Abstract
Deep brain stimulation (DBS) of the ventral intermediate nucleus (VIM) of the thalamus improves essential tremor. Suppression of the amplitude of the postural tremor component with VIM DBS depends on stimulation frequency. The purpose of this study is to determine the effect of DBS frequency on the intention tremor component, i.e. tremor that is enhanced by target-directed movement, and to compare it to the effect of DBS frequency on postural tremor in people with essential tremor. We measured tremor frequency and amplitude during trials of postural holding and voluntary reaching between two targets at 10 different stimulation frequency settings between 0 and 185 Hz. Tremor frequency did not change with changes in stimulation frequency. Amplitude suppression of both intention and postural tremor depended on stimulation frequency. Maximal tremor reduction occurred at approximately 130 Hz for both forms of tremor. However, at optimal frequencies the percent reduction in tremor amplitude relative to the DBS OFF condition was greater for postural than for intention tremor. These results suggest that VIM DBS stimulation frequencies near 130Hz may provide maximal control of intention and postural tremor. Identification of optimal stimulation settings should consider assessment of intention tremor not just postural tremor, as intention tremor may not be as well controlled as postural tremor but may be a better gauge for functional benefit.
Keywords: essential tremor, deep brain stimulation, kinematics
Introduction
Essential tremor (ET) is characterized by both postural and kinetic tremor (Findley et al. 1993, Smaga 2003). Specifically, the kinetic component includes intention tremor, defined as tremor that increases with target-directed movements (Smaga 2003). Many individuals with essential tremor (ET) have noted remarkable reduction of tremor with deep brain stimulation of the ventral intermediate nucleus (VIM) of the thalamus (Benabid et al. 1997, Koller et al. 1997, Schuurman et al. 2000). In our clinical practice, we typically set stimulator frequency at 185 Hz, while adjusting voltage and pulse width to optimize tremor control. We have previously described that using 185 Hz stimulation frequency may not be necessary since postural tremor improves incrementally with stimulation frequencies up to 100 Hz, and frequencies above 100 Hz appear to confer little additional suppression of postural tremor for most people (Ushe et al. 2004, 2006). However, the effects of stimulation frequency on the intention tremor component of ET have yet to be determined.
The purpose of this study was to compare the effects of stimulation frequency on postural and intention tremor in people with ET. Intention tremor is particularly important because it can have a profound effect on functional activities such as feeding, shaving, and brushing one’s teeth. Therefore, control of postural tremor alone may not provide adequate functional benefit for many people. If intention tremor, like postural tremor, responds to lower stimulation frequencies then such lower frequency settings would lead to substantial prolongation of battery life thereby reducing the need for surgical battery replacement (Ushe et al. 2006). We hypothesized that intention tremor suppression, depends on stimulation frequency, with higher frequencies yielding better tremor control. We further hypothesized that the relationship between stimulation frequency and tremor amplitude would not be identical for intention and postural tremor, with intention tremor being less well-controlled than postural tremor. This hypothesis was based on our clinical observation that several ET patients on DBS therapy whose DBS frequency was dropped from 185 Hz to lower frequencies continued to have adequate postural tremor control but often reported worsening of their ability to handle their activities of daily living at home.
Methods
Subjects
Eleven subjects with clinically diagnosed ET, defined as tremor that is maximal with sustained posture and remains prominent during intentional movement (Findley et al. 1993), were recruited for this study (Table 1). Subjects with history or evidence of other neurological conditions were excluded. All subjects previously had a DBS electrode (Medtronic model 3387 lead, Medtronic Inc., Minneapolis, MN) implanted into the left VIM at Washington University in St. Louis at least 8 months prior to this study with either a Soletra or Itrel II pulse generator (Medtronic model 7426 and 7424, respectively) placed subcutaneously below the clavicle. Targeting of VIM nucleus was performed under T2-weighted MRI guidance using a Leksell stereotactic head frame. The second contact from the distal tip of the electrode was implanted along the anterior commissure/posterior commissure axial plane, 12 to 14 mm lateral to the midline or 11 mm lateral to the edge of the third ventricle, and posterior to the midcommissural point at ¼ of the distance between the anterior commissure and the posterior commissure. Using a hand held stimulator (Medtronic screener model 3628, Medtronic Inc., MN), macrostimulation was performed to confirm adequate electrode location as evidenced by right hand tremor control, lack of DBS-induced adverse effects and appearance of paresthesias in the right hand first (rather than in the face or leg). DBS voltage was increased slowly by increments of 0.1 volt up to 2.0 volts maintaining a constant pulse width of 120 microseconds and pulse frequency of 185 Hz. The electrode was moved anteriorly or posteriorly as needed if the sensory thresholds when testing the deepest contact were not between 0.5 to 1.0 volts. All subjects had good clinical response to the left VIM DBS as evidenced by the improvement in total Fahn-Tolosa-Marin Tremor Rating Scale (TRS) scores postoperatively (Table 1) (Fahn et al. 1993). Average right upper extremity score (sum of postural and intention tremor scores for the right upper extremity) improved from 2.6 ± 1.7 before to 0.3 ± 0.5 after surgery. Average activities of daily living score improved from 12.5 ± 5.3 to 3.5 ± 2.9 after surgery. The magnitude of these changes in TRS scores are in keeping with other published studies that have examined VIM stimulation for essential tremor (Ondo et al. 1998, 2001, Pahwa et al. 1999). All subjects signed an informed consent prior to participating in this research that was approved by the Human Research Protection Office of Washington University.
