Abstract
This study examined the preventive effects of the local application of mitomycin C (MMC), 5-fluorouracil (5-FU), and cyclosporine A (CsA) in minimizing spinal epidural fibrosis in a rat laminectomy model. Thirty-two 2-year-old male Wistar albino rats, each weighing 400 ± 50 g, were divided into four equal groups: sham, MMC, 5-FU, and CsA. Each rat underwent laminectomy at the L5–L6 lumbar level. Cotton pads (4 × 4 mm2) soaked with MMC (0.5 mg/ml), 5-FU (5 ml/mg), or CsA (5 mg/ml) were placed on the exposed dura for 5 min. Thirty days after surgery, the rats were killed and the epidural fibrosis, fibroblast density, inflammatory cell density, and arachnoid fibrosis were quantified. The epidural and arachnoid fibroses were reduced significantly in the treatment groups compared to the sham group. Fibroblast cell density and inflammatory cell density were decreased significantly in the MMC and 5-FU groups, but were similar in the sham and CsA groups. The decreased rate of epidural fibrosis was promising. Further studies in humans are needed to determine the short- and long-term complications of the agents used here.
Keywords: Epidural fibrosis, Mitomycin C, 5-Fluorouracil, Cyclosporine A
Introduction
Postoperative epidural fibrosis is the deposition of fibrous tissue in the epidural space, adjacent to the dura mater, following surgical laminectomy. It is associated with an increased complication rate in revision spine surgery, particularly with the increased occurrence of dural tears, nerve root injury, and bleeding [6, 19]. In addition, it has been reported to be the cause of symptoms in 8–14% of patients with failed back surgery syndrome (FBSS) [7, 28].
After establishing a correlation between epidural scars and a poor clinical outcome following surgery, surgeons and researchers have sought treatment strategies that would prevent scar formation without compromising the wound healing process [29]. Several materials have been investigated, including Adcon-L®, fat graft, Gelfoam®, Gore-Tex membrane, silastic, and sodium hyaluronate [1, 5, 13, 26, 40, 41]. Although a large variety of materials implanted on the dura have achieved moderate success in animal models, their clinical use is yet to be successful [1, 10, 16, 22, 37, 41].
Mitomycin C (MMC) is an ankylosing chemotherapeutic agent that potentially suppresses fibroblast proliferation after surgery [15]. It has been used extensively to reduce the scar formation associated with ophthalmologic and otolaryngological surgery [9, 20, 30, 32, 43]. The local application of MMC prevents epidural fibrosis in rat and rabbit laminectomy models [12, 27].
5-Fluorouracil (5-FU) is a chemotherapeutic agent that exerts its antiproliferative effect by antagonizing pyrimidine metabolism. Experimentally, 5-FU is a very effective inhibitor of fibroblast growth. In vitro and in vivo studies demonstrate that a 5-min exposure results in growth arrest and may have a long-lasting effect on cultured human Tenon’s fibroblasts. 5-FU also specifically interferes with qualitative fibroblast functions. It decreased the mitotic rate and thymidine uptake of healing corneal epithelium in an experimental rabbit model. It also inhibited cell replication during the S (DNA synthesis) and G2 (RNA synthesis) phases [23].
Cyclosporine A (CsA) is an immunosuppressive agent that is widely used to prevent rejection following organ or bone marrow transplantation. There is growing evidence that it also affects the biological functions of some non-immune cell types [35]. CsA also inhibits fibroblast proliferation in vitro in rabbit subconjunctival fibroblasts, depending on the dose and duration of application [4].
This study examined the possible preventive effects of MMC, 5-FU, and CsA in minimizing spinal epidural fibrosis in a rat laminectomy model.
Materials and methods
Thirty-two 2-year-old male Wistar albino rats, each weighing 400 ± 50 g, were used in this study, which was approved by the animal ethics committee of our medical school (100/12, 09.09.2005).
