Abstract
Primary malignant spinal tumors and solitary vertebral metastases of selected tumor entities in the thoracolumbar spine are indications for total en bloc spondylectomy (TES). This study aimed to describe our oncological and surgical management and to analyze the treatment results by management with TES for extra- and intracompartmental solitary spinal metastases and primary malignant vertebral bone tumors. In 15 patients (3 malignant bone tumors and 12 solitary metastases), tumors were distributed in the thoracic (n = 8) and lumbar (n = 7) spine. Tumors were classified as intra- (n = 8) and extracompartmental (n = 7). All patients underwent TES via a laterally extended posterior approach followed by dorsoventral reconstruction. Function and quality of life were assessed by Oswestry disability index (ODI) and SF-36 score. At follow-up (100%; mean: 33 ± 22 months), 11 patients had no evidence of disease. Two patients were alive with the disease and two were dead of the disease (no primary bone tumors). Histology revealed negative margins (R0) in all patients with wide (n = 11) and marginal (n = 4) resections. Two patients developed pulmonal metastases of which they died at 4 and 16 months of survival. No local recurrence was observed. Major complications did not occur. TES resulted in an acceptable outcome in the quality of life and function. TES is a demanding procedure reaching wide to marginal resections in a curative approach. In conjunction with multimodal therapies, local recurrences can effectively be prevented while control of distant disease needs to be improved. Proper selection of adequate patients combined with careful surgical planning are prerequisites for low complication rates, acceptable function and improved overall prognosis.
Keywords: En bloc spondylectomy, Solitary metastases, Primary malignant tumors, Thoracolumbar spine
Introduction
To date, it is generally accepted that wide surgical margins, in combination with multimodality therapy, represent an essential precondition for disease-free survival in patients suffering from primary and secondary malignant bone tumors. In comparison to the extremities, where improved reconstruction of neurovascular and soft tissue structures with vascular grafts, neural transplants and pedicled or free flaps regularly allow wide resections and increased limb salvage, the resection of malignant tumors of the spine is incomparably more difficult [3, 34]. The topographic vicinity of vital and central neurovascular structures, i.e., spinal cord, nerve roots, aorta, caval vein, etc., to the vertebral column profoundly complicate the achievement of wide resection margins for malignant vertebral tumors. Intraoperative tumor cell contamination may unavoidably occur in some cases and rare tumor entities exist in which marginal to intralesional resection does not necessarily lead to a palliative approach. Nevertheless, intralesional surgery predisposes the patient to local recurrences and poor prognosis. To reduce the surgery-induced tumor cell dissemination, decrease local recurrence rates and to improve survival times, different techniques [8, 15, 16, 20, 23, 25, 26, 32] for wide resections at the spine, involving either en bloc total [32] or hemilaminectomy [18] followed by en bloc corporectomy and dorso-ventral stabilization have been developed. In early 1980, Stener and Roy-Camille were the first to describe en bloc spondylectomy via a posterior approach after complete resection of the dorsal vertebral structures, i.e., both laminae, spinous process and parts of the pedicles, resulting in an oncological adequate resection for primary bone tumor locations not extending beyond the vertebral body [23, 26]. Based on these reports, several authors have begun to develop further techniques for en bloc vertebrectomy encompassing variable indications, approaches, stabilization techniques and yielding promising results in view of local control and overall prognosis [6, 20, 27, 32, 38]. Tomita et al. reported a technique with dorsal en bloc resection after transpedicular osteotomy followed by ventral vertebrectomy [32]. Significant progress in decision making for surgical therapy of spinal malignancies was made by the surgical classification for tumors of the spine described by Boriani et al. [8]. Depending on the stage, localization and extension of the tumor within the affected vertebra, the approach and mode of resection can be deduced. In addition, applying the oncological principles of muskuloskeletal surgery of the extremities to the spine, Tomita et al. have modified the surgical staging system of Enneking et al. [13] and developed a surgical classification of vertebral tumors, considering tumors strictly located in the vertebral body, pedicle or laminae as intra-, and malignancies spreading beyond the vertebral cortex into the spinal canal (epidural space), paraspinal area or disk space as extracompartmental [32]. While wide resections for tumor not penetrating the cortex of the vertebra are definitely feasible, attainment of wide resection margins for extracompartmentally located spinal tumors is profoundly complicated and may require additional resection of adjacent tissue layers functioning as a biological border for invading tumor cells. Furthermore, as described by Krepler et al., extracompartmental tumor growth into the spinal canal with invasion of the dura can necessitate en bloc spondylectomy with dura resection and replacement, again underscoring the essential importance of wide margins in the surgical treatment of malignant spinal bone tumors [19]. However, extracompartmental tumor growth with circumferential dura invasion or anterior invasion around major vascular structures does not allow for wide resection, but verges on the limits of what is surgically feasible and oncologically useful.
Based upon these reports, the aim of the current study was to present our results of total en bloc spondylectomy (TES) for patients with solitary spinal metastasis and primary malignant vertebral tumors.