Table 1.
Subject Demographics and Original Stimulator Settings
| Project | Age (yr) | Sex | Time from DBS surgery (months) | Duration illness (yrs) |
Pre-Op Total TRS (max 144) |
Post-Op Total TRS (max 144) |
Familial (Yes/No) |
Stimulator Voltage (volts) |
Stimulator Pulse Width (μsec) |
Stimulator Frequency (Hz) |
Comments |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 79 | F | 63 | 39 | 61 | 20 | Y | 3.5 | 60 | 185 | Dilateral |
| 2 | 68 | M | 56 | 63 | 26 | 8 | N | 4.5 | 60 | 185 | Unilateral |
| 3 | 81 | F | 63 | 11 | 29 | 9.5 | N | 3.8 | 90 | 185 | Unilateral |
| 4 | 68 | M | 9 | 31 | 42 | 26 | N | 3.2 | 90 | 185 | Unilateral |
| 5 | 77 | F | 68 | 38 | 79 | 19 | N | 4.0 | 120 | 185 | Dilateral |
| 6 | 64 | M | 72 | 20 | 29 | 12.5 | N | 4.5 | 120 | 185 | Dilateral |
| 7 | 81 | M | 32 | 39 | 29 | 14.5 | N | 3.2 | 60 | 185 | Dilateral |
| 8 | 79 | F | 16 | 38 | 49 | 34 | N | 3.6 | 60 | 185 | Dilateral |
| 9 | 77 | M | 80 | 25 | 66 | 12.5 | N | 4.5 | 90 | 185 | Dilateral |
| 10 | 69 | M | 57 | 25 | 38 | 10.5 | Y | 3.0 | 90 | 185 | Unilateral |
| 1 | 51 | F | 8 | 21 | 44 | 17 | Y | 2.8 | 60 | 185 | Unilateral |
| Mean | 72.2 | 5F/6M | 47.6 | 31.8 | 44.6 | 16.7 | 2Y/9N | 3.7 | 81.8 | 185 | 6B/5U |
|
| |||||||||||
| D | 9.3 | 26.4 | 13.9 | 17.5 | 7.8 | 0.62 | 23.6 | 0 | |||
Measurement Equipment
Tremor was assessed using a 3-D motion capture system (Motion Analysis Corporation, Santa Rosa, CA). Reflective markers were placed on the right upper extremity to track the movement of the limb using an eight-camera digital system with a resolution of 1mm. Data were sampled at 100Hz.
Paradigm
Each subject was seated in a straight-back chair with feet flat on the floor. A strap then was placed across the torso to keep the subject firmly in place. Reflective markers were placed on bony landmarks of the right upper extremity to track movement of the entire arm from shoulder to index finger. Each subject was then tested to determine the position that maximized postural tremor of the right arm with the left stimulator turned off. The position that evoked maximal postural tremor was used to assess their postural tremor. For most individuals, this position was having the arm held straight in front of the body and the shoulder flexed to 90 degrees. Two individuals had more tremor in a saluting position (i.e., with the shoulder and elbow flexed to 90 degrees and the hand held with the palm facing downward). We assessed intention tremor by having each subject reach repeatedly between their chin and a target placed directly in front of the right shoulder at shoulder height and arm’s length from the subject. For individuals with bilateral stimulators, the right stimulator was turned off at least 15 minutes prior to testing and remained off throughout the experiment.
