Abstract
The clinical phenotype of interleukin 12 receptor β1 chain (IL-12Rβ1) deficiency and the function of human IL-12 in host defense remain largely unknown, due to the small number of patients reported. We now report 41 patients with complete IL-12Rβ1 deficiency from 17 countries. The only opportunistic infections observed, in 34 patients, were of childhood onset and caused by weakly virulent Salmonella or Mycobacteria (Bacille Calmette-Guérin -BCG- and environmental Mycobacteria). Three patients had clinical tuberculosis, one of whom also had salmonellosis. Unlike salmonellosis, mycobacterial infections did not recur. BCG inoculation and BCG disease were both effective against subsequent environmental mycobacteriosis, but not against salmonellosis. Excluding the probands, seven of the 12 affected siblings have remained free of case-definition opportunistic infection. Finally, only five deaths occurred in childhood, and the remaining 36 patients are alive and well. Thus, a diagnosis of IL-12Rβ1 deficiency should be considered in children with opportunistic mycobacteriosis or salmonellosis; healthy siblings of probands and selected cases of tuberculosis should also be investigated. The overall prognosis is good due to broad resistance to infection and the low penetrance and favorable outcome of infections. Unexpectedly, human IL-12 is redundant in protective immunity against most microorganisms other than Mycobacteria and Salmonella. Moreover, IL-12 is redundant for primary immunity to Mycobacteria and Salmonella in many individuals and for secondary immunity to Mycobacteria but not to Salmonella in most.
Keywords: Mycobacteria, Salmonella, immunodeficiency, interleukin 12 receptor, interferon γ
Introduction
Mendelian susceptibility to mycobacterial disease (MSMD; Mendelian inheritance in man [MIM] 209950) (1) is a rare syndrome which results in predisposition to clinical disease caused by poorly virulent mycobacterial species such as the Bacillus Calmette-Guérin (BCG)* vaccines (2) and nontuberculous environmental Mycobacteria (EM; reference 3). Patients are also susceptible to virulent Mycobacterium tuberculosis (1). Unlike patients with classic immunodeficiencies, these patients rarely present other unusual infectious diseases, with the exception of extra-intestinal nontyphoid salmonellosis, which affects less than half of them. Histopathological and clinical heterogeneity are suggestive of genetic heterogeneity. Some sporadic and most familial cases suggest autosomal recessive heredity, but the syndrome segregates in an autosomal dominant (4) or X-linked recessive (5) pattern in some families.
Five disease-causing autosomal genes have been found, and allelic heterogeneity accounts for the existence of nine defined disorders, all of which result in impaired IFN-γ–mediated immunity (1). Null recessive mutations in the IFN-γ-receptor ligand-binding chain (IFN-γR1)-encoding gene (IFNGR1) abolish either receptor expression (6, 7) or the binding of surface-expressed receptors to IFN-γ (8). Partial recessive (9) and dominant (4) IFN-γR1 deficiencies have also been described. Different recessive mutations in the gene of the IFN-γ signaling chain (IFN-γR2), IFNGR2, are responsible for complete (10) or partial (11) IFN-γR2 deficiency. A dominant mutation in STAT1, resulting in partial STAT-1 (signal transducer and activator of transcription-1) deficiency and impaired cellular responses to IFN-γ, has also been identified in other kindreds (12).
The remaining two genetic defects result in normal cellular responses to IFN-γ, but abnormal IL-12–dependent production of IFN-γ. A few children are homozygous for null mutations in IL12B, encoding the p40 subunit of IL-12 (13–15). Null recessive IL12RB1 mutations have been identified in other patients with IL-12 receptor β1 chain deficiency (15–21). These studies (13–21) only addressed case reports or small cohorts of patients, making it difficult to estimate the range of pathogenic microorganisms and the severity of the clinical course. These issues are of both clinical and immunological importance, given the central role attributed to IL-12 in Th1 development and immunity to various pathogens (22–24). Here, we describe the IL12RB1 genotype and the cellular and clinical phenotypes of 41 patients with IL-12Rβ1 deficiency.
Materials and Methods
Subjects, Kindreds, and Statistical Methods.
We investigated 120 unrelated patients, including five previously described patients (16, 19, 20). Our study was conducted according to the principles expressed in the Helsinki Declaration, with informed consent obtained from each patient or the patient's family. The proportion of infection-free individuals, survival, and penetrance were calculated by the Kaplan-Meier method, and the differences between curves were assessed by the log-rank test. All calculations were performed with the Lifetest procedure of SAS version 8.2 (SAS).
Cell Culture and Stimulation.
Epstein-Barr virus-transformed lymphoblastoid cell lines (EBV-B cell lines) were cultured as described previously (14). Whole blood samples were diluted 1:2 in RPMI 1640 (Invitrogen) and infected by incubation with live M. bovis BCG at a multiplicity of infection of 20:1, alone or with recombinant IL-12p70 (20 ng/ml; R&D Systems), for 48 h. Alternatively, PBMCs were cultured in RPMI 1640 supplemented with 10% heat-inactivated pooled human AB serum, and activated by incubation with PHA P 1/700 (Bacto PHA-P; Becton Dickinson) for 72 h. T cell blasts were restimulated every 48 h with IL-2 (40 IU/ml; Chiron Corp.).
ELISA and Flow Cytometry.
Cell culture supernatants were assayed for IFN-γ by ELISA, according to the kit manufacturer's recommendations (Pelikin Compact, CLB). IFN-γ concentration was calculated per 1 million PBMCs. T cell blasts and/or EBV-B cells were first incubated with an IL-12Rβ1–specific mouse IgG1 mAb (24E6), an IL-12Rβ1–specific rat IgG2a mAb (2B10), or isotypic control mAbs, then with a biotinylated rat anti–mouse Ab or a biotinylated mouse anti–rat Ab, and finally with streptavidin-PE (all reagents were obtained from BD Biosciences/Becton Dickinson). Signals were analyzed with a FACScan™ machine, using CELLQuest™ software (Becton Dickinson).
DNA and RNA Extraction, cDNA Synthesis, and PCR Amplification.
Genomic DNA and total RNA were extracted from EBV-transformed B cells or T cell blasts, as described previously (14). RNA was reverse transcribed in the presence of oligo (dT) with Superscript II reverse transcriptase (Invitrogen; reference 14). The IL12RB1 cDNA, coding exons and flanking intron regions were amplified using pairs of primers and PCR conditions available upon request.
Single-stranded Chain Polymorphism and Sequencing.
