Abstract
Antigen-specific T helper type 1 (Th1) cells mediate protective immunity against a range of infectious diseases, including that caused by Bordetella pertussis. Distinct T cell subtypes that secrete interleukin (IL)-10 or tumor growth factor (TGF)-β are considered to play a role in the maintenance of self-tolerance. However, the antigens recognized by these regulatory T cells in vivo have not been defined. Here we provide the first demonstration of pathogen-specific T regulatory type 1 (Tr1) cells at the clonal level and demonstrate that these cells are induced at a mucosal surface during an infection where local Th1 responses are suppressed. Tr1 clones specific for filamentous hemagglutinin (FHA) and pertactin were generated from the lungs of mice during acute infection with B. pertussis. The Tr1 clones expressed T1/ST2 and CC chemokine receptor 5, secreted high levels of IL-10, but not IL-4 or interferon (IFN)-γ, and suppressed Th1 responses against B. pertussis or an unrelated pathogen. Furthermore, FHA inhibited IL-12 and stimulated IL-10 production by dendritic cells (DCs), and these DCs directed naive T cells into the regulatory subtype. The induction of Tr1 cells after interaction of a pathogen-derived molecule with cells of the innate immune system represents a novel strategy exploited by an infectious pathogen to subvert protective immune responses in vivo.
Keywords: CD4+ T cells, bacterial infection, mucosal immunity, immunosuppression, IL-12
Introduction
CD4+ T cell subtypes have been distinguished on the basis of their cytokine profile, function, and more tentatively by surface antigen expression. Th1 and Th2 clones have been generated against a range of infectious pathogens and although distinct subtypes of T cells with regulatory function, termed Tr1* (1–4) or Th3 (4–6) cells, that secrete high levels of IL-10 or TGF-β respectively, have been described; these cells have been more difficult to culture in vitro. OVA-specific Tr1 clones have been generated in vitro by culture of naive T cells in the presence of IL-10 and have been shown to be capable of inhibiting Th1-mediated colitis in a murine model. However, little is known about the induction of T regulatory cells in vivo, the antigens they recognized or the factors that control their differentiation from naive T cells.
Dendritic cells (DCs) are considered to be the key antigen-presenting cell for activation of naive T cells (7). Binding of conserved pathogen-derived molecules to pathogen recognition receptors (PRRs), including Toll-like receptors, on DCs activates transcription of genes coding for MHC and costimulatory molecules and in the production of inflammatory cytokines (8, 9). This process of maturation is accompanied by migration of DCs to the local lymph node where they function as an APC to activate naive T cells (7). Certain pathogen-derived molecules, notably LPS from Gram-negative bacteria and CpG motifs from bacterial DNA, can stimulate immature bone marrow–derived DCs into a phenotype, termed DC1, that selectively enhance the differentiation of naive T cells into Th1 cells (9, 10). Conversely cholera bacterial toxins and molecules from helminth pathogens appear to be capable of driving immature DCs into a phenotype, termed DC2, that selectively enhance Th2 induction (7, 10, 11). While there have been no definitive reports on a DC phenotype that drives Tr1 or Th3 cells, it has been shown that repeated stimulation of naive human T cells with allogeneic immature DCs preferentially activated IL-10–producing nonproliferating CD4+ T cells (12).
Bordetella pertussis causes a protracted and severe disease, which is often complicated by secondary infection and pneumonia, and can have a lethal outcome in young children (13). Recovery from infection is associated with the development of B. pertussis–specific Th1 cells (14, 15) and these cells play a critical role in clearance of the bacteria from the respiratory tract (14, 16, 17). However, antigen-specific Th1 responses in the lung and local lymph nodes are severely suppressed during the acute phase of infection (18). B. pertussis has evolved a number of strategies to circumvent protective immune responses (19). We have found that one virulence factor, filamentous hemagglutinin (FHA), is capable of inhibiting LPS-driven IL-12 production by macrophages, IL-12 and IFN-γ production in a murine model of septic shock (20), and Th1 responses to an unrelated pathogen, influenza virus, when administered simultaneously to the respiratory tract (21). FHA is considered to function primarily as an adhesin, mediating binding of B. pertussis to the β2-integrin (CR3, CD11b/CD18, αMβ2) via binding to leukocyte response integrin (αVβ3, CD61) and the integrin-associated protein (CD47) complex (22). In the present study we addressed the hypothesis that FHA may contribute to suppressed Th1 responses during acute infection with B. pertussis by the induction of T cells with regulatory activity, as a result of its interaction with cells of the innate immune system. We demonstrate that FHA interacts directly with DCs to induce IL-10 and inhibit LPS-induced IL-12 and inflammatory chemokine production. The DCs generated after interaction with FHA selectively stimulates the induction of Tr1 cells from naive T cells. Tr1 clones specific for FHA and pertactin (PRN) from B. pertussis were generated from the lungs of acutely infected mice. These Tr1 cells secreted high levels of IL-10 and inhibited protective Th1 responses against B. pertussis in vitro and in vivo. Our study demonstrates a novel function for Tr1 cells, exploited by a respiratory pathogen to evade protective immunity, and provides evidence that these regulatory cells are induced by DCs in which IL-10 production is activated and IL-12 suppressed after interaction with a pathogen-derived molecule.
Materials and Methods
Animals.
Female BALB/c mice purchased from Harlan UK. BALB/c mice expressing a transgene for the DO11.10 TCR specific for amino acids 323–339 of OVA and I-Ad (DO.11.10 TCR transgenic [Tg] mice; reference 23) were obtained from the Biomedical Services, John Radcliffe Hospital, Oxford, UK, with the permission of Dr. Fiona Powrie. All mice maintained according to EU regulations and experiments were performed under licence from the Department of Health and with approval of the NU1 Maynooth Biology Department ethics committee. Mice were 6–8 wk old at the initiation of experiments.
Bacteria and Reagents.
B. pertussis parental strain BP338 (Nalr derivative of Tohama 1) and FHA-depleted mutant BPM409 (24) were grown at 36°C in Stainer-Scholte medium. Heat-killed B. pertussis for use in T cell assays were prepared by incubation of cells at 90°C for 20 min and B. pertussis sonic extract was prepared as described previously (14). Purified FHA and PRN were prepared from B. pertussis Tohama 1 strain (14), and were free of other B. pertussis proteins by analysis on SDS-PAGE and free of LPS by analysis with an E-toxate kit (Sigma-Aldrich). E. coli LPS (serotype 127:B8), Phosphorothioate-stabilized oligodeoxynucleotide-containing CpG motifs (CpG-ODN) (5′GCTAGACGTTAGCGT) were synthesized by Sigma-Aldrich. OVA peptide 323–339 was synthesized by MWG-Biotech AG.
B. pertussis Respiratory Challenge.
Bacteria from a 48 h culture were concentrated to 2 × 1010/ml in PBS with 1% casein. Aerosol challenge was administered over 15 min using a nebulizer (0.5 ml/min). The course of B. pertussis infection was followed by performing CFU counts on lungs from groups of four mice at various times after aerosol challenge. Lungs were aseptically removed and homogenized in 1 ml of sterile physiological saline with 1% casein on ice. 100 μl of undiluted homogenate or of serially diluted homogenate from individual lungs was spotted in triplicate onto Bordet-Gengou agar plates, and the number of CFU was estimated after 4 d of incubation at 37°C. Results are reported as the mean number of B. pertussis CFU for individual lungs from four mice per experimental group per time point.
Effect of FHA on DC Cytokine Production and Maturation.
Bone marrow–derived immature DCs were prepared by culturing bone marrow cells obtained from the femur and tibia of BALB/c mice in RPMI-1640 and 10% FCS supplemented with 5–10% of a supernatant from a GM-CSF expressing cell line (provided by Nathalie Winter, Institute Pasteur, Paris, France, with permission of David Gray, University of Edinburgh, Edinburgh, UK). Cells were washed and recultured with fresh RPMI/10% FCS containing 10% GM-CSF cell supernatant every 3 d for a period of 8 d. Bone marrow–derived DCs (106/ml) were cultured at 37°C for 2 h in the presence or absence of FHA (0.01–5 μg/ml) before stimulation with LPS from Escherichia coli (1 μg/ml) and murine IFN-γ (20 ng/ml) or CpG-ODN (1 μg/ml). In certain experiments anti–IL-10 mAb (10 μg/ml) was added with the FHA. Alternatively, bone marrow–derived DCs were incubated with sonic extracts of B. pertussis BP338 or FHA− mutant BPM409. Supernatants were removed after 24 h for analysis of cytokine and chemokine concentrations or cells were recovered, washed, and used for Immunofluorescence analysis. IL-12p40, IL-12p70, IL-10, TGF-β, TNF-α, CCL3 (MIP-1α), CCL4 (MIP-1β), and MIP-2 concentrations were measured by immunoassay using pairs of antibodies, purchased from BD PharMingen or R&D Systems (IL-12p40 and p70), as described (20).
Generation of T Cell Clones from B. pertussis–infected Mice.
T cells were isolated from lung parenchyma and alveolar spaces from mice 3 wk after aerosol challenge with B. pertussis as described previously (18). Lung T cells (105/ml) were cultured with splenic APCs (irradiated spleen cells, 2 × 106/ml) and FHA (5 μg/ml) or PRN (5 μg/ml). IL-10 (200 pg/ml) was added with PRN but not with FHA for the first two rounds of antigen stimulation. After two rounds of antigen stimulation T cell lines were cloned by limiting dilution. T cell clones were generated and maintained by culture with FHA or PRN (1 μg/ml) and splenic APC for 4–5 d, followed by 5–7 d culture with feeder cells and IL-2. Th1 and Th2 clones specific for B. pertussis or influenza virus antigens were generated as described previously (25, 26) from spleens of mice that had recovered from respiratory infection with the respective pathogen.
T Cell Cytokine Production.