Postural and intention tremor were evaluated at 10 different stimulator frequency settings, namely 0, 30, 45, 60, 75, 90, 100, 130, 145, and 185 Hz. At each of these settings, three 15-second trials of postural tremor and three 15-second trials of repeated reaching to assess intention tremor were recorded. Trials alternated between postural and reaching tasks, and the block of 6 trials at each successive setting alternately began with either a postural or a reaching trial. The order of frequency settings was randomized. After each change in frequency setting there was a five minute delay prior to testing. The original stimulator settings (see Table 1) of each individual were restored at the end of the study.
Data Analysis
The excursion of the index finger was taken as an indicator of overall movement of the arm. Index finger data were first analyzed to determine the dominant frequency of tremor for each trial. A 1.5 Hz high-pass 5th order Butterworth filter was applied to all trials. This filter allowed for removal of low frequency components related to the intended movements during reaching trials. Data for the entire 15 seconds of each trial then were assessed using a Fourier transform analysis to determine dominant frequency for each trial (Datapac 2K2, Run Technologies, Mission Viejo, CA). One-way repeated measures ANOVAs with Holm-Sidak posthoc tests were used to test for differences in tremor frequency across different stimulator settings for intention and postural tremor (P < 0.05).
Tremor amplitude was assessed using average excursion of the index finger in the vertical dimension per unit time, as vertical movements were confirmed to be the largest component of the tremor for all subjects. The index finger movement was taken as an indicator of overall movement of the arm. Raw, unfiltered data were used for this analysis. For postural trials the entire 15 seconds