Abstract
Centrosomes mature as cells enter mitosis, accumulating γ-tubulin and other pericentriolar material (PCM) components. This occurs concomitant with an increase in the number of centrosomally organized microtubules (MTs). Here, we use RNA-mediated interference (RNAi) to examine the role of the aurora-A kinase, AIR-1, during centrosome maturation in Caenorhabditis elegans. In air-1(RNAi) embryos, centrosomes separate normally, an event that occurs before maturation in C. elegans. After nuclear envelope breakdown, the separated centrosomes collapse together, and spindle assembly fails. In mitotic air-1(RNAi) embryos, centrosomal α-tubulin fluorescence intensity accumulates to only 40% of wild-type levels, suggesting a defect in the maturation process. Consistent with this hypothesis, we find that AIR-1 is required for the increase in centrosomal γ-tubulin and two other PCM components, ZYG-9 and CeGrip, as embryos enter mitosis. Furthermore, the AIR-1–dependent increase in centrosomal γ-tubulin does not require MTs. These results suggest that aurora-A kinases are required to execute a MT-independent pathway for the recruitment of PCM during centrosome maturation.
Keywords: microtubule; mitosis; cell cycle; cancer
Introduction
In metazoans, centrosomes consist of a pair of centrioles surrounded by electron-dense pericentriolar material (PCM)* that directs the assembly of microtubules (MTs). During cell division, centrosomes undergo a series of structural changes (for review see Fry et al., 2000). During the G2/M transition, coincident with the activation of Cdk1, centrosomes mature, accumulating γ-tubulin and other PCM components and increasing in size and nucleating capacity (for review see Palazzo et al., 2000). Centrosomes separate before spindle assembly with timing that varies among cell types (Callaini et al., 1997; Keating and White, 1998; Fry et al., 2000) and organize the poles of the mitotic spindle after the breakdown of the nuclear envelope. The regulation of these cell cycle–dependent events remains largely unknown, although mitotic kinases likely play a key role (Fry et al., 2000; Nigg, 2001).
In addition to responding to cell cycle transitions, centrosomes also contribute to cell cycle progression. Recent experiments have shown that centrosomes are required for cells to enter S phase (Hinchcliffe et al., 2001; Khodjakov and Rieder, 2001). In addition, Cdk1 and polo-like kinase, two mitotic kinases that participate in a positive feedback loop regulating mitotic entry, accumulate at centrosomes (Glover et al., 1998; Ohi and Gould, 1999; Nigg, 2001). This has led to the speculation that centrosomes might accelerate the G2/M transition by facilitating the rapid coactivation of cell cycle regulators in proximity to target proteins involved in spindle assembly (Ohi and Gould, 1999).
In vertebrate cells, centrosome maturation occurs late in the G2/M transition. The amount of γ-tubulin at centrosomes increases 3–5-fold in late prophase (Khodjakov and Rieder, 1999), coincident with an increase in the size of centrosomal asters (Palazzo et al., 2000). Polo-like kinases have been directly implicated in PCM recruitment during maturation (Glover et al., 1998). Mutants in Drosophila polo fail to recruit γ-tubulin and the centrosomal protein CP190 (Sunkel and Glover, 1988; Donaldson et al., 2001), and injection of antibodies to Plk-1 into immortalized human tissue culture cells results in small centrosomes that fail to recruit γ-tubulin and MPM-2 phosphoepitopes (Lane and Nigg, 1996).
Aurora-A serine/threonine kinases are a recently emerging family of mitotic kinases that localize to centrosomes. Aurora-A kinases have been implicated in centrosome separation and spindle assembly (for review see Bischoff and Plowman, 1999; Giet and Prigent, 1999; Nigg, 2001). Interestingly, aurora-A is amplified frequently in human malignancies, and its overexpression can transform cells (Bischoff and Plowman, 1999). The activity of aurora-A kinase peaks during the G2/M transition (for review see Bischoff and Plowman, 1999), making it an attractive candidate for regulating centrosome maturation.
To analyze the role of aurora-A in centrosome maturation, we focus on the first mitotic division of the Caenorhabditis elegans embryo. A major experimental advantage of C. elegans is the ability to specifically destroy the mRNA transcript derived from any gene by dsRNA-mediated interference (RNAi) (Montgomery and Fire, 1998). Injection of dsRNA into adult hermaphrodites results in the formation of oocytes containing cytoplasm essentially cleared of the targeted protein within 20–30 h. Here, we combine RNAi of aurora-A with live and fixed assays for centrosome assembly and function to reveal a fundamental role for aurora-A in centrosome maturation.