T cell clones (105/ml) and APCs (irradiated spleen cells 2 × 106/ml) or ex vivo spleen cells were cultured with medium only or B. pertussis antigen (106 killed bacteria/ml). Supernatants were removed after 1 d to test for IL-2 production or after 3 d to test for IL-4, IL-5, IL-10, IFN-γ, and TGF-β. IL-2 concentrations were determined using the CTLL bioassay and other cytokines by specific immunoassays as described (18). T cell proliferation was determined by [3H]thymidine incorporation as described (14). The effect of Tr1 cells on cytokine production by Th1 or Th2 cells was assessed in coculture experiments where the cells were cultured in the same wells or in a Transwell system. Tr1 clones (105/ml or 3 × 105/ml) were cultured alone or in the presence of Th1 or Th2 clones (105/ml) and APCs with the relevant antigen(s), either in the same wells or separated by a semi-permeable membrane. In certain experiments a neutralizing anti–IL-10 mAb (10 μg/ml) was added to the cultures. Supernatants were removed after 3 d and tested for cytokine production and cells were pulsed with [3H]thymidine after 4 d to test for proliferation.
Effect of Tr1 Clones on Th1 Responses and B. pertussis Infection In Vivo.
Spleen cells were prepared from naive BALB/c mice or mice that had recovered from infection with B. pertussis 6 wk after challenge (immune spleen cells). Adoptive cell transfers was performed into recipient mice that had been sublethally irradiated to ablate their immune system (14). Groups of 12 recipient mice, irradiated with 6 Gy total body irradiation 24 h earlier, were injected intravenously with either naive spleen cells (2 × 107), immune spleen cells (2 × 107) alone, or with Tr1 clones (2 × 105). Mice were challenge by aerosol inoculation with B. pertussis 4 h later (14). Four mice from each experimental group mice were killed 7, 14, and 21 d after challenge. Lungs were removed to determine B. pertussis CFU counts and spleen to determine T cell responses. A proportion of the naive and immune spleen cells, alone or with Tr1 clones, were also cultured with B. pertussis antigen before in vivo transfer to determine the effect of Tr1 on polyclonal T cell responses in vitro.
Bronchoalveolar Lavage.
Bronchoalveolar lavage (BAL) fluids were obtained by injecting and aspirating in 0.5 ml volumes (total 4–5 ml) of warm RPMI-1640 via cannulation of the trachea. Cells from the lavage fluids were recovered by centrifugation at 300 g for 5 min, and resuspended in RPMI-1640 with 8% FCS. For detection of cytokine concentrations, BAL fluids were concentrated fivefold using Centiprep-10 concentrators (Amicon Ltd.).
Generation of Tr1 Lines In Vitro from DO11.10 TCR Tg Mice.
GM-CSF amplified immature bone marrow–derived DCs were cultured for 24 h with OVA peptide (0, 0.5, 5, 50 μg/ml) and FHA (5 μg/ml), CpG-ODN (1 μg/ml), FHA, and anti–IL-10 (10 μg/ml) or medium only. Cells were washed and cultured at 105/ml with naive T cells purified from DO11.10 TCR Tg mice (106/ml). After 4 d, fresh medium and IL-2 was added to the cultures and fresh DCs, preincubated for 24 h with OVA peptide and modulators as for the primary stimulation, were added after a further 8 d. In the third and subsequent rounds of stimulation, surviving T cells were cultured at 105/ml with OVA peptide (0.5, 5, 50 μg/ml) and splenic APCs (2 × 106/ml). Supernatants were removed after the second round of antigen stimulation and concentrations of IL-4, IL-5, IL-10, and IFN-γ determined by immunoassay.
Immunofluorescence Analysis.
Phenotypic analysis of T cell clones was performed using PerCP-conjugated anti-CD3 (clone 145–2C11), PE-conjugated anti-CD28 (clone 37.51), anti-CCR5 (clone C34–3448), anti–CTLA-4 (clone UC10–4F10–11), PE/FITC-conjugated anti-CD4 (clone GK1.5), FITC-conjugated anti-CD25 (clone 7D4), and biotinylated rat anti–mouse T1/ST2 (clone 3E10 (27), provided by Millennium Pharmaceuticals Inc., Cambridge, MA) plus Extravidin-PE (Sigma-Aldrich). Expression of DC surface markers was assessed using biotinylated anti-CD11c (clone HL3) with Streptavidin-PerCP, PE-conjugated anti-CD80 (16–10A1), anti-CCR5 (clone C34–3448), FITC-conjugated anti-CD86 (clone GL1), and FITC-conjugated anti I-Ad/I-Ed (clone 2G9). All antibodies, except anti-T1/ST2, were purchased from BD PharMingen. Cells incubated with an isotype matched directly conjugated antibody with irrelevant specificity acted as a control. After incubation for 30 min at 4°C, cells were washed and Immunofluorescence analysis was performed on a FACScan™ (Becton Dickinson) and analyzed using CELLQuest™ software. 10,000 cells were analyzed per sample.
Statistical Analysis.
Results, expressed as mean ± SE, were compared using the Student's t test. P values <0.05 were considered statistically significant.
Results
FHA Stimulates IL-10 Production by DCs and Inhibits LPS-induced IL-12 and CCL3 Production.
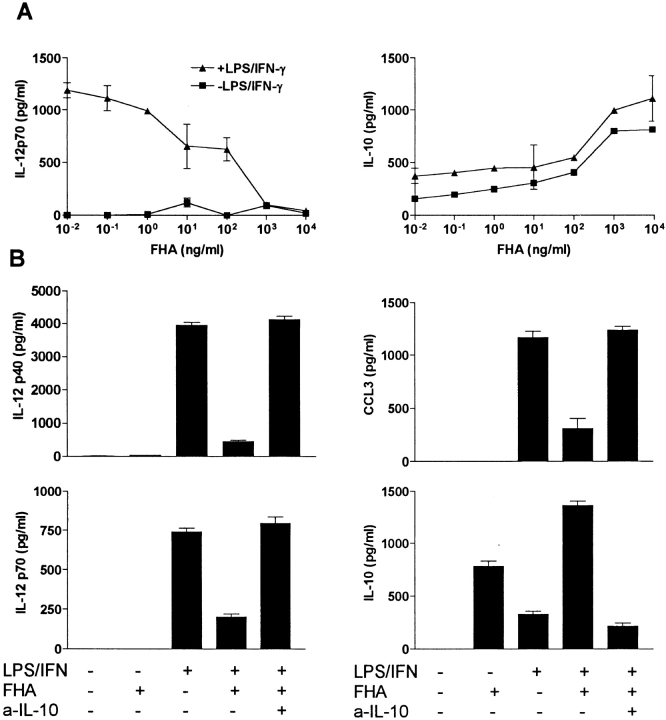
We have demonstrated previously that FHA inhibits macrophage IL-12 production in vitro and IL-12 and IFN-γ production in vivo in response to LPS (20). In this study we examined its effect on cytokine production and maturation of DCs. Incubation of immature bone marrow–derived DCs with FHA stimulated significant levels of IL-10 (Fig. 1 A). Preincubation of DCs with FHA suppressed LPS and IFN-γ–induced production of IL-12 p40 and IL-12p70 (Fig. 1 A) and the β-chemokine CCL3 (Fig. 1 B), but did not affect the production of TNF-α or MIP-2 or the viability of the DCs (data not shown). The suppression of IL-12 and CCL3 was reversed in the presence of a neutralizing anti–IL-10 antibody (Fig. 1 B). Incubation of immature bone marrow–derived DCs with LPS and IFN-γ (Fig. 2) or CpG-ODN (data not shown) for 24 h resulted in enhanced expression of MHC class II, B7–1, B7–2, CD40, and decreased expression of CCR5. Stimulation of DCs with FHA significantly enhanced CD86, moderately enhanced CD40, but did not affect expression of CD80 or CCR5 and compared with LPS and IFN-γ, FHA only had a marginal effect on MHC class II expression (Fig. 2). Thus, FHA-stimulated DCs have a distinct phenotype to the DC1 subtype and although they have certain features of immature DCs they may represent a distinct subtype of DCs.
Figure 1.
FHA inhibits DC IL-12 and CCL3 production by inducing IL-10. (A) Bone marrow–derived DCs were stimulated with FHA alone or with FHA after 2 h with LPS (1 μg/ml) and IFN-γ (20 ng/ml). After 24 h of culture, supernatants were removed and IL-12p70 and IL-10 were determined by specific immunoassays. (B) DCs were preincubated with FHA (5 μg/ml) with or without anti–IL-10, followed 2 h later with LPS (1 μg/ml) and IFN-γ (20 ng/ml). After 24 h of culture, supernatants were removed and IL-12p70, IL-12p40, CCL3, and IL-10 determined by specific immunoassays. Results are representative of six experiments.
Figure 2.