The rats were divided into four equal groups: sham, MMC, 5-FU, and CsA. Anaesthesia was induced with an intraperitoneal application of ketamine (8 mg/100 g; Eczacibasi, Istanbul, Turkey). Each rat underwent laminectomy at the L5–L6 lumbar levels. The lumbar area of each rat was shaved, and the operative field was prepared in a sterile manner using povidone–iodine solution. A dorsal skin incision was made with a number-15 blade and continued down to the spinous process. The paraspinal muscles were stripped away from the lamina and spinous process. Under an operating microscope, laminectomies at L5 and L6 were performed with a 1 mm rongeur. The ligaments at L5–L6 were removed, and the underlying dura mater was exposed for 4 × 4 mm2 widening. Throughout the procedure, haemostasis was maintained by irrigation with saline and surgical gauze. Bipolar cautery, bone wax, Surgicel, or other hemostatic materials were not used. When the laminectomy site was free of active haemorrhage, it was irrigated with 10 ml of saline. To this point, the procedure was the same for all rats. Subsequently, the rats were assigned randomly to four groups of eight animals each. Cotton pads (4 × 4 mm2) soaked with MMC (0.5 mg/ml), 5-FU (5 ml/mg), or CsA (5 mg/ml) were applied on the exposed dura for 5 min. Then, the soaked cotton wool was removed and the laminectomy site was irrigated with 10 ml of saline to wash off any leftover agent. In the fourth, sham group, the exposed dura mater was only irrigated with 10 ml of saline solution. In all groups, the fascia at the laminectomy site was marked with 3/0 silk to facilitate the harvest of pathological specimens. The wound was then closed in layers using the same suture material in each animal. There were no complications or adverse effects from the surgery or the application of antifibrotic agents. No prophylactic antibiotics were used.
The rats were killed 30 days after surgery with a lethal dose (100 mg/kg) of sodium pentobarbital. The lumbar spine was removed en bloc with the paraspinal musculature and fixed in 10% buffered formalin solution for 1 week and then placed in decalcifying solution until complete decalcification. The laminectomy site was identified and four 2-mm-thick sections were obtained. Each section was embedded in paraffin and serial sections (5 μm) were cut with a microtome and stained with haematoxylin and eosin (HE) for examination. All the sections were examined by a pathologist (SC) who was blinded to the groups.
In the histopathological evaluation, epidural fibrosis, fibroblast density, inflammatory cell density, and arachnoid fibrosis were investigated.
Epidural fibrosis was graded based on the scheme devised by He et al. [16]: Grade 0, the dura is free of scar tissue; Grade 1, only thin fibrous bands are observed between the scar tissue and dura; Grade 2, continuous adherence is observed in less than two-thirds of the laminectomy defect; and Grade 3, scar tissue adherence is large, affecting more than two-thirds of the laminectomy defect, or the adherence extends to the nerve roots.
To quantify the epidural cell density (fibroblasts and inflammatory cells) in the scar tissue, the cells in three different areas (two borders and the centre of the laminectomy defect) were counted and the mean was calculated. The fibroblast and inflammatory cell densities were graded as follows: Grade 1, less than 100 fibroblasts/inflammatory cells per ×400 field; Grade 2, 100 to 150 fibroblasts/inflammatory cells per ×400 field; Grade 3, more than 150 fibroblasts/inflammatory cells per ×400 field.
The presence and degree of arachnoid fibrosis were also investigated and graded as 0 = absent, 1 = minimal, 2 = moderate, and 3 = severe.
Statistical analysis
The data were analysed using the Kruskal–Wallis non-parametric test. The Mann–Whitney U test was used to compare the data between groups. P < 0.05 was deemed statistically significant. The statistical analysis was performed using the Statistical Package for the Social Sciences (SPSS), version 11.0.
Results
In all animals, the postoperative course was uneventful except for a skin infection in one rat in the sham group. This superficial wound infection was controlled with povidone–iodine, without using antibiotics. No sign of neurological deficit or cerebrospinal leak was observed in any rat.