Methods
In a 6-year period, 15 Patients (eight males, seven females; mean age: 46.6 ± 20 years, range: 5–72) underwent TES by one surgical team. Three patients suffering from primary malignant tumors and 12 patients with solitary vertebral metastases were included. No patients with recurrent or residual disease were investigated. Tumors were equally distributed in the thoracic (n = 8) and lumbar (n = 7) spine. According to the surgical classification of vertebral tumors [32, 34], the anatomic sites of eight tumors were considered as intra- and of seven tumors as extracompartmental. Five patients were treated with surgical treatment alone and another ten patients underwent neoadjuvant or adjuvant postoperative therapy, i.e., systemic polychemotherapy and/or radiation therapy (Table 1). Two patients (no. 1 and 3) with a primary malignant vertebral tumor became symptomatic with progressive paraplegic symptoms (Table 1). Thus, an intralesional emergency laminectomy for decompression was performed as the primary operative procedure. In all other patients, diagnosis was secured by CT-assisted transpedicular biopsy. One patient suffering from post chemotherapy residual tumor mass in the Th10 vertebral body was included due to vital tumor cells in the CT-guided biopsy, elevating HCG-levels and positive PET scan [4, 17]. Apart from conventional radiographs in two planes for surgical planning of preoperative staging, CT-scans of the chest, abdomen and the spine levels affected by and neighboring to the tumor lesion were included. In addition, a technetium bone scintigraphy was performed in all patients for the assessment of secondary lesions. Furthermore, in patients displaying extracompartmental tumor location [32, 34], an additional MRI scan of the affected spine levels was performed. All patients were evaluated using the revised scoring system of metastatic spine tumor prognosis, according to Tokuhashi et al. [30].
Table 1.
Patient characteristics (no. 1–3: primary bone tumors; 4–15: solitary metastases), tumor type, location, primary tumor site, type according to the surgical classification of spinal tumors of Tomita [32], revised Tokuhashi prognostic score [30], disease-free survival before the appearance of metastases, preoperatively diagnosed neurological symptoms, previous therapy, surgical margins and current oncological status
| Number | Sex/age | Histology | Site/resected segment | Primary tumor site (adjuvant therapy) | Classification (Tomita) | Tokuhashi prognostic score (metastatic tumors) | Diseases-free survival before metastasis (months) | Preoperative neurological symptoms | Multimodal therapy chemo: C radiation: R | Surgical margins | Cumulative disease specific survival (months) | Cumulative metastases free survival (months) | Oncolog status |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | M/55 | Malignant fibrous histiocytoma | Th 2/3 | – | 6 | – | – | Paraparesis emergency laminectomy | C (neoadjuvant) | Marginal | 52 | 52 | NED |
| 2 | F/33 | Pleomorphous sarcoma | Th 9/10/11 | – | 6 | – | – | – | C (neoadjuvant) | Wide | 51 | 51 | NED |
| 3 | M/12 | Osteosarcoma | Th 1 | – | 5 | – | – | Paraparesis emergency laminectomy | C + R (neoadjuvant) | Marginal | 40 | 40 | NED |
| 4 | F/54 | Leiomyosarcoma | L 1 | Left thigh (no) | 2 | 12 | 98 | Radicular (L1) | – | Wide | 57 | 57 | NED |
| 5 | M/56 | Renal cell Ca | Th 8 | Left kidney (no) | 2 | 13 | 5 | – | R | Wide | 16 | 12 | DOD |
| 6 | F/59 | Leiomyosarcoma | L 2/3 | Retroperitoneum (no) | 6 | 11 | 66 | – | C + R | Wide | 4 | 2 | DOD |
| 7 | M/5 | Teratoma | L 2 | Left groin (radiation) | 4 | 12 | 23 | – | C | Marginal | 75 | 75 | NED |
| 8 | M/22 | Seminoma | Th 9 | Right testes (chemotx) | 4 | 11 | 12 | – | C + R (neoadjuvant) | Marginal | 44 | 44 | NED |
| 9 | F/58 | Mamma-Ca | Th 11 | Right mamma (tamoxifen) | 2 | 15 | 56 | – | – | Wide | 44 | 44 | NED |
| 10 | M/44 | Renal cell Ca | L 4 | Right kidney (no) | 2 | 11 | 12 | – | C | Wide | 25 | 3 | AWD |
| 11 | F/52 | Mamma-Ca | L 5 | Right mamma (tamoxifen) | 2 | 14 | 46 | – | C | Wide | 41 | 4 | AWD |
| 12 | F/72 | Malignant schwannoma | L2 | Left forearm (no) | 5 | 12 | 44 | – | R | Marginal | 19 | 19 | NED |
| 13 | F/67 | Mamma-Ca | Th10 | Left mamma (no) | 2 | 15 | 81 | – | – | Wide | 18 | 18 | NED |
| 14 | M/62 | Renal cell Ca | Th6 | Right kidney (no) | 2 | 13 | 180 | – | – | Wide | 4 | 4 | NED |
| 15 | M/49 | Renal cell Ca | Th9 | Left kidney (no) | 2 | 13 | 151 | – | – | Wide | 2 | 2 | NED |
Patient no. 2 received mulitvisceral resection (Th 9/10/11, lower lung lobe, diaphragm, parts of the chest wall en bloc) DOD dead of disease, AWD alive with disease, NED no evidence of disease
Surgical procedure
Under general anesthesia, eight patients were placed on the side and seven patients in a prone position. After a dorsomedian skin incision, the paraspinal muscles were detached from the spinous processes and the laminae as well as the facet joints; however, in cases with extracompartmental tumor manifestation/extension overlying soft tissue layers were left untouched. In 11 patients, the dorsal approach was extended laterally to the side unaffected by the tumor. In two patients (no. 10 and 11), a dorsal lumbar approach and instrumentation was combined by ventral retroperitoneal approach and subsequent resection and stabilization. In the thoracic spine, the dorsal parts of ribs adjacent to the costotransverse joint were resected in order to reach access to the ventral aspect of the affected vertebrae. En bloc laminectomy was performed when tumor growth did not extend into the laminae, as judged from coronar MRI-/CT scans. In case of tumor invasion in either one of the laminae, hemilaminectomy of the unaffected side was performed. Open, cut cross sections and bone surfaces were immediately sealed with bone wax. Intervertebral dissection of the neighboring disk spaces and of the posterior and partially anterior longitudinal ligaments was performed carefully with a specific chisel-like dissector and rongeur. The resection of the anterior column of the affected vertebrae was preceded by gentle and blunt dissection in the interface between the anterior aspect of the vertebral body and the pleura, aorta as well as the iliopsoas muscle in the thoracic and lumbar spine, respectively. In the thoracic spine, the nerve roots were transected if root infiltration by the extracompartmental tumor was present. Intraoperatively, multiple frozen specimens were sent for histopathological evaluation to exclude tumor infiltration and to determine the status of resection margins (wide, marginal or intralesional).
