Abstract
To assess whether the postural function is impaired by comparing the performances in upright standing at ground and bevel levels in adult subjects with spina bifida occulta (SBO). Eighty subjects with SBO (38 with minor type and 42 with major type) and 35 healthy control subjects participated in the study. All participants performed ten tests while standing upright on a platform at ground level (0°, baseline) and on a beveled surface (with their feet in dorsiflexion and plantarflexion at 10° and 20°). Tests were done with their eyes open and closed. The postural sway was examined using a force platform (CATSYS, Danish) that records sway intensity and velocity. Sway intensity and sway velocity were universally associated with group, degree of bevel, open- or closed-eyes condition, and dorsiflexion or plantarflexion after adjusting for age and gender. With respect to sway intensity, the differences of minor or major SBO group were significantly decreased at different bevel degrees when compared with control groups, whereas the differences between minor and major SBO were significant differences at 10° and 20°. With respect to sway velocity, the differences of major SBO group were significantly decreased at different bevel degrees when compared with minor SBO and control groups, whereas the difference in minor SBO was only significant at 0° when compared with control. Group differences (minor SBO vs. control, major SBO vs. control) showed a significant decrease in sway velocity when comparing at 10° than at 0° and at 20° than at 0°. In all subjects with SBO, the sway intensity/velocity values obtained with open eyes and with plantarflexion had lower values, when compared with values obtained with closed eyes and with dorsiflexion. This study supports the hypothesis that SBO impairs control of postural sway in both the resting upright and stressful postures. Our results imply that the larger the bone defect at the lumbosacral midline, the more the group differences in different stressful conditions. Both velocity and intensity were able to reflect the function of the postural sway from our results. This is the first report to add the bevel degree and foot position, as well as visual input as being the part of the study in investigating the postural sway.
Keywords: Spina bifida occulta, Sway intensity, Sway velocity, Bevel level, Posture
Introduction
Spina bifida occulta (SBO) is a developmental anomaly that results in defects of the spine that are limited to a failure of vertebral arch bony growth and fusion. On radiologic studies, SBO appears as a cleft or gap between the vertebral spinous process and laminae, but protrusion of the cord is not observed because the neural tube is closed. SBO most frequently occurs in the lumbosacral spine, such as at L5, S1, or S2 alone or at S1 and S2 [17]. Prevalence of SBO in the general population is 23% in England [17], 16.3% in Turkey [1], and 18–34% in the United States [2].
Symptoms of SBO may be absent, minimal or severe depending on the extent of neural involvement and on whether the disease is static or slowly progressive. Some authors have dismissed SBO as a finding too common to be of importance, especially if it occurs without low back pain. This assertion agrees with the results of surveys in the general population [18], and in athletes [42], soldier volunteers [2], and skiers [35].
Increasing evidence links SBO to specific anomalies and clinically important manifestations, including skin stigmata in the lumbosacral region, neurogenic foot deformities, bladder and bowel disturbances, intraspinal lipoma, tethered cord syndrome [10, 41], a low-placed conus medullaris [33], disk pathology, lumbar spondylolysis, foot deformities, and syringomyelia [8, 21]. In a recent survey examining the prevalence of secondary impairment in young adults with all types of spina bifida, Verhoef et al. [44] reported that although almost half of the patients with SBO (n = 37) have foot deformities, 95% of them are either normal ambulators or community ambulators. One of the noticeable things in the study was the problem with sitting balance that is not present in their SBO patients. Nevertheless, our interest focused on balance in standing rather than sitting, and we know of no study referencing the standing balance of adult subjects with SBO.
Fidas et al. [17] have described four X-ray patterns of spinal abnormalities in the formation of the posterior arch, that is SBO. Of them, type 1 is a minimal defect in L5 or S1. Type 2 is cross-over of overlapping laminae in L5 or S1. Type 3 is a symmetric, narrow or wide midline defect in L5 and/or S1. Type 4 is an asymmetric midline defect in L5 or S1. All of their patterns show a minor body defect, without descriptive occurrence of SBO patterns elongating in the entire sacrum. We encountered two subjects whose plain KUB films showed a large longitudinal defect in the posterior part of the entire sacrum and was misdiagnosed to have had a laminectomy previously. We also found a cadaver picture with a similar large defect on the Internet [27]. We wondered whether the entire major cleft is different from a minor cleft; therefore, we attempted to investigate the difference between the two kinds of SBO.
Documented parameters measuring postural sway include sway velocity and intensity [15, 30, 36]. However, any one of three ways, foot position, bevel plane, as well as visual input, may be a confounding factor [13, 15, 28, 34]. Therefore, considering the variances of age, gender, visual input, foot position, and bevel angles, we conducted this study to determine if postural function is impaired in subjects with SBO and if so, to observe its possible influence during challenge testing. We hypothesized that SBO impairs control of postural sway in both the resting upright and stressful positions.
Materials and methods
Subjects and control groups
Subjects with SBO (code 756.17 in the International Classification of Diseases, 9th Revision, Clinical Modification) [7] were surveyed in our hospital whether being an inpatient or outpatient between September 2000 and February 2004. All subjects received orthopedic and neurologic examinations. The exclusion criteria were skeletal anomaly, foot shape/formation malformations, sensory impairment, cutaneous stigmata in the form of lipoma, tufts of hair, or dermal sinuses on the back, history of surgery in lower trunk or pelvis, history of rheumatologic or immune disorder(s), pathologic conditions in the lumbosacral spine from previous computed axial tomography (CAT) scan or magnetic resonance imaging (MRI), such as posterior or ectopic herniation of an intervertebral disk, remarkable scoliosis, spondylolysis, or spondylolisthesis. Any possibility of the tethered cord syndrome or even DeAnquin’s disease would be excluded. In order to exclude some hidden or new onset injuries in the back or spine, we checked all of the cases using MRI or CAT scan prior to testing.
