Abstract
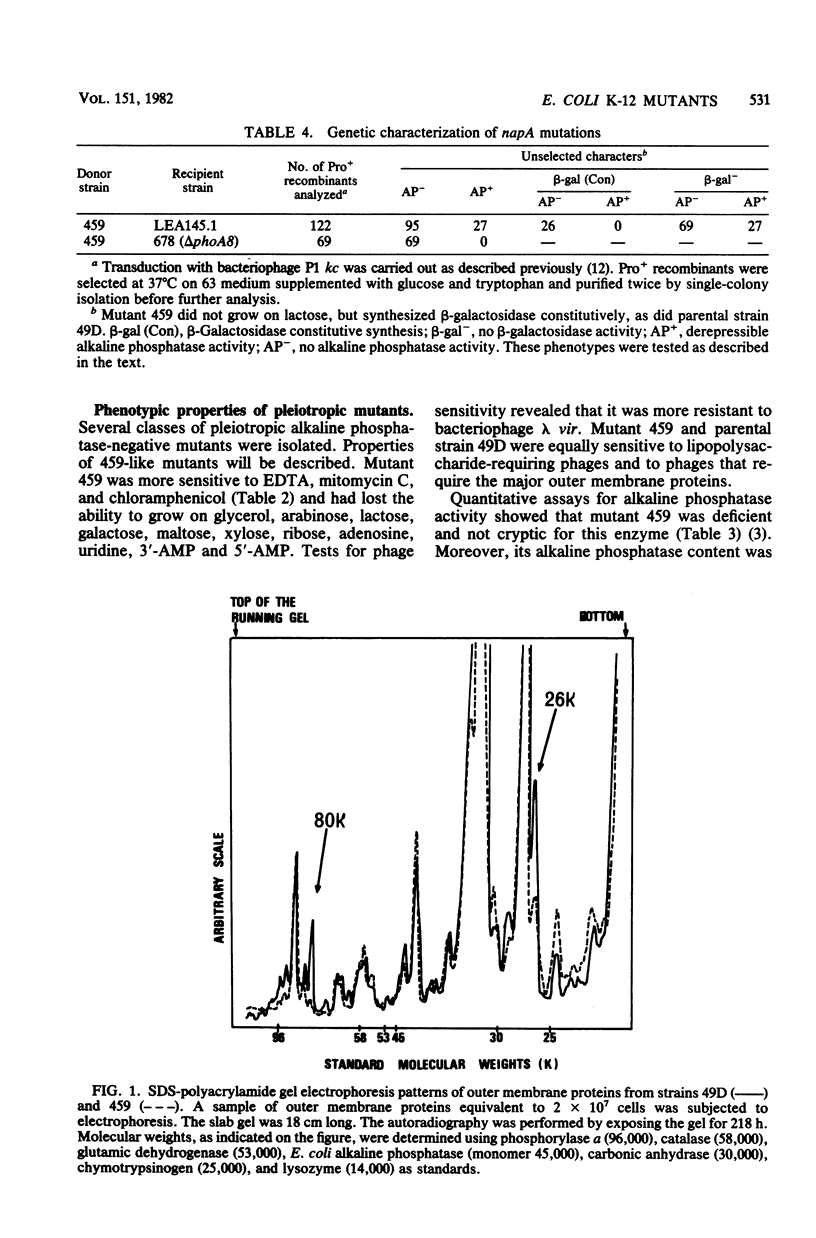
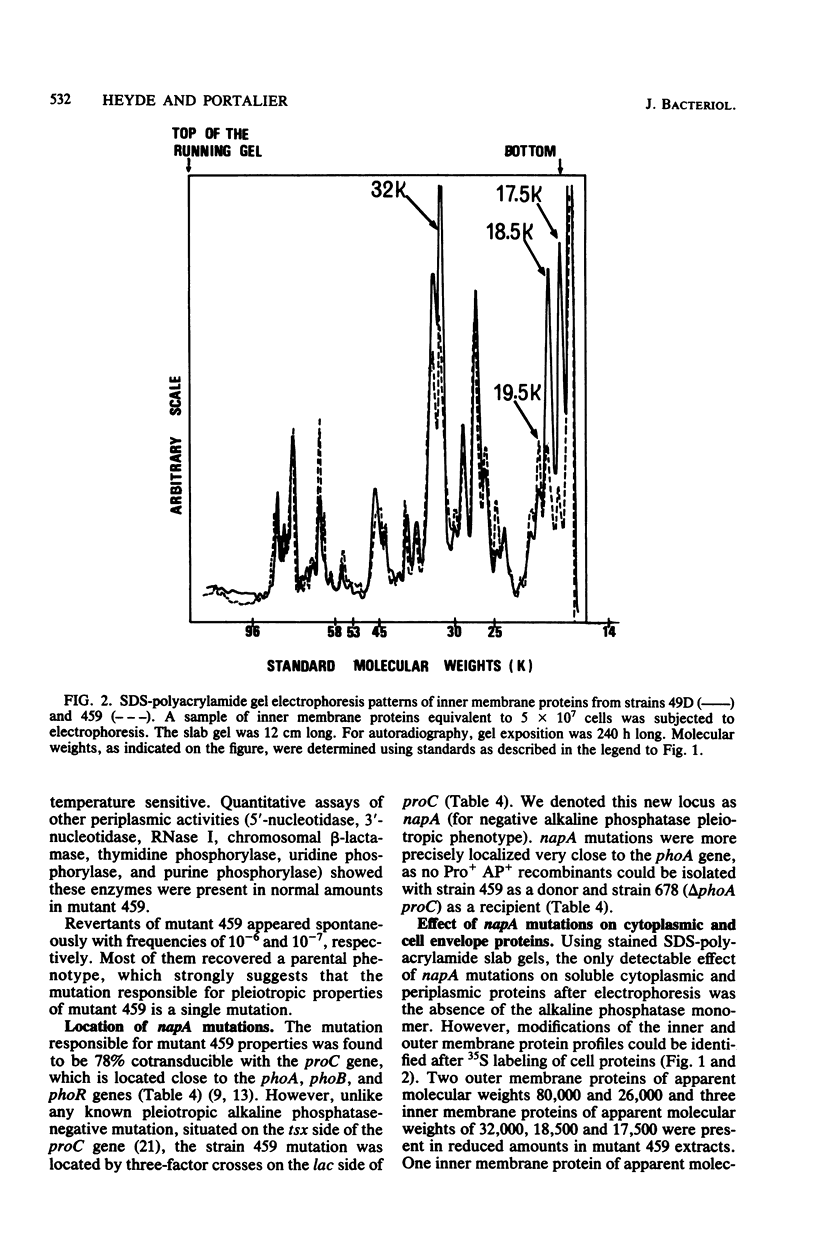
Escherichia coli K-12 mutants showing reduced alkaline phosphatase activity were isolated as 5-fluorouracil-plus-adenosine-resistant derivatives of a upp pho (either phoS or phoT) strain. One class of these mutants displayed a temperature-sensitive alkaline phosphatase-negative phenotype, a pleiotropic defect for growth on some substrates, an increased sensitivity to toxic compounds (e.g., EDTA, mitomycin, and chloramphenicol), and alterations in the expression of some membrane proteins. It phenotypically differed from previously described mutants. The mutation was located at min 8.5 close to the phoA gene and defines a new genetic locus we called napA (for negative alkaline phosphatase pleiotropic phenotype). As these mutants have lost the ability to grow on lactose and galactose, Lac+ and Gal+ revertants were isolated that simultaneously recovered the parental phenotype.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ahmad S. I., Pritchard R. H. A map of four genes specifying enzymes involved in catabolism of nucleosides and deoxynucleosides in Escherichia coli. Mol Gen Genet. 1969 Aug 15;104(4):351–359. doi: 10.1007/BF00334234. [DOI] [PubMed] [Google Scholar]
- Bachmann B. J., Low K. B. Linkage map of Escherichia coli K-12, edition 6. Microbiol Rev. 1980 Mar;44(1):1–56. doi: 10.1128/mr.44.1.1-56.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beacham I. R., Kahana R., Levy L., Yagil E. Mutants of Escherichia coli K-12 "cryptic," or deficient in 5'-nucleotidase (uridine diphosphate-sugar hydrolase) and 3'-nucleotidase (cyclic phosphodiesterase) activity. J Bacteriol. 1973 Nov;116(2):957–964. doi: 10.1128/jb.116.2.957-964.