Before segmental resection of the vertebral body was completed, unilateral posterior instrumentation (CT-based navigation of pedicle screws) was performed in order to achieve stability of the spine. After gentle dissection of the spinal cord from the surrounding epidural venous plexus and the ligamentous tissue in the spinal canal using a thin nerve dissector, the entire vertebra was mobilized step-by-step followed by meticulous bipolar coagulation of bleeding branches of the venous plexus. Finally, the vertebra was rotated along its longitudinal axis in a direction opposite to the unilateral instrumentation without compression or injury of the spinal cord, leading to a circumferential decompression of the spinal cord and a segmental total en bloc vertebrectomy (Fig. 1).
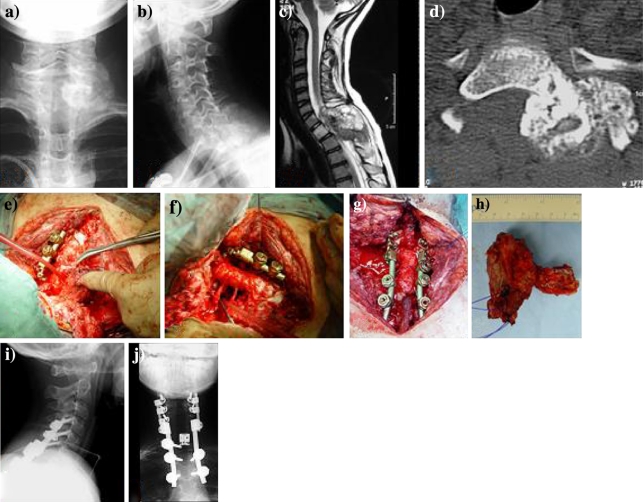
Fig. 1.
Patient no. 3. Extracompartmental osteosarcoma affecting the Th1 vertebra in a male 12-year-old child, symptomatic with acute paraparesis and necessitating emergency laminectomy. Front (a) and lateral (b) radiographs show a large radiodense tumor mass extending dorsal and left to Th1. Sagittal MRI scan prior to laminectomy (c) revealed complete destruction of the dorsal elements, spinal canal involvement and marked compression of the spinal cord. Following emergency laminectomy and neoadjuvant multimodal therapy (COSS 96 and radiation), coronar CT-scans (d) demonstrate a decrease in size and increase in sclerosis of the tumor tissue. (e) Intraoperative view showing rotation of the Th1 vertebra around the longitudinal axis. After en bloc excision (f) posterior cervicothoracal reconstruction with anterior expandable cage interposition (g) was performed. Specimen (h) of Th1-vertebral osteosarcoma originating from and extending dorsolaterally to the pedicle after en bloc excision, showing histologically tumor-free margins (posterior view). (i), (j) Postoperative biplanar radiographs following dorsoventral instrumentation
Reconstruction of the spine was performed by completion of the posterior instrumentation, two or one level (if one vertebra was resected) below and above the spondylectomy in the thoracic and lumbar spine, respectively. Anterior stabilization was carried out using expandable cages filled with and surrounded by cancellous bone graft or bone substitutes as well as an additional ventral angular stable plate system between the neighboring healthy vertebrae.
Postoperative management was characterized by wound drainage for 3–5 days. Patients were mobilized after 1 week and were instructed to wear a thoracolumbar corset for 2–3 months if tumor resection of the lumbar spine had been performed.
Histological assessment and surgical classification
All resected specimens were analyzed histologically in order to determine the surgical margins. Tumor extension was assessed as either intra- or extracompartmental according to the classification by Tomita et al. [32, 34]. The resection was classified as wide when the distance between tumor tissue and excision margin was oncologically sufficient and/or an intact anatomical barrier (e.g., compartmental cortical wall of the vertebra) was present. The surgical margin was defined as intralesional if visible tumor tissue was present, the tumor was cut through during the operation or the excision margins were positive at the microscopic level. If the margin was less than wide, but more than intralesional (thin tumor-free tissue layer or capsule), the resection status was classified as marginal [29].
Follow-up
All patients were followed routinely by bidimensional X-ray and CT-scan of the thoracic or lumbar spine and chest as well as bone scintigraphy (radionuclid imaging, technetium bone scan) during two postoperative years every 3 months, followed by 6-month intervals during the later course.
Oncological outcome
Local recurrence-free survival, distant relapse-free survivals and disease-specific overall survival were assessed using the SPSS program (MicroSoft Windows release 6.1; SPSS Inc., Chicago, IL, USA). Survival data were collected from the start of operative treatment.