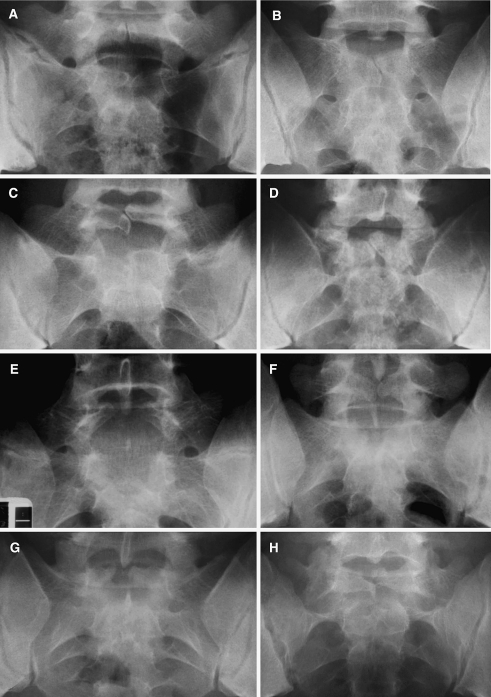
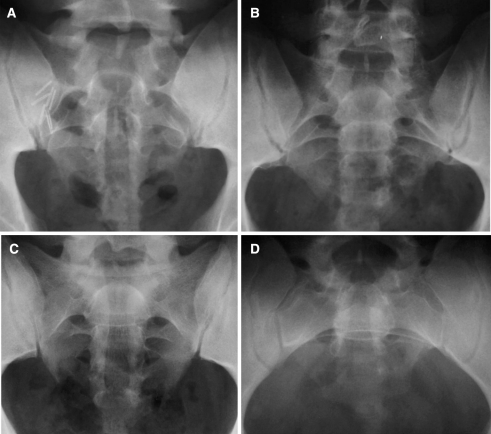
A radiologist from our hospital surveyed the conventional radiographies of the lumbosacral spine of all participants, who were then divided into two experimental groups according to the radiologic characteristics of SBO, that is, minor or major bony defects/clefts. Subjects with any of the four types of L5 and/or S1 SBO as described by Fidas et al. [17] were included in the minor group (Fig. 1). Subjects with defects involving S1, S2, and/or L5, combined with a high level of opening of the posterior sacral arc in S3 (e.g., SBO in the entire sacrum) were included in the major group (Fig. 2).
Fig. 1.
Four patterns of minor SBO. Type 1 is minimal defect in L5 (a) or S1 (b). Type 2 is cross-over of overlapping laminae in L5 (c) or S1 (d). Type 3 is a symmetric, wide midline defect in L5-S1 (e) or a narrow defect in L5-S1 (f). Type 4 is an asymmetric midline defect in S1 (g, h)
Fig. 2.
Various patterns of major SBO involving the entire sacrum (a–d). Please note that surgical metallic clips show in the right lower quadrant of abdomen after previous appendicitis in a. Bony defect extends up to L5 in b
We enrolled 80 subjects (73 male, 7 female) aged 19–32 years. The minor group included 38 subjects aged 20–26 years (median age, 22.6 years; body weight, 53.6 ± 5.8 kg), and the major SBO group included 42 subjects aged 19–32 years (median age, 23.4 years; body weight, 54. 6 ± 4.1 kg). As a control group, 35 healthy subjects (30 male, 5 female) aged 18–28 years (median age, 23.2 years; body weight, 57. 6 ± 2.6 kg) were enrolled.
All participants provided written informed consent, and the human ethics committee of our medical center approved this study, which conformed to the principles of the Declaration of Helsinki.
Study design
This cross-sectional cohort study with repeated measurements was conducted to evaluate sway parameters at 0° (baseline at ground level) and at two bevel degrees (10° and 20°).
Studies were performed by having the participants stand upright in a relaxed manner on a firm surface. All participants were barefoot and permitted to choose a comfortable foot position with the soles of their feet opened at an angle of about 30°. Postural sway was tested by asking the subject to stand upright motionless and quiet on two legs on a force platform at 0° (i.e., level to the ground) for 60 s. The test was performed under two conditions: with the subject’s eyes open and with their eyes closed. Subjects were asked to stare at a cross sign in front of them during testing.
Postural sway was then measured under eight support-surface conditions. An angle-adjustable frame was fabricated to control a platform to disrupt proprioceptive input for postural control (in the sagittal direction). The experimental bevel angles were created by inclining the platform. Subjects were instructed to stand upright on the platform beveled at 10° or 20° with their ankles in plantarflexion or dorsiflexion. Dorsiflexion was achieved by having the subjects turn on the platform 180° relative to their original position. Each condition was tested twice for 60 s, once with the subjects’ eyes open and once with their eyes closed, and the subject was allowed to rest for 3–5 min after each test. All participants were tested individually for approximately 40–50 min. Subjects were allowed to recover from their abnormal posture by contracting their ankle and/or abdominal muscles in the beveled standing position.
This procedure was the same as that Després et al. [15] described, except that we used an inclined surface rather than a foam pad. Standing upright on a beveled platform is disruptive to the proprioceptive feedback of postural control [13, 28, 34].
Measurements
Analysis of postural sway was performed using a portable CATSYS force platform system (Danish Product Development, Denmark), which was based on a data logger to communicate with a computer via a serial cable. It recorded signal data from a force plate by means of test software. Using this system, we obtained the sway parameters of intensity and velocity. This platform has been shown to be reliable and reproducible as a measure of postural sway [15, 32]. We performed four parameters of sway measurements, and this article reports the results of sway velocity and intensity.
The definitions of the sway parameters followed those of Després et al. [15] and Nadeau [32]. Sway intensity was defined as the root mean square of acceleration, recorded in the 0.2–10.2 Hz band during the testing period, and was determined by analyzing the trajectory using the fast Fourier transformation method as equipped in the signal processor. Sway velocity was defined as the average travel speed of the center of pressure in the horizontal force plate plane and was calculated by dividing the total length of the trajectory (in millimeters) of the center of pressure by the recording period length (in seconds). Sway velocity and intensity has been known to be able to reflect the function of postural sway [15, 30, 36].