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blobel G., Dobberstein B. Transfer of proteins across membranes. I. Presence of proteolytically processed and unprocessed nascent immunoglobulin light chains on membrane-bound ribosomes of murine myeloma. J Cell Biol. 1975 Dec;67(3):835–851. doi: 10.1083/jcb.67.3.835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bracha M., Yagil E. Genetic mapping of the phoR regulator gene of alkaline phosphatase in Escherichia coli. J Gen Microbiol. 1969 Nov;59(1):77–81. doi: 10.1099/00221287-59-1-77. [DOI] [PubMed] [Google Scholar]
- Brickman E., Beckwith J. Analysis of the regulation of Escherichia coli alkaline phosphatase synthesis using deletions and phi80 transducing phages. J Mol Biol. 1975 Aug 5;96(2):307–316. doi: 10.1016/0022-2836(75)90350-2. [DOI] [PubMed] [Google Scholar]
- Hammer-Jespersen K., Munch-Petersen A., Schwartz M., Nygaard P. Induction of enzymes involed in the catabolism of deoxyribonucleosides and ribonucleosides in Escherichia coli K 12. Eur J Biochem. 1971 Apr 30;19(4):533–538. doi: 10.1111/j.1432-1033.1971.tb01345.x. [DOI] [PubMed] [Google Scholar]
- Inouye H., Beckwith J. Synthesis and processing of an Escherichia coli alkaline phosphatase precursor in vitro. Proc Natl Acad Sci U S A. 1977 Apr;74(4):1440–1444. doi: 10.1073/pnas.74.4.1440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kreuzer K., Pratt C., Torriani A. Genetic analysis of regulatory mutants of alkaline phosphatase of E. coli. Genetics. 1975 Nov;81(3):459–468. doi: 10.1093/genetics/81.3.459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lazzaroni J. C., Portalier R. C. Genetic and biochemical characterization of periplasmic-leaky mutants of Escherichia coli K-12. J Bacteriol. 1981 Mar;145(3):1351–1358. doi: 10.1128/jb.145.3.1351-1358.