Abstract
The CD4+ T lymphocyte plays a central role in host defense against Pneumocystis pneumonia but has received only limited attention in rats. CD4+ T-cell-depleting (OX-38) and nondepleting (W3/25) monoclonal antibodies, which recognize an identical or adjacent epitope, were administered for up to 14 weeks to Lewis rats that had been exposed to Pneumocystis. While OX-38 produced a greater decrease in circulating CD4+ cells than W3/25, both antibody treatments resulted in similar effects on the health of the rats and the levels of Pneumocystis pneumonia, which were milder than those found with corticosteroids. W3/25 also did not enhance the severity of Pneumocystis pneumonia achieved with corticosteroids alone. We conclude that CD4+ cell function is more important than CD4+ cell number in host defense against Pneumocystis in the rat and that this new model permits study of opportunistic infections in the rat without the confounding effects of corticosteroids.
Pneumocystis is an extracellular fungus of low virulence that causes pneumonia in immunocompromised individuals, such as human immunodeficiency virus-positive patients and cancer and organ transplant patients. Various immunodeficient and immunosuppressed rat and mouse models have been used to study the interaction between Pneumocystis and host (9, 11, 31, 32). The corticosteroid (CS)-treated rat is the original animal model used to study Pneumocystis pneumonia and is the source of most available information on the epidemiology, immunopathogenesis, diagnosis, and therapy of the disease (3, 9). However, this model is limited by the broad immunosuppressive effects of steroids on the immune system, including lymphocyte depletion and impairment of function, reduced leukocyte chemotaxis and phagocytosis, and deficient antibody (Ab) production (38).
Clinical and experimental studies have shown that CD4+ T lymphocytes play a central role in host defenses against Pneumocystis (14, 25, 29, 37). Pneumocystis pneumonia can be induced in mice exposed to the organism by the administration of GK1.5, a rat immunoglobulin G2b (IgG2b) monoclonal antibody (MAb) specific for CD4+ cells (31). This model has proven to be popular because it circumvents the need for CS immunosuppression; however, no such model exists in rats. Given the increasing evidence of the genetic diversity and host specificity of Pneumocystis (6, 33), it cannot be assumed that the results obtained in one animal species can be applied to another. The development of a CD4+ depletion model in the rat would be important in studying the role of CD4+ T cells in the rat.
MAbs have been produced to rat CD4+ cells, and their properties have been analyzed by in vitro or short-term in vivo studies (2, 5, 26, 39). These Abs are of two general types: depleting Abs, which trigger cell lysis; and nondepleting Abs, which cause downregulation of CD4 antigen expression resulting in inadequate T-cell receptor (TCR)-antigen-major histocompatibility complex class II interaction (34). These Abs have been used in a variety of autoimmune or other disease studies, including studies of rat adjuvant arthritis (23), organ transplantation (17), and allergic encephalomyelitis (34). Yet, there is little published information about the use of these Abs in an infectious disease model.
We undertook the present study to determine if the administration of MAbs to CD4+ cells can induce Pneumocystis pneumonia in rats. We chose two widely used MAbs that recognize an identical or adjacent epitope on the CD4+ molecule (15): W3/25 (mouse IgG1), a nondepleting MAb that downregulates CD4+ cell function, and OX-38 (mouse IgG2a), a depleting MAb.
MATERIALS AND METHODS
Animals.
Male Lewis rats were acquired from Charles River Laboratories (Hollister, Calif.). Male and female Long-Evans rats were bred and raised at the Veterans Affairs Medical Center, Cincinnati, Ohio. All rats were 6 to 8 weeks of age and weighed 125 to 150 g at the beginning of the experiments. The animals were housed in microisolator cages in a bioBubble (Fort Collins, Colo.) to control aerosol contamination and were nourished with autoclaved food and water. Ampicillin (1 mg ml−1; TEVA Pharmaceuticals, Sellersville, Pa.) was given in the water to control secondary bacterial infections. All rats used in this study were exposed to Pneumocystis by being housed with CS-treated rats with active Pneumocystis pneumonia. Pneumocystis pneumonia was induced in rats by subcutaneous injections of methylprednisolone acetate (4.0 mg/0.2 ml/week; Depo-Medrol; Pharmacia and Upjohn Co., Kalamazoo, Mich.), as described previously (35). All animals were handled according to institutionally recommended guidelines.
Anti-rat CD4+ MAbs.
The W3/25 and OX-38 hybridomas were obtained from the European Collection of Animal Cell Cultures (ECACC) Centre for Applied Microbiology & Research (CAMR) (Wiltshire, United Kingdom) and shipped to the National Cell Culture Center (Minneapolis, Minn.). MAbs W3/25 (mouse IgG1, nondepleting, anti-rat CD4+) and OX-38 (mouse IgG2a, depleting, anti-rat CD4+), which recognize an identical or adjacent epitope on the rat CD4+ molecule based on binding studies (15), were produced from static culture, concentrated by tangential flow, and purified by protein A chromatography by the National Cell Culture Center. The W3/25 and OX-38 Abs were diluted in phosphate-buffered serum (PBS) to a final concentration of 1 mg/ml and were administered by intraperitoneal (i.p.) injection.