Single-stranded chain polymorphism (SSCP) analysis and PCR were performed with pairs of intron primers flanking each IL12RB1 exon, under conditions available upon request. PCR products were sequenced by dideoxynucleotide termination with nested primers (available upon request) and the BigDye terminator kit. PCR products were sequenced on an ABI Prism 3100 apparatus, and analyzed with Sequencing Analysis software (Applied Biosystems).
Results
We investigated 120 unrelated probands, including 100 with MSMD syndrome (disseminated BCG or EM disease with or without salmonellosis), and 20 with only nontyphoid extraintestinal salmonellosis, as we recently described a child with salmonellosis as the sole infectious disease and IL-12p40 deficiency (14).
Detection of IL12RB1 Mutations and Intrafamilial Segregation.
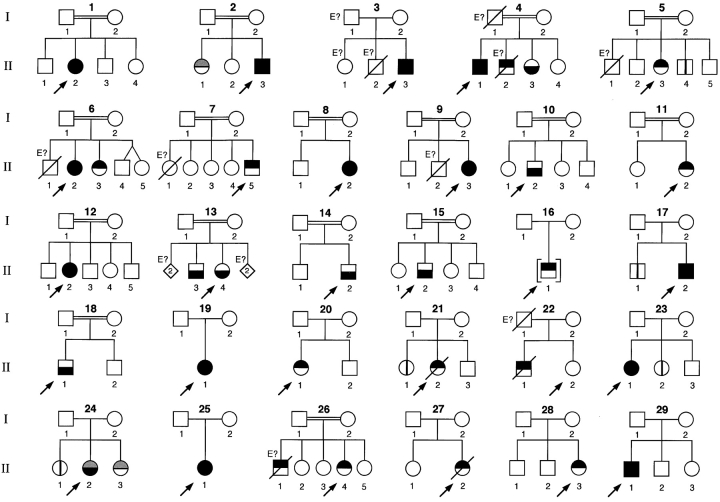
The 17 IL12RB1 coding exons and flanking intron regions were analyzed by SSCP. 25 patients displayed a unique SSCP pattern, each for only one exon (Kindreds 1–9, 11–16, 18-26, 29; Table I and Fig. 1) . Three additional patients (17.II.2, 27.II.2, 28.II.3) displayed unique SSCP patterns for two separate exons, and in one patient (10.II.2), exons 8 to 13 were not amplified. All coding exons were sequenced in these 29 patients with abnormal SSCP patterns. Introns 7 and 13 were also sequenced in patient 10.II.2, and introns 12 and 13 in patient 19.II.1 (owing to the identification of a cDNA of low molecular weight, see below). The patients were found to be homozygous, or compound heterozygous for nonsense (n = 4), splice (n = 4), and missense (n = 6) mutations, small insertions (n = 1), large deletions (n = 2), and deletions/insertions (n = 4; a total of 21 mutant alleles; Table I). None of the mutations were found in 50 unrelated healthy individuals from the corresponding ethnic group. We then amplified and sequenced the full-length IL12RB1 cDNA coding region for the 29 probands. IL12RB1 transcripts of low molecular weight were found in patients 10.II.2 and 19.II.1. In patients homozygous for splice mutations, no full-length mRNAs were found. All parents tested were heterozygous for one IL12RB1 mutation. Only 12 of the 61 known siblings could not be genotyped (designated “E?” in Fig. 1). Two had died of proven BCG (26.II.1) or EM (4.II.2) infection and therefore probably carried the disease-associated IL12RB1 genotype. 10 of the 49 siblings tested, from nine kindreds, carried two mutant IL12RB1 alleles, raising the number of affected siblings to 12. Altogether, we report 41 individuals from 29 kindreds demonstrated (n = 39) or thought (n = 2) to be homozygous or compound heterozygous for IL12RB1 mutations, including 29 probands and 12 siblings.
Table I.
Genotypes and Clinical Phenotypes of Patients with IL-12 Receptor β1 Deficiency
| Kindred
|
Patientsa
|
Mutationsb
|
Originc
|
Follow-upd
|
Agee(yr)
|
BCGf
|
EMf
|
Mtbf
|
Salmonella
f
|
|---|---|---|---|---|---|---|---|---|---|
| 1 | II.2 | K305X | Morocco | Alive | 23 | D | – | – | S. typhimurium |
| 2 | II.1 | R213W | Morocco | Alive | 26 | Resistant | – | GI tract | – |
| II.3 | Alive | 15 | D | – | – | S. typhimurium | |||
| 3 | II.3 | Y367C | Cameroon | Alive | 2 | D | – | – | S. dublin |
| 4 | II.1 | 1623_1624delinsTT | Cyprus | Alive | 33 | Resistant | M. avium M. triplex | – |
S. enteritidis
|
| II.2 | Deceased | 8 | nv | M. avium | – | – | |||
| II.3 | Alive | 27 | Resistant | – | – | S. enteritidis | |||
| 5 | II.3 | 783+1G>A | Turkey | Alive | 15 | D | – | – | – |
| II.4 | Alive | 12 | nv | – | – | – | |||
| 6 | II.2 | 783+1G>A | Turkey | Alive | 14 | D | – | – | S. enteritidis |
| II.3 | Alive | 9 | D | – | – | – | |||
| 7 | II.5 | R173P | Turkey | Alive | 14 | Resistant | M. chelonae | – | – |
| 8 | II.2 | R173P | Turkey | Alive | 9 | D | – | – | S. enteritidis |
| 9 | II.3 | 557_563delins8 | Turkey | Alive | 12 | D | – | – | S. enteritidis |
| 10 | II.2 | 700+362-1619-944del | Israel | Alive | 4 | nv | – | – | S. group D |
| 11 | II.2 | 1190-1G>A | Saudi Arabia | Alive | 3 | D | – | – | – |
| 12 | II.2 | C186S | Qatar | Alive | 7 | D | – | – | S. enteritidis |
| 13 | II.3 | C186S | Qatar | Alive | 7 | Resistant | – | S. spp | |
| II.4 | Alive | 4 | Resistant | – | S. typhimurium | ||||
| 14 | II.2 | 1791+2T>G | Iran | Alive | 8 | nv | – | – | S. enteritidis |
| 15 | II.2 | S321X | Pakistan | Alive | 19 | Resistant | – | – | S. enteritidis |
| 16 | II.1 | 1791+2T>G | Sri-Lanka | Alive | 18 | D | – | – | – |
| 17 | II.1 | [Q32X+1623_1624delinsTT] | France | Alive | 18 | Resistant | – | – | – |
| II.2 | Alive | 13 | nv | M. genavense | - | S. enteritidis | |||
| 18 | II.1 | Q376X | France | Alive | 30 | Resistant | – | – | S. dublin |
| 19 | II.1 | [1745_1746insCA+1483+182-1619-1073del] | France | Alive | 31 | D | – | – |
S. dublin
|
| 20 | II.1 | Q32X | France | Alive | 6 | D | – | – | – |
| 21 | II.1 | Q32X | Belgium | Alive | 16 | nv | – | – | – |
| II.2 | Deceased | 7 | nv | M. avium | – | – | |||
| 22 | II.1 | 1623_1624delinsTT | Germany | Deceased | 3.5 | nv | M. avium | – | – |
| 23 | II.1 | 1623_1624delinsTT | Germany | Alive | 10 | D | – | – | S. enteritidis |
| II.2 | Alive | 7 | nv | – | – | – | |||
| 24 | II.1 | 1791+2T>G | Spain | Alive | 16 | nv | – | – | – |
| II.2 | Alive | 14 | nv | – | Lungs | S. enteritidis | |||
| II.3 | Alive | 7 | nv | D | – | ||||
| 25 | II.1 | 1791+2T>G | Spain | Alive | 4 | nv | M. avium | – | S. enteritidis |
| 26 | II.1 | 549+2T>C | Bosnia- Herzegovina |
Deceased | 4 | D | – | – | – |
| II.4 | Alive | 6 | nv | M. spp | – | – | |||
| 27 | II.2 | [1440_1447delins16+Q171P] | Slovakia | Deceased | 2 | D | – | – | – |
| 28 | II.3 | [1007_1008delinsG+Q171P] | Slovakia | Alive | 3 | D | – | – | – |
| 29 | II.1 | L77P | Brazil | Alive | 24 | D | – | – | S. typhimurium |
Children 3.II.2, 5.II.1, 6.II.1, 7.II.1 and 9.II.2 (Fig. 1) died during their first year of life of unknown causes; they were not available for genetic analysis (no DNA available), and thus were not included in the present series of patients as their phenotype and genotype were unclear. In contrast, two patients with the known phenotype of BCG and/or EM infection were included despite their genotype not being known (4.II.2 and 26.II.1).