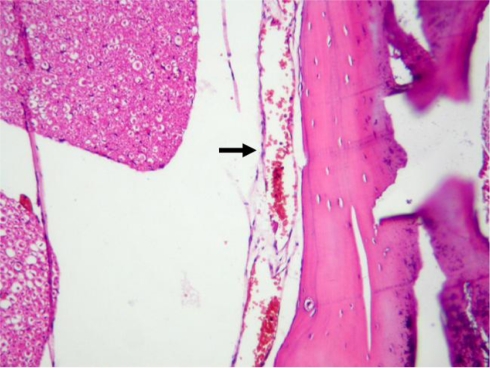
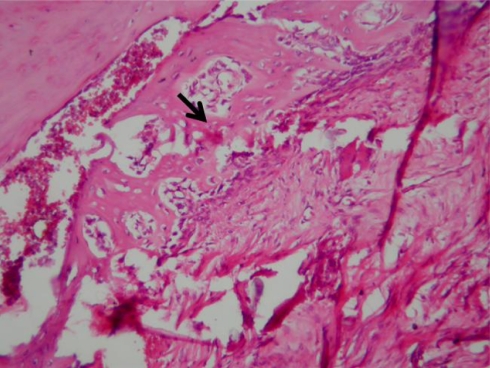
The histological parameters for the groups are summarised in Table 1. Grade 3 epidural fibrosis was observed in all of the sham rats (Fig. 1). Epidural fibrosis was significantly greater in the sham group than in all of the treatment groups (P = 0.000, P = 0.000, and P = 0.000 for MMC, 5-FU, and CsA, respectively; Fig. 2). The arachnoid fibrosis was Grade 3 in all of the sham rats. The arachnoid fibrosis was significantly denser in the sham group than in all of the treatment groups (P = 0.000, P = 0.000, and P = 0.000 for MMC, 5-FU, and CsA, respectively). The fibroblast cell density was significantly lower in the MMC and 5-FU groups compared with the sham group (Fig. 3), whereas it was similar in CsA and sham groups (Fig. 4; P = 0.001, P = 0.005, and P = 0.065 for MMC, 5-FU, and CsA, respectively). The inflammatory cell density was significantly decreased in the MMC and 5-FU groups compared with the sham group, whereas it was similar in the CsA and sham groups (P = 0.002, P = 0.002, and P = 0.234 for MMC, 5-FU, and CsA, respectively).
Table 1.
Histopathologic examination results of sham and treatment groups
| Groups | Epidural fibrosis in dura mater | Fibroblast cell density | Inflammatory cell density | Arachnoidal fibrosis |
|---|---|---|---|---|
| Sham | ||||
| Sh1 | 3 | 3 | 2 | 3 |
| Sh2 | 3 | 3 | 2 | 3 |
| Sh3 | 3 | 3 | 2 | 3 |
| Sh4 | 3 | 2 | 2 | 3 |
| Sh5 | 3 | 3 | 2 | 3 |
| Sh6 | 3 | 2 | 2 | 3 |
| Sh7 | 3 | 3 | 2 | 3 |
| Sh8 | 3 | 3 | 2 | 3 |
| MMC | ||||
| MMC1 | 2 | 1 | 1 | 1 |
| MMC2 | 1 | 2 | 1 | 1 |
| MMC3 | 1 | 1 | 1 | 1 |
| MMC4 | 1 | 1 | 2 | 1 |
| MMC5 | 1 | 1 | 1 | 2 |
| MMC6 | 1 | 1 | 1 | 2 |
| MMC7 | 1 | 2 | 1 | 1 |
| MMC8 | 1 | 1 | 1 | 1 |
| 5-FU | ||||
| 5-FU1 | 2 | 2 | 2 | 1 |
| 5-FU2 | 1 | 1 | 1 | 1 |
| 5-FU3 | 1 | 2 | 1 | 2 |
| 5-FU4 | 1 | 2 | 1 | 2 |
| 5-FU5 | 0 | 2 | 1 | 2 |
| 5-FU6 | 1 | 1 | 1 | 1 |
| 5-FU7 | 1 | 2 | 1 | 2 |
| 5-FU8 | 1 | 2 | 1 | 2 |
| CsA | ||||
| CsA1 | 1 | 3 | 3 | 2 |
| CsA2 | 2 | 2 | 2 | 2 |
| CsA3 | 1 | 1 | 1 | 1 |
| CsA4 | 1 | 2 | 1 | 1 |
| CsA5 | 1 | 3 | 2 | 2 |
| CsA6 | 0 | 2 | 1 | 1 |
| CsA7 | 1 | 1 | 1 | 1 |
| CsA8 | 1 | 2 | 2 | 2 |
Sh Sham, MMC Mitomisin C, 5-FU 5-Flurourasil, CsA Cyclosporin A
Fig. 1.