Preliminary studies were conducted to determine the optimal dose regimen for the MAbs. A review of the literature indicated that MAbs to rat CD4+ cells are administered either on a milligram-per-rat or milligram-per-kilogram basis. We found that W3/25 administered at a dose of 1.0 mg (7.4 mg/kg of body weight) once a week downregulated CD4+ molecule expression in the spleen, cervical lymph nodes, and peripheral blood of Lewis rats. This effect was not enhanced by larger doses (e.g., 2.0 mg or 14.4 mg/kg or 3.0 mg or 22.2 mg/kg) of W3/25 or more frequent administration (two or three times per week). We also found that 1.0 mg of W3/25 MAb was sufficient to downregulate CD4+ for at least 2 weeks following a single administration in both spleen and peripheral blood. Therefore, we chose to use administrations of W3/25 or OX-38 MAbs at 1.0 mg/week/rat in our study. Other investigators have previously shown that similar administrations of W3/25 MAb (28) or OX-38 MAb (8) to rats are effective anti-CD4+ treatments.
Tissue processing and lymphocyte isolation.
Rats were anesthetized by i.p. injection with 16.0 mg of Telazol per kg (1:1 tiletamine HCl-zolazepam HCl; Lederle Parenterals, Inc., Puerto Rico) and 4.8 mg of xylazine HCl (2-[2,6-dimethylphenylamino]-4H-5,6-dihydrothiazine) per kg (Sigma, St. Louis, Mo.). Body weight was determined, and the spleens and lungs were excised. Lymphocytes were obtained from the spleens by homogenization with a Tenbroeck tissue grinder (Wheaton, Millvill, N.J.) and filtered through a 0.4-mm-diameter cell strainer. Erythrocytes were lysed with 0.85 M ammonium chloride, and the remaining cells were washed and resuspended in RPMI 1640 medium supplemented with 2 mM glutamine, 2.5 × 10−5 M β-mercaptoethanol (β-ME), 100 U of penicillin G sodium, 100.0 μg of streptomycin sulfate, 0.25 μg of amphotericin B, and 10% fetal calf serum.
Flow cytometry.
Spleen cells from individual rats in an experimental group were analyzed separately or were pooled to compensate for reduced numbers of lymphocytes in immunosuppressed rats (38). Preliminary testing showed that the mean lymphocyte percentage of individual samples was very similar to the lymphocyte percentage of the pooled group sample. Cells were suspended in 25 ml of RPMI medium, and counts (number of cells per milliliter) were determined with a hemocytometer. The number of cells per milliliter was multiplied by 25 ml and divided by the number of spleens (usually 4). The total cell count per spleen is reported in Table 1. The cells were 95% viable as determined by trypan blue dye exclusion. Viable splenocytes were plated at 3.0 × 105 cells per well in 96-well, round-bottom plates. Pooled group specimens were plated in triplicate. The cells were stained with 50.0 ng of OX-35 (fluorescein isothiocyanate [FITC]-conjugated anti-rat CD4+, which binds to a different epitope from OX-38 and W3/25), OX-8 (FITC-conjugated anti-rat CD8a+), R73 (FITC-conjugated anti-rat TCR), and G4.18 (phycoerythrin [PE]-conjugated anti-rat CD3+) Abs (Pharmingen, San Diego, Calif.) for 30 min on ice, washed, and then resuspended in 10% buffered formalin. Samples were analyzed on a Coulter EPICS IV flow cytometer (XL-MCL; Coulter Immunology Corp., Hialeah, Fla.) by selecting 104 cells in a lymphocyte forward-versus-side light-scatter region, and results were plotted on log fluorescence histograms. The numbers of CD4 and CD8 cells were expressed as the number of cells per spleen. All data were reported as the mean number of cells ± standard deviation.
TABLE 1.
Effects of CS and anti-CD4+ Ab treatments on Lewis rat splenocyte populations
| Treatmenta | Total no. of cells (106)/spleen (%)b,c
|
CD4/CD8 ratioc | |
|---|---|---|---|
| CD4 | CD8 | ||
| PBS (1.0 ml) | 52 ± 30 (32) | 15 ± 1.5 (9.4) | 3.5 ± 0.20 |
| CS (4.0 mg) | 0.36 ± 0.24 (6.1) | 0.15 ± 0.06 (2.5) | 2.4 ± 0.15 |
| W3/25 (1.0 ml) | 20 ± 2.8 (18) | 23 ± 7.6 (21) | 0.88 ± 0.04 |
| OX-38 (1.0 ml) | 6.3 ± 0.73 (5.6) | 35 ± 7.81 (31) | 0.18 ± 0.0035 |
| W3/25 + OX38 (1.0 ml each) | 9.1 ± 2.6 (11) | 24 ± 5.87 (29) | 0.37 ± 0.02 |
Four rats per group were given the treatment specified for 8 weeks. A hemocytometer was used to determine the total number of cells per spleen as follows: PBS, 160 × 106; CS, 5.8 × 106; W3/25, 110 × 106; OX-38, 110 × 106; and W3/25 plus OX-38, 84 × 106.
The results represent the mean ± standard deviation number of CD4+ or CD8+ cells counted per spleen in a lymphocyte forward-versus-side light-scatter region by flow cytometry analysis. The percentage of CD4 or CD8 cells per spleen is given in parentheses.