Mutation nomenclature follows the recommendations of Antonarakis and den Dunnen (reference 52). Mutation Q214R initially found in patient 4.II.1 (16), was subsequently found to be a polymorphism. “Delins” indicates mutations combining the deletion and insertion of nucleotides.
The clinical features of kindreds 1, 4, and 5 were initially reported in 1998 (reference 16), kindred 2 in 2001 (reference 20), kindred 4 in 1995 (reference 3) and 2000 (reference 18), kindred 5 in 1988 (reference 53), kindred 7 in 2001 (reference 19), and kindred 21 in 1997 (reference 54). The countries of residence in some cases differ from the countries of origin: kindred 1 lives in France, kindreds 4 and 15 live in the United Kingdom, kindreds 5 and 6 living in Sweden and originating from Turkey are of the Kurd ethnic group, kindred 9 originating from Turkey lives in Germany, kindred 10 originating from and living in Israel is of the Bedouin ethnic group, kindred 14 lives in USA, kindred 16 lives in Switzerland.
Deceased or alive.
Age at death or at the time of writing this report.
BCG, Bacille Calmette-Guérin; D, Disseminated; EM, Environmental Mycobacteria; Mtb, Mycobacterium tuberculosis; nv, not vaccinated with BCG; Resistant, no adverse reaction to BCG vaccination; M. spp, patient 26.II.4 was not BCG-vaccinated and suffered from chronic disseminated granulomatous disease that responded to empirical anti-mycobacterial treatment (rifampicin, isoniazid, ethambutol, amikacin); no mycobacterial species were visible or cultured, suggesting atypical mycobacteriosis. Patient 10.II.2 also had Kingella kingae infections, and patient 29.II.1 Paracoccidioides brasiliensis infections. Patient 4.II.1, previously reported not to have been vaccinated (references 3, 16, and 18), had in fact been inoculated with BCG at 5 yr of age. Sera from 26, 25, and 22 patients were tested for Ab against non-typhoid Salmonella, Toxoplasma gondii, and cytomegalovirus, respectively. The detailed clinical features of all patients will be reported elsewhere.
Figure 1.
Pedigrees of 29 kindreds with IL-12 receptor β1 deficiency. Each kindred is designated by a capital letter (1–29), each generation by a roman numeral (I–II), and each individual by an Arabic numeral (from left to right). Symbols are partitioned in two parts by a horizontal line: the upper part indicates infections with Mycobacteria (in black, patients with BCGosis or atypical mycobacteriosis; in gray, patients with tuberculosis); the lower part indicates infections with Salmonella (in black, nontyphoid salmonellosis). The proband are indicated by an arrow. Individuals whose genetic status could not be evaluated are indicated by the symbol “E?”. Asymptomatic individuals carrying two mutant IL12RB1 alleles are represented by a vertical line.
Impaired Surface Expression and Function of IL-12Rβ1.
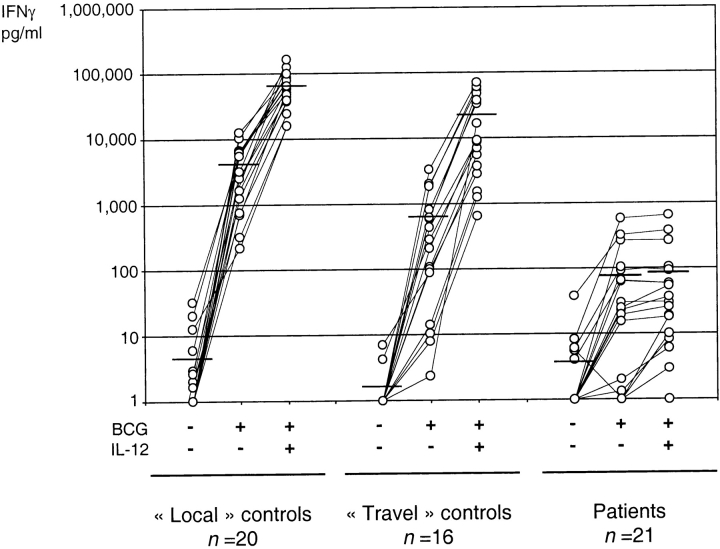
We assessed IL-12Rβ1 expression on the surface of T cell blasts (kindreds 9, 14, 15, 22, 25), EBV-transformed B cells (kindreds 8, 11, 19, 20, 29), or both (kindreds 3, 6, 10, 12, 16, 17, 23, 24, 26–28), from 21 of the 24 newly diagnosed probands, by flow cytometry with specific antibodies. Consistent with the five patients previously reported (16, 19, 20), no IL-12Rβ1 molecules were detected on the surface of cells from all but one patient. Patient 10.II.2, carrying a large in-frame deletion, presented an expression of the β1 chain, both on T cell blasts and EBV-transformed B cells, detectable with one monoclonal antibody (24E6), but not with another (2B10; data not depicted). Cells from the three remaining probands (kindreds 13, 18, 21) were not tested for IL-12Rβ1 expression, but were homozygous for mutations found in other kindreds (Table I). The cells from all 21 patients tested, including the proband from kindred 10 with residual IL-12Rβ1 expression produced less IFN-γ than the “travel” and “local” laboratory controls in response to BCG (mean of 76 versus 664 and 4,109 pg/ml), and production was not increased by IL-12 (mean of 87 versus 22,432 and 65,636 pg/ml; Fig. 2) . Some patients were not tested with this assay (kindreds 4, 7, 12, 13, 14, 18, 22, 29). However, kindreds 4, 7, 14, 18, and 22 carry the same mutation as other patients (Table I). We and others have demonstrated that T cells homozygous for the IL12RB1 missense mutation R213W (kindred 2) are complemented by transfection with a wild-type IL12RB1 allele (20, 21). Overall, we have demonstrated complete IL-12Rβ1 deficiency in 29 patients (absence of detectable IL-12Rβ1 surface expression, n = 28, absence of response to IL-12, n = 27, or both, n = 27).