Results and discussion
AIR-1 localizes to centrosomes and is required for spindle assembly
The C. elegans homologue of aurora-A, AIR-1, localizes to centrosomes and is required for normal spindle assembly (Schumacher et al., 1998). To investigate the role of aurora-A in the centrosome cycle, we first analyzed its localization during the first embryonic division. We raised an antibody to AIR-1 that detects a single band of ∼40 kD on Western blots. This band is reduced >90% in air-1(RNAi) worms (Fig. 1 A), confirming the specificity of our antibody. During fertilization, a sperm-derived centrosome enters the egg (which lacks centrosomes) and duplicates, resulting in two small centrosomes positioned between the sperm pronucleus and the cortex. AIR-1 localizes to centrosomes in early embryos (Fig. 1 B, top) and also weakly to astral and cytoplasmic MTs. In wild-type, the maternal pronucleus migrates toward the sperm pronucleus as the chromosomes condense. The pronuclei fuse, their nuclear envelopes break down, and the first mitotic spindle assembles. In mitotic embryos, AIR-1 concentrates in the centers of the asters (Fig. 1 B, bottom) in a donut-shaped region peripheral to γ-tubulin staining (Fig. 1 C) and also localizes along the base of astral MTs. The pattern of localization of AIR-1 is specific, since it is not observed in air-1(RNAi) embryos.
Figure 1.
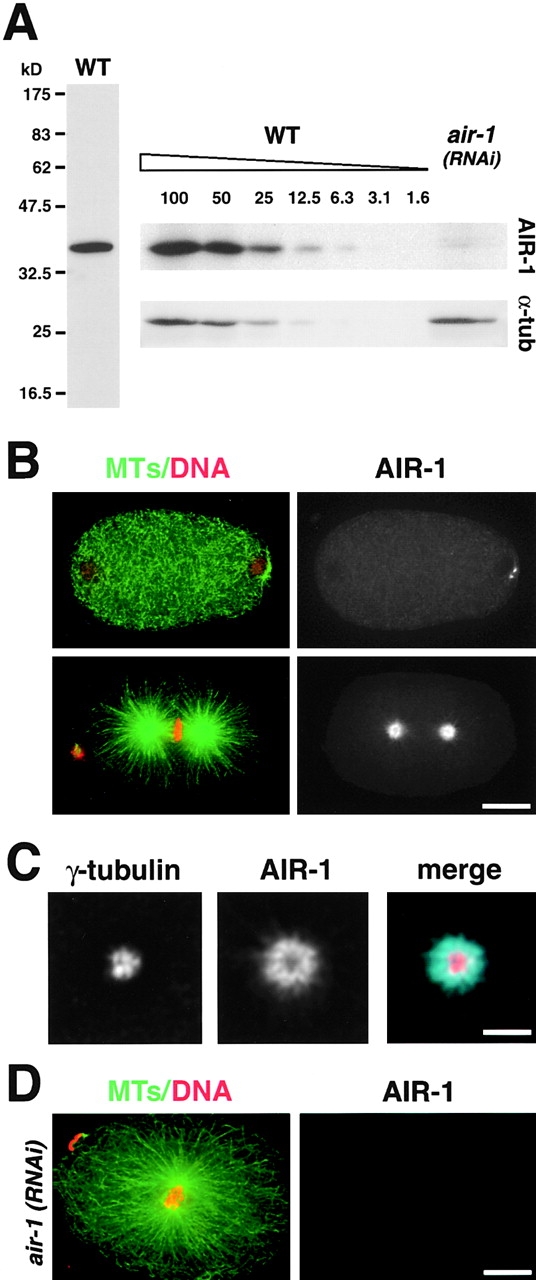
AIR-1 localizes to centrosomes and is required for spindle assembly. (A) The AIR-1 antibody detects a single band of ∼40 kD in extracts prepared from wild-type worms (left). (Right) Comparison of air-1(RNAi) worm extract with serial dilutions of wild-type extract (numbers indicate percentage of amount loaded in 100% lane) indicates >90% depletion of AIR-1. α-Tubulin was used as a loading control. (B) Wild-type embryos stained for MTs, DNA (left, green and red), and AIR-1 (right). A recently fertilized embryo (top) and a metaphase embryo (bottom) are shown. (C) A single deconvolved focal plane showing a centrosome from a metaphase embryo stained for γ-tubulin and AIR-1. (D) A mitotic air-1(RNAi) embryo stained for MTs, DNA (left, green and red), and AIR-1 (right). Bars: (B and D) 10 μm; (C) 2.5 μm.
In air-1(RNAi) embryos judged to be mitotic by the presence of condensed chromosomes, normal spindles were never observed. Instead, a pair of closely apposed centrosomal asters was associated with the condensed chromosomes (Fig. 1 D). These results confirm an essential role for AIR-1 in spindle assembly and are reminiscent of observations in Xenopus and Drosophila where inhibition of aurora-A function results in monopolar spindles with unseparated centrosomes (Glover et al., 1995; Roghi et al., 1998).