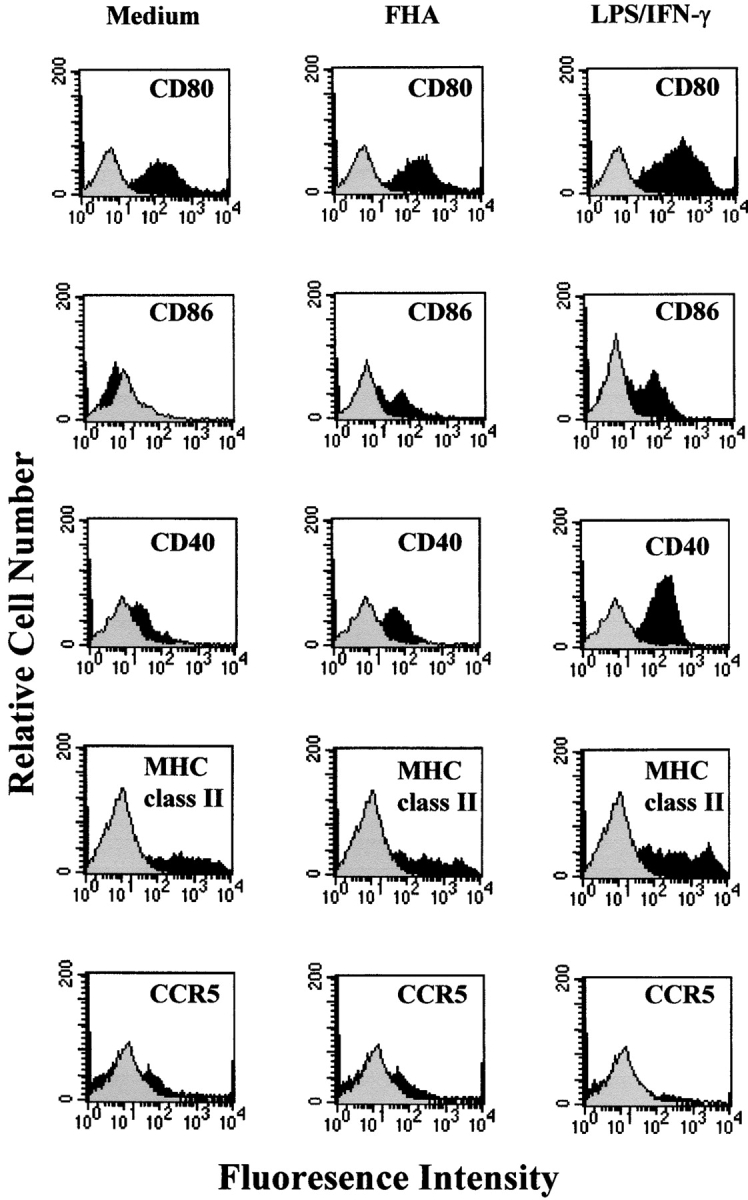
Influence of FHA on DC maturation. Bone marrow–derived DCs were stimulated with LPS (1 μg/ml) and IFN-γ (20 ng/ml), FHA (5 μg/ml), or medium. After 24 h of culture, cells were washed and immunofluorescence analysis performed for CD80, CD86, CD40, MHC class II, and CCR5 (black histograms) or isotype-matched control antibodies (gray histograms). Profiles are shown for a single experiment, and are representative from four experiments.
B. pertussis-specific Tr1 Clones Generated from the Respiratory Tract in the Presence of Exogenous or Endogenous IL-10.
As FHA stimulated IL-10 production (Fig. 1), which appears to be required for Tr1 induction (1), and we had previously shown that local Th1 responses were suppressed in vivo during acute infection with B. pertussis (18), we examined the hypothesis that the suppression was mediated by the induction of pathogen-specific Tr1 cells. Tr1 cell lines and clones specific for FHA and PRN were successfully generated from lung T cells isolated from mice 21 d after challenge with B. pertussis. Four PRN-specific Tr1-like T cell lines were generated from lung tissue by culturing purified lung T cells with antigen in the presence of IL-10 and restimulation with antigen, splenic APCs, and IL-10. Four FHA-specific Tr1-like T cell lines were generated from lung T cells in the absence of exogenous IL-10. Here the IL-10 was produced endogenously by FHA-stimulated APCs in the culture. Using the same culture protocols Tr1-type T cells could not be expanded from lungs of naive mice or from spleen of B. petrussis–infected or convalescent mice. PRN and FHA-specific T cell clones were generated from the established lung-derived T cell lines by cloning at limiting dilution. The cloning efficiency was low, with only two clones, from the four T cell lines at 10 cell/well (96 wells per line), successfully maintained in culture and expanded into 24-well plates and eventually into 25-ml flasks. Clone FHA1.1 was specific for FHA and clone TEK.1 was specific for PRN, neither clone responded to other B. pertussis or unrelated antigens (influenza virus, diphtheria toxoid, or ovalbumin). The Tr1 clones were found to be slow cycling cells and proliferated 10–20-fold less efficiently than Th1 clones. Nevertheless, they did secrete high levels of cytokines after stimulation with specific antigen and APCs.
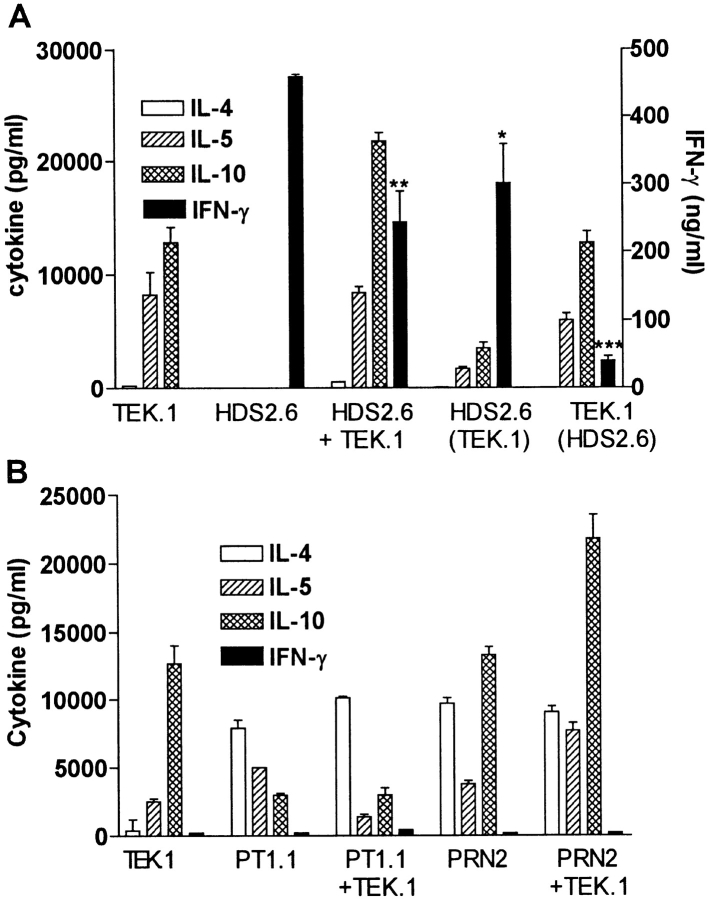
The Tr1 clones secreted high levels of IL-10 and IL-5, but very low levels of IFN-γ and undetectable IL-2 or IL-4 (Fig. 3 A). One of the two Tr1 clones examined in detail also secreted moderate levels of TGF-β. In contrast, Th1 clones generated from spleens of mice infected with B. pertussis or influenza virus antigens secreted IFN-γ and IL-2 but no IL-4, IL-5, IL-10, or TGF-β, whereas Th2 clones generated from spleen of mice infected with B. pertussis secreted high levels of IL-4, IL-5, moderate IL-10, and no IFN-γ, IL-2, or TGF-β (Fig. 3 A). A phenotypic analysis of the Tr1 clones demonstrated that they shared certain surface marker characteristics of both Th1 and Th2 cells (CD4+, CD25+), but could be distinguished from Th1 cells on the basis of the expression of T1/ST2 and from Th2 cells by the high expression of CCR5 (Fig. 3 B). Furthermore expression of CD28 and CTLA-4 was weak on Tr1 clones, significantly lower than that observed for Th1 or Th2 clones.
Figure 3.
