Abstract
PURPOSE Guidelines encourage primary care clinicians to document smoking status when obtaining patients’ blood pressure, temperature, and pulse rate (vital signs), but whether this practice promotes cessation counseling is unclear. We examined whether the vital sign intervention influences patient-reported frequency and intensity of tobacco cessation counseling.
METHODS This study was a cluster-randomized, controlled trial conducted in the Virginia Ambulatory Care Outcomes Research Network (ACORN). At intervention practices, nurses and medical assistants were instructed to assess the tobacco use status of every adult patient and record it with the traditional vital signs. Control practices did not use any systematic tobacco screening or identification system. Outcomes were the proportion of smokers reporting clinician counseling of any kind and the frequency of 2 counseling subcomponents: simple quit advice and more intensive discussion.
RESULTS A total of 6,729 adult patients (1,149 smokers) at 18 primary care practices completed exit questionnaires during a 6-month comparison period. Among 561 smokers at intervention practices, 61.9% reported receiving any counseling, compared with 53.4% of the 588 smokers at control practices, for a difference of 8.6% (P = .04). The effect was largely restricted to simple advice, which was reported by 59.9% of intervention patients and 51.5% of control patients (P=.04). There was no significant increase in more extensive discussion, with 32.5% and 29.3% of patients at intervention and control practices, respectively, reporting this type of counseling (P=.18).
CONCLUSIONS The vital sign intervention promotes tobacco counseling at primary care practices through a modest increase in simple advice to quit. When implemented as a stand-alone intervention, it does not appear to increase intensive counseling.
Keywords: Smoking, tobacco, smoking cessation, vital signs, vital sign intervention, health behavior, counseling, primary care, office visits, practice-based research
INTRODUCTION
Few interventions are more important to public health than promoting cessation of tobacco use.1 Tobacco use is the leading cause of death in the United States, claiming an estimated 440,000 lives per year.2,3 Health professionals play a key role in promoting tobacco cessation: smokers cite physician advice as a major determinant in quitting,4,5 and even simple advice from physicians has been shown to significantly increase abstinence rates.6 If clinicians helped as few as 10% of smokers to quit, 3.5 million smokers would become tobacco free each year.7
In 1996, the US Department of Health and Human Services (DHHS) issued formal guidelines urging primary care clinicians to adopt officewide systems, such as the vital sign intervention, to assess smoking status systematically.8 This recommendation was reiterated and updated in 2000,9 and it was incorporated by the National Committee for Quality Assurance into its Health Plan Employer Data and Information Set (HEDIS). A survey in 2000 showed that 43% of health plans required clinicians to record smoking status as part of patients’ vital signs (blood pressure, temperature, and pulse rate).10
Scientific evidence to support this recommendation was limited. The DHHS guideline based its recommendation on 3 studies11–13 reporting increased counseling at practices that adopted the vital sign intervention, but these studies relied on before-and-after designs, were subject to confounding and limited generalizability, or both. Studies published after the release of the DHHS guidelines found that the vital sign intervention did not increase subsequent counseling.14–17
Well-designed studies are needed both to confirm whether the vital sign intervention increases counseling and to clarify the intensity of any resultant counseling as defined by the “5As” framework (Table 1 ▶). Because simple advice to quit (A2) is less effective in promoting abstinence than more intensive counseling (A3–5),18–21 knowing whether the vital sign intervention increases not only A2 but also A3–5 is essential. We therefore conducted a cluster-randomized, controlled trial of the vital sign intervention comparing patient-reported rates of tobacco cessation counseling at practices that asked each adult patient about tobacco use when measuring vital signs (intervention) and at practices that continued their usual routine (control). We examined not only the overall counseling rates (A2–5) but also the intensity of counseling (A2 vs A3–5).
Table 1.
The 5A Counseling Framework Recommended in the US Department of Health and Human Services Practice Guideline on Smoking Cessation Counseling9
| A | Activity | Description |
| A1 | Ask | Identify and document tobacco use status for every patient at every visit |
| A2 | Advise | In a clear, strong, and personalized manner, urge every tobacco user to quit |
| A3 | Assess | Determine whether the tobacco user is willing to make a quit attempt at this time |
| A4 | Assist | For the patient willing to make a quit attempt, use counseling and pharmacotherapy to help him or her quit |
| A5 | Arrange | Schedule follow-up contact, in person or by telephone, preferably within the first week after the quit date |
METHODS
This study was approved by the institutional review boards of Virginia Commonwealth University and Bon Secours Richmond Health System.