AIR-1 is required to maintain centrosome separation during spindle assembly
To examine the kinetics of centrosome separation, we filmed air-1(RNAi) embryos expressing GFP–α-tubulin (Fig. 2, A and B). In wild-type, the two centrosomes are located on opposite sides of the nucleus and maintain a constant separation just before and after nuclear envelope breakdown (NEBD). The distance between the centrosomes begins to increase late in metaphase and continues to increase as the spindle elongates during anaphase (Oegema et al., 2001). In air-1(RNAi) embryos, centrosomal asters were detected associated with sperm pronuclei as early as 600 s before NEBD (Fig. 2 A, −563 s). The asters remained associated with the sperm pronucleus and always separated (Fig. 2 A, −282 s). Cytoplasmic MTs depolymerized in both wild-type (Fig. 2 A, −446 compared with +0 s) and air-1(RNAi) embryos (Fig. 2 A, −563 s compared with +97 s) as they entered mitosis. However, subsequent to NEBD the asters in air-1(RNAi) embryos collapsed together (collapse occurs between +97 and +177 s; Fig. 2 A), resulting in two small relatively bright asters lying side by side. In some cases, the asters moved apart slightly during late anaphase/telophase. In other examples, the two asters remained unseparated and moved around the embryo or rocked back and forth coincident with the onset of cortical contractions. These results suggest that AIR-1 is not necessary for initial separation of centrosomes in C. elegans but is required to maintain centrosome separation during spindle assembly.
Figure 2.
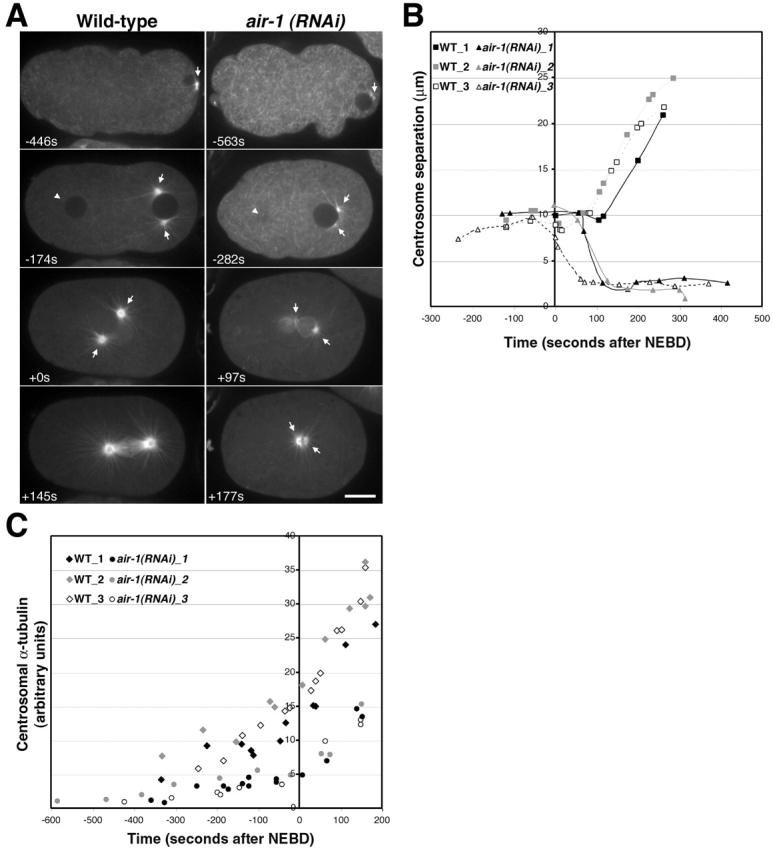
AIR-1 is required to maintain centrosome separation during spindle assembly. (A) Panels summarize time-lapse recordings of wild-type (left) and air-1(RNAi) embryos (right) expressing GFP–α-tubulin. Times are seconds after NEBD. In wild-type (left), unseparated asters (−446 s, arrow) associate with the sperm pronucleus. (−174 s) The asters separate and position themselves on opposite sides of the sperm pronucleus. The maternal pronucleus is also indicated (arrowhead). (+0 s) The pronuclei meet, move with the asters (arrows) to the center of the embryo, and the nuclear envelope breaks down. (+145) After NEBD, the asters increase in size and become the poles of the mitotic spindle. In air-1(RNAi) embryos (right), the asters (arrows) separate as in wild-type (−563 s and −282 s). The maternal pronucleus (arrowhead) is out of focus. After NEBD, the centrosomal asters (arrows) collapse toward each other till they sit side by side (+97 s and +177 s). See also videos 1 and 2 available at http://www.jcb.org/cgi/ content/full/jcb.200108051/DC1. (B) Kinetic traces of centrosome separation. Centrosome separation was measured for each time point where both centrosomes were in focus in seven wild-type and seven air-1(RNAi) embryos. Three examples are shown for each. Times are with respect to NEBD. (C) Kinetic traces of centrosomal α-tubulin fluorescence. Centrosomal fluorescence was quantitated in five wild-type and six air-1(RNAi) embryos. Three traces are shown for each. Bar, 10 μm.
We noticed that mitotic centrosomal asters in air-1(RNAi) embryos appeared less robust than in wild-type. To quantify this, we measured centrosomal α-tubulin fluorescence (Fig. 2 C). In wild-type, centrosomal α-tubulin fluorescence increased during the interval between 300 s before NEBD and anaphase onset (∼155 s after NEBD). In air-1(RNAi) embryos, centrosomal α-tubulin fluorescence also increased but ultimately reached only ∼40% of wild-type levels. This suggests that mitotic centrosomes in air-1(RNAi) embryos organize fewer MTs than in wild-type.