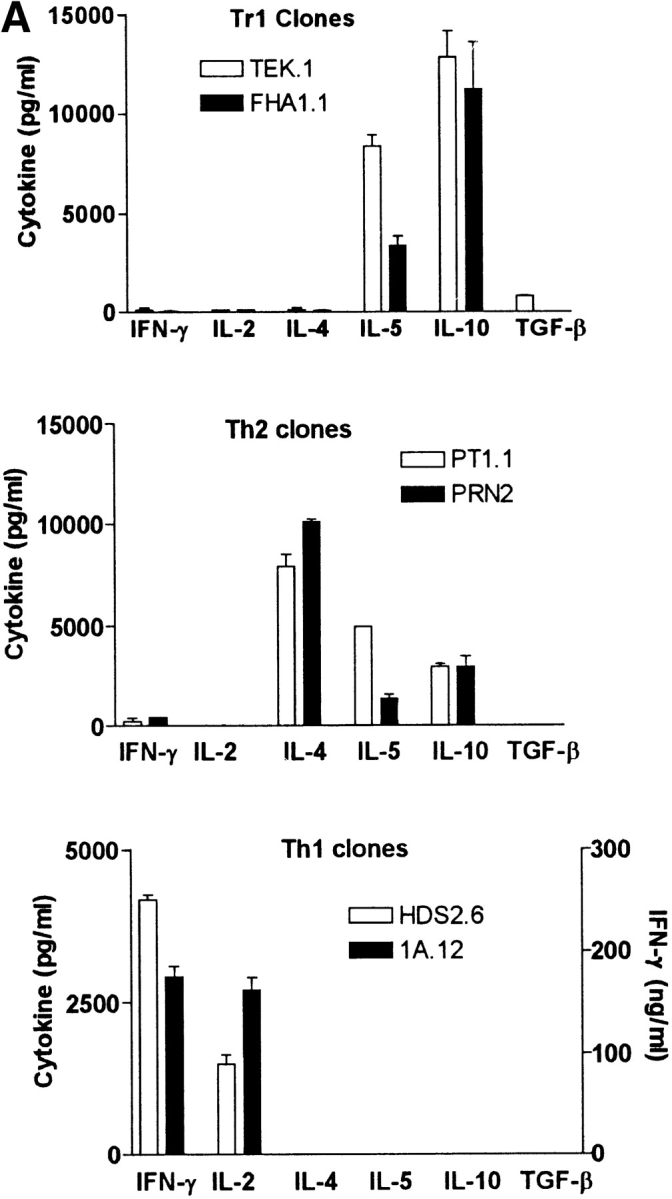
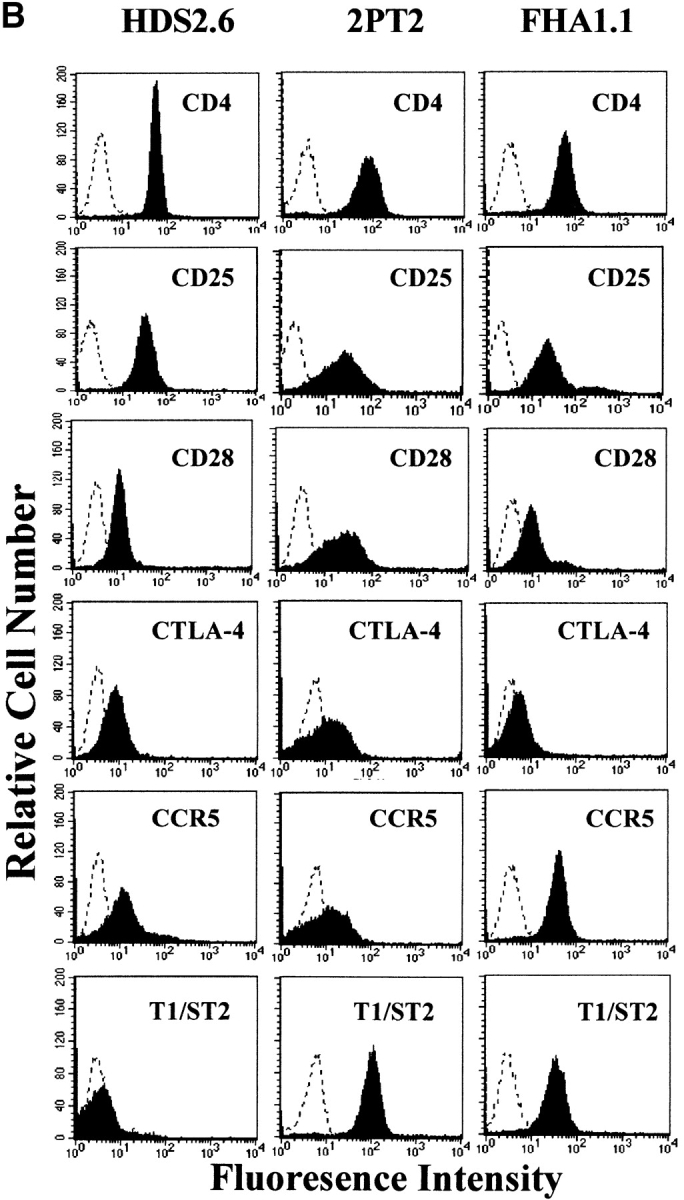
Cytokine production and phenotypic analysis of Tr1 clones generated from the respiratory tract of mice infected with B. pertussis. (A) Tr1 clones were generated from lungs of mice 21 d after aerosol challenge with B. pertussis. TEK.1, specific for PRN was cloned from a T cell line generated by stimulation with antigen, APCs, and IL-10 (200 pg/ml). FHA1.1, specific for FHA, was generated by stimulation with antigen and APCs in the absence of exogenous IL-10. Th1 clones, HDS2.6 and 1A.12, specific for influenza virus HA and B. pertussis, respectively, and Th2 clones PT1.1 and PRN2, specific for pertussis toxin and PRN, respectively, were generated from spleens of influenza virus or B. pertussis–infected mice. T cell clones were stimulated with antigen and APCs. IL-2 concentrations were determined in 24 h supernatants by CTLL bioassay and IFN-γ, IL-4, IL-5, and IL-10 concentrations in 3-d supernatants were determined by immunoassay. (B) Th1 clone HDS2.6, Th2 clone 2PT2 (specific for pertussis toxin), and Tr1 clone FHA1.1 were labeled with antibodies specific for CD4, CD25, CD28, CTLA4, CCR5, and T1/ST2 and immunofluoresence analysis performed on a FACScan™. The dotted line indicated staining with isotype matched control antibody. T cell clones were used at the end of the resting stage of the growth cycle, 7 d after addition of IL-2. Results are representative of three experiments and results for B are shown for one Tr1 clone only, but were essentially identical to that found for the second Tr1 clone.
Tr1 Clones Suppress Protective Th1, but Not Th2, Responses against B. pertussis.
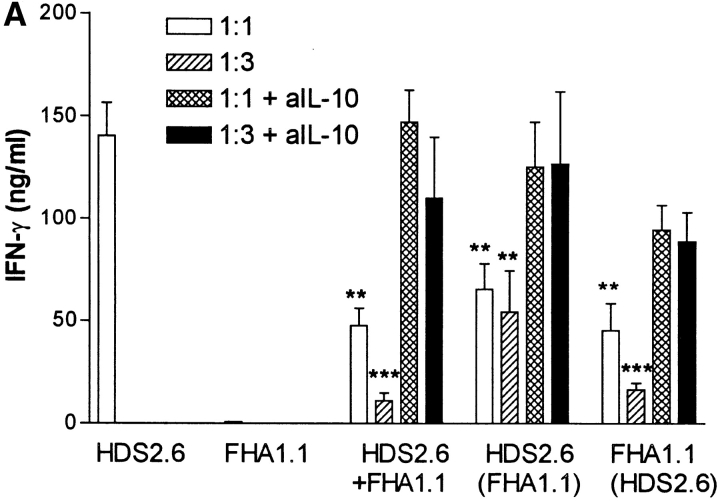
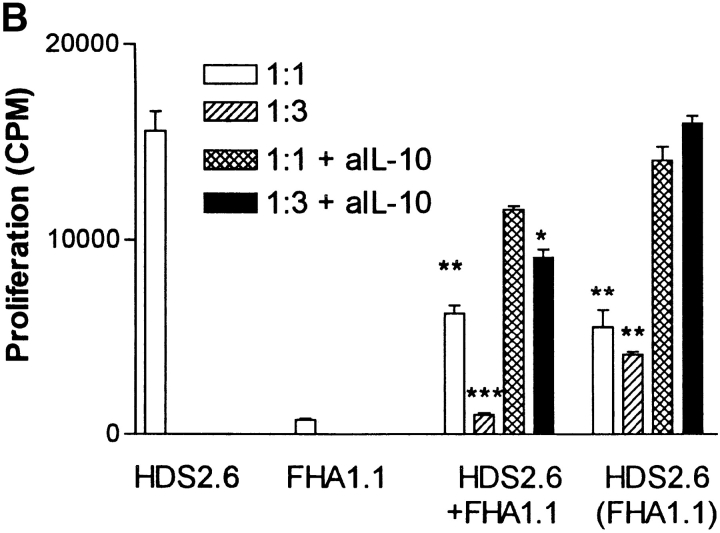
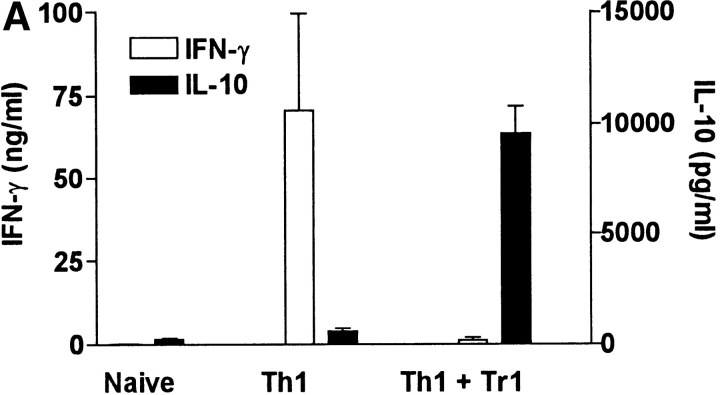
Having demonstrated that B. pertussis–specific Tr1 cells isolated from the lung during this stage of infection secreted IL-10 in response to specific antigen stimulation, we examined the hypothesis that suppression of local Th1 responses during B. pertussis infection (18) was mediated in part by Tr1 cells. We first demonstrated that the Tr1 clones were capable of inhibiting cytokine production by a Th1 clone against an unrelated antigen, influenza virus hemagglutinin (HA). IFN-γ production by the HA-specific Th1 clone HDS2.6 was significantly suppressed after coincubation with the PRN-specific Tr1 clone TEK.1 (Fig. 4 A) or FHA-specific Tr1 clone FHA1.1 (Fig. 5 A). Using Tr1 clone FHA1.1 we also observed significant inhibition of proliferation (Fig. 5 B) and IL-2 production by the Th1 clone (Th1 clone alone, 1,400 pg/ml IL-2; Th1 with Tr1, 150 pg/ml). The suppression was particularly pronounced when the ratio of Tr1:Th1 cells was increased from 1:1 to 3:1 (Fig. 5). Furthermore, the suppression was still observed in a Transwell system where the Th1 and Tr1 clones were separated by a semi-permeable membrane (Figs. 4 A and 5). Addition of a neutralizing anti–IL-10 antibody to the cultures abrogated the suppressive effect of the Tr1 clone (Fig. 5). Some reduction in IL-5 and IL-10 was observed across the membrane on the Th1, but not the Tr1, side of the membrane (Fig. 4 A). This may reflect incomplete equilibration of the cytokines across the membrane; in later experiments twice daily mixing of the cultures allowed more complete equilibration and less significant differences between cytokine concentrations either side of the membrane (Fig. 5, and data not shown). In contrast, the Tr1 clone had no effect on IL-4 production by Th2 clones specific for the same or unrelated B. pertussis antigens (Fig. 4 B).
Figure 4.