Figure 2.
Impaired cellular response to interleukin-12. Production of IFN-γ by whole blood cells from 20 healthy “local” positive controls (fresh blood), from 16 healthy “travel” positive controls and from 21 patients, either unstimulated (−) or stimulated with BCG alone or with BCG plus recombinant IL-12p70. Fresh, heparinized blood from 7 patients and all “travel” controls were shipped to Paris within 12 to 48 h, where the experiments were performed. Blood from four patients and all “local” control was drawn in Paris and readily stimulated. The supernatants were harvested after 48 h of activation for cytokine quantification by ELISA. The horizontal bars represent the arithmetic mean of the values.
Onset and Outcome of Infections in IL-12Rβ1–deficient Patients.
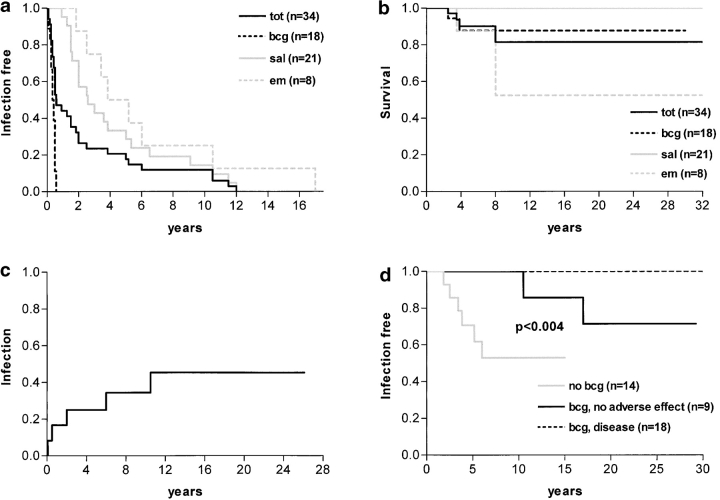
We first analyzed the 34 individuals who developed case-definition opportunistic infections caused by weakly virulent microorganisms (BCG, EM, and nontyphoid Salmonella; Table I). 25 of these 34 patients had been inoculated with live BCG in childhood. Only 18 developed disseminated BCG disease, within six months of inoculation (Fig. 3 a). No recurrence of BCG disease was observed in the 16 patients who survived the initial BCG infection. Eight patients suffered EM disease. EM disease began before the age of six years, in all but two patients (4.II.1 and 7.II.5) who developed M. avium and M. fortuitum infections at the ages of 17 and 10 yr, respectively. One of these two patients (4.II.1), unlike the others, suffered a second EM disease, caused by M. triplex. Finally, disseminated nontyphoid salmonellosis occurred in 21 of 34 patients. Salmonellosis was the only infectious disease for seven patients. First Salmonella infection occurred at or before the age of 12 yr. Eight patients had recurrent salmonellosis (8/21, 38%), involving the same serotype, whereas BCG and EM infections were not recurrent. Nevertheless, we found high titers of Salmonella-specific antibodies in the sera of 4 deficient patients with no history of clinical salmonellosis. Only five of the 34 infected IL-12Rβ1-deficient patients died due to BCG (n = 2) or EM (n = 3; a mortality rate among symptomatic patients of only 15% [5/34]) (Fig. 3 b). All deaths occurred before 8 yr of age. No death was attributable to salmonellosis. Thus, infections caused by weakly virulent Mycobacteria and Salmonella in IL12Rβ1-deficient patients generally have a childhood onset and a favorable clinical outcome, particularly once patients have entered their teens.
Figure 3.
Epidemiological features of IL-12Rβ1 deficiency. First onset (a) and outcome (b) of case-definition opportunistic infectious diseases in 34 deficient patients, according to infections: BCG (broken black line), EM (broken gray line), Salmonella (solid gray line), and all 3 infections (solid black line). (c) Penetrance of case-definition infectious diseases in the 12 IL-12Rβ1–deficient siblings (excluding all probands). (d) Variations in onset of EM disease among the 41 deficient patients, who had been vaccinated with BCG and suffered BCG disease (broken black line, n = 18), who had been vaccinated with BCG without developing BCG disease (resistance to BCG, solid black line, n = 9), or who had not been vaccinated with BCG (solid gray line, n = 14).
Clinical Penetrance and Impact of BCG on Clinical Phenotype.
We determined the clinical penetrance of IL12Rβ1 deficiency, by excluding all probands (Fig. 3 c). Five (42%) of the 12 affected siblings were completely free of unusual infections at their last follow-up visit (mean duration of follow-up: 13.8 yr, range: 7–18 yr). None of the other 7 siblings had more than one infection. Two of the six siblings who had been vaccinated had disseminated BCG disease. Three additional siblings developed infections with weakly virulent microorganisms, EM in one case and Salmonella in the other two. Overall, the penetrance of opportunistic infections was estimated to be 45.3% (confidence interval [CI] 95%: 14.2%-75.4%) when calculated with survival analysis techniques to account for differences in follow-up period (Fig. 3 c). Finally, tuberculosis occurred in 2 other siblings (2.II.1, 24.II.3). We then determined the impact of BCG on the clinical phenotype for the 41 patients described herein. BCG inoculation or disease had no impact on the occurrence (and age at onset) of Salmonella infection, or the occurrence of tuberculosis. In contrast, none of the 18 patients with BCG disease developed EM disease (interval between onset of BCG disease and last follow-up: 3–31 yr, mean 12.8 yr). Only two (4.II.1 and 7.II.5) of the nine patients resistant to BCG (BCG inoculation without BCG disease) suffered EM disease, with late onset of the disease. Six (43%) of the 14 patients who had not been inoculated with BCG suffered EM disease (Fig. 3 d), with early onset of the disease. The difference in age at onset of EM disease between the three groups of patients was highly significant (P < 0.004; Fig. 3 d). This difference was particularly clear if patients with BCG disease were compared with patients not inoculated with BCG (P < 0.004). The difference between patients resistant to BCG and nonvaccinated patients was not significant (P = 0.09). Finally, the difference in incidence of EM disease between BCG-inoculated patients (with or without BCG disease) and nonvaccinated patients was highly significant (P < 0.002).