AIR-1 is required to recruit centrosomal γ-tubulin during maturation
The small centrosomal asters in mitotic air-1(RNAi) embryos suggested a role for AIR-1 in centrosome maturation. To determine if AIR-1 has a role in the accumulation of centrosomal γ-tubulin during this transition, we filmed air-1(RNAi) embryos expressing both GFP histone, as a marker for cell cycle progression, and GFP–γ-tubulin (Fig. 3 A). γ-Tubulin was visible shortly after fertilization at the still tiny centrosomes in both wild-type and air-1(RNAi) embryos (Fig. 3 A, −484 s and −608 s). The cell cycle progressed in air-1(RNAi) embryos as evidenced by DNA condensation, the release of cortical contraction at the end of pseudocleavage, NEBD, the onset of cortical contractions coincident with the initiation of cytokinesis in wild-type, and reformation of the nuclear envelope. However, the dramatic accumulation of centrosomal γ-tubulin that occurs in wild-type during the interval between 300 s before NEBD and anaphase onset (Fig. 3 A, compare –256 s with +122 s) was completely absent in air-1(RNAi) embryos (Fig. 3 A, right, all panels). Quantification of integrated fluorescence intensity showed that centrosomal γ-tubulin fluorescence does not change significantly as air-1(RNAi) embryos enter mitosis (Fig. 3 B). In summary, our results show that the cell cycle progresses in air-1(RNAi) embryos. However, the dramatic increase in centrosomal γ-tubulin, a hallmark of centrosome maturation, never occurs.
Figure 3.
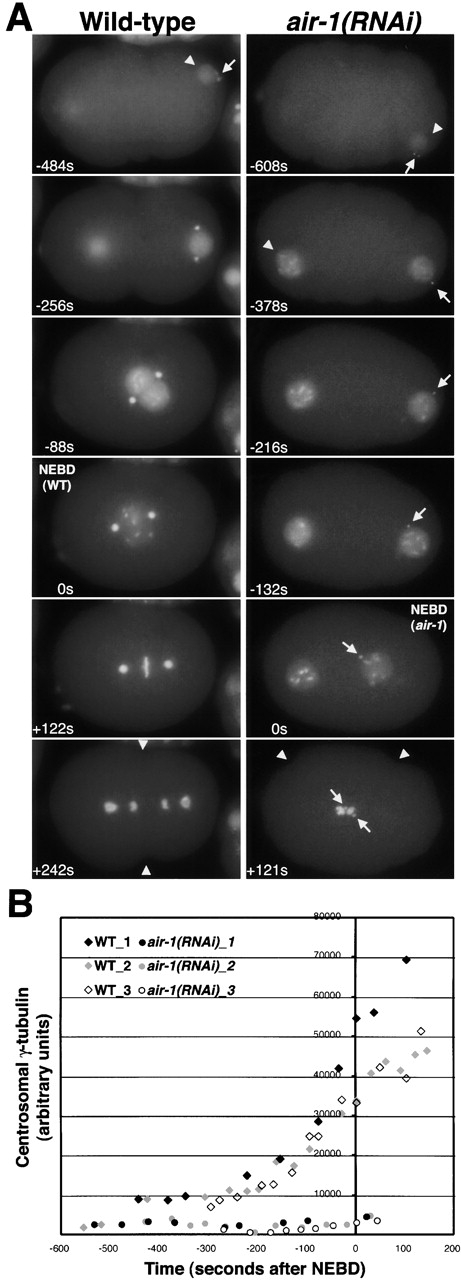
AIR-1 is required for the accumulation of centrosomal γ-tubulin during maturation. (A) Panels summarize time-lapse recordings of wild-type (left) and air-1(RNAi) embryos (right) expressing GFP histone and GFP–γ-tubulin. Both sequences start at a similar early embryonic stage, judged by the extent of cortical ruffling, the small size of the sperm pronuclei (arrowheads), and the lack of separation of γ-tubulin–labeled centrosomes (arrow). The sequences end with onset of cytokinesis (wild-type, +242 s) or cortical contractions (air-1(RNAi), +121 s, arrowheads). The duration of both recordings is ∼12 min. Times are seconds after NEBD. The times in the corresponding panels differ by ∼2 min because NEBD is slightly delayed in air-1(RNAi) embryos. See videos 3–6 available at http://www.jcb.org/cgi/content/full/jcb.200108051/DC1. (Left) In wild-type, the DNA condenses, beginning before the migration of the pronuclei toward each other and continuing during migration. This occurs concomitant with an increase in the amount of centrosomal γ-tubulin (compare −256 s with −88 s). The accumulation of centrosomal γ-tubulin continues after NEBD as the mitotic spindle assembles (+122 s). (Right) In air-1(RNAi) embryos, the chromosomes condense with timing similar to wild-type, but γ-tubulin fails to accumulate at centrosomes (arrows, all panels). The migration of the maternal pronucleus toward the sperm pronucleus is lethargic, probably due to the failure of centrosomes to nucleate robust mitotic asters, but the two pronuclei eventually move toward each other, and the nuclear envelope breaks down (0 s). Chromosomes never align properly, but cortical contractions begin coincident with cytokinesis in wild-type embryos (+121 s). Cytokinesis does not succeed in air-1(RNAi) embryos (unpublished data). (B) Kinetic traces of centrosomal γ-tubulin fluorescence. Centrosomal fluorescence was quantified in 7 wild-type and 10 air-1(RNAi) embryos. Three traces are shown for each. Bar, 10 μm (as in Fig. 2).