Tr1 clones inhibit cytokine production by Th1 but not Th2 clones. (A) PRN-specific Tr1 clone TEK.1 (105/ml) and influenza virus HA-specific Th1 clone HDS2.6 (105/ml) were cultured alone or together with PRN (5 μg/ml) and/or influenza virus (10 HAU/ml) and splenic APCs (2 × 106/ml). Supernatants were removed after 2–3 d and concentrations of IL-4, IL-5, IL-10, and IFN-γ determined by immunoassays. In the cocultures, Tr1 and Th1 cells were cultured in the same well, HDS2.6 plus TEK.1, or separated by a semi-permeable membrane, where supernatants were sampled from the Th1 side, HDS2.6 (TEK.1) or the Tr1 side, TEK.1 (HDS2.6). *P < 0.05, **P < 0.01, ***P < 0.001 HDS2.6 alone versus cocultured with TEK.1. (B) PRN-specific Tr1 clone TEK.1 was cultured with antigen alone or with PT-specific Th2 clone PT1.1 or PRN-specificTh2 clone PRN2 and cytokine production assessed by immunoassay. Results are representative of two experiments.
Figure 5.
Suppression of Th1 cell proliferation and IFN-γ production by Tr1 clone is mediated by IL-10. FHA-specific Tr1 clone FHA 1.1 and influenza virus HA-specific Th1 clone HDS2.6 (105/ml) were cultured alone (105/ml) or together at a ratio of 1:1 (105/ml) or 3:1 (3 × 105/ml: 105/ml) with FHA (5 μg/ml) and/or influenza virus (10 HAU/ml) and splenic APCs (2 × 106/ml). Cocultures were also performed in the presence a neutralizing anti-IL-10 antibody (10 μg/ml). Supernatants were removed after 3 d and concentrations of IFN-γ determined by immunoassays. (A) Cells were harvested after 4 d and proliferation assessed by [3H]thymidine incorporation (B). In the cocultures Tr1 and Th1 cells were cultured in the same well, HDS.6 plus FHA1.1, or separated by a semi-permeable membrane, where supernatants was sampled from the Th1 side, HDS2.6 (FHA1.1) or the Tr1 side, FHA1.1 (HDS2.6). Proliferation could only be assessed on the Th1 side of the membrane. *P < 0.05, **P < 0.01, ***P < 0.001 HDS2.6 alone versus cocultured with FHA1.1. Results are representative of two experiments.
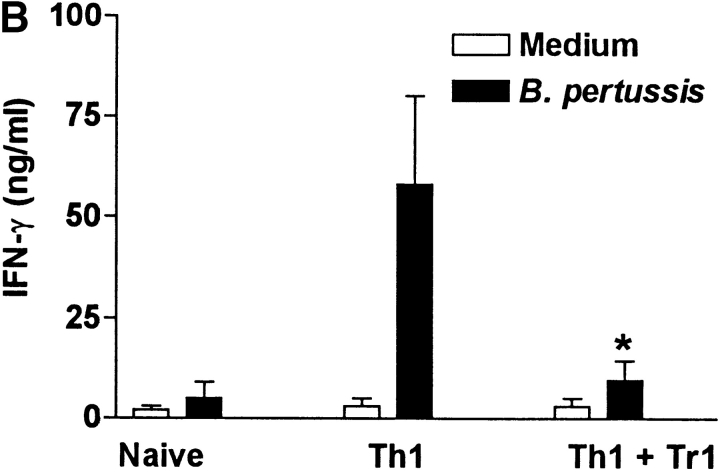
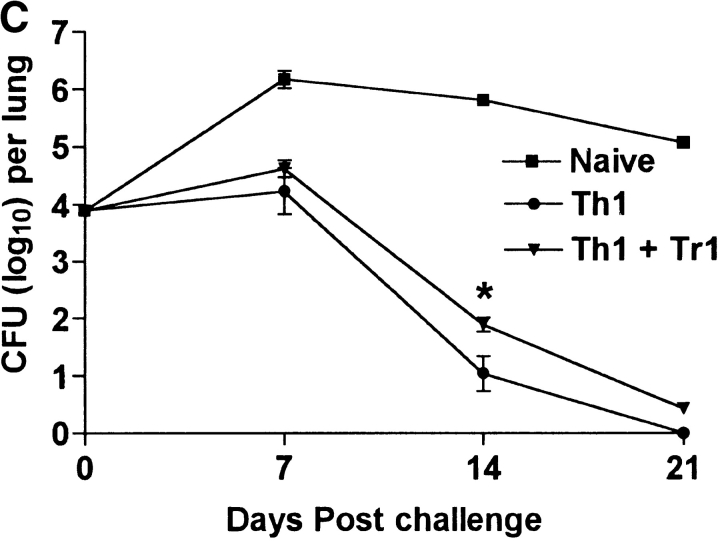
We next examined the effect of the Tr1 clones on B. pertussis–specific Th1 responses in vitro and in vivo. Splenic T cells from mice that had cleared an infection with B. pertussis secreted high levels of IFN-γ and low or undetectable IL-4, IL-5, or IL-10, (Fig. 6 A), a classic Th1-type response; we have previously demonstrated that CD4+ T cells are the major source of this IFN-γ (14). In contrast, spleen cells from naive mice did not secrete cytokines in response to B. pertussis antigen. Coincubation of the B. pertussis–specific Th1 cells with Tr1 clone TEK.1 completely abrogated IFN-γ production. In contrast, significant levels of IL-10 were observed in the supernatants of the cocultured cells (Fig. 6 A). The effect of Tr1 cells on B. pertussis–specific Th1 cells and protection in vivo was determined using adoptive transfer experiments with sublethally irradiated mice. After transfer of T cells from naive mice, IFN-γ could only be detected after 3 wk (data not shown) and the course of infection was severe, with significant numbers of bacteria still remaining in the respiratory tract at 3 wk (Fig. 6 C). Adoptive transfer of B. pertussis–specific Th1 cells was accompanied by significant B. pertussis–specific IFN-γ production by 7 d (Fig. 6 B) and complete bacterial clearance by 3 wk (Fig. 6 C). After coadministration with Tr1 cells, B. pertussis–specific IFN-γ was significantly suppressed (Fig. 6 B) and the rate of bacterial clearance was delayed (Fig. 6 C). The poorer proliferative capacity of the Tr1, compared with Th1, clone may have compromised their ability to exert more significant effect on the course of infection. Nevertheless the numbers of bacteria in the lungs was 4–8-fold greater after transfer of the Tr1 and Th1 cells than with the B. pertussis–specific Th1 cells alone (Fig. 6 C).
Figure 6.
Tr1 clones suppress B. pertussis–specific IFN-γ production in vitro and in vivo. (A) Spleen cells from B. pertussis convalescent mice 6 wk after challenge (designated Th1) were cultured at 2 × 106/ml with antigen (B. pertussis sonic extract, 5 μg/ml) alone or with Tr1 clone TEK.1 (105/ml) and PRN (5 μg/ml). Spleen cells from naive mice served as a control. Supernatants were removed after 3 d and tested for IFN-γ and IL-10 by immunoassay. (B and C) Spleen cells from naive or convalescent mice (designated Th1) were transferred into sublethally irradiated BALB/c mice (2 × 107 per mouse) alone or with Tr1 clone TEK.1 (2 × 105 per mouse). Mice were challenged with B. pertussis and killed 7, 14, and 21 d later. (B) Spleen cells were isolated after 7 d and stimulated with B. pertussis antigen and IFN-γ concentrations determined in supernatants 3 d later. (C) Lungs were removed and the number of viable B. pertussis determined by performing CFU counts. Results are mean (SD) for four mice per group at each time point. *P < 0.05, Th1 vs. Th1 plus Tr1. The results shown in A are representative of two experiments and due to the large numbers of cells required the experiment shown in B and C were performed once.
Enhanced Th1 Responses and Bacterial Clearance with B. pertussis FHA Mutant.
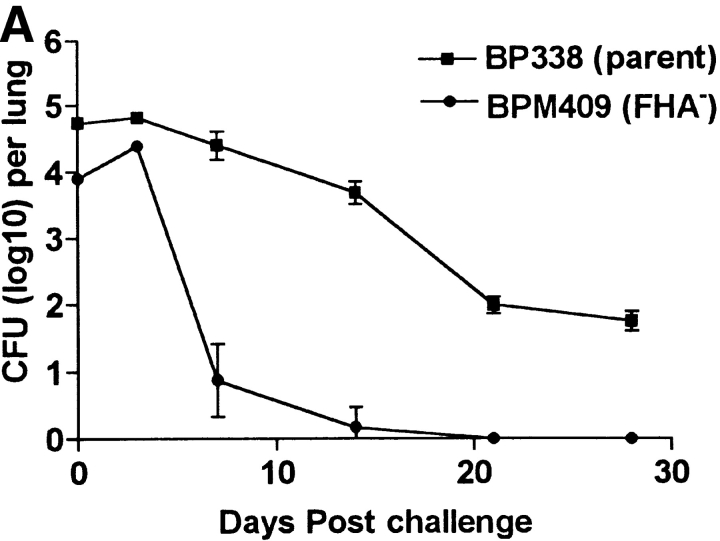
To provide further evidence that FHA modulation of regulatory cytokine production, either directly through binding to DCs, or indirectly through the induction of Tr1 cells, may contribute to the immunosuppression observed we examined immune responses and the course of infection with an FHA− mutant of B. pertussis, BPM409. The parent B. pertussis strain, BP338, caused a protracted infection in naive mice, with bacteria still detectable in the lungs for at least 5 wk after aerosol challenge (Fig. 7 A). In contrast, the FHA− mutant BPM409 was completely cleared from the lungs within 3 wk of infection (Fig. 7 A). The initial rise in bacterial counts after challenge with the FHA− mutant suggests that reduced adherence to the cells of the respiratory tract does not account for all of the loss of virulence observed.
Figure 7.