Data and statistical analyses
Generalized estimating equations (GEEs) were used to assess the association of interests (group effects, bevels effects, and group–by–degree effects) and to adjust the correlations from repeated measurements in the same subjects obtained under different conditions (bevels of 10° vs. 0° and 20° vs. 0°, plantarflexion vs. dorsiflexion, and eyes open vs. eyes closed), as well adjusted for potential confounders (age and gender). A P value of 0.05 or less indicated a statistically significant difference. Contrast estimates were performed after the final GEE models of sway intensities and sway velocities, respectively. All data analysis was assessed by PROC GENMOD (SAS, 9.13, Carry, NC, USA).
Generalized estimating equation is a more recent methodology for the analysis of many correlation structures among the values, including the ability to estimate this correlation structure rather than make assumptions [16, 48]. The reason we used GEE for statistics, rather than traditional repeated measures analysis of variance (ANOVA), is principally because the ANOVA model makes the sphericity assumption about the variance structure; however this assumption is often not met so the ANOVA model cannot accommodate generic correlation structures [25].
Results
Sway intensity and sway velocity were associated with group, degree of bevel, open- or closed-eyes condition, and dorsiflexion or plantarflexion, respectively (Tables 1, 3). The relationship between sway intensity/velocity and subject groups varied depending on the bevel. None of the subjects were injured during testing.
Table 1.
Sway intensities at different degrees of bevel angle in the three groups
| Comparison | Estimate ± SE | Z | P-value |
|---|---|---|---|
| Intercept | 2.9281 ± 0.0813 | 36.02 | < 0.0001 |
| Group | |||
| Δ (Minor SBO vs. control) at 0° | 2.5174 ± 0.2016 | 12.49 | < 0.0001 |
| Δ (Major SBO vs. control) at 0° | 3.0112 ± 0.2299 | 13.10 | < 0.0001 |
| Bevel degree | |||
| Δ (10° vs. 0°) controls | 0.7913 ± 0.0657 | 12.05 | < 0.0001 |
| Δ (20° vs. 0°) controls | 1.0524 ± 0.0813 | 12.95 | < 0.0001 |
| Eyes open vs. closed | −1.2034 ± 0.0934 | −12.89 | < 0.0001 |
| Plantarflexion vs. dorsiflexion | −0.2089 ± 0.0678 | −3.08 | 0.0021 |
| Minor SBO vs. control | |||
| Δ (10° vs. 0°) | −1.1499 ± 0.1947 | −5.91 | < 0.0001 |
| Δ (20° vs. 0°) | −1.1907 ± 0.1982 | −6.01 | < 0.0001 |
| Major SBO vs. control | |||
| Δ (10° vs. 0°) | −0.5455 ± 0.2744 | −1.99 | 0.0468 |
| Δ (20° vs. 0°) | −0.7398 ± 0.2793 | −2.65 | 0.0081 |
Statistical model was assessed by using generalized estimating equations (GEEs) and was adjusted for age, gender, and group-by-bevel degree interaction effects
SE Standard error
Table 3.
Sway velocities at different degrees of bevel angle in the three groups
| Comparison | Estimate ± SE | Z | P-value |
|---|---|---|---|
| Intercept | 8.9341 ± 0.1099 | 81.28 | < 0.0001 |
| Group | |||
| Δ (Minor SBO vs. Control) at 0° | 2.7636 ± 0.3707 | 7.45 | < 0.0001 |
| Δ (Major SBO vs. control) at 0° | 4.0881 ± 0.6000 | 6.81 | < 0.0001 |
| Bevel degree | |||
| Δ (10° vs. 0°) | 0.7340 ± 0.0979 | 7.50 | < 0.0001 |
| Δ (20° vs. 0°) | 2.0643 ± 0.1467 | 14.07 | < 0.0001 |
| Eyes open vs. closed | –3.4022 ± 0.1872 | –18.17 | < 0.0001 |
| Plantarflexion vs. dorsiflexion | –0.3217 ± 0.1090 | –2.95 | 0.0032 |
| Minor SBO vs. Control | |||
| Δ (10° vs. 0°) | –2.4165 ± 0.3844 | –6.29 | < 0.0001 |
| Δ (20° vs. 0°) | –2.4786 ± 0.3606 | –6.87 | < 0.0001 |
| Major SBO vs. control | |||
| Δ (10° vs. 0°) | –1.7854 ± 0.5033 | –3.55 | 0.0004 |
| Δ (20° vs. 0°) | –1.9780 ± 0.5002 | –3.95 | < 0.0001 |
Statistical model was assessed by using GEEs and was adjusted for age, gender, and group-by-bevel degree interaction effects
SE Standard error
Sway intensity
After we adjusted for age and gender, the relationship between sway intensity and groups was found to vary depending on the bevel angles.
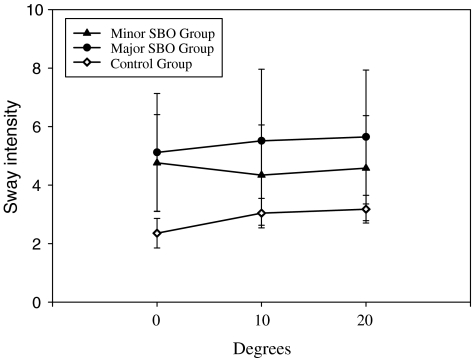
Comparing the sway intensities group differences at different bevel degrees (0°, 10°, 20°), the group differences of minor SBO group versus control were 2.52, 1.37, and 1.33 significantly decreased, whereas major SBO group versus control were 3.01, 2.47, and 2.27 significantly decreased (all P < 0.0001, Table 2, Fig. 3); however, the differences between minor and major SBO (Δ = −0.49) were not significant (P = 0.0894, Table 2) at 0°, but significant differences at 10° (Δ = −1.09, P = 0.0015, Table 2) and 20° (Δ = −0.94, P = 0.0029, Table 2), respectively.
Table 2.