were analyzed. For reaching trials, we analyzed only the portions of the trial at either end of each reach when the arm was not in transit to or from a target. We were thus able to capture the intention tremor at the targets without the confounding influence of the transitional movements between the two targets. This produced varying time intervals analyzed for each cycle of movement, but values were normalized per unit time for each cycle, and these values were then averaged to obtain a single value per trial. Given a stable and unchanging tremor frequency, as was demonstrated by Fourier transform, index finger excursion values are an appropriate indicator of tremor amplitude. Values for the three trials of postural or reaching tasks at each setting were averaged for each subject. All values were normalized to the 0 Hz (DBS OFF) setting, which was defined as 100%. Normalized values then were averaged across subjects for the postural and reaching tasks at each frequency setting. Stimulus frequency was plotted versus tremor amplitude (as reflected by normalized length of path). These curves for postural and reaching tasks were fitted with a 4-parameter sigmoidal curve of the form Y = y0 + a/(1 + exp(−(x−x0)/b)). These curves also were fitted with a linear function of the form Y = y0 + ax. Goodness of curve fits for the sigmoidal vs. linear functions were then compared to determine which best described each curve.
Results
Tremor frequency did not change with stimulation frequency. Dominant frequencies for intention and postural tremor were similar across the entire range of stimulation frequencies. The average dominant frequency ± SD across all conditions was 3.66 ± 0.09 Hz for intention tremor and 3.73 ± 0.09 Hz for postural tremor. There were no significant differences in frequency across stimulation frequencies for intention tremor (p = 0.73) or for postural tremor (p = 0.36).