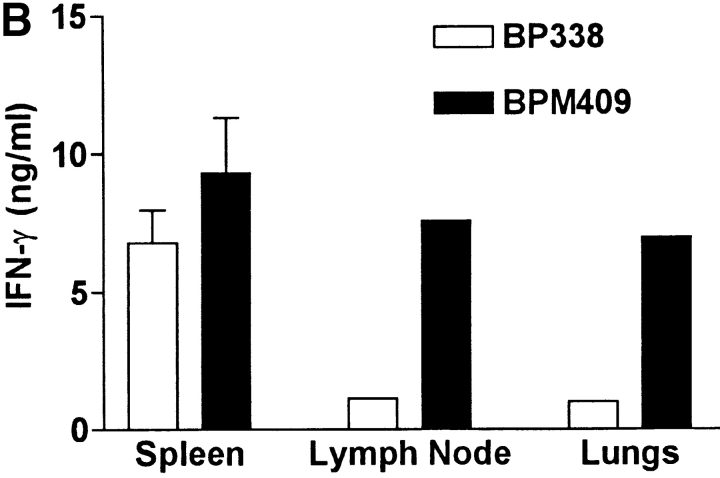
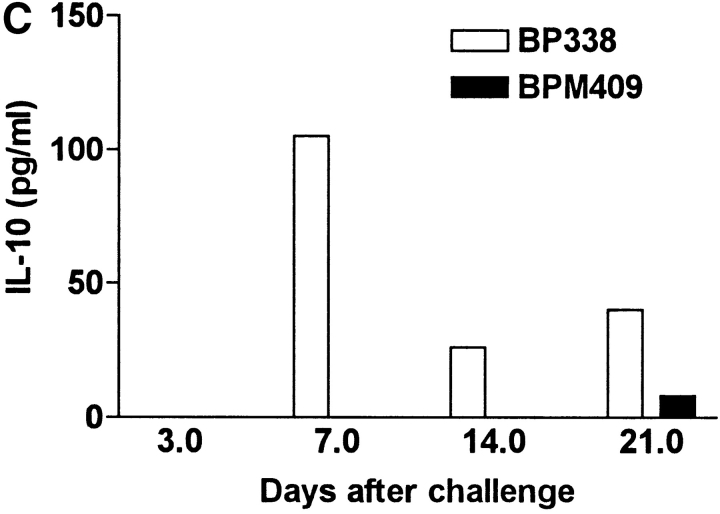
Deletion of FHA attenuates local immunosuppression and enhances B. pertussis clearance form the lungs. Groups of 24 BALB/c mice were challenged by exposure to an aerosol of B. pertussis BP338 or FHA− mutant BPM409. (A) The course of infection was followed by performing CFU counts on the lungs of four mice per group at 3 h, 7, 14, 21, and 28 d after challenge. (B) Spleen, lymph nodes, and lung mononuclear cells were prepared from mice 21 d after challenge and antigen-specific IFN-γ production was tested by culturing with B. pertussis antigen and assessing cytokine levels in supernatants 3 d later. (C) IL-10 concentrations in BAL fluid from mice 3–21 d after infection with B. pertussis BP338 or FHA− mutant BPM409. Results are representative of four experiments.
An examination of local and systemic immune responses in infected mice demonstrated that B. pertussis–specific IFN-γ production was significantly stronger in cells from the lungs and local lymph node, but not significantly different in spleens of FHA− mutant compared with the parent B. pertussis strain (Fig. 7 B). Furthermore, significant levels of IL-10 were detected in BAL fluid during the acute phase of infection with the parental BP338 strain, but not with the FHA− mutant BPM409 (Fig. 7 C). A role for FHA in suppression of protective Th1 responses during acute infection with B. pertussis in vivo was compatible with our observation that sonic extracts of FHA− mutant BPM409 stimulated higher IL-12 production by bone marrow–derived DCs than the parent strain BP338 (BP338, 710 ± 80 pg/ml; BPM409, 2,100 ± 120).
FHA-stimulated DCs Direct Naive T Cells Into a Regulatory Phenotype In Vitro.
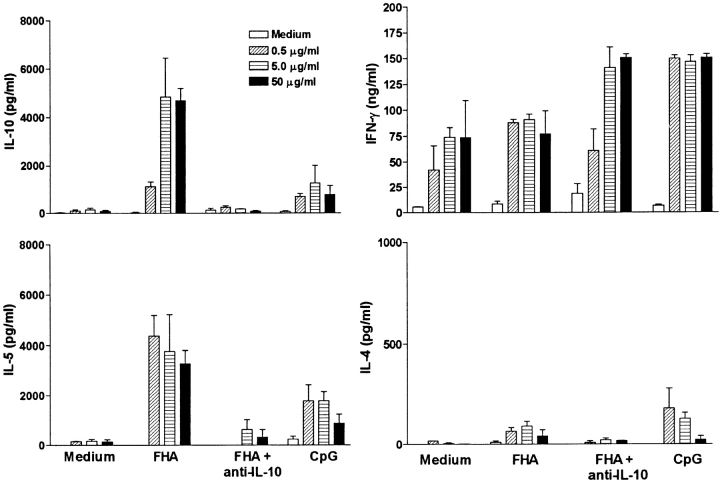
Having already demonstrated that FHA induced Tr1 cells in vivo and modifies cytokine production and costimulatory molecule expression on DCs, we sought to provide further evidence for the hypothesis that FHA may drive DCs into a phenotype that selectively enhances the induction of Tr1 cells. In these studies we used the DO11.10 OVA TCR Tg mice as a source of naive T cells. Immature bone marrow–derived DCs expanded for 8 d in the presence of GM-CSF were cultured for 24 h with OVA peptide (0, 0.5, 5, or 50 μg/ml) alone or with CpG ODN, FHA, or FHA and anti–IL-10 antibody and used as APCs for purified T cells from DO11.10 TCR Tg mice. DCs matured with CpG ODN selectively enhanced the induction of Th1-type cells, with high IFN-γ and low levels of IL-4, IL-5 and IL-10 (Fig. 8). In contrast, T cells cultured with DCs that were stimulated with OVA peptide and FHA directed the induction of Tr1-type T cells, producing high IL-10, low IL-4, and moderate levels of IL-5 and IFN-γ. Stable OVA-specific CD4+ high IL-10–secreting Tr1 cell lines were established from these cultures by periodic restimulation with OVA peptide and splenic APCs (data not shown). An examination of the suppressive effect of an OVA-specific Tr1 line on the influenza HA-specific Th1 clone revealed significant inhibition of IFN-γ production (Th1 alone, 140 ± 16; Th1 plus Tr1, 95 ± 9: Th1 plus Tr1 plus anti–IL-10, 173 ± 35 ng/ml). When anti–IL-10 antibody was added to DCs during the 24 h incubation with OVA and FHA, IL-10 and IL-5 production by DO11.10 T cells was almost completely abrogated, whereas IFN-γ production was significantly elevated (Fig. 8). This suggests that IL-10 is an essential differentiation factor for Tr1 cells. Taken together with our findings on B. pertussis described above, our results demonstrate that the induction of Tr1 cells is controlled by a subtype of DC that secretes IL-10.
Figure 8.
FHA stimulates DCs to direct naive T cells to a Tr1 subtype. OVA-specific CD4+ T cell lines were generated in vitro by stimulating purified naive T cells from DO11.10 TCR Tg mice with DCs preincubated for 24 h with OVA peptide (0, 0.5, 5, 50 μg/ml) and FHA (5 μg/ml), CpG-ODN (1 μg/ml), FHA, and anti–IL-10 (10 μg/ml) or medium only. After two rounds of antigen stimulation supernatants were removed and concentrations of cytokines determined by immunoassay. Results are representative of two experiments.
Discussion
Distinct subpopulations of CD4+ T cells have been shown to perform immunoregulatory functions in vitro and in vivo, inhibiting Th1-mediated responses, either through cell to cell contact or by releasing suppressive cytokines (1–6, 28–31). It has been suggested that regulatory T cells that secrete high levels of IL-10 or TGF-β, termed Tr1 and Th3 cells, respectively, may be prominent in the gut and in other mucosal tissues, where they play key roles in the maintenance of self-tolerance and the prevention of autoimmune diseases (4). Although it has been speculated that regulatory T cells may be directed against self-antigens, bacteria, or parasites, little is know about their antigen-specificity or induction in vivo (1, 28). The present study describes for the first time the generation of Tr1 clones specific for a infectious pathogen and demonstrate that the induction of Tr1 cells in the respiratory tract is directed by a DC that secretes IL-10 in response to a pathogen-derived molecule.
As a result of their poor proliferative capacity and great difficulty in expanding these cells in culture, only a small number of groups have successfully cultured antigen-specific Tr1 clones, mostly from naive T cells after repeated stimulation in vitro in the presence of IL-10 (1, 3). A number of studies have also been performed with polyclonal populations of T cells with regulatory activity, defined on the basis of their specific surface marker expression. CD4+CD25+ regulatory T cells, which represent 5–10% of CD4+ T cells from spleen and lymph nodes of normal mice, can suppress organ-specific autoimmune disease in mice (29, 30). T cells with a CD4+CD38+CD45RBlow phenotype have also been shown to function as regulatory cells in vivo, preventing colitis induced by CD4+ CD45RBhi T cells (28, 30). The Tr1 clones generated in the present study were CD4+, CD25+, CD28low, CTLAlow, CCR5+, T1/ST2+. The expression of T1/ST2 and CCR5 markers previously shown to be preferentially expressed on Th2 (32) and Th1 (33, 34) cells, respectively, suggesting that they represent a phenotypically, as well as functionally, distinct subtype of CD4+ T cells. T1/ST2 is a member of the IL-1 receptor family, but its ligand and precise function is still unknown. However, it has been shown to play a critical role in Th2 responses, especially in the lung, and is expressed on in vitro expanded Th2, but not Th1, cells (27, 35). As IL-10 and IL-5 production is common to both populations, and T1/ST2 is expressed on Tr1 as well as Th2 cells, it is tempting to suggest that T1/ST2 may be involved in signaling for these cytokines. The low expression of CTLA4 and CD28 is compatible with a phenotype of T cell found in the lungs during B. pertussis infection (18); expression of other costimulatory molecules is under investigation. The Tr1 clones described in this report secreted high levels of IL-10 and lower levels of TGF-β and IL-5 and suppressed proliferation, IFN-γ and IL-2 production by Th1 cells. Our results demonstrate for the first time that pathogen-specific Tr1 cells suppress Th1 responses against distinct antigens on the same or unrelated pathogens. Unlike the OVA-specific Tr1 clones generated in vitro (3), our Tr1 clones did not suppress cytokine production by Th2 cells and unlike the CD4+CD38+ CD45RBlow regulatory cells (28, 30) cell-to-cell contact was not the predominant mechanism. Suppression was observed across a semipermeable membrane and was completely reversed across the membrane and substantially reversed in cocultures by anti–IL-10, suggesting that bystander suppression was largely mediated by IL-10.