The arrow indicates increased fibrosis (Grade 3) in the dura in sham, HE ×40
Fig. 2.
Grade 0 fibrosis in the dura, the dura is free of scar tissue, it contains blood vessels only, HE ×40
Fig. 3.
Grade 1 fibroblast density, note the lower number of fibroblast cells, HE ×200
Fig. 4.
Grade 3 fibroblast density, note the increased number of fibroblast cells, HE ×200
Discussion
Postoperative epidural fibrosis following lumbar disc surgery significantly increases the hazards of revision spine surgery and contributes to the occurrence of FBSS [38, 39].
The formation of epidural scar tissue following epidural space surgery can vary. Epidural scar tissue often develops following spinal surgery when epidural fat is replaced by a haematoma. This haematoma is absorbed and replaced with granulation tissue, which matures into dense fibrotic tissue [25, 41]. Additional factors play a part in epidural fibrosis and the related symptoms, including excessive bleeding, excessive root tension, root anomalies, the use of sponges, and foreign tissue reaction [8, 11, 17]. Dense epidural fibrosis causes nerve root irritation, entrapment, restriction of nerve root mobility, and direct dural compression [16, 33, 41], which is the main reason for back and leg pain. Regardless of whether or not it is one of the important causes of FBSS, the existence of epidural fibrosis is related to a poorer surgical outcome and increased complication rates in patients requiring reoperation [3, 21, 34].
Because of its relationship to FBSS and its association with the increased complication rate in revision spine surgery, many treatment strategies that might prevent epidural fibrosis have been investigated. Methods include microdiscectomy, anti-inflammatory medication, and the use of biological or synthetic materials, including fat grafts [14, 24, 25, 36], absorbable gelatine sponges, silastic membrane [36], polylactic acid [31], polymethyl methacrylate, carboxymethylcellulose, sodium hyaluronate, and Adcon-L [2, 16]. Most of these materials have been moderately effective or ineffective [12, 27]. The studies that reported effective outcomes were either not done in humans or involved small series. Despite intensive effort, no drug or material is routinely used to prevent or decrease epidural fibrosis following spinal surgery, which is the procedure that neurosurgeons most commonly perform.
In this study, we examined the effects of the local application of MMC, 5-FU, and CsA in reducing epidural fibrosis in a rat laminectomy model. We postulated that because the meninges, cornea, and sclera originate from mesoderm, and MMC and 5-FU are effective at preventing scar formation in ophthalmologic operations [12, 18, 23], they might be useful for preventing epidural fibrosis. Although there are no studies on the effect of CsA in preventing epidural fibrosis, we postulated that it might also inhibit fibroblasts via a similar mechanism.