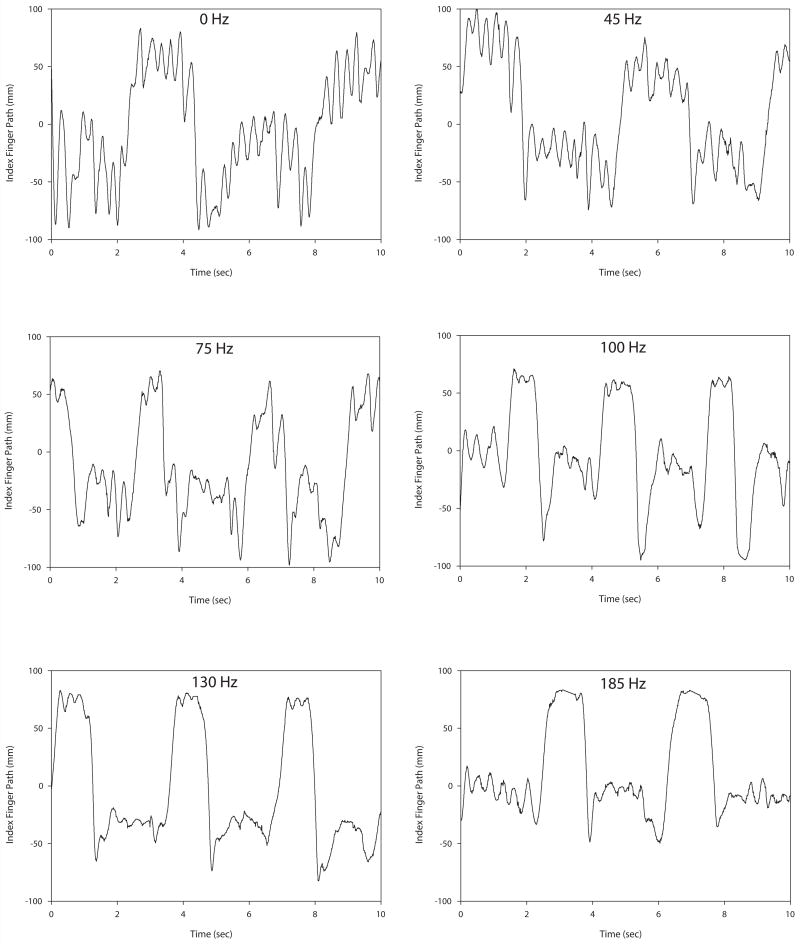
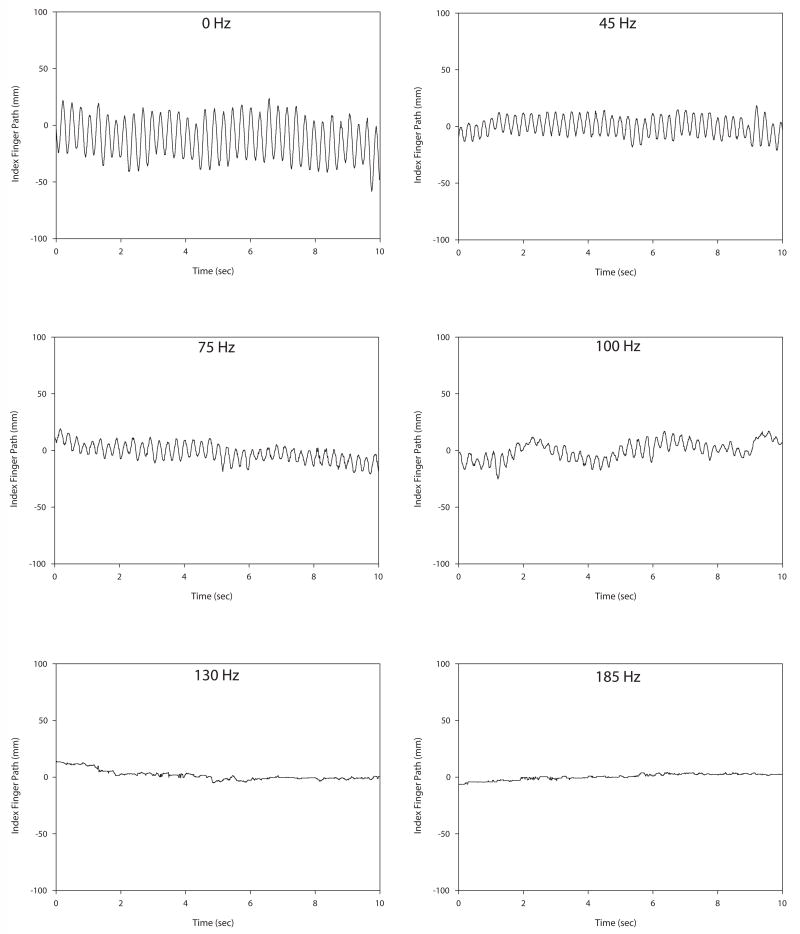
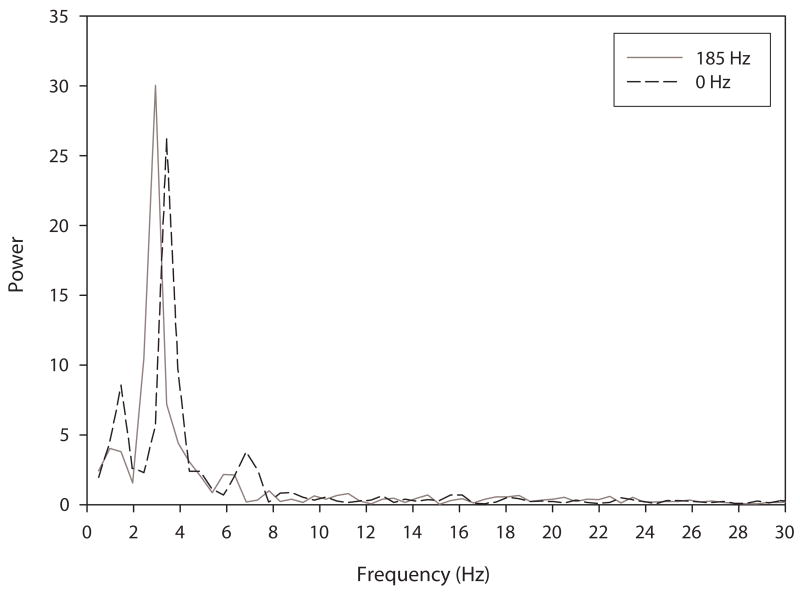
Tremor amplitude decreased with increasing stimulation frequency for both intention and postural tremor. Figure 1 illustrates intention tremor at a variety of frequency settings for a single subject. Note that tremor is most apparent during the plateaus, as there are the ends of the movement where the finger is approaching the target. (It is these plateaus that were used in the analysis.) Tremor amplitude decreased with increasing stimulation frequency up to 130 Hz. Beyond 130 Hz tremor amplitude remained relatively unchanged in this individual. Figure 2 illustrates postural tremor at a variety of frequency settings for an individual subject. Note that postural tremor amplitude decreased with increasing stimulation frequency up to 130 Hz. Beyond 130 Hz there was no additional benefit. Figure 3 shows the FFT spectra for the same 0 Hz and 185 Hz postural tremor trials shown in Figure 2. This figure confirms the lack of a substantial change in peak spectral frequency across the two extremes of frequency settings. This lack of change in peak frequency was present despite a substantial change in tremor amplitude between the 0 Hz and 185 Hz frequency settings.
Figure 1.
Illustration of intention tremor in an individual subject across six different stimulation frequencies. Tremor magnitude decreases as stimulation frequency increases up to 130 Hz, after which little additional reduction in tremor occurred in this individual. Note that tremor is most apparent at the two extremes of the reach as the subject approaches the chin or the target, but is not obvious during the movements between targets.
Figure 2.
Illustration of postural tremor in an individual subject across six different stimulation frequencies. Tremor magnitude decreases as stimulation frequency increases up to 130 Hz, after which little additional reduction in tremor occurred in this individual.