Functional outcome and quality of life assessment
Examinations at follow-up included subjective and objective measures using the Oswestry-Disability Index [14], ranging from 0 to maximum 100 (effect units). Postoperative quality of life was assessed by the validated psychometric questionnaire SF-36, which is based on the American short-form-36 health survey [36, 37] and has been psychometrically validated [1, 5, 10]. As the SF-36 score considers 36 items in eight different scales of subjective physical and mental health as well as social and emotional aspects, it is generally considered to reliably assess disease-related quality of life. The results for SF-36 of patients following en bloc spondylectomy were compared to established baseline values for normal (healthy) German population [9, 10] and patients with chronic back pain [9]. To assess the correlation between functional outcome (ODI) and disease-related quality of life (SF-36), linear regression analysis was used.
Results
Tumor characteristics and pathological findings
Histological diagnosis of the primary spinal bone tumors (localized disease, no distant or regional metastatic lesions) in three patients identified one osteosarcoma (Th 1), one malignant fibrous histiocytoma (Th 2/3) and one pleomorphous sarcoma (Th 9/10/11). Primary tumors of the 12 patients with solitary spinal metastases included three breast cancer, four hypernephroma (renal cell cancer), two leiomyosarcoma, one malignant schwannoma, one teratoma and one seminoma. Despite this heterogeneity in terms of underlying histopathological primary tumor type (six different histologies in 12 patients), pretreatment evaluation using the revised Tokuhashi-score revealed a score range between 11 and 15 in all patients, thus predicting a 1-year survival period in more than 95% and suggesting excisional surgery [30, 31].
All patients with primary bone tumors of the spine were preoperatively treated according to established neoadjuvant polychemotherapy protocols. Furthermore, one patient with primary osteosarcoma of Th 1 was additionally pretreated with radiotherapy. Patient demographics, tumor characteristics, previous therapy, follow-up results and current oncological status are shown in Table 1.
Mortality and morbidity
The mean duration of the operation was 9.2 ± 3.1 h (range 5.3–16.4). The mean amount of transfused blood substitutes for erythrocyte concentrate units was 15.7 ± 14.1 (range 2–52) and 20 ± 15.3 (range 3–56) for fresh frozen plasma units. The mean arterial concentration of hemoglobin at the beginning of the operation (g/dl) was 11.4 ± 2.3 (range 7.9–15.4) and 8.8 ± 2.2 (range 5.0–12.7) at the end of the operation. Patients were treated in a intensive care unit for a mean postoperative duration of 4 ± 2 days (range 2–8). None of the patients died in the direct postoperative period. One patient (no. 6) developed a deep infection on day 8 postoperatively, which subsided following twice-repeated revisions, debridement and wound drainage. Another patient (no. 13) required one revision surgery for treating superficial wound healing disturbances with a final uneventful course. In patient 7 (a 5-year-old child), increasing skeletal and spinal growth led to progressive kyphosis of the lumbar spine, which necessitated a corrective surgical procedure with complete implant removal, exchange of the expandable vertebral replacement cage and new positioning of the dorsal instrumentation. Patient no. 3 developed temporary postoperative neurological deterioration with hypesthesia and slight reduction in motor strength of the finger flexion muscles (Dig IV and V) of the right hand (C8 nerve root), becoming asymptomatic over a 6-week period.
Overall and disease-free survival
All patients with primary bone tumors were free of recurrence and have currently no evidence of disease 39–52 months after surgery with a mean follow-up of 47.7 ± 7.0 months. Mean survival time for patients with solitary metastases was 29.0 ± 23.1 months (range: 2–75). Two patients with initially solitary metastasic lesion of the lumbar and thoracic spine developed progressive pulmonal metastatic disease, 2 and 12 months after surgery, of which they died 4 and 16 months postoperatively. The overall mean survival of all patients was 32.7 ± 22.0 (range: 2–75, 95% confidence intervals 19–39 months). See Table 1.
Local Tumor recurrence
At the time of the last follow-up, none of the patients with primary bone tumors or solitary spinal metastatic lesions developed local tumor recurrence 2–75 months after surgery.
Distant metastases
Eleven patients are currently free from distant metastases 2–75 months after surgery. Four patients who underwent en bloc spondylectomy for solitary metastases (two renal cell carcinoma, one breast cancer, one leiomyosarcoma) developed lung metastases. Two of these patients died of systemic metastatic disease (lung and brain) 4 and 16 months after surgery, whereas the other two patients are currently alive with disease (Table 1). The mean survival time, free from distant metastases in these four patients is 5.3 ± 4.5 months (range: 2–12). The three patients with primary bone tumors are free from distant metastases.