In this study, epidural and arachnoid fibrosis was significantly lower with MMC administration (Grade 0) than in the sham group, although neither pathological feature was inhibited completely. Similar results have been reported with the local administration of 0.05 to 0.2 mg/ml MMC [12, 27]. The administration of 0.01 mg/ml MMC was found to be less effective than higher doses, although it resulted in markedly less epidural fibrosis compared to the sham group [27]. MMC might cause dose-related complications even after local application. Serious complications associated with its use have been reported in ophthalmologic surgery, including epithelial defects, delayed wound healing, corneal perforation, necrotizing keratitis, and persistent hypotonic maculopathy [20, 30, 32, 42].
There were significant decreases in fibroblast and inflammatory cell density, which support the results of Dogulu et al. [12]. Both 5-FU and MMC are antifibrotic agents that have been used in glaucoma surgery to modulate wound healing and improve the surgical outcome. These two agents were first used as adjuncts to trabeculectomy in the early 1980s [44]. 5-FU is used to produce a controlled, scar-limited fistula in glaucoma filtration. Experimentally, 5-FU has been a very effective inhibitor of fibroblast growth.
The results in the 5-FU group were similar to those in the MMC group, with 5-FU successfully preventing epidural fibrosis and reducing fibroblast cell density. We found no studies on the effect of 5-FU on epidural fibrosis in a Medline search. Early findings regarding the antifibrotic effects of 5-FU in glaucoma surgery were encouraging, although the long-term results have been less convincing when it is used in glaucoma filtration. Despite the initial good results, trabeculectomies with 5-FU injections drift towards failure over time [44]. As a result, the long-term effects of the local application of 5-FU for preventing epidural fibrosis need further investigation.
The CsA is a powerful immunosuppressive agent that is widely used for preventing allograft rejection and treating autoimmune diseases. Clinical and experimental data suggest that it also acts on connective tissue [35]. We investigated the influence of CsA on fibrosis formation. To our best knowledge, this is the first study to examine this topic. Although CsA resulted in decreased epidural and arachnoid fibrosis, it had no ability to prevent increases in the densities of fibroblast and inflammatory cells.
Conclusion
In this study, the treatment groups showed promising decreases in the rate of epidural fibrosis. However, further animal trials, safety studies, or long-term effective studies are necessary before human trials should be considered. Further studies should examine different dosages of drugs to determine the short- and long-term complications and their potential in clinical practice.
Footnotes
Presented as special poster in 2006 Annual Meeting of the Spine Society of Europe, October 25–28, İstanbul, Turkey.
References
- 1.Abitbol JJ, Lincoln TL, Lind BI, Amiel D, Akeson WH, Garfin SR. Preventing postlaminectomy adhesion. A new experimental model. Spine. 1994;19:1809–1814. doi: 10.1097/00007632-199408150-00004. [DOI] [PubMed] [Google Scholar]