P < 0.001 for each comparison of mean cell number versus PBS-treated controls.
Lung processing and Pneumocystis carinii enumeration.
The right lung from each animal was used for quantitation of Pneumocystis cysts and nuclei as previously described (16, 35, 36). Lungs were minced in 10.0 ml of phosphate-buffered saline (PBS), homogenized in a Stomacher 80 (Tekmar, Inc., Gehanna, Ohio), and filtered through gauze. These preparations were then treated with 0.85 M ammonium chloride and washed, and slides were prepared for enumeration. The stains used to identify Pneumocystis were cresyl echt violet (CEV), which selectively stains the cell wall of the cyst; and Diff-Quick (DQ), which stains the nuclei of all developmental forms. Three 10-μl drops were placed on a glass slide, the slides were stained, and 10 randomly scanned fields were read from each drop. The lower limit of detection by this evaluation is log10 5.24 (5.26 × 105) organisms per lung.
Histological analysis.
A portion of the left lung lobe was excised and immersed in 10% buffered formalin. Lung histological sections were stained with hematoxylin-eosin (H+E) and Grocott-Gomori methenamine-silver nitrate (GMS) as described elsewhere (35).
Statistical analysis.
Statistical analyses were performed by using Graphpad Instat and Prism (San Diego, Calif.) software. Data were compared by analysis of variance followed by the Student-Neuman-Keuls test for multiple comparisons (35). A P value < 0.05 was considered significant.
RESULTS
Effects of nondepleting rat CD4+ W3/25 MAb administration and depleting rat CD4+ OX-38 MAb administration.
We hypothesized that the CD4+ T-cell-reducing effect of the OX-38 and W3/25 MAbs would render our Lewis rat model susceptible to Pneumocystis. To test this, Lewis rats received weekly administration of either 1.0 mg of OX-38, 1.0 mg of W3/25, or a combination of both Abs (1.0 mg each). As positive and negative controls for Pneumocystis infection, rats received weekly i.p. administration of methylprednisolone (4.0 mg/0.2 ml) or 1.0 ml of PBS, respectively. All rats were exposed to Pneumocystis-infected rats and were sacrificed at different time points.
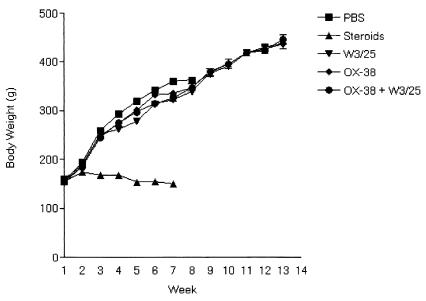
PBS-treated rats were healthy and increased their mean body weight by 130% over 8 weeks (Fig. 1). CS-treated rats experienced the wasting effects typical of CS treatment and did not gain any weight over 7 weeks. The CD4+-reducing Abs did not appear to have an effect on overall health, because the rats treated with W3/25, OX-38, and OX-38 plus W3/25 had increases in their weight for the first 8 weeks (130, 110, and 120%, respectively), and these increases continued for 14 weeks (190, 170, and 190%, respectively). The slightly lower weight gain of anti-CD4+-treated rats compared to PBS-treated rats has been noted in previous experiments in this laboratory (data not shown).
FIG. 1.
Sequential changes in mean body weight of Lewis rats during various i.p. treatments. PBS (1.0 ml), methylprednisolone (4.0 mg), 1.0 OX-38 (1.0 mg), W3/25 (1.0 mg), and OX-38 plus W3/25 (1.0 mg) were administered weekly to Lewis rats. Rats were weighed weekly; each data point represents the mean body weight (g) of at least 4 to 14 rats.
The effects of different immunosuppressive regimens on splenocyte cell populations are outlined in Table 1. The CS-treated rats lowered the total spleen cell count from 160 × 106 cells in the PBS-treated controls to 5.8 × 106 cells. Less reduction was found in W3/25-treated rats (110 × 106), OX-38-treated rats (110 × 106), and the combined W3/25-OX-38-treated rats (84 × 106). CS-treated rats experienced a significant reduction in the CD4+ cell population (81% decrease compared to PBS-treated control rats; P < 0.001). The CD4+ reduction achieved with OX-38 alone (83% decrease compared to the level in PBS-treated controls; P < 0.001) was greater than the decrease achieved with either W3/25 alone (44% decrease; P < 0.001) or with combined W3/25 plus OX-38 (67% decrease; P < 0.001). CS-treated rats showed a significant decrease in splenic CD8+ cells as well (73% decrease compared to the level in PBS-treated controls; P < 0.001). A significant increase in the splenic CD8+ T-cell population was observed in rats treated with OX-38 (230% increase; P < 0.001), W3/25 (120% increase; P < 0.001), and the combined anti-CD4+ MAbs (210% increase; P < 0.001). The increased CD8+ T-cell numbers seen in both OX-38- and W3/25-treated animals were proportional to the decreased CD4+ T-cell numbers, indicating that CD4+ T cells were actually depleted after treatment with the W3/25 Ab. This CD4+ T-cell reduction effect has been observed in other studies after multiple MAb injections (23).