Setting
Eighteen practices were recruited as research sites under the auspices of the Virginia Ambulatory Care Outcomes Research Network (ACORN), a practice-based research network.22 We initially identified 84 practices within a 25-mile radius of downtown Richmond, Virginia, with at least 1 general internal medicine or family medicine physician on a list of current and former community preceptors of first- or second-year Virginia Commonwealth University medical students. Practices with the full-time equivalent of at least 2 clinicians (family physicians, general internists, nurse-practitioners, or physician’s assistants) who specialized in primary care and who provided care to adult patients (wholly or in part) were eligible. We included 1 practice with several primary care clinicians and 1 non–primary care clinician, but excluded the specialist clinician from the study. We excluded residency programs, clinics serving special populations (eg, urgent care centers, indigent/free clinics, student health centers), practices not under the auspices of the overseeing institutional review boards (see above), or practices with an existing systematic tobacco identification and reminder system. The recruitment process entailed a letter of invitation from the principal investigator (S.F.R.) followed by telephone calls to lead physicians and meetings with some office managers and head nurses (A.E.B.). Practices were offered $500 for participation and an additional $1,000 if assigned to the intervention group.
Data Collection
Counseling rates were determined by patient self-report: exit questionnaires administered at the conclusion of their office visit asked adult patients to describe the counseling they had just received. Trained research assistants (RAs) rotating among practices distributed these questionnaires in person. In advance, the project coordinator asked practices to identify potential visit dates during the study period when at least 2 clinicians would be seeing patients but did not reveal on which of these suggested dates RAs would be on site. The number of days that RAs visited each practice was periodically adjusted to balance the number of smokers surveyed at each practice.
RAs attempted to approach each adult patient as he or she exited the clinic area. Patients were eligible if they verbally confirmed that they were aged 18 years or older and had seen a clinician (physician, nurse-practitioner, or physician’s assistant) that day. The RA invited patients to complete a self-administered questionnaire, providing limited assistance with uncompleted entries and offering no cessation counseling. Informed consent was obtained through an RA statement and a written preamble preceding the questionnaire.
The 10-item questionnaire was designed to obtain patients’ perspective on whether A1–5 had occurred. Although we created individual questions for A1 (Did a nurse or doctor ask you today if you smoke?) and A2 (If you smoke, did your doctor advise you today to stop smoking?), adapted from previous instruments,23,24 we produced a composite question for A3–5 (If you smoke, did your doctor talk with you today about ideas or plans to help you quit smoking?) out of concern that too lengthy a questionnaire would compromise patient participation. The questionnaire included additional questions about patient age and sex, smoking status, counseling about other health behaviors, and the nature of the visit and clinician.
Although not part of the data for this study’s primary outcomes, additional data were obtained from patients who indicated that they were current smokers and agreed to take home a more detailed postal questionnaire to complete and mail back. The postal questionnaire asked in greater detail about counseling activities at the visit and about patient and visit factors associated with cessation counseling. Further details about the postal questionnaire and its results will be published separately.
Preintervention Period and Randomization
Before conducting the intervention, we determined each practice’s baseline rate of providing cessation counseling (A2–5) by surveying a cross-sectional sample of visiting smokers. This preintervention questionnaire was fielded during a 6-week period (November 2003 to January 2004) to obtain the data needed to control for practice-level variation in our block randomization (see “Data Analysis”). We divided the 18 practices into 5 matched groups with similar preintervention counseling rates by applying a centroid hierarchical clustering method,25 modified to require 2 or more practices within each group. We used Microsoft Excel (Microsoft Corp, Redmond, Washington) and a random number generator to randomize practices within the matched groups to the intervention or control condition. Allocation was concealed; once it was determined by the program, investigators informed practices which condition they had been assigned to. The performance of the intervention was evident to clinicians, patients, and investigators, and in this regard the trial was unblinded.
Intervention
During a 3-week period preceding the launch of the intervention, the project coordinator (A.E.B.) conducted a 1-hour training session at intervention practices on how to implement the vital sign procedure. We invited all nurses and medical assistants responsible for processing arriving patients and escorting them to examination rooms (rooming staff); most office managers and some practice medical directors and physicians also attended the sessions. The trainer gave rooming staff a vital sign rubber stamp (Figure 1 ▶) and a description of the study, and instructed them to modify their procedures in 3 ways: (1) stamp the encounter/progress notes where other vital signs were normally recorded, (2) ask every adult patient at every visit whether they used tobacco, and (3) record the answer within the stamped imprint. One intervention practice did not use the rubber stamp but opted to print new progress notes with a modified vital sign format. The intervention did not address the counseling practices of staff or clinicians.
Figure 1.

Imprint on medical record produced by vital sign stamp.