Discussion
We report here the first large series of patients with complete IL-12Rβ1 deficiency, including 41 deficient individuals from 29 unrelated kindreds (Table I and Fig. 1). The kindreds originate from 17 countries, in Africa, America, Asia, and Europe. Seven other unrelated patients reported elsewhere (15, 17, 18, 21) raise the total number of cases to 48, from 36 kindreds, in 20 countries. The cellular phenotype of IL-12-receptor β1 chain–deficient patients is uniform: all patients tested failed to respond to IL-12. The relative contributions to the clinical phenotype of the lack of response of NK cells (16) and of T lymphocytes (15–18, 21) to IL-12 are unclear. The receptor for IL-23, another IFN-γ–inducing cytokine, contains the IL-12Rβ1 subunit (25), suggesting that patients with IL-12Rβ1 deficiency probably also suffer from IL-23 receptor deficiency.
The results obtained in this study confirm that IL-12Rβ1 deficiency is principally associated with mycobacterial diseases (Table I). If we include the seven patients reported elsewhere (15, 17, 18, 21), 33 of the 48 patients have had BCG (n = 22) or EM (n = 11) disease. We also report three patients from two kindreds with M. tuberculosis disease. Thus, a diagnosis of IL-12 receptor deficiency should be considered in patients with unusually severe forms of tuberculosis (20). Mice with a disrupted IL12B gene (26) are susceptible to BCG (27), M. tuberculosis (27–29) and, to a lesser extent, to M. avium (30–32). Our results extend these observations, and highlight the crucial role of IL-12 in immunity to BCG, several EM and M. tuberculosis in humans.
However, IL-12Rβ1–deficient patients are less susceptible to Mycobacteria than was initially thought (15–21). 9 of 27 children inoculated with BCG did not suffer adverse effects. Only 8 of the 41 patients (19%) in this series, and 11 of the 48 (23%) including the cases reported elsewhere (17, 18, 21), developed EM infections. Excluding the probands, only 2 of 6 siblings vaccinated with BCG and 1 of 12 IL-12Rβ1–deficient siblings developed BCG and EM disease, respectively. Paradoxically, these data indicate that most IL-12Rβ1–deficient patients do not have the original phenotype of BCG/EM disease or salmonellosis. Healthy siblings of probands should therefore be investigated. IL-12 is surprisingly redundant in protective immunity against primary infection by weakly virulent Mycobacteria in a majority of individuals.
In our survey, the age at onset of EM disease differed between patients with and without BCG disease. If we include the other IL-12Rβ1– and IL-12p40–deficient cases (13–15, 17, 18, 21), there are 63 patients in total, 36 of whom had BCG disease. None of these 36 patients developed EM disease, while among the 27 patients who did not develop any BCG disease 12 developed EM disease (P < 10−6, Fisher's exact test). This indicates that BCG disease protects against subsequent EM illness in patients with IL-12 deficiency or IL-12R deficiency. Antibiotic treatment against BCG probably played a minor role in preventing EM disease, as it was discontinued after a few years in all patients. Moreover, the impact of BCG inoculation itself (with or without BCG disease) was also significant in our series. Similarly, EM disease was protective in itself. In contrast, BCG inoculation and EM disease had no effect on the incidence of salmonellosis. IL-12 is thus redundant for adaptive, secondary immunity to Mycobacteria in most, if not all, patients.
The prognosis of IL-12Rβ1 deficiency is good due to the low clinical penetrance of primary mycobacterial infection, acquired protective immunity to secondary mycobacterial infection, and the lack of life-threatening nonmycobacterial infections. Only 5 of our 41 (12%) IL-12Rβ1–deficient patients died of BCG or EM infection, before the age of 8 yr. The overall mortality rate for all known IL-12Rβ1–deficient patients is only 15% (7/48), increasing to 21% (13/63) if we include IL-12p40–deficient children (13–15, 17, 18, 21). Outcome improved with age, and our patients (now aged 2–33 yr) are healthy and off all treatment. These results should help us to predict clinical outcome for individual patients. They also indicate that immune mechanisms progressively develop ways of compensating for the lack of IL-12–mediated immunity.
21 of the 41 patients (51%) in our series had had extra-intestinal infections caused by Salmonella, as had 4 of the other 7 IL-12Rβ1–deficient patients (15, 17, 18, 21) and 5 of the 15 IL-12p40–deficient patients (a total of 30 out of 63, 48%; references 13–15). Of clinical interest, five children presented salmonellosis as the only infectious disease. IL-12 has been shown to play a major role in host defense against Salmonella in IL-12p40-KO mice (33, 34). Our observation extends this findings in humans. However, not all Salmonella-infected patients developed clinical disease (Table I). IL-12 was redundant in primary immunity against Salmonella in some patients. Salmonellosis recurrence was frequent (8/21, 38%), suggesting that IL-12 is required for secondary immunity to Salmonella in many patients. These data suggest that different immune mechanisms control secondary immunity to Salmonella and Mycobacteria.
There were no other unusually severe infections in our 41 patients, as in the other seven patients (15, 17, 18, 21; Table I). CMV infection had been well controlled in at least 14 patients (Table I). Other common viruses did not cause severe disease in our patients. One patient suffered benign infection with Kingella kingae. There were no other diseases caused by common bacteria, including macrophage-infecting species such as Listeria and Nocardia. One IL-12–deficient patient had one episode of nocardiosis (14). Paracoccidioides brasiliensis infection in one curable IL-12Rβ1–deficient patient had a symptomatic yet benign course, and oral candidiasis was diagnosed in a few patients. Other fungi represented no threat to IL-12Rβ1–deficient patients. Toxoplasma gondii infection had an uneventful course in at least five patients. Other common opportunistic protozoa were not pathogenic.