Interestingly, we also noticed that the interval between NEBD and the onset of cortical contractions was slightly shorter in air-1(RNAi) embryos (168 ± 64 s; Fig. 3 A, air-1(RNAi), 0 s and +121 s) than in wild-type (268 ± 13 s; Fig. 3 A, wild-type, 0 s and +242 s). This result suggested that NEBD might be delayed. Consistent with this, in air-1(RNAi) embryos we often observed fully condensed rod-like chromosomes in nuclei that had not yet broken down (Fig. 3 A; −132 s; videos 4, 5, and 6 available at http://www.jcb.org/cgi/content/full/jcb.200108051/DC1), whereas in wild-type completion of condensation to form rod-like chromosomes usually occurs coincident with NEBD (Fig. 3 A, 0 s; video 3 available at http://www.jcb.org/cgi/content/full/jcb.200108051/DC1). From these results, we conclude that NEBD is delayed by ∼1–2 min with respect to other cell cycle events in air-1(RNAi) embryos.
AIR-1 is required to recruit other PCM components during centrosome maturation
The results above showed that a GFP–γ-tubulin fusion protein fails to accumulate at mitotic centrosomes in air-1(RNAi) embryos. To confirm this for endogenous γ-tubulin, we fixed air-1(RNAi) embryos and stained them for DNA, MTs, γ-tubulin, and AIR-1 (Fig. 4 A). In mitotic air-1(RNAi) embryos, AIR-1 was depleted to undetectable levels (Fig. 1 D), and γ-tubulin staining foci were much smaller than in wild-type (Fig. 4 A), consistent with the live analysis. Interestingly, each centrosome in the air-1(RNAi) embryos often appeared as a pair of small dots of γ-tubulin staining (Fig. 4 A, bottom right, inset). These dots are likely to be γ-tubulin associated with paired centrioles that have not yet separated.
Figure 4.
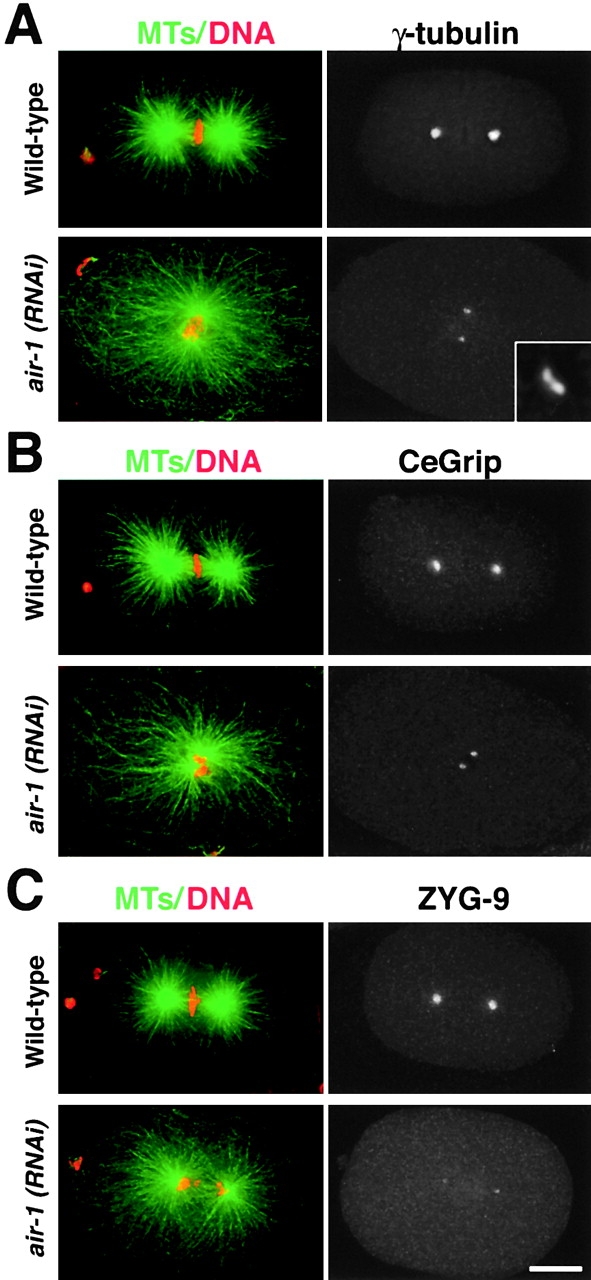
AIR-1 is required for the accumulation of γ-tubulin, CeGrip, and ZYG-9 during centrosome maturation. Wild-type and air-1(RNAi) embryos stained for MTs and DNA (left, green and red) and for either γ-tubulin (A), CeGrip (B), or ZYG-9 (C) are shown. Inset in A is magnified 5.5-fold. Bar, 10 μm.