No interventions were undertaken at control practices. In addition, we requested that control practices not adopt a systematic tobacco identification or reminder system during the comparison period.
Comparison Period
The comparison period lasted 6 months (February through August 2004) and targeted a new cross-sectional sample of patients. On the first day of the intervention, we faxed to practices a boldly printed “first day” notice for distribution to rooming staff as a reminder about the protocol. To reinforce continued performance of the intervention, we faxed reports at 6 weeks, 3 months, and 5 months to the office manager at intervention sites, giving only the frequency of patient-reported performance of the intervention. One intervention practice experienced a 2-month delay in launching the intervention.
Power Analysis
We accounted for both between-practice variation and variation among patients within practices. Because the standard deviation for between-practice variation in the delivery of A2–5 was not available in the literature, we constructed estimates using quit advice data (A2) collected over several years by a large health maintenance organization. We estimated variation for control and intervention practices, respectively, by examining their data before and after the distribution of a smoking cessation guideline. The estimated standard deviation for between-practice variation in A2 was 10% to 12% and 12% to 14%, respectively, for intervention practices and control practices. We reasoned that because A2 is the most frequently delivered subcomponent of counseling,11,13,26–28 its standard deviation would reasonably approximate variation rates for counseling in general. For control practices, we assumed a counseling rate of 50% and a between-practice standard deviation of 14%. Given these assumptions, the intra-cluster correlation coefficient is 0.073. To estimate points on the power curve, we simulated a sufficient number of 2-stage samples, using the estimated parameters and varying effect sizes, to achieve a margin of error of 1% or less. We derived the power curve from the proportion of simulations in which our primary hypothesis would be supported at the .05 level (1-sided test). Our simulation predicted 80% power to detect a 12% effect size if we enrolled 18 practices and surveyed 27 preintervention period smokers and 81 comparison period smokers per practice.
Data Analysis
The main outcome measure (A2–5) was counted when patients gave affirmative responses to the question addressing A2, the question addressing A3–5, or both. The unit of analysis was the practice, because practices rather than patients or clinicians were randomized. We used an intention-to-treat analysis; at the practice that experienced a 2-month delay in implementing the intervention, data collected during the delay were included as comparison period data. We excluded data from 2 surveyed patients (1 from each study arm) because their 2 clinicians were not represented in both the preintervention and comparison periods.
The number of smokers surveyed at each practice was not uniformly distributed over time, which could have resulted in bias from temporal trends. To account for this possibility, we weighted the observations within each practice over the comparison period so that the cumulative sampling volume had a constant slope over time. As we also intended to incorporate preintervention counseling rates in the analysis and recognized that the survey volumes attributed to each clinician within practices might differ between the preintervention and comparison periods, we weighted the preintervention data to match the proportions observed in the comparison period.
Using a nested, hierarchical logistic regression model,29 we accounted for variation among practices, variation among clinicians within practices, and variation among a clinician’s patients. The model’s independent variables were group assignment, the logit of the weighted preintervention counseling rate, and the matched group. We used the sandwich variance estimator,29 which is robust against heteroscedasticity and overdispersion (intracluster correlation). Statistical significance was determined by computing the 1-sided P value associated with the coefficient of the treatment group independent variable. We used SAS version 9.1 (SAS Institute Inc, Cary, North Carolina, 2002–2003) for power calculations and summary statistics. We used SUDAAN version 8.0 (Research Triangle Institute, Research Triangle Park, NC, 2001) for statistical significance testing.
RESULTS
The 18 eligible, participating practices included 14 family medicine practices, 3 internal medicine practices, and 1 practice featuring both specialties. Two practices were urban, 2 were rural, and 14 were suburban. Only 1 practice used electronic medical records.
Preintervention Period
Counseling rates during the preintervention period were determined by administration of the exit questionnaire for 6 weeks to 2,145 patients (384 smokers) at the 18 practices. Counseling rates (the proportion of office visits in which A2–5 was delivered) fell into 5 ranges: 33% to 38% (5 practices), 50% to 53% (3 practices), 56% to 60% (3 practices), 62% to 65% (5 practices), and 71% to 73% (2 practices). These 5 ranges defined the matched groups.
The randomization procedure assigned 9 practices to the vital sign intervention, and there was no attrition of intervention or control practices before or after randomization (Figure 2 ▶). The 1 practice with an electronic medical record system was randomized to the control group. Preintervention survey data revealed no statistically significant differences between the intervention and control practices with regard to practice size, clinician or patient characteristics, and frequency of tobacco counseling activities before implementing the intervention, with the exception that intervention patients were an average of 2.4 years younger than control patients (P = .001) (Table 2 ▶).
Figure 2.