Figure 3.
FFT plots for the 0 Hz and 185 Hz postural tremor trials shown in Fig. 2. There is no substantial change in peak spectral frequency, which is in the range of 3–4 Hz for both trials, despite the substantial reduction in tremor amplitude at 185 Hz.
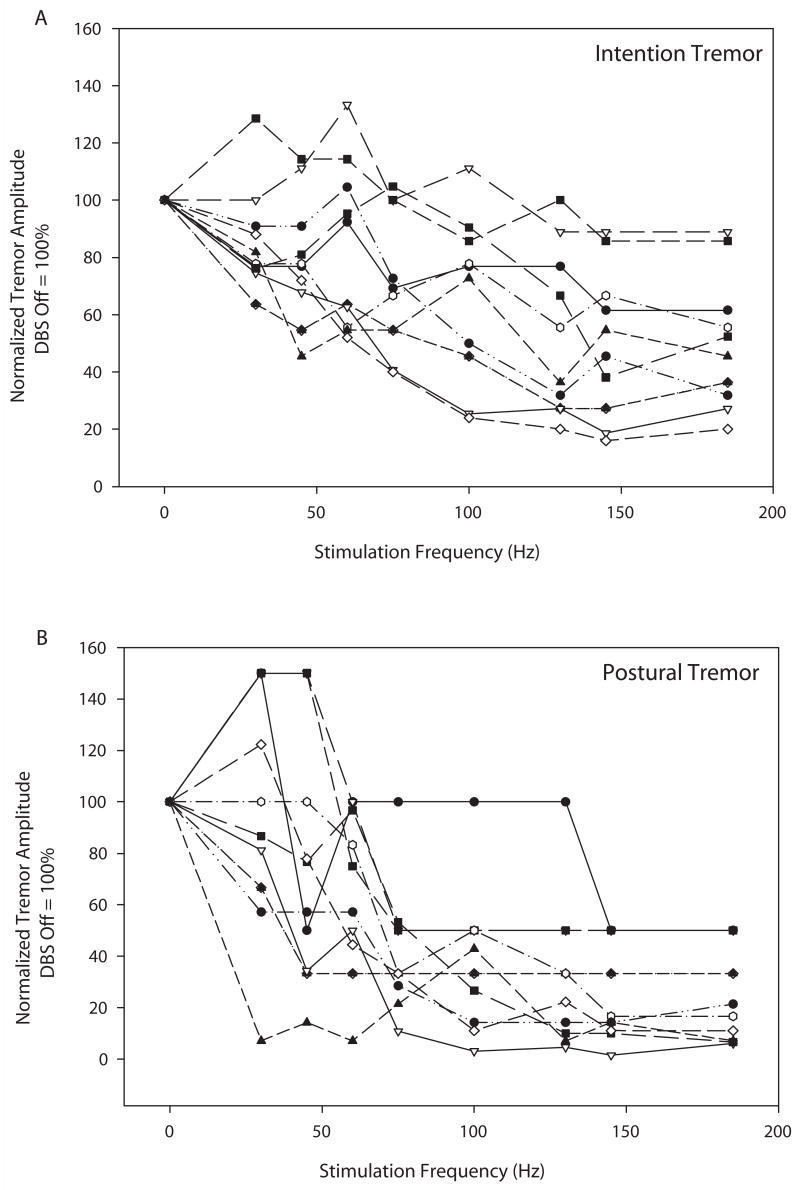
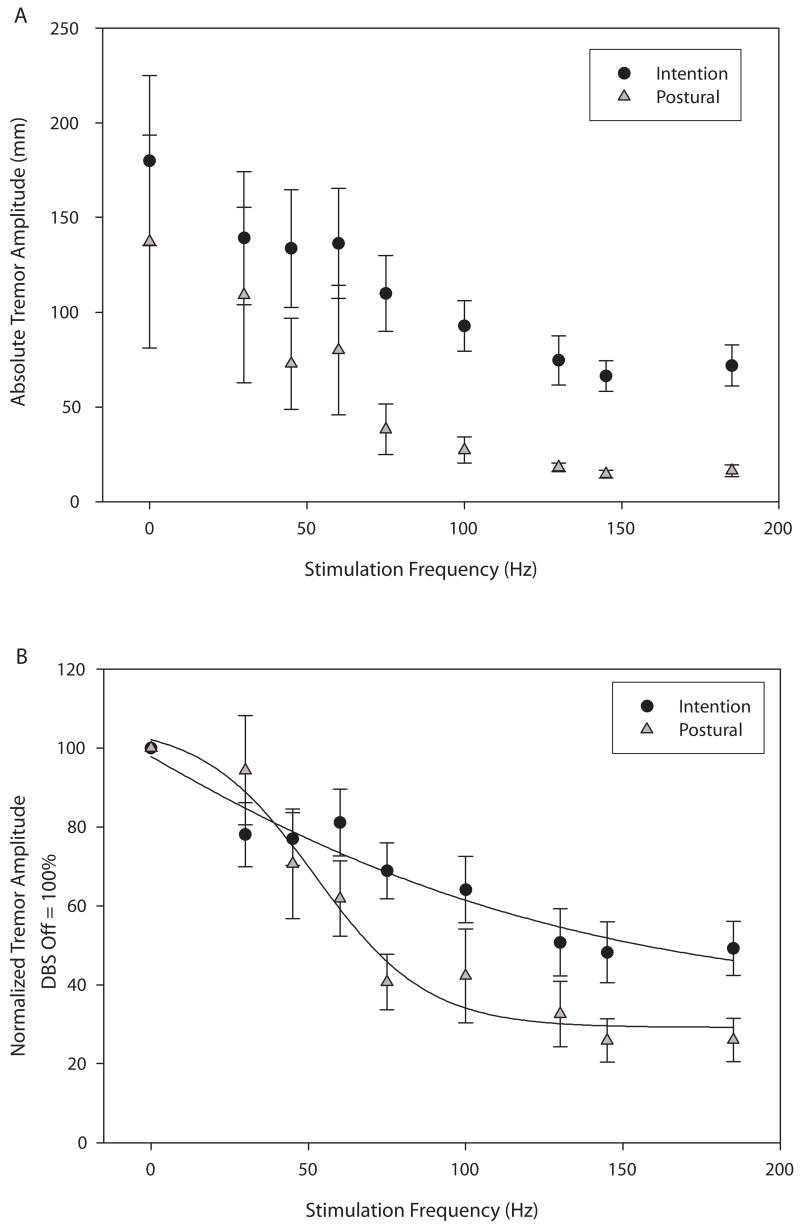
Figure 4 illustrates each individual’s tremor normalized tremor amplitude (DBS OFF = 100%) for intention (4A) and postural (4B) tremor, providing a sense of the variability of response among subjects. Figure 5 shows group average data for both absolute and normalized tremor amplitude. The absolute amplitude of intention tremor was greater than that of postural tremor (Fig. 5A) but similar decrements in tremor amplitude were noted for both forms of tremor as stimulation frequency increased. Note that these non-normalized group data show that maximal tremor reduction was reached at 130 Hz with no additional substantial decrement in either intention or postural tremor as stimulation frequency increased beyond 130 Hz. When normalized to the DBS OFF condition, the data again demonstrate a lack of substantial tremor amplitude decrement at stimulation frequencies beyond 130 Hz. Both intention and postural tremor showed inverse sigmoidal relationships between tremor amplitude and stimulation frequency (Fig. 5B). The sigmoidal relationship was more pronounced for postural than for intention tremor, but both the postural and intention curves were better fit with a 4-parameter sigmoidal curves than they were with linear curves. Adjusted R2 values for the linear vs. sigmoidal fit of the postural curve were 0.83 and 0.95, respectively. Adjusted R2 values for the linear vs. sigmoidal fit of the intention curve were 0.88 and 0.90, respectively.