Contrast estimate results from sway intensities GEE model
| Label | Estimate ± SE | 95% CL | χ2 | P-value | |
|---|---|---|---|---|---|
| Between—group differences at different bevel degrees | |||||
| Minor SBO vs. Control at 0° | 2.5174 ± 0.2016 | 2.1223 | 2.9126 | 155.90 | < 0.0001 |
| Major SBO vs. Control at 0° | 3.0112 ± 0.2299 | 2.5605 | 3.4619 | 171.49 | < 0.0001 |
| Minor vs. Major SBO at 0° | −0.4938 ± 0.2907 | −1.0635 | 0.0760 | 2.89 | 0.0894 |
| Minor SBO vs. Control at 10° | 1.3675 ± 0.2006 | 0.9744 | 1.7607 | 46.48 | < 0.0001 |
| Major SBO vs. Control at 10° | 2.4657 ± 0.2838 | 1.9095 | 3.0218 | 75.50 | < 0.0001 |
| Minor vs. Major SBO at 10° | −1.0981 ± 0.3451 | −1.7745 | −0.4218 | 10.13 | 0.0015 |
| Minor SBO vs. Control at 20° | 1.3268 ± 0.1788 | 0.9763 | 1.6773 | 55.04 | < 0.0001 |
| Major SBO vs. Control at 20° | 2.2714 ± 0.2654 | 1.7512 | 2.7916 | 73.23 | < 0.0001 |
| Minor vs. Major SBO at 20° | −0.9446 ± 0.3167 | −1.5654 | −0.3239 | 8.90 | 0.0029 |
| Within—group variations | |||||
| Minor SBO, 10° vs. 0° | −0.3586 ± 0.1816 | −0.7146 | −0.0026 | 3.90 | 0.0484 |
| Minor SBO, 20° vs. 0° | −0.1383 ± 0.1818 | −0.4945 | 0.2180 | 0.58 | 0.4469 |
| Major SBO, 10° vs. 0° | 0.2458 ± 0.2667 | −0.2769 | 0.7684 | 0.85 | 0.3567 |
| Major SBO, 20° vs. 0° | 0.3126 ± 0.2696 | −0.2157 | 0.8409 | 1.34 | 0.2462 |
| Control, 10° vs. 0° | 0.7913 ± 0.0657 | 0.6626 | 0.9200 | 145.14 | < 0.0001 |
| Control, 20° vs. 0° | 1.0524 ± 0.0813 | 0.8931 | 1.2117 | 167.69 | < 0.0001 |
Contrasts and estimates were statistically assessed in the GEE model listed in Table 1 by using SAS 9.13
GEE Generalized estimating equations, SE standard error, CL confidence limits
Fig. 3.
Sway intensities of the study groups at different bevel degrees of the platform
Comparing each group’s own sway intensity at 0° (within group variation), we found that the minor SBO group had 0.36 a significantly lower intensity at 10° (P = 0.0484, Table 2) but not significant at 20° (P = 0.4469, Table 2). There were no significant variations in major SBO group, either at 10° (P = 0.3567, Table 2) or 20° (P = 0.2462, Table 2). However, the variation in the control group was significantly increased 0.79 at 10° and 1.05 at 20° (P < 0.0001, Table 2, respectively).
Moreover, values obtained with open eyes and with plantarflexion had 1.2 lower (P < 0.0001, Table 1) and 0.2 lower (P = 0.0021, Table 1) sway intensity, when compared with sway intensity values obtained with closed eyes and with dorsiflexion, respectively.
Sway velocity
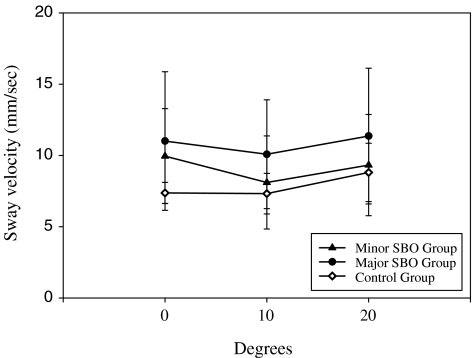
After we adjusted for age and gender, the relationship between sway velocity and subject groups varied with the different bevels. Group differences (minor SBO vs. control, major SBO vs. control) showed a significant decrease when comparing at 10° than at 0° and at 20° than at 0° (Table 3, Fig. 4).
Fig. 4.
Sway velocities of the study groups at different bevel degrees of the platform
Comparing the sway velocities group differences at different bevel degrees (0°, 10°, 20°), the group differences of major SBO group versus control were 4.08, 2.30, and 2.11 significantly decreased (all P < 0.0001, Table 4, Fig. 4). Although the difference between minor SBO group versus control was significant at 0° (Δ = 2.76, P < 0.0001, Table 4), there were not significant differences at 10° (P = 0.3939, Table 4) and 20° (P = 0.4660, Table 4), respectively. However, the differences between minor SBO and major SBO were marginally significant at 0° (Δ = −1.32, P = 0.0574, Table 4) and significant at 10° (Δ = −1.96, P = 0.0010, Table 4) and 20° (Δ = −1.83, P = 0.0021, Table 4), respectively.
Table 4.