The number of kindreds investigated from various genetic backgrounds and exposed to a variety of environmental conditions, together with the number of siblings affected, ruling out a major ascertainment bias, make it possible to conclude that mycobacteriosis and salmonellosis are the most frequent infectious diseases in patients with IL-12Rβ1 deficiency. Our study does not exclude the possibility that other infectious or non-infectious diseases may occur in other patients with this genetic defect, or in our patients later in life. The apparent resistance of IL-12Rβ1–deficient patients to most microorganisms nevertheless contrasts with results obtained in mice. Indeed, IL12p40 KO mice are susceptible to almost all microorganisms tested (35–51). The lack of a requirement for IL-12 and probably IL-23 for protective immunity against most microorganisms in humans may reflect the natural (1), as opposed to experimental, course of most infections in man.
Acknowledgments
We would like to thank the patients and their families, the members of the Laboratory of Human Genetics of Infectious Diseases, Dr. Dominique Recan for the EBV transformation of B cells, and Dr. Sandra Weller for invaluable advice in flow cytometry.
C. Fieschi was supported by the Association pour la Recherche contre le Cancer (ARC), S. Dupuis by the Ministere de l'Education Nationale, de la Recherche, et de la Technologie (MENRT), and J. Bustamante by the Fondation Schlumberger. This work was supported by the Fondation BNP-Paribas, the Fondation Schlumberger, and the Sequella Foundation.
F. Altare's present address is Institute of Pharmacology and Structural Biology, CNRS UMR 5089, 31077 Toulouse, France.
Footnotes
Abbreviations used in this paper: BCG, Bacille Calmette-Guérin; EM, environmental Mycobacteria; SSCP, single-stranded conformation polymorphism.
References
- 1.Casanova, J.L., and L. Abel. 2002. Genetic dissection of immunity to mycobacteria: The human model. Annu. Rev. Immunol. 20:581–620. [DOI] [PubMed] [Google Scholar]
- 2.Casanova, J.L., S. Blanche, J.F. Emile, E. Jouanguy, S. Lamhamedi, F. Altare, J.L. Stephan, F. Bernaudin, P. Bordigoni, D. Turck, et al. 1996. Idiopathic disseminated bacillus Calmette-Guerin infection: a French national retrospective study. Pediatrics. 98:774–778. [PubMed] [Google Scholar]
- 3.Levin, M., M.J. Newport, S. D'Souza, P. Kalabalikis, I.N. Brown, H.M. Lenicker, P.V. Agius, E.G. Davies, A. Thrasher, N. Klein, and J. Blackwell. 1995. Familial disseminated atypical mycobacterial infection in childhood: a human mycobacterial susceptibility gene? Lancet. 345:79–83. [DOI] [PubMed] [Google Scholar]
- 4.Jouanguy, E., S. Lamhamedi-Cherradi, D. Lammas, S.E. Dorman, M.-C. Fondanèche, S. Dupuis, R. Döffinger, F. Altare, J.-F. Emile, J. Girdelstone, et al. 1999. A human IFNGR1 small deletion hotspot associated with dominant susceptibility to mycobacterial infection. Nat. Genet. 21:370–378. [DOI] [PubMed] [Google Scholar]
- 5.Frucht, D.M., and S.M. Holland. 1996. Defective monocyte costimulation for IFN-gamma production in familial disseminated Mycobacterium avium complex infection: abnormal IL-12 regulation. J. Immunol. 157:411–416. [PubMed] [Google Scholar]
- 6.Newport, M.J., C.M. Huxley, S. Huston, C.M. Hawrylowicz, B.A. Oostra, R. Williamson, and M. Levin. 1996. A mutation in the interferon-gamma-receptor gene and susceptibility to mycobacterial infection. N. Engl. J. Med. 335:1941–1949. [DOI] [PubMed] [Google Scholar]
- 7.Jouanguy, E., F. Altare, S. Lamhamedi, P. Revy, J.F. Emile, M. Newport, M. Levin, S. Blanche, E. Seboun, A. Fischer, and J.L. Casanova. 1996. Interferon-gamma-receptor deficiency in an infant with fatal bacille Calmette-Guerin infection. N. Engl. J. Med. 335:1956–1961. [DOI] [PubMed] [Google Scholar]
- 8.Jouanguy, E., S. Dupuis, A. Pallier, R. Doffinger, M.C. Fondaneche, C. Fieschi, S. Lamhamedi-Cherradi, F. Altare, J.F. Emile, P. Lutz, et al. 2000. In a novel form of IFN-gamma receptor 1 deficiency, cell surface receptors fail to bind IFN-gamma. J. Clin. Invest. 105:1429–1436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Jouanguy, E., S. Lamhamedi-Cherradi, F. Altare, M.C. Fondaneche, D. Tuerlinckx, S. Blanche, J.F. Emile, J.L. Gaillard, R. Schreiber, M. Levin, et al. 1997. Partial interferon-gamma receptor 1 deficiency in a child with tuberculoid bacillus Calmette-Guerin infection and a sibling with clinical tuberculosis. J. Clin. Invest. 100:2658–2664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Dorman, S.E., and S.M. Holland. 1998. Mutation in the signal-transducing chain of the interferon-gamma receptor and susceptibility to mycobacterial infection. J. Clin. Invest. 101:2364–2369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Döffinger, R., E. Jouanguy, S. Dupuis, M.C. Fondanèche, J.L. Stéphan, J.F. Emile, S. Lamhamedi, F. Altare, A. Pallier, G. Barcenas-Morales, et al. 2000. Partial interferon gamma receptor signalling chain deficiency in a patient with bacille Calmette-Guérin and Mycobacterium abscessus infection. J. Infect. Dis. 181:379–384. [DOI] [PubMed] [Google Scholar]
- 12.Dupuis, S., C. Dargemont, C. Fieschi, N. Thomassin, S. Rosenzweig, J. Harris, S.M. Holland, R.D. Schreiber, and J.L. Casanova. 2001. Impairment of mycobacterial but not viral immunity by a germline human STAT1 mutation. Science. 293:300–303. [DOI] [PubMed] [Google Scholar]