Little is known about APCs and cytokines that promote the induction of regulatory T cells in vivo. The production of IL-12 and IL-4 by DCs or other innate cells and activation of DC maturation into DC1 and DC2 by binding of pathogen-derived molecules appears to be a key process in directing naive T cells to differentiate into Th1 and Th2 cells. LPS, dsRNA, and CpG ODN promote maturation of DC1, which direct naive T cells to Th1 subtype (7, 9, 10). In contrast, phosphorylcholine-containing glycoproteins derived from nematode parasites, cholera toxin or yeast hyphae promote DC2, which selectively activate Th2 cells (7, 11, 36). The results of the present study demonstrate that activation of DCs that secrete IL-10, but not IL-12, can direct naive T cells to a Tr1 subtype and that the pathogen-derived molecule FHA from B. pertussis can provide the stimulus. FHA binds to leukocyte response integrin (αVβ3, CD61) and integrin-associated protein (CD47) complex and stimulates bacterial adherence to the β2-integrin CR3 (CD11b/CD18, αMβ2) (22). Engagement of CR3 or CD47 on macrophages or DCs with specific antibodies or natural ligands has been shown to suppress IL-12 production (37–39), and this may represent a physiological phenomenon for normal control of immune responses in vivo. Our study demonstrates that B. pertussis FHA can exploit this function by binding to these integrins on DCs, inducing the production of IL-10, and inhibiting inflammatory chemokine and IL-12 production and other aspects of DC1 maturation. It has been suggested that immature DCs may promote the expansion of IL-10–secreting or regulatory T cells (12, 40). While FHA-activated DCs may have features of immature DCs, they appear to represent a distinct subtype of DCs, with enhanced expression of CD86 and CD40 over that seen on immature DCs and in addition secrete high levels of IL-10 after appropriate stimulation.
Analagous to the role of IL-12 in driving Th1 responses, IL-10 appears to be the critical cytokine for the induction of Tr1 cells. Interestingly most Tr1 clones generated to date have employed IL-10 as a stimulating cytokine in the culture medium (1, 3). The PRN-specific Tr1 clones generated in this study also required IL-10, but significantly, the FHA-specific Tr1 clones were generated in the absence of exogenous IL-10; here the IL-10 was produced endogenously by FHA-stimulated APCs. Addition of an anti–IL-10 antibody during culture of DCs with FHA and OVA peptide abrogated the ability of the DCs to activate OVA-specific Tr1 cells from the DO11.10 TCR Tg mice. However, IL-10 was not required for continuous culture of PRN- or FHA-specific Tr1 clones. This suggests that IL-10 is a critical differentiation but not growth factor for Tr1 cells. Recently it has been demonstrated that human CD4+ T cells primed in the presence of IL-10 and IFN-α differentiate into Tr1 cells (41). Immature DCs from the respiratory tract express high levels of IL-10 mRNA (42). Likewise, immature DCs from liver (43) or Peyer's patch (44) secrete high levels of IL-10 and selectively induce IL-10 secreting alloreactive T cells or Th2 cells, respectively. This may represent the default position at a mucosal surface, resulting in a natural propensity to stimulate regulatory T cells after stimulation with an appropriate self or foreign antigen, a strategy for the maintenance of immunological tolerance. We found that Tr1 clones could be generated at low frequency from the lungs of B. pertussis–infected mice and Th1 clones at higher frequency from spleen, but B. pertussis–specific T cells with Tr1-type cytokine profile could not be generated from spleen of infected or convalesecent mice or from the lungs of naive mice. These findings suggests that in addition to the maintenance of tolerance, induction of regulatory T cells at mucosal surfaces may be a strategy evolved by pathogens to suppress protective Th1 responses at the local site of infection.
Antigen-specific Th1 responses in the lungs and local lymph nodes are suppressed during acute infection with B. pertussis (18). We found that deletion of FHA from B. pertussis partially reversed the immunosuppression in vivo and enhanced the clearance of B. pertussis from the lungs. Although we do not rule out the role of FHA in mediating adherence to cells in the respiratory tract (22) or the role of other virulence factors (19), whose influence would be removed as a result of earlier bacterial clearance, our data suggests that FHA has a major influence on immune responses to B. pertussis or other pathogens in the lung. Indeed we have already demonstrated that intranasally administered FHA with influenza virus can suppress virus-specific IFN-γ production in the local lymph nodes (21). During active infection with B. pertussis, the bacteria secrete high levels of FHA, which bind to CD61–CD47 complex and CD11b/CD18 (22) expressed on DC (39). Immature DCs are normally resident in the respiratory tract of rats and mice (41, and unpublished data). We have observed transient recruitment and maturation of DCs in the lungs after infection with B. pertussis and maturation was more pronounced in mice infected with the FHA− mutant (unpublished data). Furthermore, IL-12 production by DCs in vitro was stronger with the FHA− mutant than with the parent B. pertussis strain. Taken together with our demonstration of inhibition of IL-12 and inflammatory chemokines and stimulation of IL-10–producing DCs that directs the induction of Tr1 cells, the net effect of FHA is to inhibit the induction, activation and recruitment of Th1 cells. This represents a novel immune subversion strategy employed by a respiratory tract pathogen to prolong its survival in the host.
Acknowledgments
We thank T. Coyle, D. Gray, F. Powrie, and N. Winter for reagents and mice and E. Lavelle for help with dendritic cell culture.
This work was supported by grants from The Wellcome Trust, the Health Research Board of Ireland, and Science Foundation Ireland.
Footnotes
Abbreviations used in this paper: BAL, bronchoalveolar lavage; CpG-ODN, oligodeoxynucleotide-containing CpG motifs; DC, dendritic cell; FHA, filamentous hemagglutinin; HA, hemagglutinin; PRN, pertactin; Tg, transgenic; Tr1, T regulatory 1.
References
- 1.Groux, H., A. O'Garra, M. Bigler, M. Rouleau, S. Antonenko, J.E. De Vries, and M.G. Roncarolo. 1997. A CD4+ T-cell subset inhibits antigen-specific T-cell responses and prevents colitis. Nature. 389:737–742. [DOI] [PubMed] [Google Scholar]
- 2.Cavani, A., F. Nasorri, C. Prezzi, S. Sebastiani, C. Albanesi, and G. Girolomoni. 2000. Human CD4+ T lymphocytes with remarkable regulatory functions on dendritic cells and nickel-specific Th1 immune responses. J. Invest. Dermatol. 114:295–302. [DOI] [PubMed] [Google Scholar]
- 3.Cottrez, F., S.D. Hurst, R.L. Coffman, and H. Groux. 2000. T regulatory cells 1 inhibit a Th2-specific response in vivo. J. Immunol. 165:4848–4853. [DOI] [PubMed] [Google Scholar]
- 4.Roncarolo, M.G., and M.K. Levings. 2000. The role of different subsets of T regulatory cells in controlling autoimmunity. Curr. Opin. Immunol. 12:676–683. [DOI] [PubMed] [Google Scholar]
- 5.Chen, Y., V.K. Kuchroo, J. Inobe, D.A. Hafler, and H.L. Weiner. 1994. Regulatory T cell clones induced by oral tolerance: suppression of autoimmune encephalomyelitis. Science. 265:1237–1240. [DOI] [PubMed] [Google Scholar]
- 6.Fukaura, H., S.C. Kent, M.J. Pietrusewicz, S.J. Khoury, H.L. Weiner, and D.A. Hafler. 1996. Induction of circulating myelin basic protein and proteolipid protein-specific transforming growth factor-β1-secreting Th3 T cells by oral administration of myelin in multiple sclerosis patients. J. Clin. Invest. 98:70–77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Moser, M., and K.M. Murphy. 2000. Dendritic cell regulation of TH1-TH2 development. Nat. Immunol. 1:199–205. [DOI] [PubMed] [Google Scholar]
- 8.Medzhitov, R., and C.A. Janeway. 1997. Innate immunity: the virtues of a nonclonal system of recognition. Cell. 91:295–298. [DOI] [PubMed] [Google Scholar]
- 9.Hemmi, H., O. Takeuchi, T. Kawai, T. Kaisho, S. Sato, H. Sanjo, M. Matsumoto, K. Hoshino, H. Wagner, K. Takeda, and S. Akira. 2000. A Toll-like receptor recognizes bacterial DNA. Nature. 408:740–745. [DOI] [PubMed] [Google Scholar]
- 10.Whelan, M., M.M. Harnett, K.M. Houston, V. Patel, W. Harnett, and K.P. Rigley. 2000. A filarial nematode-secreted product signals dendritic cells to acquire a phenotype that drives development of Th2 cells. J. Immunol. 164:6453–6460. [DOI] [PubMed] [Google Scholar]
- 11.Gagliardi, M.C., F. Sallusto, M. Marinaro, A. Langenkamp, A. Lanzavecchia, and M.T. De Magistris. 2000. Cholera toxin induces maturation of human dendritic cells and licences them for Th2 priming. Eur. J. Immunol. 30:2394–2403. [DOI] [PubMed] [Google Scholar]