Contrast estimate results from sway velocities intensities GEE model
| Label | Estimate ± SE | 95% CL | χ2 | P-value | |
|---|---|---|---|---|---|
| Between—group differences at different bevel degrees | |||||
| Minor SBO vs. Control at 0° | 2.7636 ± 0.3707 | 2.0370 | 3.4902 | 55.57 | < 0.0001 |
| Major SBO vs. Control at 0° | 4.0881 ± 0.6000 | 2.9121 | 5.2640 | 46.42 | < 0.0001 |
| Minor vs. Major SBO at 0° | −1.3244 ± 0.6970 | −2.6905 | 0.0416 | 3.61 | 0.0574 |
| Minor SBO vs. Control at 10° | 0.3471 ± 0.4072 | −0.4509 | 1.1452 | 0.73 | 0.3939 |
| Major SBO vs. Control at 10° | 2.3027 ± 0.4405 | 1.4393 | 3.1662 | 27.32 | < 0.0001 |
| Minor vs. Major SBO at 10° | −1.9556 ± 0.5932 | −3.1181 | −0.7930 | 10.87 | 0.0010 |
| Minor SBO vs. Control at 20° | 0.2851 ± 0.3911 | −0.4814 | 1.0515 | 0.53 | 0.4660 |
| Major SBO vs. Control at 20° | 2.1100 ± 0.4665 | 1.1958 | 3.0243 | 20.46 | < 0.0001 |
| Minor vs. Major SBO at 20° | −1.8250 ± 0.5939 | −2.9891 | −0.6609 | 9.44 | 0.0021 |
| Within—group variations | |||||
| Minor SBO, 10° vs. 0° | −1.6825 ± 0.3713 | −2.4102 | −0.9549 | 20.54 | < 0.0001 |
| Minor SBO, 20° vs. 0° | −0.4143 ± 0.3315 | −1.0641 | 0.2355 | 1.56 | 0.2114 |
| Major SBO, 10° vs. 0° | −1.0514 ± 0.4825 | −1.9972 | −0.1056 | 4.75 | 0.0293 |
| Major SBO, 20° vs. 0° | 0.0862 ± 0.4749 | −0.8444 | 1.0169 | 0.03 | 0.8559 |
| Control, 10° vs. 0° | 0.7340 ± 0.0979 | 0.5420 | 0.9259 | 56.18 | < 0.0001 |
| Control, 20° vs. 0° | 2.0643 ± 0.1467 | 1.7768 | 2.3518 | 198.03 | < 0.0001 |
Contrasts and estimates were statistically assessed in the GEE model listed in Table 3 by using SAS 9.13
GEE Generalized estimating equations, SE standard error, CL confidence limits
Comparing each group’s own sway velocity at 0° (within group variation), we found that the minor SBO group had significant lower velocities at 10° (P < 0.0001, Table 4) but not significant at 20° (P = 0.2114, Table 4). There were significant variations in the major SBO group at 10° (P = 0.0293, Table 4) but not at 20° (P = 0.8559, Table 4). However, the variation in the control group was significantly increased 0.73 at 10° and 2.06 at 20° (all P < 0.0001, Table 4).
Moreover, velocity values obtained with open eyes and with plantarflexion had 3.4 lower (P < 0.0001, Table 3) and 0.32 lower (P = 0.0032, Table 3) sway velocity, when compared with sway velocity values obtained with closed eyes and with dorsiflexion, respectively.
Discussion
To our knowledge, this was the first study of changes in postural sway observed on the ground and in stressful positions in subjects with SBO. We compared postural sway in subjects with SBO and in healthy control subjects when they maintained an upright posture in ten experimental conditions designed to challenge the roles that vision (eyes open vs. closed) and proprioception (platform bevel and dorsiflexion vs. plantarflexion) play. From the results of postural sway, we can see that sway intensity and sway velocity were associated with group, degree of bevel, open- or closed-eyes condition, and dorsiflexion or plantarflexion, respectively (Tables 1, 3). With respect to sway intensity, the differences of the minor or the major SBO groups were significantly decreased at different bevel degrees when compared with control group (Table 2, Fig. 3), whereas the differences between minor and major SBO were significant at 10° and 20°. With respect to sway velocity, the differences of the major SBO group were significantly decreased at different bevel degrees when compared with the minor SBO and the control group, whereas the difference in the minor SBO was only significant at 0° when compared with control (Table 4, Fig. 4). The statistical evidences imply a difference between minor SBO and major SBO, irrespective of viewing from sway intensity or sway velocity. The sway intensity/velocity values obtained with open eyes and with plantarflexion had lower values, when compared with values obtained with closed eyes and with dorsiflexion (Tables 1, 3). The fact means visual input and proprioception may universally alter the postural sway. The 10° and 20° bevels of the standing platform altered body sway parameters, as compared with measurements when the platform was at 0°. Also the sway parameters of subjects with major SBO were significantly larger than those of subjects with minor SBO or control subjects during the upright standing tests. Our results seem to imply that the larger the bone defect at the lumbosacral midline, the more the group differences in different stressful conditions.
Postural sway characteristics due to designed experimental conditions depend on many variables. The first is proprioception. Humans maintain a balanced posture by integrating sensory inputs to correct static and dynamic postures and by maintaining gaze orientation [3]. The vestibular, proprioceptive, and visual systems clearly contribute to postural control to provide stabilization at low frequencies of sway. Therefore, to maintain postural stability, an increase in postural stiffness should be accompanied by a corresponding increase in damping [39, 40]. Maintenance of upright posture is based on patency of the bony structures and related soft tissues (e.g., joint capsules, ligaments) that contribute to the spinal-stabilizing system in the trunk [24]. Among the spinal soft tissues, the lumbar back muscles play an important role in controlling lumbar segmental stability [4, 12, 31, 37, 38, 45].
The somatosensory feedback system plays a dominant role in maintaining an upright posture when healthy peoples stand on a force-measuring platform with a fixed back support [19], which system is provided mostly by proprioceptive receptors in the lower limb muscle and trunk muscle [11, 26, 43]. Therefore, when a person stands on a solid, stable support, all of these signals work simultaneously and coherently, allowing them to determine the position of the projection of the center of a suprapedal mass relative to the foot [22]. Allum et al. [6] declared that the proprioceptive inputs from the trunk or hip may be more important than those from the lower legs in triggering postural corrections. O’Sullivan et al. [37] found that the internal oblique, superficial lumbar multifidus, and thoracic erector spinae muscles have significantly decreased EMG activity during passive sway standing compared with active upright standing. They concluded that the lumbopelvic stabilizing musculature is active in maintaining optimally aligned, upright postures, and that activation of specific lumbopelvic muscles differs in sway standing and upright postures. Ali et al. [5] also found that positioning of the torso on the pelvis in the sagittal and coronal planes is an essential element of postural control in a sitting position as well as in a standing position.