Functional outcome
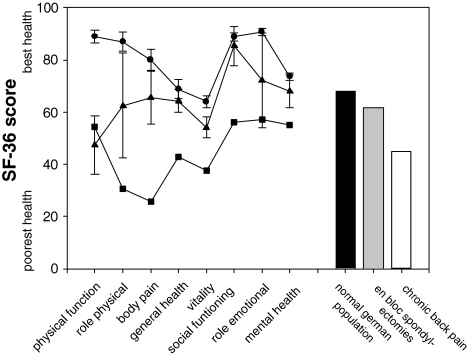
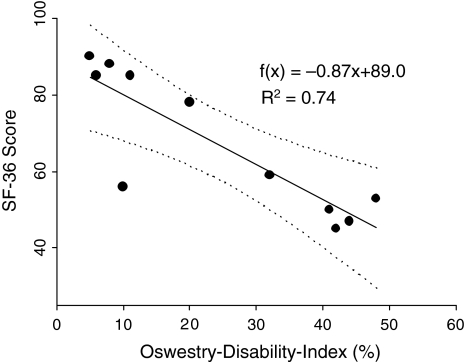
Evaluation of the subjective impairment of function using the Oswestry-low-back-pain-disability questionnaire [14] resulted in mean ODI values of 17.8 ± 16.5% (range 6–48, 95% confidence interval 9–45). Disease-related quality of life, as assessed by the validated psychometric questionnaire SF-36 [37], was found to be decreased for all individual scales, mostly pronounced for physical scales, when compared to German normal population, but markedly exceeded values for chronic back pain patients (Fig. 2). An inverse correlative relationship between functional outcome (ODI) and disease-related quality of life (SF-36 score) was found (Fig. 3).
Fig. 2.
SF-36 health profile for different scales (line scatter plots) and averaged for all scales (bars) in patients following en bloc spondylectomy (triangle up, gray bar) when compared with the general German population norms (circle, filled bar) and chronic back pain patients (square, open bar) [9, 10]
Fig. 3.
Regression analysis of functional outcome (Oswestry Disability Index) on disease-related quality of life (SF-36) for the surviving patients. Dotted lines represent the 95% confidence intervals; R2 is the coefficient of determination. f(x) = − 0.87x + 89.0; R2 = 0.74
Discussion
En bloc resection in patients with primary malignant tumors and solitary metastasis of the spine is now attracting major interest, as its outcome increasingly seems to be superior to piecemeal resection. Direct support for this notion has initially come from en bloc resections of primary spinal neoplasms and vertebral bone tumors. Notably, Boriani et al. compared surgical results of intralesional curettage and en bloc spondylectomy for chondrosarcoma of the spine in 10 and 12 patients, respectively [7]. The authors were able to demonstrate that after more than 5 years follow-up, only 3 of 12 patients developed local relapse after TES; while after curettage, all of the 10 patients suffered from at least one local recurrence and 80% of the patients died of disease. Interpretation of these striking differences included less violation of the tumor border, minimized tumor cell contamination and decreased tumor tissue derived blood loss. Application of these principles on solitary spinal metastatic disease have reinforced the concept of aggressive surgical treatment in selected patients, with biologically favorable cancers, who have the potential of long-term survival [3, 28, 39]. Despite conclusive evidence for significantly improved survival is missing to date, Sundaresan et al. have shown that among 80 patients with solitary spinal metastases who underwent gross resection of tumor by various techniques, the rate of locoregional recurrences for the entire series was 32%, whereas only 17% of the patients who underwent en bloc resection experienced local recurrence [28]. The authors of this study furthermore demonstrated median survival time after en bloc spondylectomy of more than 30 months and nearly 20% surviving more than 5 years. These findings underscore the results reported by Tomita et al. who demonstrated median survival times in patients after en bloc resection of 38 months [33–35].
Based on these promising results, Tomita et al. were the first to extend the surgical oncological concept of en bloc spondylectomy also to the treatment strategy of solitary spinal metastases of selected patients with specific tumor entities [33–35]. According to these studies, there is general consensus that indication for en bloc spondylectomy requires careful patient selection for which the following criteria might be helpful: (1) no underlying tumor type, which is considered to be a systemic disease, (2) tumor types that are known to be biologically favorable and to have a prolonged course (e.g., renal cell or mamma carcinoma), (3) radical treatment of the original/primary tumor, (4) long period between treatment of primary tumor and diagnosis of solitary metastatic disease, and (5) verification of isolated/solitary metastatic disease in CT scans, bone scan and/or PET.
In particular, for breast carcinoma patients, advances in the multidisciplinary approach to treatment, i.e., chemo/hormonal therapy and radiation, have enabled clinicians to improve outcomes [11]. Nevertheless, TES is reported to additionally improve local control and long-term outcome in patients who otherwise were treated with palliative excision surgery or radiochemotherapy [34]. Furthermore, Tomita et al. have developed a prognostic scoring system for spinal metastases [33–35] by revealing a relationship between grades of malignancy and the extent of visceral and further bone metastases on the one hand and long-term local controls as well as disease-free survival on the other. Based on the prognostic score and expected survival periods, treatment goals could be defined and the appropriate oncological surgical strategy, ranging from simple spinal cord decompression to en bloc spondylectomy, can be selected. In this context, Sakaura et al. reported on the outcome of TES for solitary metastasis of the thoracolumbar spine with a local recurrence in two patients, 23 and 25 months after surgery, and dead of disease in 5 of 12 patients [24]. These reports are partly consistent with our results showing no local tumor relapse and dead of disease after a mean survival of 10 months in 2 of 12 patients. Based on the observation that local recurrence of solitary metastases occurred in cases of extracompartimental extension, Sakaura et al. proposed that TES should be limited to intracompartmental solitary lesions. Although in none of our patients with extracompartmental metastatic location local tumor relapse occurred, distant metastases developed in four patients (36%) with initially solitary metastatic lesions. Therefore, efforts must be undertaken to effectively improve postoperative control of distant disease.