Figure 4.
Illustration of individual tremor amplitudes for intention (4A) and postural (4B) tremor across stimulation frequencies. Values are normalized to the DBS OFF condition which was set at 100%.
Figure 5.
Illustration of average absolute tremor amplitudes for intention (black circles) and postural (gray triangles) tremor across stimulation frequencies (5A). Values represent the average length of path of the index finger per second. Figure 5B illustrates average normalized tremor amplitudes (DBS OFF = 100%). The intention tremor data points were best fit by the curve Y = 35.5 + 172.1/(1+exp(− (X+48.6)/− 85.8)). The postural tremor data points were best fit by the curve Y = 29.2 + 76.8/(1+exp(− (X−52.1)/− 17.8)). Graphed values are means +/− SE.
Discussion
Our results suggest that the intention and postural components of essential tremor respond similarly to changes in stimulation frequency, while dominant tremor frequency remains unchanged across stimulation frequencies. On average, VIM DBS stimulation frequencies of 130 Hz provided near maximal suppression of either intention or postural tremor amplitude. Higher stimulation frequencies of 145 and 185 Hz provided no substantial additional benefit. There was, however, variability among subjects in terms of the frequency beyond which no additional benefit occurred.
Our results suggest that a DBS frequency of approximately 130 Hz may be a reasonable starting point for programming of thalamic stimulators in the clinical treatment of essential tremor. Voltage and pulse width could then be optimized with frequency being held fixed, as suggested in DBS programming algorithms (Volkmann et al. 2006). Frequency changes could then be instituted as needed from the 130 Hz level based upon each individual’s response.
All individuals in our sample had a frequency setting of 185 Hz, as part of routine clinical care. Reduction of the frequency setting to 130 Hz, as supported by our results and previous work on postural tremor, could extend expected battery life by approximately 30% for these individuals (Ushe et al. 2004, 2006). This would lead to fewer pulse generator replacements, thereby reducing medical costs and patient risk. Although maximum benefit was, on average, obtained at 130 Hz for both intention and postural tremor, the percent reduction in tremor amplitude relative to the DBS OFF condition was greater for postural than for intention tremor. This suggests that, at least for stimulation of the VIM thalamus, intention tremor is not suppressed as much as postural tremor even at optimal stimulation frequencies. As such, when attempting to optimize DBS settings the assessment of intention tremor may be critical. Although postural tremor may be substantially reduced at a particular stimulation frequency, intention tremor may be less well-controlled by VIM stimulation and be more disabling to the individual in daily life. To date, VIM DBS remains the most commonly performed procedure for the treatment for ET despite newer studies suggesting that DBS in the area of the STN may be more effective at controlling action and intention tremor (Plaha et al. 2004, Herzog et al. 2007).
We were limited by the short delay between setting DBS frequency and the tremor sampling. Response to DBS may change with chronic stimulation as has been noted in the case of Parkinson disease subthalamic nucleus DBS (Temperli et al. 2003). Also, because this was our first investigation of intention tremor and was being conducted for research purposes outside of subjects’ routine clinical visits, we had to restore all subjects to their original setting of 185 Hz at the completion of the study. As such, we do not have long-term follow-up data regarding patient satisfaction at lower frequency settings.