Based on our results, we suggest that the bevel disrupted proprioceptive information for postural control. Theoretically, proprioceptive system should compensate for the lack of visual and vestibular feedback when subjects stand with their eyes closed or for alterations in foot position from 0° (ground level) [20]. However, measured parameters of postural sway were still significantly different in subjects with SBO than in control subjects. We assume SBO may hinder function of the proprioceptive sensors and believed it is the main reason to cause such differences because the structural difference among our three groups was SBO only in the lower trunk. Viewing from the anatomical point, there are many musculatures, such as the lower parts of erector spinae and multifidus muscles located in the exact areas of the posterior vertebral arch [12] where SBO happens. Based on our data, we suggest that major SBO increases the loss of proprioception in the lumbar back muscles more than minor SBO; therefore, we assume the larger the bony defect in the lumbosacral midline, the more the group differences in different stressful positions. Impairment of postural sway due to probable loss of back extensor muscle function resulting from SBO might be a characteristic correlate of this condition when the participant is in a stressful position.
The second variable that influences postural sway is the muscle contraction timing and magnitude of the ankle dorsiflexors and plantarflexors. It has been reported that the muscles of the lower limb have a role in controlling sway and orientation in the sagittal plane of standing subjects [23]. Winter et al. [46] used an inverted pendulum model to show that ankle stiffness is sufficient to control posture during quiet standing in healthy subjects. Nevertheless, we did not measure surface EMG activity of the dorsiflexors and plantarflexors. We instead adopted 60-s testing in ten identical conditions in the three groups, who had no foot problems, in order to equalize the confounding factor of ankle muscles and to emphasize the SBO. All of our participants adopted a stiffening strategy (contraction of the leg muscles) and might have done so to help compensate for a loss of posture or to create secondary effects necessary to maintain a new position. Nonetheless, this cannot explain why sway intensity and velocity were larger in subjects with SBO than in control subjects at variable bevels, with more changes in the major SBO group, even when they stood under similar stressful positions.
The third variable is the possible involvement of abdominal bracing, which is isometric contraction of all the muscles of the abdominal wall. Kavcic et al. [29] preformed a quantitatively compared lumbar spine stability resulting from muscle-activation patterns measured when subjects performed selected stabilization exercises. They found that an abdominal brace is a maneuver that subjects perform to increase the level of spine stability during different tasks. We allowed our participants to contract their abdominal muscles during testing, so we anticipated that they might adopt this abdominal brace to compensate for a loss of posture in high stress conditions. However, even in the identical stressful conditions, the trends of sway parameters of SBO groups were not the same as those of control groups.
Some authors might add the consideration of other variables affecting specific postural control, which are based on two hypotheses—sensory re-weighting and load compensation [39, 40]. The former is an alteration of the relative contributions of proprioceptive and graviceptive information to postural control, and the latter is an increase in the overall gain of the neural controller to produce sufficient corrective torque [40]. We realized that when our subjects were performing the tests, corrective torque may have been generated to counter the destabilizing torque due to gravity. We cannot ignore the involvement of the two hypotheses in organizing the sensory and motor systems that contribute to postural stability, however, the two hypotheses can only be explained when the subjects are performing dynamic tasks, not like the static ones in our study.
Our high degree of beveling was also similar to the platform that Carpenter et al. [14] used in examining kinetic and kinematic parameters in young healthy adults. They performed static posturography during quiet standing under three conditions of increasing postural threat: ground level (low threat), 81 cm above ground level with or without support (medium or high threat). They found that the condition of high threat is accompanied by relatively large increases in EMG activation of tibialis anterior (36.5%) and rectus femoris (30.0%), when compared to standing in the low threat condition. Irrespective of the differences in study design between the two studies, one finding which has remained consistent is that when vision was not available participants expressed an inferior sway pattern in conditions of stressful postures [14].
We did not distinguish differences in the sway parameters according to the four types of minor SBO that Fidas et al. [17] have described but, mostly important, we can distinguish the difference between minor and major SBO. There were differences in sway intensity and sway velocity between the three groups at measured ground level (0°), but, unexpectedly, the trend pattern of sway in the SBO groups at 10° and 20° did not follow the trend of the control groups. Of them, the trends of sway intensity and velocity in the major SBO group declined in the 10° and inclined in the 20°. In the minor SBO group, however, a similar trend demonstrated in sway velocity from 10° to 20°, but not in sway intensity from 10° to 20° (Figs. 3, 4). It is now hard to explain the mechanism that contributes to the different sway patterns between the two SBO groups.
Our findings were correlated with the observation that both sway intensity and velocity are the most sensitive and reliable measures of standing steadiness [15, 30, 36]. Either velocity or intensity was able to reflect the function of the postural sway from our results measured in the control group. According to the values of the postural sway of control group in the consecutive bevel levels from 0° to 20°, we can see the obvious larger differences in intensity between 0° and 10°, and those in velocity between 10° and 20°. The above truth seemed to mean that sway intensity was useful to detect low stressful position, and sway velocity was more useful to detect high stressful position. Therefore standing upright could be a reproducible way of measuring postural sway, in spite of standing on foam and/or an uneven plane.
There have been no reports of studying the difference in the bevel plane and foot position in the SBO groups before, but now we have the results showing a difference when considering the confounding factors of bevel and foot position. We suggest, in the future, that factors of bevel plane and foot position should be considered and added to the sway postural study.
There might be some problems we did not consider in the study, such as the role of upper limbs [9, 47]. This suggests a future orientation for us in another way to investigate SBO.
Conclusion
In general, the observed postural stability of subjects with minor or major SBO when standing upright at various bevels was inferior to that of the control group. There was a significant difference in sway parameters between minor or major SBO compared with healthy subjects. This study supports the hypothesis that SBO impairs control of postural function in both the resting upright and stressful postures, which mechanism might be contributed by a loss of proprioception in the back extensor musculatures.
Acknowledgments
We wish to thank the faculty of Department of Radiology for their assistance in the survey of radiological films.