We achieved ten wide and five marginal resections, i.e., negative margins (R0) in all patients. Nevertheless, postoperative radiotherapy was performed in five patients and chemotherapy in eight patients, who had either extracompartmental tumors or took part in adjuvant chemotherapy study protocols. At a mean follow-up of 32 months, we had no local recurrence. In the series by Abe et al., local recurrence was found in three cases with a mean follow-up of 38 months [2]. The low recurrence rate in our study, with no re-operation required for local relapse, may be due to both, the consequent multimodality treatment concept and the negative resection margins in all patients. Furthermore, according to our experience, postoperative radiation treatment of the spine shows a positive effect with negligible decrease in function and morbidity. Therefore, after complete wound healing, we included postoperative radiation in patients with marginal resections and when the potential for local recurrence was perceived to be increased.
Wound healing problems and one deep infection occurred in our patients requiring multiple surgeries. These complications were probably due to intensive soft tissue manipulation in order to reach mobility of the affected vertebra for passage of the myelon through the laminectomy gap. Additional causative factors may include long operation durations, marked blood loss and extended surgical approaches, sometimes necessitating a limited thoracotomy. From our experience, a detailed perioperative antibiotic scheme with repeated intraoperative administrations for prophylaxis of infection [22], intra- and postoperative blood transfusions and intensive deep/superficial wound drainage kept for at least 3–5 days [12] for prevention of postoperative hematoma should be performed.
Apart from increasing lumbar kyphotic malalignment with subsequently resulting implant dislocation due to progressive skeletal growth in a child, we had no implant failure or secondary spinal instability in our patients. One favored hypothesis for this observation is the fact that we used a combination of an expandable vertebral replacement cage (filled with autologous bone graft or bone substitutes) and angular stable plate fixation for ventral spinal fusion of the resected vertebral levels in 9 of 15 patients in addition to dorsal stabilization. Expandable cages allow an exact intraoperative adaptation of the cage size on the extent of intervertebral resection, without injury to the endplates of adjacent vertebrae. In addition, cage expansion in situ allows easier cage positioning, increased intervertebral anchorage and enhanced pretension of the anterior column, thus enhanced spinal stability. Ventral stabilization was completed by autologous and homologous cancellous bone graft surrounding the cage and the use of an angular stable anterolateral plate stabilization with fixation of locking head screws in the neighboring vertebral bodies, additionally bridging and stabilizing the resection defect.
Most studies analyzing the results by management with en bloc spondylectomy focus on the oncological outcome without assessing postoperative function and quality of life. As measured by Oswestry-low-back-pain-disability index and the SF-36 health survey, the present study shows that en bloc spondylectomy results in acceptable function as well as physical and mental health (Fig. 2). Results of the validated psychometric questionnaire SF-36 demonstrate that in relation to the normal population, the impact of en bloc spondylectomy on physical components (physical function, role physical, body pain) is more pronounced than on mental scales (vitality, social functioning, role emotional, mental health), which have been nearly comparable to normative values. This notion is underscored by the inverse correlation between the ODI and SF-36 score (Fig. 3) of the surviving patients, indicating the causal role of physical dysfunction (increased ODI) for decreased SF-36 (reduced quality of life) after en bloc resections when compared to the normal population.
Conclusion
This study agrees with previous investigations [2, 6, 18, 21, 24, 32, 34, 38] supporting en bloc spondylectomy for treatment of primary malignant vertebral bone tumors and solitary spinal metastasis of certain tumor entities. En bloc spondylectomy followed by dorsoventral reconstruction allowed radical resections with negative margins in all patients. The present results of a very low local recurrence rate in all patients should be interpreted with caution, as some patients have a follow-up period of less than 18 months and no conclusions can be drawn for these patients in terms of long-term tumor-free survival. However, despite a mean follow-up time of only 33 months, heterogeneity of histological diagnoses and a relatively small number of patients in the current study, long-term local relapse-free survival could be achieved. While en bloc spondylectomy is a technically demanding and risky operation, it allows in attaining wide to marginal resections for primary tumors and solitary metastatic lesions of the spine. However, exact preoperative diagnostic imaging and planning has to precede a realistic evaluation if wide to marginal resections can be achieved in order to justify the risk for the patient. Furthermore, careful selection of patients, consideration of underlying tumor type and extensive experience in spine surgery and reconstruction are essential preconditions for low complication rates, acceptable function and increased overall outcome. Finally, despite excellent local control rates, the limitation for long-term disease-free survival in our patients is distant metastases. Consequently, systemic therapies need to be developed aimed at targeting pathways involved in postoperative metastatic spread and to modify them to impair disseminated tumor growth.
Footnotes
Sources of support: Deutsche Forschungsgemeinschaft (Scha 930/1-1).
Contributor Information
Ingo Melcher, Email: ingo.melcher@charite.de.
Alexander C. Disch, Phone: +49-30-450652136, FAX: +49-30-450652958, Email: alexander.disch@charite.de
Cyrus Khodadadyan-Klostermann, Email: cyrus.khodadadyan@charite.de.
Ulrich Stöckle, Email: ulrich.stoeckle@charite.de.
Norbert P. Haas, Email: norbert.haas@charite.de
Klaus-Dieter Schaser, Email: klaus-dieter.schaser@charite.de.
