183–190. doi: 10.4049/jimmunol.164.1.183. [DOI] [PubMed] [Google Scholar]
- Takahashi T., Tagami T., Yamazaki S., Uede T., Shimizu J., Sakaguchi N., Mak T.W., Sakaguchi S. Immunologic self-tolerance maintained by CD25+CD4+ regulatory T cells constitutively expressing cytotoxic T lymphocyte-associated antigen 4. J. Exp. Med. 2000;192:303–310. doi: 10.1084/jem.192.2.303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jonuleit H., Schmitt E., Schuler G., Knop J., Enk A.H. Induction of interleukin 10-producing, nonproliferating CD4+ T cells with regulatory properties by repetitive stimulation with allogeneic immature human dendritic cells. J. Exp. Med. 2000;192:1213–1222. doi: 10.1084/jem.192.9.1213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shevach E.M. Regulatory T cells in autoimmmunity. Annu. Rev. Immunol. 2000;18:423–449. doi: 10.1146/annurev.immunol.18.1.423. [DOI] [PubMed] [Google Scholar]
- Jonuleit H., Kuhn U., Muller G., Steinbrink K., Paragnik L., Schmitt E., Knop J., Enk A.H. Pro-inflammatory cytokines and prostaglandins induce maturation of potent immunostimulatory dendritic cells under fetal calf serum-free conditions. Eur. J. Immunol. 1997;27:3135–3142. doi: 10.1002/eji.1830271209. [DOI] [PubMed] [Google Scholar]
- Jonuleit H., Tuting T., Steitz J., Bruck J., Giesecke A., Steinbrink K., Knop J., Enk A.H. Efficient transduction of mature CD83+ dendritic cells using recombinant adenovirus suppressed T cell stimulatory capacity. Gene Ther. 2000;7:249–254. doi: 10.1038/sj.gt.3301077. [DOI] [PubMed] [Google Scholar]
- Jonuleit T., van der Kuip H., Miething C., Michels H., Hallek M., Duyster J., Aulitzky W.E. Bcr-Abl kinase down-regulates cyclin-dependent kinase inhibitor p27 in human and murine cell lines. Blood. 2000;96:1933–1939. [PubMed] [Google Scholar]
- Erlich H.A. Polymerase chain reaction. J. Clin. Immunol. 1989;9:437–447. doi: 10.1007/BF00918012. [DOI] [PubMed] [Google Scholar]
- Read S., Malmstrom V., Powrie F. Cytotoxic T lymphocyte-associated antigen 4 plays an essential role in the function of CD25+CD4+ regulatory cells that control intestinal inflammation. J. Exp. Med. 2000;192:295–302. doi: 10.1084/jem.192.2.295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sakaguchi S., Sakaguchi N., Asano M., Itoh M., Toda M. Immunologic self-tolerance maintained by activated T cells expressing IL-2 receptor α-chains (CD25). Breakdown of a single mechanism of self-tolerance causes various autoimmune diseases. J. Immunol. 1995;155:1151–1164. [PubMed] [Google Scholar]
- Salomon B., Lenschow D.J., Rhee L., Ashourian N., Singh B., Sharpe A., Bluestone J.A. B7/CD28 costimulation is essential for the homeostasis of the CD4+CD25+ immunoregulatory T cells that control autoimmune diabetes. Immunity. 2000;12:431–440. doi: 10.1016/s1074-7613(00)80195-8. [DOI] [PubMed] [Google Scholar]
- Itoh M., Takahashi T., Sakaguchi N., Kuniyasu Y., Shimizu J., Otsuka F., Sakaguchi S. Thymus and autoimmunityproduction of CD25+CD4+ naturally anergic and suppressive T cells as a key function of the thymus in maintaining immunologic self-tolerance. J. Immunol. 1999;162:5317–5326. [PubMed] [Google Scholar]
- Rieser C., Bock G., Klocker H., Bartsch G., Thurnher M. Prostaglandin E2 and tumor necrosis factor α cooperate to activate human dendritic cellssynergistic activation of interleukin 12 production. J. Exp. Med. 1997;186:1603–1608. doi: 10.1084/jem.186.9.1603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jonuleit H., Wiedemann K., Muller G., Degwert J., Hoppe U., Knop J., Enk A.H. Induction of IL-15 messenger RNA and protein in human blood-derived dendritic cellsa role for IL-15 in attraction of T cells. J. Immunol. 1997;158:2610–2615. [PubMed] [Google Scholar]
- Stoll S., Jonuleit H., Schmitt E., Muller G., Yamauchi H., Kurimoto M., Knop J., Enk A.H. Production of functional IL-18 by different subtypes of murine and human dendritic cells (DC)DC-derived IL-18 enhances IL-12-dependent Th1 development. Eur. J. Immunol. 1998;28:3231–3239. doi: 10.1002/(SICI)1521-4141(199810)28:10<3231::AID-IMMU3231>3.0.CO;2-Q. [DOI] [PubMed] [Google Scholar]
- Walunas T.L., Bluestone J.A. CTLA-4 regulates tolerance induction and T cell differentiation in vivo. J. Immunol. 1998;160:3855–3860. [PubMed] [Google Scholar]