References
- 1.Akar N, Cavdar AO, Arcasoy A. High incidence of neural tube defects in Bursa, Turkey. Paediatr Perinat Epidemiol. 1988;2:89–92. doi: 10.1111/j.1365-3016.1988.tb00181.x. [DOI] [PubMed] [Google Scholar]
- 2.Albano JP, Shannon SG, Alem NM, Mason KT. Injury risk for research subjects with spina bifida occulta in a repeated impact study: a case review. Aviat Space Environ Med. 1996;67:767–769. [PubMed] [Google Scholar]
- 3.Alessandrini M, D’Erme G, Bruno E, Napolitano B, Magrini A. Vestibular compensation: analysis of postural re-arrangement as a control index for unilateral vestibular deficit. Neuroreport. 2003;14:1075–1079. doi: 10.1097/00001756-200305230-00034. [DOI] [PubMed] [Google Scholar]
- 4.Alexiev AR. Some differences of the electromyographic erector spinae activity between normal subjects and low back pain patients during the generation of isometric trunk torque. Electromyogr Clin Neurophysiol. 1994;34:495–499. [PubMed] [Google Scholar]
- 5.Ali AS, Rowen KA, Iles JF. Vestibular actions on back and lower limb muscles during postural tasks in man. J Physiol. 2003;546.2:615–624. doi: 10.1113/jphysiol.2002.030031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Allum JHJ, Bloem BR, Carpenter MG, Hulliger M, Hadders-Algra M. Proprioceptive control of posture: a review of new concepts. Gait Posture. 1998;8:214–242. doi: 10.1016/S0966-6362(98)00027-7. [DOI] [PubMed] [Google Scholar]
- 7.American Medical Association (2005) International classification of diseases, 9th revision, clinical modification: physician ICD-9-CM. AMA, Chicago
- 8.Avrahami E, Frishman E, Fridman Z, Azor M. Spina bifida occulta of S1 is not an innocent finding. Spine. 1994;19:12–15. doi: 10.1097/00007632-199401000-00003. [DOI] [PubMed] [Google Scholar]
- 9.Beaudoin L, Zabjek KF, Leroux MA, Coillard C, Rivard CH. Acute systematic and variable postural adaptations induced by an orthopaedic shoe lift in control subjects. Eur Spine J. 1999;8:40–45. doi: 10.1007/s005860050125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Berbrayer D. Tethered cord syndrome complicating spina bifida occulta: a case report. Am J Phys Med Rehabil. 1991;70:213–214. doi: 10.1097/00002060-199108000-00010. [DOI] [PubMed] [Google Scholar]
- 11.Bloem BR, Allum JHJ, Carpenter MG, Verschuuren JJGM, Honegger F. Triggering of balance corrections and compensatory strategies in a patient with total leg proprioceptive loss. Exp Brain Res. 2002;142:91–107. doi: 10.1007/s00221-001-0926-3. [DOI] [PubMed] [Google Scholar]
- 12.Bogduk N, Macintosh JE, Pearcy MJ. A universal model of the lumbar back muscles in the upright position. Spine. 1992;17:897–913. doi: 10.1097/00007632-199208000-00007. [DOI] [PubMed] [Google Scholar]
- 13.Brumagne S, Cordo P, Verschueren S. Proprioceptive weighting changes in persons with low back pain and elderly persons during upright standing. Neurosci Lett. 2004;366:63–66. doi: 10.1016/j.neulet.2004.05.013. [DOI] [PubMed] [Google Scholar]
- 14.Carpenter MG, Frank JS, Silcher CP, Peysar GW. The influence of postural threat on the control of upright stance. Exp Brain Res. 2001;138:210–218. doi: 10.1007/s002210100681. [DOI] [PubMed] [Google Scholar]
- 15.Després C, Lamoureux D, Beuter A. Standardization of a neuromotor test battery: the CATSYS system. Neurotoxicology. 2000;21:725–735. [PubMed] [Google Scholar]
- 16.Diggle PJ, Liang KY, Zeger SL. Analysis of Longitudinal Data, 1st edn. New York, Oxford: Oxford University Press; 1994. [Google Scholar]
- 17.Fidas A, MacDonald HL, Elton RA, Wild SR, Chisholm GD, Scott R. Prevalence and patterns of spina bifida occulta in 2707 normal adults. Clin Radiol. 1987;38:537–542. doi: 10.1016/S0009-9260(87)80150-2. [DOI] [PubMed] [Google Scholar]
- 18.Frymoyer JW, Newberg A, Pope MH, Wilder DG, Clements J, MacPherson B. Spine radiographs in patients with low-back pain: an epidemiological study in men. J Bone Joint Surg Am. 1984;66:1048–1055. [PubMed] [Google Scholar]
- 19.Fukuoka Y, Nagata T, Ishida A, Minamitani H. Characteristics of somatosensory feedback in postural control during standing. IEEE Trans Neural Syst Rehabil Eng. 2001;9:145–153. doi: 10.1109/7333.928574. [DOI] [PubMed] [Google Scholar]
- 20.Gill KP, Callaghan MJ. The measurement of lumbar proprioception in individuals with and without low back pain. Spine. 1998;23:371–377. doi: 10.1097/00007632-199802010-00017. [DOI] [PubMed] [Google Scholar]
- 21.Gregerson DM. Clinical consequences of spina bifida occulta. J Manipulative Physiol Ther. 1997;20:546–550. [PubMed] [Google Scholar]
- 22.Gurfinkel VS, Ivanenko YuP, Levik YuS, Babakova IA. Kinesthetic reference for human orthograde posture. Neuroscience. 1995;68:229–243. doi: 10.1016/0306-4522(95)00136-7. [DOI] [PubMed] [Google Scholar]
- 23.Gurfinkel VS, Ivanenko YuP, Levik YuS The contribution of foot deformation to the changes of muscular length and angle in the ankle joint during standing in man. Physiol Res. 1994;43:371–377. [PubMed] [Google Scholar]
- 24.Holm S, Indahl A, Solomonow M. Sensorimotor control of the spine. J Electromyogr Kinesiol. 2002;12:219–234. doi: 10.1016/S1050-6411(02)00028-7. [DOI] [PubMed] [Google Scholar]