A comparison of the CD4+/CD8+ ratios is also shown in Table 1. CS-treated rats had a lower CD4+/CD8+ ratio than control rats, as was previously seen in our studies (5). W3/25-treated rats and W3/25-OX-38-treated rats exhibited reduced CD4+/CD8+ ratios in the spleen compared to controls, but the greatest reduction of the CD4+/CD8+ ratio was seen with the OX-38-treated rats compared to controls.
Mild P. carinii infection susceptibility in W3/25- and OX-38-treated Lewis rats.
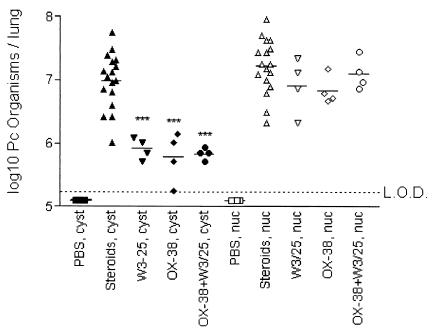
Our CS-treated Lewis rats typically perish after 9 to 10 weeks of treatment due to the wasting effects of the steroids, so all of the CS-treated rats were sacrificed at the week 8 time point. These steroid-treated rats were infected with Pneumocystis (Fig. 2), evidenced by detection of both Pneumocystis cysts (log 7.0 ± 0.44 per lung [mean ± standard deviation]) and nuclei (7.2 ± 0.43 per lung), while PBS-treated, healthy rats were not infected with Pneumocystis. When anti-CD4+ MAb-treated rat lungs were processed for Pneumocystis detection at week 8, Pneumocystis cysts and nuclei were detected in the OX-38-treated rats (cysts, 5.8 ± 0.40 per lung; and nuclei, 6.9 ± 0.23 per lung), W3/25-treated rats (5.9 ± 0.17 and 6.9 ± 0.44 per lung), and OX-38-W3/25-treated rats (5.8 ± 0.09 and 7.1 ± 0.26 per lung). The effect of anti-CD4+ MAb administration appeared to be greater on the trophic form of Pneumocystis than on the cyst, as evidenced by the similar burden of nuclei (indicating all morphological forms of Pneumocystis) between CS- and anti-CD4+ MAb-treated rats. Similar Pneumocystis cyst and nucleus burdens were observed in rats treated with the anti-CD4+ MAbs for 12 to 14 weeks (data not shown); the cyst and nucleus burdens detected at 12 to 14 weeks were (5.2 ± 0.36 cysts per lung and 6.6 ± 0.28 nuclei per lung for OX-38-treated rats, <5.2 cysts per lung and 6.54 ± 0.45 nuclei per lung for W3/25-treated rats, and 5.3 ± 0.35 cysts per lung and 6.6 ± 0.31 nuclei per lung for OX-38-W3/25-treated rats.
FIG. 2.
Pneumcystis organism burden of Lewis rats following anti-CD4+ Ab treatments after 8 weeks. PBS (1.0 ml), methylprednisolone (4.0 mg), OX-38 (1.0 mg), W3/25 (1.0 mg), and 1.0 mg of OX-38 plus W3/25 were administered weekly to Lewis rats. After 8 weeks of treatment, rats were sacrificed, and lungs were processed and analyzed for organism burden. Pneumocystis cyst results were obtained from CEV-stained slides prepared from processed lungs; Pneumocystis nucleus (nuc) results were obtained from DQ-stained slides prepared from processed lungs. Each data point represents the respective organism burden of one rat. L.O.D., limit of (microscopic) detection (log10 5.24 organisms per lung). The horizontal bar represents the mean. ***, P < 0.001 compared to steroid-treated controls.
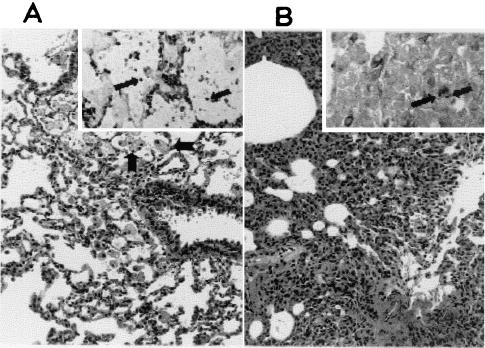
Silver-stained lung sections of CS-treated rats revealed numerous Pneumocystis cysts arranged in typical clusters within alveoli (Fig. 3). In contrast, lung sections of the anti-CD4+ MAb-treated (OX-38, W3/25, and OX-38 plus W3/25) rats only showed scattered cysts, and no cysts were seen in the PBS-treated animals. The frequencies of cysts found in lung sections from rats treated with CD4+-depleting and -nondepleting MAbs were similar. Lung sections stained with H+E revealed typical intra-alveolar foamy exudates with increased macrophages in the CS-treated rats but not in the anti-CD4+ MAb- or PBS-treated rats. The host inflammatory response in all groups was modest and characterized by a nonspecific mononuclear cell infiltrate with occasional perivascular inflammation (Fig. 3).
FIG. 3.