References
- 1.Aaronson NK, Acquadro C, Alonso J, Apolone G, Bucquet D, Bullinger M, Bungay K, Fukuhara S, Gandek B, Keller S, et al. International quality of life assessment (IQOLA) project. Qual Life Res. 1992;1(5):349–351. doi: 10.1007/BF00434949. [DOI] [PubMed] [Google Scholar]
- 2.Abe E, Sato K, Tazawa H, Murai H, Okada K, Shimada Y, Morita H. Total spondylectomy for primary tumor of the thoracolumbar spine. Spinal Cord. 2000;38(3):146–152. doi: 10.1038/sj.sc.3100968. [DOI] [PubMed] [Google Scholar]
- 3.Aebi M. Spinal metastasis in the elderly. Eur Spine J. 2003;12(Suppl 2):S202–S213. doi: 10.1007/s00586-003-0609-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Albers P, Melchior D, Muller SC. Surgery in metastatic testicular cancer. Eur Urol. 2003;44(2):233–244. doi: 10.1016/S0302-2838(03)00252-5. [DOI] [PubMed] [Google Scholar]
- 5.Alonso J, Ferrer M, Gandek B, Ware JE, Jr, Aaronson NK, Mosconi P, Rasmussen NK, Bullinger M, Fukuhara S, Kaasa S, Leplege A. Health-related quality of life associated with chronic conditions in eight countries: results from the international quality of life assessment (IQOLA) project. Qual Life Res. 2004;13(2):283–298. doi: 10.1023/B:QURE.0000018472.46236.05. [DOI] [PubMed] [Google Scholar]
- 6.Boriani S, Biagini R, Iure F, Bertoni F, Malaguti MC, Di Fiore M, Zanoni A. En bloc resections of bone tumors of the thoracolumbar spine. A preliminary report on 29 patients. Spine. 1996;21(16):1927–1931. doi: 10.1097/00007632-199608150-00020. [DOI] [PubMed] [Google Scholar]
- 7.Boriani S, Iure F, Bandiera S, Campanacci L, Biagini R, Di Fiore M, Bandello L, Picci P, Bacchini P. Chondrosarcoma of the mobile spine: report on 22 cases. Spine. 2000;25(7):804–812. doi: 10.1097/00007632-200004010-00008. [DOI] [PubMed] [Google Scholar]
- 8.Boriani S, Weinstein JN, Biagini R. Primary bone tumors of the spine. Terminology and surgical staging. Spine. 1997;22(9):1036–1044. doi: 10.1097/00007632-199705010-00020. [DOI] [PubMed] [Google Scholar]
- 9.Bullinger MKI (1998) Fragebogen zum Gesundheitszustand. Hogrefe; Göttingen: Pages
- 10.Bullinger MKI, Ware JE. Der deutsche SF-36 health survey. Z Gesundheitswiss. 1995;3:21–36. [Google Scholar]
- 11.Castiglione-Gertsch M, Gelber RD, O’Neill A, Coates AS, Goldhirsch A. Systemic adjuvant treatment for premenopausal node-negative breast cancer. The international breast cancer study group. Eur J Cancer. 2000;36(4):549–550. doi: 10.1016/S0959-8049(00)00006-X. [DOI] [PubMed] [Google Scholar]
- 12.Eichholz KM, Ryken TC. Complications of revision spinal surgery. Neurosurg Focus. 2003;15(3):E1. doi: 10.3171/foc.2003.15.3.1. [DOI] [PubMed] [Google Scholar]
- 13.Enneking WF, Spanier SS, Goodman MA. A system for the surgical staging of musculoskeletal sarcoma. Clin Orthop Relat Res. 1980;153:106–120. [PubMed] [Google Scholar]
- 14.Fairbank JC, Couper J, Davies JB, O’Brien JP. The Oswestry low back pain disability questionnaire. Physiotherapy. 1980;66(8):271–273. [PubMed] [Google Scholar]
- 15.Fourney DR, Abi-Said D, Rhines LD, Walsh GL, Lang FF, McCutcheon IE, Gokaslan ZL. Simultaneous anterior-posterior approach to the thoracic and lumbar spine for the radical resection of tumors followed by reconstruction and stabilization. J Neurosurg. 2001;94(Suppl 2):232–244. doi: 10.3171/spi.2001.94.2.0232. [DOI] [PubMed] [Google Scholar]
- 16.Gokaslan ZL, York JE, Walsh GL, McCutcheon IE, Lang FF, Putnam JB, Jr, Wildrick DM, Swisher SG, Abi-Said D, Sawaya R. Transthoracic vertebrectomy for metastatic spinal tumors. J Neurosurg. 1998;89(4):599–609. doi: 10.3171/jns.1998.89.4.0599. [DOI] [PubMed] [Google Scholar]
- 17.Herr HW, Sheinfeld J, Puc HS, Heelan R, Bajorin DF, Mencel P, Bosl GJ, Motzer RJ. Surgery for a post-chemotherapy residual mass in seminoma. J Urol. 1997;157(3):860–862. doi: 10.1016/S0022-5347(01)65065-1. [DOI] [PubMed] [Google Scholar]
- 18.Krepler P, Windhager R, Bretschneider W, Toma CD, Kotz R. Total vertebrectomy for primary malignant tumours of the spine. J Bone Joint Surg Br. 2002;84(5):712–715. doi: 10.1302/0301-620X.84B5.12684. [DOI] [PubMed] [Google Scholar]
- 19.Krepler P, Windhager R, Toma CD, Kitz K, Kotz R. Dura resection in combination with en bloc spondylectomy for primary malignant tumors of the spine. Spine. 2003;28(17):E334–E338. doi: 10.1097/01.BRS.0000090504.32585.AC. [DOI] [PubMed] [Google Scholar]