To further investigate the role of AIR-1 in the recruitment of PCM components, we tested whether two additional centrosomal proteins CeGrip, a C. elegans homologue of Spc98p (unpublished data; B. Haberman, personal communication), and ZYG-9 (Matthews et al., 1998) also require AIR-1 to accumulate at mitotic centrosomes. We fixed and stained wild-type and air-1(RNAi) embryos for CeGrip (Fig. 4 B) and ZYG-9 (Fig. 4 C). Centrosomes labeled with either marker also remained tiny in mitotic air-1(RNAi) embryos, demonstrating that AIR-1 is required for the centrosomal accumulation of three PCM components during maturation.
AIR-1–dependent recruitment of centrosomal γ-tubulin does not require MTs
Our results show that AIR-1 is required for the increase in centrosomally organized MTs and for the accumulation of centrosomal γ-tubulin as embryos enter mitosis. To test the possibility that the decrease in centrosomally organized MTs is responsible for the failure to accumulate centrosomal γ-tubulin, we treated wild-type and air-1(RNAi) embryos with nocodazole to depolymerize MTs and stained them for MTs, DNA, γ-tubulin, and AIR-1 (Fig. 5). MTs were completely depolymerized in nocodazole-treated embryos with only centrosomes detectable using an α-tubulin antibody (Fig. 5, top). Embryos were treated with nocodazole for 7 min before fixation (see Materials and methods). Since pronuclear migration requires MTs, we analyzed embryos with condensed chromosomes in which the pronuclei were still separated. This separation indicates that MT depolymerization occurred at a stage similar to the −256 s panel in Fig. 3 A, which is before the accumulation of additional centrosomal γ-tubulin. Despite complete depolymerization of MTs, centrosomes in wild-type embryos accumulated γ-tubulin and AIR-1 to normal levels during the 7 min incubation in nocodazole. In contrast, centrosomes in nocodazole-treated air-1(RNAi) embryos remained small, similar to those in untreated air-1(RNAi) embryos. From these results, we conclude that the AIR-1–dependent accumulation of centrosomal γ-tubulin during maturation does not require MTs.
Figure 5.
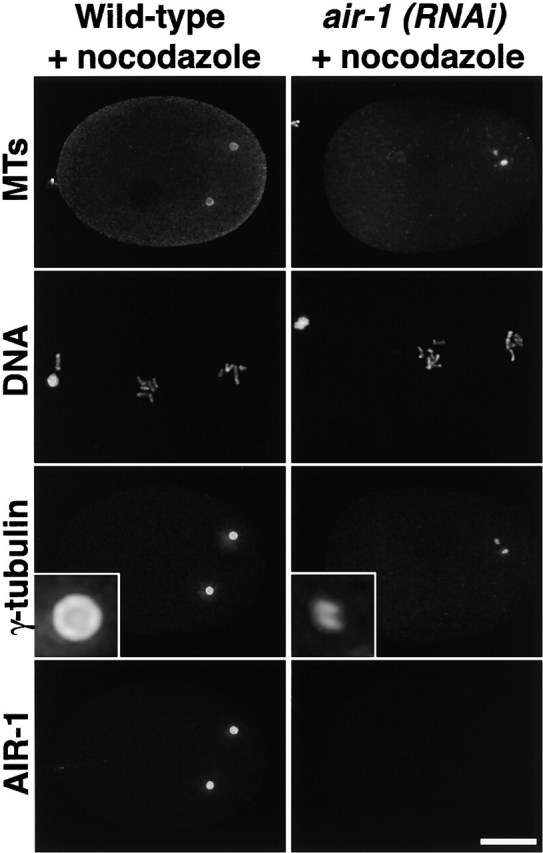
The effect of AIR-1 depletion on the accumulation of centrosomal γ-tubulin is MT independent. Wild-type (left) and air-1(RNAi) embryos (right) were treated with nocodazole, fixed, and stained for MTs, DNA, γ-tubulin, and AIR-1. γ-Tubulin staining is reduced dramatically, and two small foci of γ-tubulin staining are observed for each centrosome in air-1(RNAi) embryos (compare wild-type and air-1(RNAi) insets). Insets are magnified 5.5-fold. Bar, 10 μm.
Cumulatively, our results demonstrate that the aurora-A kinase, AIR-1, is required to execute a MT-independent pathway for the recruitment of PCM components during centrosome maturation. Spindle assembly also fails in air-1(RNAi) embryos. One possibility, suggested by our results, is that centrosome maturation is a prerequisite for spindle assembly. Our kinetic analysis of centrosome dynamics revealed that AIR-1 is not required for centrosome separation but instead is required to prevent the centrosomes from collapsing together after NEBD. In Drosophila and Xenopus, inhibition of aurora-A function results in the generation of monopolar spindles with unseparated centrosomes (Glover et al., 1995; Roghi et al., 1998). Kinetic studies during a single cell cycle will be needed to determine if these phenotypes result from an initial failure of centrosome separation or, as we have found for C. elegans, from a failure to maintain centrosome separation during the early stages of spindle assembly. Differences in the onset of the mitotic phenotype associated with inhibition of aurora-A could also arise from variations in the timing of centrosome separation among cell types. This event occurs relatively early in the mitotic cycle in C. elegans and Drosophila embryos and just before spindle assembly in vertebrate tissue culture cells (Callaini et al., 1997; Keating and White, 1998; Fry et al., 2000).