Photomicrographs of representative lung sections from a CS-treated rat and anti-CD4+ MAb-treated rat stained with H+E and GMS. A lung section from a CS-treated rat following 8 weeks of treatment is represented in panel A. A lung section from a W3/25 MAb-treated rat following 12 weeks of treatment is represented in panel B. The photos were taken with a Zeiss Axioscope microscope (Carl Zeiss, Inc., Germany) with an attached Spot 2e digital camera (Diagnostic Instruments, Inc., Sterling Heights, Mich.). The larger panels are stained with H+E (magnification, ×200); the inset figures stained with GMS (magnification, ×400) indicate many Pneumocystis cysts in a CS-treated rat (A) and occasional cysts in a W3/25 MAb-treated rat (B). Arrows (insets) indicate cysts; notched arrows (A) indicate macrophages in the vicinity of Pneumocystis cysts.
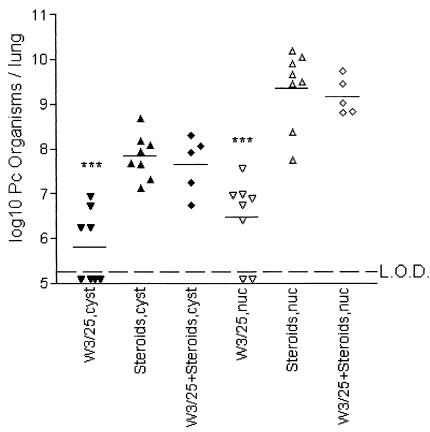
Another experiment was performed to investigate the effects of W3/25 in a different rat species and to determine if this MAb increased the level of Pneumocystis infection in CS-treated rats. Long-Evans rats were treated with weekly administrations of W3/25 (1.0 mg), CS (4.0 mg), or a combination of W3/25 and CS for 9 weeks (Fig. 4). Animals were sacrificed 7 to 9 weeks after treatments, and their lungs were examined for organism burden. The CS-treated rats were positive for Pneumocystis cysts (7.8 ± 0.50 per lung) and nuclei (9.4 ± 0.86 per lung). The organism burden of CS-treated rats was not increased when W3/25 was administered along with the steroids (7.7 ± 0.65 and 9.2 ± 0.42 per lung, respectively). As in the previous experiment with Lewis rats, the W3/25-treated Long-Evans rats also showed a mild susceptibility to Pneumocystis infection (5.8 ± 0.80 and 6.5 ± 0.91 per lung) that was significantly less (P < 0.001) than that of steroid-treated rats and steroid-W3/25-treated rats. In contrast to the previous experiment shown in Fig. 2, the cyst and nucleus burdens were significantly lower in W3/25-treated rats than in CS-treated rats.
FIG. 4.
Pneumocystis organism burden of Long-Evans rats following W3/25, methylprednisolone, or combined W3/25- methylprednisolone treatment. Rats received either 1.0 mg of W3/25, 4.0 mg of methylprednisolone acetate (Depo-Medrol), or both. Rats were sacrificed after 7 to 9 weeks of treatment. Each data point represents the respective organism burden of one rat. The horizontal bar represents the mean. ***, P < 0.001 compared to steroid-treated controls.
DISCUSSION
Rodent models of Pneumocystis pneumonia have been powerful tools to study the disease. Due to the plentiful supply of inbred strains and ease of genetic manipulation, mouse models of Pneumocystis pneumonia are more numerous than rat models (3). Besides the immunosuppressed (CS treated) rat, the only other rat model of Pneumocystis pneumonia is the athymic (nude) rat. While there are two 1993 reports of spontaneous and inoculated Pneumocystis pneumonia infection of athymic Rowett nude (Han:RNU rnu/rnu) rats and New Zealand nude (Han:NZNU rnuN/rnuN) rats (10, 27), there have been no further published results concerning the development of this model. More recently, the rnu/rnu rat strain has been used in Pneumocystis studies, but these nude rats were treated with dexamethasone before inoculation with Pneumocystis (1, 7).
The present study was undertaken to develop a new rat model of Pneumocystis pneumonia by administration of MAbs to CD4+ cells. These MAbs have mainly been used in short-term experiments and to study autoimmune and related disorders. W3/25 and OX-38, the MAbs selected for analysis, recognize an identical or adjacent epitope (21) and thus provided a unique opportunity to compare the effects of CD4+ depletion and CD4+ antigen downregulation on this disease. Since Pneumocystis replicates slowly in the lungs, the different effects of these MAbs on CD4+ cells could be monitored over a lengthy period of time.
The data in the present study have shown that the OX-38 MAb had a greater effect on the number of circulating and splenic CD4+ cells than the W3/25 MAb. Yet, the use of both MAbs resulted in a modest level of Pneumocystis pneumonia that was considerably lower than the level that could be achieved with CS. The Pneumocystis organism burden was not increased by coadministration of W3/25 and OX-38 or by longer periods of Ab treatment (up to 14 weeks). These MAbs also did not enhance the level of Pneumocystis pneumonia induced by CS alone. There were discrepancies observed in the effectiveness of anti-CD4+ MAb treatment on Pneumocystis nucleus burdens in our two different animal models, and these may be explained by rat strain Pneumocystis susceptibility differences or by the diverse burdens of Pneumocystis infection found in the different experiments. Other reports have shown that variations in Pneumocystis cyst and nucleus burden susceptibility can occur following certain treatments, such as immune cell adoptive transfer and echinochandin drugs (30, 41)
The lower organism burden in the anti-CD4+-treated rats compared to CS-treated rats suggests that components other than CD4+ cells play an important role in host defenses against Pneumocystis in this model. Contributions of CD8+ T cells (4), alveolar macrophages (20), B cells (19), the CD40-CD40 ligand interaction (40), lung surfactant proteins (18), Igs (12), cytokines (24, 42), and chemokines (43) have all been reported in mice and/or rats. Only the CD4+ T-cell contribution to host defense is affected by the anti-CD4+ Abs. While CD4+ reduction or depletion may lead to dysregulation of the immune system (such as CD4+ T-cell-macrophage interactions or cytokine production), the other components in host defense are not directly affected by the anti-CD4+ Abs.