- 20.Magerl F, Coscia MF. Total posterior vertebrectomy of the thoracic or lumbar spine. Clin Orthop Relat Res. 1988;232:62–69. [PubMed] [Google Scholar]
- 21.Murakami H, Kawahara N, Abdel-Wanis ME, Tomita K. Total en bloc spondylectomy. Semin Musculoskelet Radiol. 2001;5(2):189–194. doi: 10.1055/s-2001-15679. [DOI] [PubMed] [Google Scholar]
- 22.Rimoldi RL, Haye W. The use of antibiotics for wound prophylaxis in spinal surgery. Orthop Clin North Am. 1996;27(1):47–52. [PubMed] [Google Scholar]
- 23.Roy-Camille R, Saillant G, Bisserie M, Judet T, Hautefort E, Mamoudy P. [Total excision of thoracic vertebrae (author’s transl)] Rev Chir Orthop Reparatrice Appar Mot. 1981;67(3):421–430. [PubMed] [Google Scholar]
- 24.Sakaura H, Hosono N, Mukai Y, Ishii T, Yonenobu K, Yoshikawa H. Outcome of total en bloc spondylectomy for solitary metastasis of the thoracolumbar spine. J Spinal Disord Tech. 2004;17(4):297–300. doi: 10.1097/01.bsd.0000096269.75373.9b. [DOI] [PubMed] [Google Scholar]
- 25.Stener B. Complete removal of vertebrae for extirpation of tumors. A 20-year experience. Clin Orthop Relat Res. 1989;245:72–82. [PubMed] [Google Scholar]
- 26.Stener B. Total spondylectomy in chondrosarcoma arising from the seventh thoracic vertebra. J Bone Joint Surg Br. 1971;53(2):288–295. [PubMed] [Google Scholar]
- 27.Sundaresan N, Rosen G, Huvos AG, Krol G. Combined treatment of osteosarcoma of the spine. Neurosurgery. 1988;23(6):714–719. doi: 10.1097/00006123-198812000-00005. [DOI] [PubMed] [Google Scholar]
- 28.Sundaresan N, Rothman A, Manhart K, Kelliher K. Surgery for solitary metastases of the spine: rationale and results of treatment. Spine. 2002;27(16):1802–1806. doi: 10.1097/00007632-200208150-00021. [DOI] [PubMed] [Google Scholar]
- 29.Talac R, Yaszemski MJ, Currier BL, Fuchs B, Dekutoski MB, Kim CW, Sim FH. Relationship between surgical margins and local recurrence in sarcomas of the spine. Clin Orthop Relat Res. 2002;397:127–132. doi: 10.1097/00003086-200204000-00018. [DOI] [PubMed] [Google Scholar]
- 30.Tokuhashi Y, Matsuzaki H, Oda H, Oshima M, Ryu J. A revised scoring system for preoperative evaluation of metastatic spine tumor prognosis. Spine. 2005;30(19):2186–2191. doi: 10.1097/01.brs.0000180401.06919.a5. [DOI] [PubMed] [Google Scholar]
- 31.Tokuhashi Y, Matsuzaki H, Toriyama S, Kawano H, Ohsaka S. Scoring system for the preoperative evaluation of metastatic spine tumor prognosis. Spine. 1990;15(11):1110–1113. doi: 10.1097/00007632-199011010-00005. [DOI] [PubMed] [Google Scholar]
- 32.Tomita K, Kawahara N, Baba H, Tsuchiya H, Fujita T, Toribatake Y. Total en bloc spondylectomy. A new surgical technique for primary malignant vertebral tumors. Spine. 1997;22(3):324–333. doi: 10.1097/00007632-199702010-00018. [DOI] [PubMed] [Google Scholar]
- 33.Tomita K, Kawahara N, Baba H, Tsuchiya H, Nagata S, Toribatake Y. Total en bloc spondylectomy for solitary spinal metastases. Int Orthop. 1994;18(5):291–298. doi: 10.1007/BF00180229. [DOI] [PubMed] [Google Scholar]
- 34.Tomita K, Kawahara N, Kobayashi T, Yoshida A, Murakami H, Akamaru T. Surgical strategy for spinal metastases. Spine. 2001;26(3):298–306. doi: 10.1097/00007632-200102010-00016. [DOI] [PubMed] [Google Scholar]
- 35.Tomita K, Toribatake Y, Kawahara N, Ohnari H, Kose H. Total en bloc spondylectomy and circumspinal decompression for solitary spinal metastasis. Paraplegia. 1994;32(1):36–46. doi: 10.1038/sc.1994.7. [DOI] [PubMed] [Google Scholar]
- 36.Ware JE., Jr SF-36 health survey update. Spine. 2000;25(24):3130–3139. doi: 10.1097/00007632-200012150-00008. [DOI] [PubMed] [Google Scholar]
- 37.Ware JE, Jr, Sherbourne CD. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med Care. 1992;30(6):473–483. doi: 10.1097/00005650-199206000-00002. [DOI] [PubMed] [Google Scholar]
- 38.Weinstein JN, McLain RF. Primary tumors of the spine. Spine. 1987;12(9):843–851. doi: 10.1097/00007632-198711000-00004. [DOI] [PubMed] [Google Scholar]
- 39.Yao KC, Boriani S, Gokaslan ZL, Sundaresan N. En bloc spondylectomy for spinal metastases: a review of techniques. Neurosurg Focus. 2003;15(5):E6. doi: 10.3171/foc.2003.15.5.6. [DOI] [PubMed] [Google Scholar]