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakata A., Peterson G. R., Brooks E. L., Rothman F. G. Location and orientation of the phoA locus on the Escherichia coli K-12 linkage map. J Bacteriol. 1971 Sep;107(3):683–689. doi: 10.1128/jb.107.3.683-689.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Osborn M. J., Gander J. E., Parisi E., Carson J. Mechanism of assembly of the outer membrane of Salmonella typhimurium. Isolation and characterization of cytoplasmic and outer membrane. J Biol Chem. 1972 Jun 25;247(12):3962–3972. [PubMed] [Google Scholar]
- Osborn M. J., Wu H. C. Proteins of the outer membrane of gram-negative bacteria. Annu Rev Microbiol. 1980;34:369–422. doi: 10.1146/annurev.mi.34.100180.002101. [DOI] [PubMed] [Google Scholar]
- Szmelcman S., Hofnung M. Maltose transport in Escherichia coli K-12: involvement of the bacteriophage lambda receptor. J Bacteriol. 1975 Oct;124(1):112–118. doi: 10.1128/jb.124.1.112-118.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wanner B. L., Latterell P. Mutants affected in alkaline phosphatase, expression: evidence for multiple positive regulators of the phosphate regulon in Escherichia coli. Genetics. 1980 Oct;96(2):353–366. doi: 10.1093/genetics/96.2.353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wickner W. The assembly of proteins into biological membranes: The membrane trigger hypothesis. Annu Rev Biochem. 1979;48:23–45. doi: 10.1146/annurev.bi.48.070179.000323. [DOI] [PubMed] [Google Scholar]
- Worcel A., Burgi E. Properties of a membrane-attached form of the folded chromosome of Escherichia coli. J Mol Biol. 1974 Jan 5;82(1):91–105. doi: 10.1016/0022-2836(74)90576-2. [DOI] [PubMed] [Google Scholar]
- Yagil E., Bracha M., Lifshitz Y. The regulatory nature of the phoB gene for alkaline phosphatase synthesis in Escherichia coli. Mol Gen Genet. 1975;137(1):11–16. doi: 10.1007/BF00332537. [DOI] [PubMed] [Google Scholar]