Another potential reason may relate to the level of CD4+ depletion brought on by the anti-CD4+ Abs. In contrast to mice, in which the GK1.5 MAb used to develop Pneumocystis pneumonia results in no detectable CD4+ cells (31), both MAbs used in the present study did not completely eliminate the CD4+ cells. Although we explored a broad range of doses of W3/25 and a mainstream dose of OX-38 similar to those used by other investigators (9, 30), we found that higher or more frequent doses did not improve the level of CD4+ depletion. The possibility that higher doses of OX-38 or other MAbs could enhance our results will require further investigation. It is also feasible that the pool of CD4+ T cells that remains following anti-CD4+ Ab treatments is sufficient to mount a cellular response to Pneumocystis, as was demonstrated in our ex vivo and in vitro studies.
Despite the different immunosuppressive effects of the Abs used in this study, Pneumocystis susceptibility remained the same following CD4+ molecule downregulation or CD4+ cell depletion. The fact that W3/25 and OX-38 Abs had similar effects in our model suggests that susceptibility to Pneumocystis pneumonia is related not only to the number of CD4+ T cells (25), but also to defects in CD4+ function. Support for this idea comes from the occurrence of Pneumocystis pneumonia in knockout mice that lack TCR function (13) or experience interruption of the CD40-CD40L pathway (22). On the other hand, our study has also shown that W3/25 has some cell-depleting properties in the spleen.
The new rat model of Pneumocystis pneumonia described here offers a number of advantages over the CS-treated model. Since the anti-CD4+ MAbs result in less debilitation than CS, longer experiments can be conducted. The new model permits the study of antibodies and other factors involved in host defenses against Pneumocystis without the confounding effects of corticosteroids. The CD4+ model will now permit comparison of the effects of another form of immunosuppression on Pneumocystis antigenic expression and variation, as well as on the efficacy of anti-Pneumocystis drugs in rats. Finally, the new model may be helpful in studying other opportunistic infections that are better suited for rat than mouse models.
Acknowledgments
We gratefully acknowledge the technical assistance of Randy Thomas, Chris Woods, and Mary Palascak and Peter Ciraolo of Robert Franco's laboratory.
This work was supported by the Medical Research Service, Department of Veterans Affairs, and by the Public Service contracts AI 75319 and AI 25467 and grant RO1 HL64570 from the National Institutes of Health.
Editor: T. R. Kozel
REFERENCES
- 1.Aliouat, E. M., A. Martinez, E. Jimenez, E. Dei-Cas, C. Mullet, P. Delcourt, and D. Gargallo-Viola. 1997. Development of pneumocystosis animal models: corticosteroid-treated Wistar rat, SCID mouse and nude rat. J. Eukaryot. Microbiol. 44:41S-42S. [DOI] [PubMed]
- 2.Arima, T., M. Lehmann, and M. W. Flye. 1997. Induction of donor specific transplantation tolerance to cardiac allografts following treatment with nondepleting (RIB 5/2) or depleting (OX-38) anti-CD4 mAb plus intrathymic or intravenous donor alloantigen. Transplantation 63:284-292. [DOI] [PubMed] [Google Scholar]
- 3.Armstrong, M. Y. and M. T. Cushion. 1994. Animal models, p. 181-222. In P. D. Walzer (ed.), Pneumocystis carinii pneumonia, 2nd ed. Marcel Dekker, Inc., New York, N.Y.