- 13.Altare, F., D. Lammas, P. Revy, E. Jouanguy, R. Döffinger, S. Lamhamedi, P. Drysdale, D. Tollner, J. Girdlestone, P. Darbyshire, et al. 1998. Inherited interleukin 12 deficiency in a child with bacille Calmette-Guérin and Salmonella enteritidis disseminated infection. J. Clin. Invest. 102:2035–2040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Picard, C., C. Fieschi, F. Altare, S. Al-Jumaah, S. Al-Hajjar, J. Feinberg, S. Dupuis, C. Soudais, I.Z. Al-Mohsen, E. Genin, et al. 2002. Inherited interleukin-12 deficiency: IL12B genotype and clinical phenotype of 13 patients from six kindreds. Am. J. Hum. Genet. 70:336–348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Elloumi-Zghal, H., M.R. Barbouche, J. Chemli, M. Bejaoui, A. Harbi, N. Snoussi, S. Abdelhak, and K. Dellagi. 2002. Clinical and genetic heterogeneity of inherited autosomal recessive susceptibility to disseminated Mycobacterium bovis Bacille Calmette-Guerin infection. J. Infect. Dis. 185:1468–1475. [DOI] [PubMed] [Google Scholar]
- 16.Altare, F., A. Durandy, D. Lammas, J.F. Emile, S. Lamhamedi, F. Le Deist, P. Drysdale, E. Jouanguy, R. Döffinger, F. Bernaudin, et al. 1998. Impairment of mycobacterial immunity in human interleukin-12 receptor deficiency. Science. 280:1432–1435. [DOI] [PubMed] [Google Scholar]
- 17.de Jong, R., F. Altare, I.A. Haagen, D.G. Elferink, T. Boer, P.J. van Breda Vriesman, P.J. Kabel, J.M. Draaisma, J.T. van Dissel, F.P. Kroon, et al. 1998. Severe mycobacterial and Salmonella infections in interleukin-12 receptor-deficient patients. Science. 280:1435–1438. [DOI] [PubMed] [Google Scholar]
- 18.Verhagen, C.E., T. de Boer, H.H. Smits, F.A. Verreck, E.A. Wierenga, M. Kurimoto, D.A. Lammas, D.S. Kumararatne, O. Sanal, F.P. Kroon, et al. 2000. Residual type 1 immunity in patients genetically deficient for interleukin 12 receptor beta1 (IL-12Rbeta1): evidence for an IL-12Rbeta1-independent pathway of IL-12 responsiveness in human T cells. J. Exp. Med. 192:517–528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Aksu, G., C. Tirpan, C. Cavusoglu, S. Soydan, F. Altare, J.L. Casanova, and N. Kutukculer. 2001. Mycobacterium fortuitum-chelonae complex infection in a child with complete interleukin-12 receptor beta 1 deficiency. Pediatr. Infect. Dis. J. 20:551–553. [DOI] [PubMed] [Google Scholar]
- 20.Altare, F., A. Ensser, A. Breiman, J. Reichenbach, J.E. Baghdadi, A. Fischer, J.F. Emile, J.L. Gaillard, E. Meinl, and J.L. Casanova. 2001. Interleukin-12 receptor beta1 deficiency in a patient with abdominal tuberculosis. J. Infect. Dis. 184:231–236. [DOI] [PubMed] [Google Scholar]
- 21.Sakai, T., M. Matsuoka, M. Aoki, K. Nosaka, and H. Mitsuya. 2001. Missense mutation of the interleukin-12 receptor beta1 chain-encoding gene is associated with impaired immunity against Mycobacterium avium complex infection. Blood. 97:2688–2694. [DOI] [PubMed] [Google Scholar]
- 22.Romani, L., P. Puccetti, and F. Bistoni. 1997. Interleukin-12 in infectious diseases. Clin. Microbiol. Rev. 10:611–636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Gately, M.K., L.M. Renzetti, J. Magram, A.S. Stern, L. Adorini, U. Gubler, and D.H. Presky. 1998. The interleukin-12/interleukin-12-receptor system: role in normal and pathologic immune responses. Annu. Rev. Immunol. 16:495–521. [DOI] [PubMed] [Google Scholar]
- 24.Murphy, K.M., and S.L. Reiner. 2002. Decision making in the immune system: the lineage decisions of helper T cells. Nat. Rev. Immunol. 2:933–944. [DOI] [PubMed] [Google Scholar]
- 25.Parham, C., M. Chirica, J. Timans, E. Vaisberg, M. Travis, J. Cheung, S. Pflanz, R. Zhang, K.P. Singh, F. Vega, et al. 2002. A receptor for the heterodimeric cytokine IL-23 is composed of IL-12Rbeta1 and a novel cytokine receptor subunit, IL-23R. J. Immunol. 168:5699–5708. [DOI] [PubMed] [Google Scholar]
- 26.Magram, J., S.E. Connaughton, R.R. Warrier, D.M. Carvajal, C.Y. Wu, J. Ferrante, C. Stewart, U. Sarmiento, D.A. Faherty, and M.K. Gately. 1996. IL-12-deficient mice are defective in IFNγ production and type 1 cytokine responses. Immunity. 4:471–481. [DOI] [PubMed] [Google Scholar]
- 27.Hölscher, C., R.A. Atkinson, B. Arendse, N. Brown, E. Myburgh, G. Alber, and F. Brombacher. 2001. A protective and agonistic function of IL-12p40 in mycobacterial infection. J. Immunol. 167:6957–6966. [DOI] [PubMed] [Google Scholar]
- 28.Cooper, A.M., J. Magram, J. Ferrante, and I.M. Orme. 1997. Interleukin 12 is crucial to the development of protective immunity in mice intravenously infected with Mycobacterium tuberculosis. J. Exp. Med. 186:39–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Cooper, A.M., A. Kipnis, J. Turner, J. Magram, J. Ferrante, and I.M. Orme. 2002. Mice lacking bioactive IL-12 can generate protective, antigen-specific cellular responses to mycobacterial infection only if the IL-12 p40 subunit is present. J. Immunol. 168:1322–1327. [DOI] [PubMed] [Google Scholar]
- 30.Doherty, T.M., and A. Sher. 1998. IL-12 promotes drug-induced clearance of Mycobacterium avium infection in mice. J. Immunol. 160:5428–5435. [PubMed] [Google Scholar]
- 31.Silva, R.A., M. Florido, and R. Appelberg. 2001. Interleukin-12 primes CD4+ T cells for interferon-gamma production and protective immunity during Mycobacterium avium infection. Immunology. 103:368–374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ehlers, S., J. Benini, H.D. Held, C. Roeck, G. Alber, and S. Uhlig. 2001. alphabeta T cell receptor-positive cells and interferon-gamma, but not inducible nitric oxide synthase, are critical for granuloma necrosis in a mouse model of mycobacteria-induced pulmonary immunopathology. J. Exp. Med. 194:1847–1859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Dybing, J.K., N. Walters, and D.W. Pascual. 1999. Role of endogenous interleukin-18 in resolving wild-type and attenuated Salmonella typhimurium infections. Infect. Immun. 67:6242–6248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lehmann, J., S. Bellmann, C. Werner, R. Schroder, N. Schutze, and G. Alber. 2001. IL-12p40-dependent agonistic effects on the development of protective innate and adaptive immunity against Salmonella enteritidis. J. Immunol. 167:5304–5315. [DOI] [PubMed] [Google Scholar]