- 2.Alkalay RN, Kim DH, Urry DW, Xu J, Parker TM, Glazer PA. Prevention of postlaminectomy epidural fibrosis using bioelastic materials. Spine. 2003;28:1659–1665. doi: 10.1097/00007632-200308010-00006. [DOI] [PubMed] [Google Scholar]
- 3.Aydin Y, Ziyal IM, Duman H, Turkmen CS, Basak M, Sahin Y. Clinical and radiological results of lumbar microdiskectomy technique with preserving of ligamentum flavum comparing to the standard microdiskectomy technique. Surg Neurol. 2002;57:5–13. doi: 10.1016/S0090-3019(01)00677-2. [DOI] [PubMed] [Google Scholar]
- 4.Bagci G, Yucel I, Duranoglu Y. The effect of cyclosporin A on cultured rabbit subconjunctival fibroblast proliferation. Ophthalmologica. 1999;213:114–119. doi: 10.1159/000027403. [DOI] [PubMed] [Google Scholar]
- 5.Barbera J, Gonzalez J, Esquerdo J, Broseta J, Barcia-Salorio JL. Prophylaxis of the laminectomy membrane. An experimental study in dogs. J Neurosurg. 1978;49:419–424. doi: 10.3171/jns.1978.49.3.0419. [DOI] [PubMed] [Google Scholar]
- 6.Benoist M, Ficat C, Baraf P, Cauchoix J. Postoperative lumbar epiduro-arachnoiditis. Diagnostic and therapeutic aspects. Spine. 1980;5:432–436. doi: 10.1097/00007632-198009000-00007. [DOI] [PubMed] [Google Scholar]
- 7.Burton CV, Kirkaldy-Willis WH, Yong-Hing K, Heithoff KB. Causes of failure of surgery on the lumbar spine. Clin Orthop Relat Res. 1981;157:191–199. [PubMed] [Google Scholar]
- 8.Chang BS, Brown PR, Sieber A, Valdevit A, Tateno K, Kostuik JP. Evaluation of the biological response of wear debris. Spine J. 2004;4(6 Suppl):239S–244S. doi: 10.1016/j.spinee.2004.07.014. [DOI] [PubMed] [Google Scholar]
- 9.Chung JH, Cosenza MJ, Rahbar R, Metson RB. Mitomycin C for the prevention of adhesion formation after endoscopic sinus surgery: a randomized, controlled study. Otolaryngol Head Neck Surg. 2002;126:468–474. doi: 10.1067/mhn.2002.124705. [DOI] [PubMed] [Google Scholar]
- 10.Cook SD, Prewett AB, Dalton JE, Whitecloud TS., 3rd Reduction in perineural scar formation after laminectomy with polyactive membrane sheets. Spine. 1994;19:1815–1825. doi: 10.1097/00007632-199408150-00005. [DOI] [PubMed] [Google Scholar]
- 11.Coskun E, Suzer T, Topuz O, Zencir M, Pakdemirli E, Tahta K. Relationships between epidural fibrosis, pain, disability, and psychological factors after lumbar disc surgery. Eur Spine J. 2000;9:218–223. doi: 10.1007/s005860000144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Dogulu F, Kurt G, Emmez H, Erdem O, Memis L, Baykaner K, Ceviker N. Topical mitomycin C-induced inhibition of postlaminectomy peridural fibrosis in rabbits. J Neurosurg. 2003;99(1 Suppl):76–79. doi: 10.3171/spi.2003.99.1.0076. [DOI] [PubMed] [Google Scholar]
- 13.Einhaus SL, Robertson JT, Dohan FC, Jr, Wujek JR, Ahmad S. Reduction of peridural fibrosis after lumbar laminotomy and discectomy in dogs by a resorbable gel (ADCON-L). Spine. 1997;22:1440–1447. doi: 10.1097/00007632-199707010-00003. [DOI] [PubMed] [Google Scholar]
- 14.Gill GG, Scheck M, Kelley ET, Rodrigo JJ. Pedicle fat grafts for the prevention of scar in low-back surgery. A preliminary report on the first 92 cases. Spine. 1985;10:662–667. doi: 10.1097/00007632-198509000-00012. [DOI] [PubMed] [Google Scholar]
- 15.Gilman AG, Rall Tw, Nies AS, Taylor P (eds) (1992) Goodman and Gilman’s “The Pharmacological Basis of Therapeutics”, 8th edn. McGraw-Hill, New York, vol 2, pp 1247–1248