NEBD is also delayed with respect to other cell cycle events in air-1(RNAi) embryos. One possibility is that aurora-A independently regulates and ensures the coordination of multiple events during mitotic entry including centrosome maturation, spindle assembly, and NEBD. A second possibility, which we favor, is that centrosome maturation is required for spindle assembly and timely NEBD. The accumulation of cell cycle regulators such as polo-like kinases and Cdk1 at centrosomes prompts the speculation that maturation might promote NEBD and spindle assembly by facilitating the rapid coactivation of cell cycle regulators in proximity to target proteins important for these processes. Alternatively, the failure of centrosome maturation might trigger a checkpoint that delays subsequent cell cycle events (Lane and Nigg, 1996).
Materials and methods
RNA-mediated interference
For production of dsRNA to air-1 (gcctctcggaaaaggaaagt, ccttgattctggcgatcaat), the primers in parentheses with tails containing T3 and T7 promoters were used to amplify regions from genomic N2 DNA or the cDNA yk364b4. PCR products were transcribed and double stranded RNA prepared as described (Oegema et al., 2001).
Live imaging and quantification of centrosomal fluorescence
Live analysis and RNAi in strains expressing GFP fusions were performed as described (Oegema et al., 2001). GFP–α-tubulin and GFP–γ-tubulin fluorescence were quantified from live recordings made using spinning disc confocal and wide-field microscopy, respectively. The bright centrosomal regions in the γ-tubulin and α-tubulin sequences were circled, the average fluorescence intensity in this region and in a similarly sized background region were determined using Metamorph software (Universal Imaging Corp.), and the integrated centrosomal fluorescence was calculated from these values.
Antibodies and fixed imaging
Antibodies to the COOH-terminal 12 amino acids of AIR-1 and the COOH-terminal 17 amino acid of CeGrip and γ-tubulin were raised, affinity purified, and directly labeled as described (Oegema et al., 2001). Antibodies to ZYG-9 were a gift from A. Pozniakovsky (Max-Planck Institute of Molecular Cell Biology and Genetics, Dresden, Germany). Embryos were fixed and processed for immunofluorescence as described (Oegema et al., 2001) and imaged using a 63×, 1.4 NA PlanApochromat lens on a Deltavision microscope. Three-dimensional wide-field data sets were computationally deconvolved and projected (Applied Precision).
Western blotting
90 air-1(RNAi) animals or wild-type controls were snap frozen in 45 μl of M9 and then lysed by addition of 45 μl at room temperature 2× sample buffer (125 mM Tris, pH 6.8, 6% SDS wt/vol, 10% β-mercaptoethanol vol/vol, 20% glycerol vol/vol) followed by sonication for 10 min at 80°C in a waterbath sonicator. Immunoblots were probed using 1 μg/ml anti–AIR-1 and detected using a HRP-conjugated secondary antibody (1:3,000; Bio-Rad Laboratories) and subsequently with DM1a (1:1,000; Sigma-Aldrich) using an alkaline-phosphatase–conjugated secondary antibody (1:2,000; Jackson ImmunoResearch Laboratories).
Nocodazole treatment
Worms were dissected on slides coated with 1 mg/ml polylysine in egg buffer (118 mM NaCl, 48 mM KCl, 2 mM CaCl2, 2 mM MgCl2, 0.025 mM Hepes, pH 7.4) containing 10 μg/ml nocodazole. A coverslip was placed on top, and gentle pressure was applied to crack embryo egg shells. Slides were incubated in the presence of nocodazole for 7 min in a humid chamber before fixation.
Online supplemental material
Videos for Fig. 2 show embryos expressing GFP–α-tubulin (video 1, wild-type, and video 2, air1[RNAi]). Videos for Fig. 3 show γ-tubulin accumulation and cell cycle progression (video 3, wild-type, and videos 4 and 6, air1[RNAi]). In video 5, videos 3 and 4 are montaged to facilitate comparison of wild-type and air-1(RNAi) embryos. The maternal pronucleus is initially on the left and the paternal pronucleus on the right in all videos. Playback rate is 10 frames/s. Videos 1–6 are available at http://www.jcb.org/cgi/content/full/jcb.200108051/DC1.
Supplemental Material
Acknowledgments
We thank Arshad Desai for microscopy assistance and many helpful discussions during the course of this work; Chris Wiese, Elly Tanaka, Martin Srayko, Stephan Grill, and Arshad Desai for comments on the article; Yuji Kohara (National Institute of Genetics, Mishima, Japan) for cDNAs used for RNA and antibody production; and Geraldine Seydoux (Johns Hopkins University, Baltimore, MD) for vectors and advice on germline expression.
K. Oegema was supported by a fellowship from the Helen Hay Whitney Foundation.
The online version of this article contains supplemental material.
Footnotes
Abbreviations used in this paper: MT, microtubule; NEBD, nuclear envelope breakdown; PCM, pericentriolar material; RNAi, RNA-mediated interference.