- 35.Lu, H., X. Yang, K. Takeda, D. Zhang, Y. Fan, M. Luo, C. Shen, S. Wang, S. Akira, and R.C. Brunham. 2000. Chlamydia trachomatis mouse pneumonitis lung infection in IL-18 and IL-12 knockout mice: IL-12 is dominant over IL-18 for protective immunity. Mol. Med. 6:604–612. [PMC free article] [PubMed] [Google Scholar]
- 36.Rottenberg, M.E., A. Gigliotti Rothfuchs, D. Gigliotti, M. Ceausu, C. Une, V. Levitsky, and H. Wigzell. 2000. Regulation and role of IFN-gamma in the innate resistance to infection with Chlamydia pneumoniae. J. Immunol. 164:4812–4818. [DOI] [PubMed] [Google Scholar]
- 37.Del Rio, L., A.J. Buendia, J. Sanchez, M.C. Gallego, M.R. Caro, N. Ortega, J. Seva, F.J. Pallares, F. Cuello, and J. Salinas. 2001. Endogenous interleukin-12 is not required for resolution of Chlamydophila abortus (Chlamydia psittaci serotype 1) infection in mice. Infect. Immun. 69:4808–4815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Simmons, C.P., N.S. Goncalves, M. Ghaem-Maghami, M. Bajaj-Elliott, S. Clare, B. Neves, G. Frankel, G. Dougan, and T.T. MacDonald. 2002. Impaired resistance and enhanced pathology during infection with a noninvasive, attaching-effacing enteric bacterial pathogen, Citrobacter rodentium, in mice lacking IL-12 or IFN-gamma. J. Immunol. 168:1804–1812. [DOI] [PubMed] [Google Scholar]
- 39.Oxenius, A., U. Karrer, R.M. Zinkernagel, and H. Hengartner. 1999. IL-12 is not required for induction of type 1 cytokine responses in viral infections. J. Immunol. 162:965–973. [PubMed] [Google Scholar]
- 40.Michailowsky, V., N.M. Silva, C.D. Rocha, L.Q. Vieira, J. Lannes-Vieira, and R.T. Gazzinelli. 2001. Pivotal role of interleukin-12 and interferon-gamma axis in controlling tissue parasitism and inflammation in the heart and central nervous system during Trypanosoma cruzi infection. Am. J. Pathol. 159:1723–1733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Müller, U., G. Kohler, H. Mossmann, G.A. Schaub, G. Alber, J.P. Di Santo, F. Brombacher, and C. Holscher. 2001. IL-12-independent IFN-gamma production by T cells in experimental Chagas' disease is mediated by IL-18. J. Immunol. 167:3346–3353. [DOI] [PubMed] [Google Scholar]
- 42.Ely, K.H., L.H. Kasper, and I.A. Khan. 1999. Augmentation of the CD8+ T cell response by IFN-gamma in IL-12-deficient mice during Toxoplasma gondii infection. J. Immunol. 162:5449–5454. [PubMed] [Google Scholar]
- 43.Mattner, F., J. Magram, J. Ferrante, P. Launois, K. Di Padova, R. Behin, M.K. Gately, J.A. Louis, and G. Alber. 1996. Genetically resistant mice lacking interleukin-12 are susceptible to infection with Leishmania major and mount a polarized Th2 cell response. Eur. J. Immunol. 26:1553–1559. [DOI] [PubMed] [Google Scholar]
- 44.Mattner, F., K. Di Padova, and G. Alber. 1997. Interleukin-12 is indispensable for protective immunity against Leishmania major. Infect. Immun. 65:4378–4383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Mencacci, A., E. Cenci, G. Del Sero, C. Fe d'Ostiani, P. Mosci, G. Trinchieri, L. Adorini, and L. Romani. 1998. IL-10 is required for development of protective Th1 responses in IL-12-deficient mice upon Candida albicans infection. J. Immunol. 161:6228–6237. [PubMed] [Google Scholar]
- 46.Decken, K., G. Kohler, K. Palmer-Lehmann, A. Wunderlin, F. Mattner, J. Magram, M.K. Gately, and G. Alber. 1998. Interleukin-12 is essential for a protective Th1 response in mice infected with Cryptococcus neoformans. Infect. Immun. 66:4994–5000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Kawakami, K., Y. Koguchi, M.H. Qureshi, A. Miyazato, S. Yara, Y. Kinjo, Y. Iwakura, K. Takeda, S. Akira, M. Kurimoto, and A. Saito. 2000. IL-18 contributes to host resistance against infection with Cryptococcus neoformans in mice with defective IL-12 synthesis through induction of IFN-gamma production by NK cells. J. Immunol. 165:941–947. [DOI] [PubMed] [Google Scholar]
- 48.Harandi, A.M., B. Svennerholm, J. Holmgren, and K. Eriksson. 2001. Interleukin-12 (IL-12) and IL-18 are important in innate defense against genital herpes simplex virus type 2 infection in mice but are not required for the development of acquired gamma interferon-mediated protective immunity. J. Virol. 75:6705–6709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Carr, J.A., J.A. Rogerson, M.J. Mulqueen, N.A. Roberts, and A.A. Nash. 1999. The role of endogenous interleukin-12 in resistance to murine cytomegalovirus (MCMV) infection and a novel action for endogenous IL-12 p40. J. Interferon Cytokine Res. 19:1145–1152. [DOI] [PubMed] [Google Scholar]
- 50.Walter, M.J., N. Kajiwara, P. Karanja, M. Castro, and M.J. Holtzman. 2001. Interleukin 12 p40 production by barrier epithelial cells during airway inflammation. J. Exp. Med. 193:339–351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Xing, Z., A. Zganiacz, J. Wang, M. Divangahi, and F. Nawaz. 2000. IL-12-independent Th1-type immune responses to respiratory viral infection: requirement of IL-18 for IFN-gamma release in the lung but not for the differentiation of viral-reactive Th1-type lymphocytes. J. Immunol. 164:2575–2584. [DOI] [PubMed] [Google Scholar]
- 52.den Dunnen, J.T., and S.E. Antonarakis. 2001. Nomenclature for the description of human sequence variations. Hum. Genet. 109:121–124. [DOI] [PubMed] [Google Scholar]
- 53.Jeppsson, O., B. Petrini, J. Andersson, N. Heurlin, and G. Malm. 1988. Defective handling of mycobacteria. Lancet. 2:570. [DOI] [PubMed] [Google Scholar]
- 54.Tuerlinckx, D., C. Vermylen, B. Brichard, J. Ninane, and G. Cornu. 1997. Disseminated Mycobacterium avium infection in a child with decreased tumour necrosis factor production. Eur. J. Pediatr. 156:204–206. [DOI] [PubMed] [Google Scholar]