- 16.He Y, Revel M, Loty B. A quantitative model of post-laminectomy scar formation. Effects of a nonsteroidal anti-inflammatory drug. Spine. 1995;20:557–563. doi: 10.1097/00007632-199503010-00010. [DOI] [PubMed] [Google Scholar]
- 17.Jayson MI. The role of vascular damage and fibrosis in the pathogenesis of nerve root damage. Clin Orthop Relat Res. 1992;279:40–48. [PubMed] [Google Scholar]
- 18.Joshi AB, Parrish RK, 2nd, Feuer WF. 2002 survey of the American Glaucoma Society: practice preferences for glaucoma surgery and antifibrotic use. J Glaucoma. 2005;14:172–174. doi: 10.1097/01.ijg.0000151684.12033.4d. [DOI] [PubMed] [Google Scholar]
- 19.Key JA, Ford LT. Experimental intervertebral-disc lesions. J Bone Joint Surg Am. 1948;30:621–630. [PubMed] [Google Scholar]
- 20.Khaw PT, Doyle JW, Sherwood MB, Grierson I, Schultz G, McGorray S. Prolonged localized tissue effects from 5-minute exposures to fluorouracil and mitomycin C. Arch Ophthalmol. 1993;111:263–267. doi: 10.1001/archopht.1993.01090020117035. [DOI] [PubMed] [Google Scholar]
- 21.Kim KD, Wang JC, Robertson DP, Brodke DS, Olson EM, Duberg AC, BenDebba M, Block KM, diZerega GS. Reduction of radiculopathy and pain with Oxiplex/SP gel after laminectomy, laminotomy, and discectomy: a pilot clinical study. Spine. 2003;28:1080–1087. doi: 10.1097/00007632-200305150-00023. [DOI] [PubMed] [Google Scholar]
- 22.Kitano T, Zerwekh JE, Edwards ML, Usui Y, Allen MD. Viscous carboxymethylcellulose in the prevention of epidural scar formation. Spine. 1991;16:820–833. doi: 10.1097/00007632-199107000-00023. [DOI] [PubMed] [Google Scholar]
- 23.Lama PJ, Fechtner RD. Antifibrotics and wound healing in glaucoma surgery. Surv Ophthalmol. 2003;48:314–346. doi: 10.1016/S0039-6257(03)00038-9. [DOI] [PubMed] [Google Scholar]
- 24.Langenskiold A, Kiviluoto O. Prevention of epidural scar formation after operations on the lumbar spine by means of free fat transplants. A preliminary report. Clin Orthop Relat Res. 1976;115:92–95. [PubMed] [Google Scholar]
- 25.LaRocca H, Macnab I. The laminectomy membrane. Studies in its evolution, characteristics, effects and prophylaxis in dogs. J Bone Joint Surg Br. 1974;56B:545–550. [PubMed] [Google Scholar]
- 26.Lee HM, Yang KH, Han DY, Kim NH. An experimental study on prevention of postlaminectomy scar formation. Yonsei Med J. 1990;31:359–366. doi: 10.3349/ymj.1990.31.4.359. [DOI] [PubMed] [Google Scholar]
- 27.Lee JY, Stenzel W, Ebel H, Wedekind C, Ernestus RI, Klug N. Mitomycin C in preventing spinal epidural fibrosis in a laminectomy model in rats. J Neurosurg. 2004;100(1 Suppl Spine):52–55. doi: 10.3171/spi.2004.100.1.0052. [DOI] [PubMed] [Google Scholar]
- 28.Long DM, Filtzer DL, BenDebba M, Hendler NH. Clinical features of the failed-back syndrome. J Neurosurg. 1988;69:61–71. doi: 10.3171/jns.1988.69.1.0061. [DOI] [PubMed] [Google Scholar]
- 29.Maroon JC, Abla A, Bost J. Association between peridural scar and persistent low back pain after lumbar discectomy. Neurol Res. 1999;21:S43–S46. doi: 10.1159/000017365. [DOI] [PubMed] [Google Scholar]
- 30.Mietz H, Krieglstein GK. Three-year follow-up of trabeculectomies performed with different concentrations of mitomycin-C. Ophthalmic Surg Lasers. 1998;29:628–634. [PubMed] [Google Scholar]