- 4.Beck, J. M., R. L. Newbury, B. E. Palmer, M. L. Warnock, P. K. Byrd, and H. B. Kaltreider. 1996. Role of CD8+ lymphocytes in host defense against Pneumocystis carinii in mice. J. Lab. Clin. Med. 128:477-487. [DOI] [PubMed] [Google Scholar]
- 5.Billingham, M. E., C. Hicks, and S. Carney. 1990. Monoclonal antibodies and arthritis. Agents Actions 29:77-87. [DOI] [PubMed] [Google Scholar]
- 6.Cushion, M. T. 1998. Taxonomy, genetic organization, and life cycle of Pneumocystis carinii. Semin. Respir. Infect. 13:304-312. [PubMed] [Google Scholar]
- 7.Dei-Cas, E., M. Brun-Pascaud, V. Bille-Hansen, A. Allaert, and E. M. Aliouat. 1998. Animal models of pneumocystosis. FEMS Immunol. Med. Microbiol. 22:163-168. [DOI] [PubMed] [Google Scholar]
- 8.Flavin, T., J. Shizuru, K. Seydel, A. Wu, N. Fujimoto, E. G. Hoyt, K. Ivens, M. Billingham, C. G. Fathman, and V. A. Starnes. 1990. Selective T-cell depletion with Ox-38 anti-CD4 monoclonal antibody prevents cardiac allograft rejection in rats. J. Heart Transplant. 9:482-488. [PubMed] [Google Scholar]
- 9.Frenkel, J. K., J. T. Good, and J. A. Shultz. 1966. Latent pneumocystis infection of rats, relapse and chemotherapy. Lab. Investig. 15:1559-1577. [PubMed] [Google Scholar]
- 10.Furuta, T., M. Fujita, K. Machii, K. Kobayashi, S. Kojima, and K. Ueda. 1993. Fatal spontaneous pneumocystosis in nude rats. Lab. Anim. Sci. 43:551-556. [PubMed] [Google Scholar]
- 11.Furuta, T., K. Ueda, S. Kyuwa, and K. Fujiwara. 1984. Effect of T-cell transfer on Pneumocystis carinii infection in nude mice. Jpn. J. Exp Med. 54:57-64. [PubMed] [Google Scholar]
- 12.Gigliotti, F., C. G. Haidaris, T. W. Wright, and A. G. Harmsen. 2002. Passive intranasal monoclonal antibody prophylaxis against murine Pneumocystis carinii pneumonia. Infect. Immun. 70:1069-1074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hanano, R., K. Reifenberg, and S. H. E. Kaufmann. 1996. Naturally acquired Pneumocystis carinii pneumonia in gene disruption mutant mice: roles of distinct T-cell populations in infection. Infect. Immun. 64:3201-3209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Harmsen, A. G., and M. Stankiewicz. 1990. Requirement for CD4+ cells in resistance to Pneumocystis carinii pneumonia in mice. J. Exp. Med. 172:937-945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Jefferies, W. A., J. R. Green, and A. F. Williams. 1985. Authentic T helper CD4 (W3/25) antigen on rat peritoneal macrophages. J. Exp. Med. 162:117-127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kim, C. K., J. M. Foy, M. T. Cushion, D. Stanforth, M. J. Linke, H. L. Hendrix, and P. D. Walzer. 1987. Comparison of histologic and quantitative techniques in evaluation of therapy for experimental Pneumocystis carinii pneumonia. Antimicrob. Agents Chemother. 31:197-201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lehmann, M., E. Graser, K. Risch, W. W. Hancock, A. Muller, B. Kuttler, H. J. Hahn, J. W. Kupiec-Weglinski, J. Brock, and H. D. Volk. 1997. Anti-CD4 monoclonal antibody-induced allograft tolerance in rats despite persistence of donor-reactive T cells. Transplantation 64:1181-1187. [DOI] [PubMed] [Google Scholar]
- 18.Linke, M. J., C. E. Harris, T. R. Korfhagen, F. X. McCormac, A. D. Ashbaugh, P. Seele, J. A. Whitsett, and P. D. Walzer. 2001. Immunosuppressed surfactant protein A-deficient mice have increased susceptibility to Pneumocystis carinii infection. J. Infect. Dis. 183:943-952. [DOI] [PubMed] [Google Scholar]
- 19.Marcotte, H., D. Levesque, K. Delanay, A. Bourgeault, R. de la Durantaye, S. Brochu, and M. C. Lavoie. 1996. Pneumocystis carinii infection in transgenic B cell-deficient mice. J. Infect. Dis. 173:1034-1037. [DOI] [PubMed] [Google Scholar]
- 20.Martin, W. J., and R. Pasula. 2000. Role of alveolar macrophages in host defense against Pneumocystis carinii. Am. J. Respir. Cell Mol. Biol. 23:434-435. [DOI] [PubMed] [Google Scholar]
- 21.Morrison, W. J., N. J. Kennedy, H. Offner, and A. A. Vandenbark. 1995. Effects of anti-CD4 antibody: enhancement of lymph node PPD-memory T cell response. Cell. Immunol. 163:106-112. [DOI] [PubMed] [Google Scholar]
- 22.Oz, H. S., W. T. Hughes, J. E. Rehg, and E. K. Thomas. 2000. Effect of CD40 ligand and other immunomodulators on Pneumocystis carinii infection in rat model. Microb. Pathog. 29:187-190. [DOI] [PubMed] [Google Scholar]
- 23.Pelegri, C., M. Castell, M. Serra, M. Rabanal, M. Rodriguez-Palmero, C. Castellote, and A. Franch. 2001. Prevention of adjuvant arthritis by the W3/25 anti-CD4 monoclonal antibody is associated with a decrease of blood CD4(+)CD45RC(high) T cells. Clin. Exp. Immunol. 125:470-477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Perenboom, R. M., P. Beckers, J. W. van der Meer, A. C. van Schijndel, W. J. Oyen, F. H. Corstens, and R. W. Sauerwein. 1996. Pro-inflammatory cytokines in lung and blood during steroid-induced Pneumocystis carinii pneumonia in rats. J. Leukoc. Biol. 60:710-715. [DOI] [PubMed] [Google Scholar]