References
- Bischoff, J.R., and G.D. Plowman. 1999. The Aurora/Ipl1p kinase family: regulators of chromosome segregation and cytokinesis. Trends Cell Biol. 9:454–459. [DOI] [PubMed] [Google Scholar]
- Callaini, G., W.G. Whitfield, and M.G. Riparbelli. 1997. Centriole and centrosome dynamics during the embryonic cell cycles that follow the formation of the cellular blastoderm in Drosophila. Exp. Cell Res. 234:183–190. [DOI] [PubMed] [Google Scholar]
- Donaldson, M.M., A.A.M. Tavares, H. Ohkura, P. Deak, and D.M. Glover. 2001. Metaphase arrest with centromere separation in polo mutants of Drosophila. J. Cell Biol. 153:663–675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fry, A.M., T. Mayor, and E.A. Nigg. 2000. Regulating centrosomes by protein phosphorylation. Curr. Top. Dev. Biol. 49:291–312. [DOI] [PubMed] [Google Scholar]
- Giet, R., and C. Prigent. 1999. Aurora/Ipl1p-related kinases, a new oncogenic family of mitotic serine-threonine kinases. J. Cell Sci. 112:3591–3601. [DOI] [PubMed] [Google Scholar]
- Glover, D.M., M.H. Leibowitz, D.A. McLean, and H. Parry. 1995. Mutations in aurora prevent centrosome separation leading to the formation of monopolar spindles. Cell. 81:95–105. [DOI] [PubMed] [Google Scholar]
- Glover, D.M., I.M. Hagan, and A.A. Tavares. 1998. Polo-like kinases: a team that plays throughout mitosis. Genes Dev. 12:3777–3787. [DOI] [PubMed] [Google Scholar]
- Hinchcliffe, E.H., F.J. Miller, M. Cham, A. Khodjakov, and G. Sluder. 2001. Requirement of a centrosomal activity for cell cycle progression through G1 into S phase. Science. 291:1547–1550. [DOI] [PubMed] [Google Scholar]
- Keating, H.H., and J.G. White. 1998. Centrosome dynamics in early embryos of Caenorhabditis elegans. J. Cell Sci. 111:3027–3033. [DOI] [PubMed] [Google Scholar]
- Khodjakov, A., and C.L. Rieder. 1999. The sudden recruitment of γ-tubulin to the centrosome at the onset of mitosis and its dynamic exchange throughout the cell cycle do not require microtubules. J. Cell Biol. 146:585–596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khodjakov, A., and C.L. Rieder. 2001. Centrosomes enhance the fidelity of cytokinesis in vertebrates and are required for cell cycle progression. J. Cell Biol. 153:237–242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lane, H.A., and E.A. Nigg. 1996. Antibody microinjection reveals an essential role for human polo-like kinase 1 (Plk1) in the functional maturation of mitotic centrosomes. J. Cell Biol. 135:1701–1713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matthews, L.R., P. Carter, D. Thierry-Mieg, and K. Kemphues. 1998. ZYG-9, a Caenorhabditis elegans protein required for microtubule organization and function, is a component of meiotic and mitotic spindle poles. J. Cell Biol. 141:1159–1168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Montgomery, M.K., and A. Fire. 1998. Double-stranded RNA as a mediator in sequence-specific genetic silencing and co-suppression. Trends Genet. 14:255–258. [DOI] [PubMed] [Google Scholar]
- Nigg, E.A. 2001. Cell division: mitotic kinases as regulators of cell division and its checkpoints. Nat. Rev. Mol. Cell. Biol. 2:21–32. [DOI] [PubMed] [Google Scholar]
- Oegema, K., A. Desai, S. Rybina, M. Kirkham, and A.A. Hyman. 2001. Functional analysis of kinetochore assembly in C. elegans. J. Cell Biol. 153:1209–1226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohi, R., and K.L. Gould. 1999. Regulating the onset of mitosis. Curr. Opin. Cell Biol. 11:267–273. [DOI] [PubMed] [Google Scholar]
- Palazzo, R.E., J.M. Vogel, B.J. Schnackenberg, D.R. Hull, and X. Wu. 2000. Centrosome maturation. Curr. Top. Dev. Biol. 49:449–470. [DOI] [PubMed] [Google Scholar]
- Roghi, C., R. Giet, R. Uzbekov, N. Morin, I. Chartrain, R. Le Guellec, A. Couturier, M. Doree, M. Philippe, and C. Prigent. 1998. The Xenopus protein kinase pEg2 associates with the centrosome in a cell cycle-dependent manner, binds to the spindle microtubules and is involved in bipolar mitotic spindle assembly. J. Cell Sci. 111:557–572. [DOI] [PubMed] [Google Scholar]
- Schumacher, J.M., N. Ashcroft, P.J. Donovan, and A. Golden. 1998. A highly conserved centrosomal kinase, AIR-1, is required for accurate cell cycle progression and segregation of developmental factors in C. elegans embryos. Development. 125:4391–4402. [DOI] [PubMed] [Google Scholar]
- Sunkel, C.E., and D.M. Glover. 1988. Polo, a mitotic mutant of Drosophila displaying abnormal spindle poles. J. Cell Sci. 89:25–38. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