- 31.Mikawa Y, Hamagami H, Shikata J, Higashi S, Yamamuro T, Hyon SH, Ikada Y. An experimental study on prevention of postlaminectomy scar formation by the use of new materials. Spine. 1986;11:843–846. doi: 10.1097/00007632-198610000-00021. [DOI] [PubMed] [Google Scholar]
- 32.Murayama T, Takahashi N, Ikoma N. Cytotoxicity and characteristics of mitomycin C. Ophthalmic Res. 1996;28:153–159. doi: 10.1159/000267896. [DOI] [PubMed] [Google Scholar]
- 33.Nussbaum CE, McDonald JV, Baggs RB. Use of Vicryl (polyglactin 910) mesh to limit epidural scar formation after laminectomy. Neurosurgery. 1990;26:649–654. doi: 10.1097/00006123-199004000-00014. [DOI] [PubMed] [Google Scholar]
- 34.Park YK, Kim JH, Chung HS. Outcome analysis of patients after ligament-sparing microdiscectomy for lumbar disc herniation. Neurosurg Focus. 2002;13(2):E4. doi: 10.3171/foc.2002.13.2.5. [DOI] [PubMed] [Google Scholar]
- 35.Petri JB, Schurk S, Gebauer S, Haustein UF. Cyclosporine A delays wound healing and apoptosis and suppresses activin beta-A expression in rats. Eur J Dermatol. 1998;8:104–113. [PubMed] [Google Scholar]
- 36.Quist JJ, Dhert WJ, Meij BP, Visser WJ, Oner FC, Hazewinkel HA, Verbout AJ. The prevention of peridural adhesions. A comparative long-term histomorphometric study using a biodegradable barrier and a fat graft. J Bone Joint Surg Br. 1998;80:520–526. doi: 10.1302/0301-620X.80B3.8010. [DOI] [PubMed] [Google Scholar]
- 37.Robertson JT, Meric AL, Dohan FC, Jr, Schweitzer JB, Wujek JR, Ahmad S. The reduction of postlaminectomy peridural fibrosis in rabbits by a carbohydrate polymer. J Neurosurg. 1993;79:89–95. doi: 10.3171/jns.1993.79.1.0089. [DOI] [PubMed] [Google Scholar]
- 38.Robertson Role of peridural fibrosis in the failed back: a review. Eur Spine J. 1996;5:S2–6. doi: 10.1007/BF00298565. [DOI] [PubMed] [Google Scholar]
- 39.Ross JS, Robertson JT, Frederickson RC, Petrie JL, Obuchowski N, Modic MT, deTribolet N. Association between peridural scar and recurrent radicular pain after lumbar discectomy: magnetic resonance evaluation. ADCON-L European Study Group . Neurosurgery. 1996;38:855–861. doi: 10.1097/00006123-199604000-00053. [DOI] [PubMed] [Google Scholar]
- 40.Songer MN, Ghosh L, Spencer DL. Effects of sodium hyaluronate on peridural fibrosis after lumbar laminotomy and discectomy. Spine. 1990;15:550–554. doi: 10.1097/00007632-199006000-00022. [DOI] [PubMed] [Google Scholar]
- 41.Songer MN, Rauschning W, Carson EW, Pandit SM. Analysis of peridural scar formation and its prevention after lumbar laminotomy and discectomy in dogs. Spine. 1995;20:571–580. doi: 10.1097/00007632-199503010-00012. [DOI] [PubMed] [Google Scholar]
- 42.Ward RF, April MM. Mitomycin-C in the treatment of tracheal cicatrix after tracheal reconstruction. Int J Pediatr Otorhinolaryngol. 1998;44:221–226. doi: 10.1016/S0165-5876(98)00061-5. [DOI] [PubMed] [Google Scholar]
- 43.Wujek JR, Ahmad S, Harel A, Maier KH, Roufa D, Silver J. A carbohydrate polymer that effectively prevents epidural fibrosis at laminectomy sites in the rat. Exp Neurol. 1991;114:237–245. doi: 10.1016/0014-4886(91)90040-J. [DOI] [PubMed] [Google Scholar]
- 44.Yoon PS, Singh K. Update on antifibrotic use in glaucoma surgery, including use in trabeculectomy and glaucoma drainage implants and combined cataract and glaucoma surgery. Curr Opin Ophthalmol. 2004;15:141–146. doi: 10.1097/00055735-200404000-00015. [DOI] [PubMed] [Google Scholar]