- 12.Jonuleit, H., E. Schmitt, G. Schuler, J. Knop, and A.H. Enk. 2000. Induction of interleukin 10-producing, nonproliferating CD4+ T cells with regulatory properties by repetitive stimulation with allogeneic immature human dendritic cells. J. Exp. Med. 192:1213–1222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cherry, J.D., P.A. Brunel, G.S. Golden, and D.T. Karzon. 1998. Report of the task force on pertussis immunization - 1988. Pediatrics. 81:939–984. [Google Scholar]
- 14.Mills, K.H.G., A. Barnard, J. Watkins, and K. Redhead. 1993. Cell-mediated immunity to Bordetella pertussis: role of Th1 cells in bacterial clearance in a murine respiratory infection model. Infect. Immun. 61:399–410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ryan, M., G. Murphy, L. Gothefors, L. Nilsson, J. Storsaeter, and K.H.G. Mills. 1997. Bordetella pertussis respiratory infection in children is associated with preferential activation of type 1 T helper cells. J. Infect. Dis. 175:1246–1250. [DOI] [PubMed] [Google Scholar]
- 16.Mahon, B.P., B.J. Sheahan, F. Griffin, G. Murphy, and K.H.G. Mills. 1997. Atypical disease after Bordetella pertussis respiratory infection of mice with targeted disruptions of interferon-γ receptor or immunoglobulin μ chain genes. J. Exp. Med. 186:1843–1851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Mills, K.H.G., M. Ryan, E. Ryan, and B.P. Mahon. 1998. A murine model in which protection correlates with pertussis vaccine efficacy in children reveals complementary roles for humoral and cell-mediated immunity in protection against Bordetella pertussis. Infect. Immun. 66:594–602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.McGuirk, P., B.P. Mahon, F. Griffin, and K.H.G. Mills. 1998. Compartmentalization of T cell responses following respiratory infection with Bordetella pertussis: hyporesponsiveness of lung T cells is associated with modulated expression of the co-stimulatory molecule CD28. Eur. J. Immunol. 28:153–163. [DOI] [PubMed] [Google Scholar]
- 19.Mills, K.H.G. 2001. Immunity to Bordetella pertussis Microbes Infect. 3:293–304. [DOI] [PubMed] [Google Scholar]
- 20.McGuirk, P., and K.H.G. Mills. 2000. Direct anti-inflammatory effect of a bacterial virulence factor: IL-10-dependent suppression of IL-12 production by filamentous hemagglutinin from Bordetella pertussis. Eur. J. Immunol. 30:415–422. [DOI] [PubMed] [Google Scholar]
- 21.McGuirk, P., P.A. Johnson, E.J. Ryan, and K.H.G. Mills. 2000. Filamentous hemagglutinin and pertussis toxin from Bordetella pertussis modulate immune responses to unrelated antigens. J. Infect. Dis. 182:1286–1289. [DOI] [PubMed] [Google Scholar]
- 22.Ishibashi, Y., S. Claus, and D.A. Relman. 1994. Bordetella pertussis filamentous hemagglutinin interacts with a leukocyte signal transduction complex and stimulates bacterial adherence to monocyte CR3 (CD11b/CD18). J. Exp. Med. 180:1225–1233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Murphy, K.M., A.B. Heimberger, and D.Y. Loh. 1990. Induction by antigen of intrathymic apoptosis of CD4+ CD8+TCRlo thymocytes in vivo. Science. 250:1720–1723. [DOI] [PubMed] [Google Scholar]
- 24.Ewanowich, C.A., A.R. Melton, A.A. Weiss, R.K. Sherburne, and M.S. Peppler. 1989. Invasion of HeLa 229 cells by virulent Bordetella pertussis. Infect. Immun. 57:2698–2704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Barnard, A., B.P. Mahon, J. Watkins, K. Redhead, and K.H.G. Mills. 1996. Th1/Th2 cell dichotomy in acquired immunity to Bordetella pertussis: variables in the in vivo priming and in vitro cytokine detection techniques affect the classification of T-cell subsets as Th1, Th2 or Th0. Immunology. 87:372–380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Johnson, P.A., M.A. Conway, J. Daly, C. Nicolson, J. Robertson, and K.H.G. Mills. 2000. Plasmid DNA encoding influenza virus haemagglutinin induces Th1 cells and protection against respiratory infection despite its limited ability to generate antibody responses. J. Gen. Virol. 81:1737–1745. [DOI] [PubMed] [Google Scholar]
- 27.Coyle, A.J., C. Lloyd, J. Tian, T. Nguyen, C. Erikkson, L. Wang, P. Ottoson, P. Persson, T. Delany, S. Lehar, et al. 1999. Crucial role of the interleukin 1 receptor family member T1/ST2 in T helper cell type 2-mediated lung mucosal immune responses. J. Exp. Med. 190:895–902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Asseman, C., S. Mauze, M.W. Leach, R.L. Coffman, and F. Powrie. 1999. An essential role for interleukin 10 in the function of regulatory T cells that inhibit intestinal inflammation. J. Exp. Med. 190:995–1004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Jordan, M.S., A. Boesteanu, A.J. Reed, A.L. Petrone, A.E. Holenbeck, M.A. Lerman, A. Naji, and A.J. Caton. 2001. Thymic selection of CD4+CD25+ regulatory T cells induced by an agonist self-peptide. Nat. Immunol. 2:301–306. [DOI] [PubMed] [Google Scholar]
- 30.Read, S., S. Mauze, C. Asseman, A. Bean, R. Coffman, and F. Powrie. 1998. CD38+ CD45RBlow CD4+ T cells: a population of T cells with immune regulatory activities in vitro. Eur. J. Immunol. 28:3435–3447. [DOI] [PubMed] [Google Scholar]
- 31.Sakaguchi, S. 2000. Regulatory T cells: key controllers of immunologic self-tolerance. Cell. 101:455–458. [DOI] [PubMed] [Google Scholar]
- 32.Lohning, M., A. Stroehmann, A.J. Coyle, J.L. Grogan, S. Lin, J.-C. Cutierrez-Ramos, D. Levinson, A. Radbruch, and T. Kamradt. 1998. T1/ST2 is preferentially expressed on murine Th2 cells, independent of interleukin 4, interleukin 5, and interleukin 10, and important for Th2 effector function. Proc. Natl. Acad. Sci. USA. 95:6930–6935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Sallusto, F., D. Lenig, C.R. Mackay, and A. Lanzavecchia. 1998. Flexible programs of chemokine receptor expression on human polarized T helper 1 and 2 lymphocytes. J. Exp. Med. 187:875–883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Sebastiani, S., P. Allavena, C. Albanesi, F. Nasorri, G. Bianchi, C. Traidl, S. Sozzani, G. Girolomoni, and A. Cavani. 2001. Chemokine receptor expression and function in CD4+ T lymphocytes with regulatory activity. J. Immunol. 166:996–1002. [DOI] [PubMed] [Google Scholar]
- 35.Townsend, M.J., P.G. Fallon, D.J. Matthews, H.E. Jolin, and A.N.J. McKenzie. 2000. T1/ST2-deficient mice demonstrate the importance of T1/ST2 in developing primary T helper cell type 2 responses. J. Exp. Med. 191:1069–1076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.d'Ostiani, C.F., G. Del Serro, A. Bacci, C. Montagnoli, A. Spreca, A. Mencacci, P. Ricciardi-Castagnoli, and L. Romani. 2000. Dendritic cells discriminate between yeasts and hyphae of the fungus Candida albicans. Implications for initiation of T helper cell immunity in vitro and in vivo. J. Exp. Med. 191:1661–1674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Marth, T., and B.L. Kelsall. 1997. Regulation of interleukin-12 by complement receptor 3 signaling. J. Exp. Med. 185:1987–1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Demeure, C.E., H. Tanaka, V. Mateo, M. Rubio, G. Delespesse, and M. Sarfati. 2000. CD47 engagement inhibits cytokine production and maturation of human dendritic cells. J. Immunol. 164:2193–2199. [DOI] [PubMed] [Google Scholar]
- 39.Armant, M., M.-N. Avice, P. Hermann, M. Rubio, M. Kiniwa, G. Delespesse, and M. Sarfati. 1999. CD47 ligation selectively downregulates human interleukin 12 production. J. Exp. Med. 190:1175–1182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Dhodapkar, M.V., R.M. Steinman, J. Krasovsky, C. Munz, and N. Bhardwaj. 2001. Antigen-specific inhibition of effector function in humans after injection of immature dendritic cells. J. Exp. Med. 193:233–238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Levings, M.K., R. Sangregorio, F. Galbiati, S. Squadrone, R. de Waal Malefyt, and M.G. Roncarolo. 2001. IFN-α and IL-10 induce the differentiation of human type 1 T regulatory cells. J. Immunol. 166:5530–5539. [DOI] [PubMed] [Google Scholar]
- 42.Stumbles, P.A., J.A. Thomas, C.L. Pimm, P.T. Lee, T.J. Venaille, S. Proksch, and P.G. Holt. 1998. Resting respiratory tract dendritic cells preferentially stimulate T helper cell type 2 (Th2) responses and require obligatory cytokine signals for induction of Th1 immunity. J. Exp. Med. 188:2019–2031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Khanna, A., A.E. Morelli, C. Zhong, T. Takayama, L. Lu, and A.W. Thomson. 2000. Effects of liver-derived dendritic cell progenitors on Th1- and Th2-like cytokine responses in vitro and in vivo. J. Immunol. 164:1346–5134. [DOI] [PubMed] [Google Scholar]
- 44.Iwasaki, A., and B.L. Kelsall. 1999. Freshly isolated Peyer's patch, but not spleen, dendritic cells produce interleukin 10 and induce the differentiation of T helper type 2 cells. J. Exp. Med. 190:229–239. [DOI] [PMC free article] [PubMed] [Google Scholar]