- 25.Phair, J., A. Munoz, R. Detels, R. Kaslow, C. Rinaldo, A. Saah, et al. 1990. The risk of Pneumocystis carinii pneumonia among men infected with human immunodeficiency virus type 1. N. Engl. J. Med. 322:161-165. [DOI] [PubMed] [Google Scholar]
- 26.Pleyer, U., J. K. Milani, A. Dukes, J. Chou, S. Lutz, D. Ruckert, H. J. Thiel, and B. J. Mondino. 1995. Effect of topically applied anti-CD4 monoclonal antibodies on orthotopic corneal allografts in a rat model. Investig. Ophthalmol. Vis. Sci. 36:52-61. [PubMed] [Google Scholar]
- 27.Pohlmeyer, G., and F. Deerberg. 1993. Nude rats as a model of natural Pneumocystis carinii pneumonia: sequential morphological study of lung lesions. J. Comp. Pathol. 109:217-230. [DOI] [PubMed] [Google Scholar]
- 28.Reynolds, J., and C. D. Pusey. 1994. In vivo treatment with a monoclonal antibody to T helper cells in experimental autoimmune glomerulonephritis in the BN rat. Clin. Exp. Immunol. 95:122-127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Roths, J. B., and C. L. Sidman. 1993. Single and combined humoral and cell-mediated immunotherapy of Pneumocystis carinii pneumonia in immunodeficient scid mice. Infect. Immun. 61:1641-1649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Schmatz, D. M., M. A. Romancheck, L. A. Pittarelli, R. E. Schwartz, R. A. Fromtling, K. H. Nollstadt, F. L. Vanmiddlesworth, K. E. Wilson, and M. J. Turner. 1990. Τreatment of Pneumocystis carinii pneumonia with 1, 3-β-glucan synthesis. Proc. Natl. Acad. Sci. USA 87:5950-5954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Shellito, J., V. V. Suzara, W. Blumenfeld, J. M. Beck, H. J. Steger, and T. H. Ermak. 1990. A new model of Pneumocystis carinii infection in mice selectively depleted of helper T lymphocytes. J. Clin. Investig. 85:1686-1693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Shultz, L. D., P. A. Schweitzer, E. J. Hall, J. P. Sundberg, S. Taylor, and P. D. Walzer. 1989. Pneumocystis carinii pneumonia in scid/scid mice. Curr. Top. Microbiol. Immunol. 152:243-249. [DOI] [PubMed] [Google Scholar]
- 33.Stringer, J. R., and P. D. Walzer. 1996. Molecular biology and epidemiology of Pneumocystis carinii infection in AIDS. AIDS 10:561-571. [DOI] [PubMed] [Google Scholar]
- 34.Stumbles, P., and D. Mason. 1995. Activation of CD4+ T cells in the presence of a nondepleting monoclonal antibody to CD4 induces a Th2-type response in vitro. J. Exp. Med. 182:5-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Theus, S. A., R. P. Andrews, P. Steele, and P. D. Walzer. 1995. Adoptive transfer of lymphocytes sensitized to the major surface glycoprotein of Pneumocystis carinii confers protection in the rat. J. Clin. Investig. 95:2587-2593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Theus, S. A., A. G. Smulian, P. Steele, M. J. Linke, and P. D. Walzer. 1998. Immunization with the major surface glycoprotein of Pneumocystis carinii elicits a protective response. Vaccine 16:1149-1157. [DOI] [PubMed] [Google Scholar]
- 37.Theus, S. A., and P. D. Walzer. 1997. Adoptive transfer of specific lymphocyte populations sensitized to the major surface glycoprotein of Pneumocystis carinii decreases organism burden while increasing survival rate in the rat. J. Eukaryot. Microbiol. 44:23S-24S. [DOI] [PubMed]
- 38.Walzer, P. D., M. LaBine, T. J. Redington, and M. T. Cushion. 1984. Lymphocyte changes during chronic administration of and withdrawal from corticosteroids: relation to Pneumocystis carinii pneumonia. J. Immunol. 133:2502-2508. [PubMed] [Google Scholar]
- 39.Webb, M., D. W. Mason, and A. F. Williams. 1979. Inhibition of mixed lymphocyte response by monoclonal antibody specific for a rat T lymphocyte subset. Nature 282:841-843. [DOI] [PubMed] [Google Scholar]
- 40.Wiley, J. A., and A. G. Harmsen. 1995. CD40 ligand is required for resolution of Pneumocystis carinii pneumonia in mice. J. Immunol. 155:3525-3529. [PubMed] [Google Scholar]
- 41.Wright, T. W., F. Gigliotti, J. N. Finkelstein, J. T. McBride, C. L. An, and A. G. Harmsen. 1999. Immune-mediated inflammation directly impairs pulmonary function, contributing to the pathogenesis of Pneumocystis carinii pneumonia. J. Clin. Investig. 104:1307-1317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Wright, T. W., C. J. Johnston, A. G. Harmsen, and J. N. Finkelstein. 1997. Analysis of cytokine mRNA profiles in the lungs of Pneumocystis carinii-infected mice. Am. J. Respir. Cell Mol. Biol. 17:491-500. [DOI] [PubMed] [Google Scholar]
- 43.Wright, T. W., C. J. Johnston, A. G. Harmsen, and J. N. Finkelstein. 1999. Chemokine gene expression during Pneumocystis carinii-driven pulmonary inflammation. Infect. Immun. 67:3452-3460. [DOI] [PMC free article] [PubMed] [Google Scholar]