- 25.Huynh H, Feldt LS. Conditions under which mean square ratios in repeated measurements designs have exact f-distributions. J Am Stat Assoc. 1970;65:1582–1589. doi: 10.2307/2284340. [DOI] [Google Scholar]
- 26.Inglis JT, Horak FB, Shupert CL, Jones-Rycewicz C. The importance of somatosensory information in triggering and scaling automatic postural responses in humans. Exp Brain Res. 1994;101:159–164. doi: 10.1007/BF00243226. [DOI] [PubMed] [Google Scholar]
- 27.Internet information: www.spoilheap.co.uk/spinbif.htm
- 28.Ivanenko YP, Solopova IA, Levik YS. The direction of postural instability affects postural reactions to ankle muscle vibration in humans. Neurosci Lett. 2000;292:103–106. doi: 10.1016/S0304-3940(00)01438-5. [DOI] [PubMed] [Google Scholar]
- 29.Kavcic N, Grenier S, McGill SM. Quantifying tissue loads and spine stability while performing commonly prescribed low back stabilization exercises. Spine. 2004;29:2319–2329. doi: 10.1097/01.brs.0000142222.62203.67. [DOI] [PubMed] [Google Scholar]
- 30.Letz R, Gerr F. Standing steadiness measurements: empirical selection of testing protocol and outcome measures. Neurotoxicol Teratol. 1995;17:611–616. doi: 10.1016/0892-0362(95)00023-2. [DOI] [PubMed] [Google Scholar]
- 31.Moseley GL, Hodges PW, Gandevia SC. External perturbation of the trunk in standing humans differentially activates components of the medial back muscles. J Physiol. 2003;547:581–587. doi: 10.1113/jphysiol.2002.024950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Nadeau V, Lamoureux D, Beuter A, Charbonneau M, Tardif R. Neuromotor effects of acute ethanol inhalation exposure in humans: a preliminary study. J Occup Health. 2003;45:215–222. doi: 10.1539/joh.45.215. [DOI] [PubMed] [Google Scholar]
- 33.Nakamura T, Sofue M, Homma T. Spina bifida occulta associated with a low-placed conus medullaris. J Spinal Disord. 1988;1:194–201. doi: 10.1097/00002517-198803000-00003. [DOI] [PubMed] [Google Scholar]
- 34.Newcomer KL, Jacobson TD, Gabriel DA, Larson DR, Brey RH, An KN. Muscle activation patterns in subjects with and without low back pain. Arch Phys Med Rehabil. 2002;83:816–821. doi: 10.1053/apmr.2002.32826. [DOI] [PubMed] [Google Scholar]
- 35.Ogon M, Riedl-Huter C, Sterzinger W, Krismer M, Spratt KF, Wimmer C. Radiologic abnormalities and low back pain in elite skiers. Clin Orthop Relat Res. 2001;390:151–162. doi: 10.1097/00003086-200109000-00018. [DOI] [PubMed] [Google Scholar]
- 36.Oppenheim U, Kohen-Raz R, Alex D, Kohen-Raz A, Azarya M. Postural characteristics of diabetic neuropathy. Diabetes Care. 1999;22:328–332. doi: 10.2337/diacare.22.2.328. [DOI] [PubMed] [Google Scholar]
- 37.O’Sullivan PB, Grahamslaw KM, Kendell M, Lapenskie SC, Moller NE, Richards KV. The effect of different standing and sitting postures on trunk muscle activity in a pain-free population. Spine. 2002;27:1238–1244. doi: 10.1097/00007632-200206010-00019. [DOI] [PubMed] [Google Scholar]
- 38.O’Sullivan PB, Twomey L, Allison GT. Dysfunction of the neuro-muscular system in the presence of low back pain: implications of physical therapy management. J Man Manipulative Ther. 1997;5:20–26. [Google Scholar]
- 39.Peterka RJ. Sensorimotor integration in human postural control. J Neurophysiol. 2002;88:1097–1118. doi: 10.1152/jn.2002.88.3.1097. [DOI] [PubMed] [Google Scholar]
- 40.Peterka RJ, Loughlin PJ. Dynamic regulation of sensorimotor integration in human postural control. J Neurophysiol. 2004;91:410–423. doi: 10.1152/jn.00516.2003. [DOI] [PubMed] [Google Scholar]
- 41.Stolke D, Zumkeller M, Seifert V. Intraspinal lipomas in infancy and childhood causing a tethered cord syndrome. Neurosurg Rev. 1988;11:59–65. doi: 10.1007/BF01795696. [DOI] [PubMed] [Google Scholar]
- 42.Sward L, Hellstrom M, Jacobsson B, Peterson L. Back pain and radiologic changes in the thoraco-lumbar spine of athletes. Spine. 1990;15:124–129. doi: 10.1097/00007632-199002000-00015. [DOI] [PubMed] [Google Scholar]
- 43.Tunik E, Poizner H, Levin MF, Adamovich SV, Messier J, Lamarre Y, Feldman AG. Arm-trunk coordination in the absence of proprioception. Exp Brain Res. 2003;153:343–355. doi: 10.1007/s00221-003-1576-4. [DOI] [PubMed] [Google Scholar]
- 44.Verhoef M, Barf HA, Post MWM, Asbeck FWA, Gooskens RHJM, Prevo AJH. Secondary impairments in young adults with spina bifida. Dev Med Child Neurol. 2004;46:420–427. doi: 10.1017/S0012162204000684. [DOI] [PubMed] [Google Scholar]
- 45.Wilke HJ, Wolf S, Claes LE, Arand M, Wiesend A. Stability increase of the lumbar spine with difference muscle groups: a biomechanical in vitro study. Spine. 1995;20:192–198. doi: 10.1097/00007632-199501150-00011. [DOI] [PubMed] [Google Scholar]
- 46.Winter DA, Patla AE, Rietdyk S, Ishac MG. Ankle muscle stiffness in the control of balance during quiet standing. J Neurophysiol. 2001;85:2630–2633. doi: 10.1152/jn.2001.85.6.2630. [DOI] [PubMed] [Google Scholar]
- 47.Zabjek KF, Leroux MA, Coillard C, Martinez X, Griffet J, Simard G, Rivard CH. Acute postural adaptations induced by a shoe lift in idiopathic scoliosis patients. Eur Spine J. 2001;10:107–113. doi: 10.1007/s005860000244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Zeger SL, Liang KY. Longitudinal data analysis for discrete and continuous outcomes. Biometrics. 1986;42:121–130. doi: 10.2307/2531248. [DOI] [PubMed] [Google Scholar]