Acknowledgments
We thank Michael Falvo, Shannon Goebel and Rachel Zapf for their assistance with data analysis, Joanne Wagner for her assistance in setting up the data analysis model. This work was supported by NIH grants K01 HD048437, R01 NS041509, and T32 HD007434; PODS II from the Foundation for Physical Therapy; the Greater St. Louis Chapter of the American Parkinson Disease Association (APDA); the APDA Center for Advanced PD Research at Washington University and the Barnes-Jewish Hospital Foundation (the Elliot H. Stein Family Fund and the Jack Buck Fund for PD Research).
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Benabid AL, Bebazziyz A, Pollak P. Mechanisms of deep brain stimulation. Mov Disord. 1997;17(Suppl 3):S73–S74. doi: 10.1002/mds.10145. [DOI] [PubMed] [Google Scholar]
- Fahn S, Tolosa E, Marin C. Clinical rating scale for tremor. In: Jankovic J, Tolosa E, editors. Parkinson’s disease and movement disorders. Baltimore: Williams and Wilkins; 1993. pp. 271–280. [Google Scholar]
- Findley LJ, Koller WC, de Witt P, et al. Classification and definition of tremor. In: Lord Walton., editor. Indications for and clinical implications of botulinum toxin therapy. Royal society of Medicine Services; London: 1993. pp. 22–23. Detchant. [Google Scholar]
- Herzog J, Hamel W, Wenzelburger, et al. Kinematic analysis of thalamic versus subthalamic neurostimulation in postural and intention tremor. Brain. 2007;130:1608–1625. doi: 10.1093/brain/awm077. [DOI] [PubMed] [Google Scholar]
- Koller W, Pahwa R, Busebark K, et al. High-frequency unilateral thalamic stimulation in the treatment of essential and parkinsonian tremor. Ann Neurol. 1997;42:292–299. doi: 10.1002/ana.410420304. [DOI] [PubMed] [Google Scholar]
- Ondo W, Almaguer M, Jankovic J, et al. Thalamic deep brain stimulation: comparison between unilateral and bilateral placement. Arch Neurol. 2001;58:218–222. doi: 10.1001/archneur.58.2.218. [DOI] [PubMed] [Google Scholar]
- Ondo W, Jankovic J, Schwartz K, et al. Unilateral thalamic deep brain stimulation for refractory essential tremor and Parkinson’s disease tremor. Neurology. 1998;51:1063–1069. doi: 10.1212/wnl.51.4.1063. [DOI] [PubMed] [Google Scholar]
- Pahwa R, Lyons KL, Wilkinson SB, et al. Bilateral thalamic stimulation for the treatment of essential tremor. Neurology. 1999;53:1447–1450. doi: 10.1212/wnl.53.7.1447. [DOI] [PubMed] [Google Scholar]
- Plaha P, Patel NK, Gill SS. Stimulation of the subthalamic region for essential tremor. J Neurosurg. 2004;101(1):48–54. doi: 10.3171/jns.2004.101.1.0048. [DOI] [PubMed] [Google Scholar]
- Schuurman PR, Bosch DA, Bossuyt PM, et al. A comparison of continuous thalamic stimulation and thalamotomy for suppression of severe tremor. N Eng J Med. 2000;342:461–468. doi: 10.1056/NEJM200002173420703. [DOI] [PubMed] [Google Scholar]
- Smaga S. Tremor. American Family Physician. 2003;68:1545–1552. [PubMed] [Google Scholar]
- Temperli P, Ghika J, Villemure JG, et al. How do parkinsonian signs return after discontinuation of subthalamic DBS? Neurol. 2003;60:78–81. doi: 10.1212/wnl.60.1.78. [DOI] [PubMed] [Google Scholar]
- Ushe M, Mink JW, Revilla FJ, et al. Effect of stimulation frequency on tremor suppression in essential tremor. Mov Disord. 2004;19:1163–1168. doi: 10.1002/mds.20231. [DOI] [PubMed] [Google Scholar]
- Ushe M, Mink JW, Tabbal SD, et al. Postural tremor suppression is dependent on thalamic stimulation frequency. Mov Disord. 2006;21:1290–1292. doi: 10.1002/mds.20926. [DOI] [PubMed] [Google Scholar]
- Volkmann J, Moro E, Pahwa R. Basic algorithms for the programming of deep brain stimulation in Parkinson’s disease. Mov Disord. 2006;21(Suppl 14):S284–289. doi: 10.1002/mds.20961. [DOI] [PubMed] [Google Scholar]