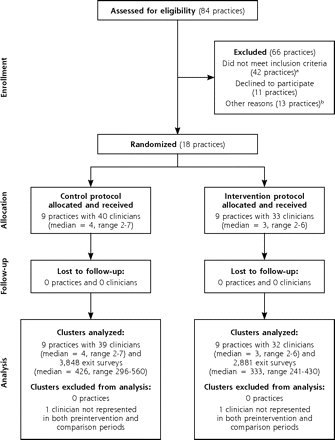
Flow diagram of the study.
Table 2.
Characteristics of Study Groups Surveyed During the Preintervention Period
| Characteristic | Control Group | Intervention Group | P Valuea |
| Practices and clinicians | |||
| Number of practices | 9 | 9 | |
| Number of clinicians per practice, median (range) | 4 (2–7) | 3 (2–6) | .28b |
| Number of clinicians | 40 | 32 | |
| Sex (male), % | 63 | 76 | .31 |
| Type of clinician, % | .30 | ||
| Family medicine physicians | 60 | 76 | |
| Internal medicine physicians | 20 | 18 | |
| Nurse-practitioners | 15 | 6 | |
| Physician’s assistants | 5 | 0 | |
| Years since licensure, % | .48 | ||
| 1–5 | 15 | 12 | |
| 6–10 | 18 | 18 | |
| 11–20 | 38 | 21 | |
| 21–30 | 25 | 42 | |
| >30 | 5 | 6 | |
| Race, % | .59 | ||
| White | 88 | 79 | |
| African American | 0 | 3 | |
| Asian | 3 | 6 | |
| Unknown | 10 | 12 | |
| Patients | |||
| Number of patients | 1,228 | 917 | |
| Age, mean (range), years | 53.0 (19–95) | 50.6 (19–95) | .001c |
| Sex (female), % | 60.3 | 60.0 | .86 |
| Visit for general checkup, % | 57.5 | 53.3 | .06 |
| Current smoker, % | 16.7 | 19.6 | .09 |
| Received cessation counseling (5A’s), % | |||
| Asked if smoking (A1) | 30.9 | 27.2 | .07 |
| Counseled to quit (A2–5)d | 52.2 | 55.9 | .47 |
| Given simple advice (A2)d | 50.7 | 53.1 | .68 |
| Had more intensive discussion (A3–5)d | 27.8 | 33.5 | .27 |
5As = ask (A1), advise (A2), assess (A3), assist (A4), and arrange (A5).
aAll proportions were tested with the Fisher exact test, except where otherwise noted.
b Kruskal-Wallis test.
c Two-sample independent t test.
d Denominator was restricted to smokers.
Comparison Period
During the comparison period, RAs spent 6 to 23 days (median, 14 days) at each practice. On the basis of the reported number of patients making visits (which did not differentiate between pediatric and adult visits), we determined that RAs approached 69% of patients (and thus a larger proportion of adults). Among potentially eligible patients at the 18 practices, 16.8% did not participate because they refused, were too ill, had language or vision difficulties, were designated by receptionists as inappropriate participants, or did not answer study-related questions (ie, smoking status, receipt of cessation counseling, and clinician seen). Participation rates did not differ between study groups. No adverse events were reported during the study.
Initial Comparisons
Across the 18 practices, 6,729 patients (1,149 smokers), including 3,848 patients (588 smokers) from control practices and 2,881 patients (561 smokers) from intervention practices, completed exit questionnaires during the comparison period. Respondent age and sex, the prevalence of current smoking, and the proportion of visits for general checkups did not differ significantly from those reported in the preintervention surveys.
The adjusted proportion of all patients who answered the question addressing A1 affirmatively was significantly higher in intervention practices than in control practices (66.0% vs 26.3%, P <.001) (Table 3 ▶), providing indirect confirmatory evidence that the vital sign intervention was delivered at intervention practices. The adjusted proportion of smokers answering the A1 question affirmatively was higher in intervention practices as well (79.5% vs 49.4%, P <.001).
Table 3.
Outcomes of the Intervention, Stratified by Counseling Activity (5A Framework)
| Adjusted Affirmative Response Rate | |||||||
| Counseling Activity | Study Outcome | Question on Exit Questionnaire | ICCa | Control Group, %b | Intervention Group, %c | Difference, % (95% CId) | P Value |
| Ask (A1) | Proxy for intervention | “Did a nurse or doctor ask you today if you smoke?” | |||||
| All patients | 0.040 | 26.3 | 66.0 | 39.7 (35.5 to ∞) | <.001 | ||
| Smokers | 0.065 | 49.4 | 79.5 | 30.1 (24.0 to ∞) | <.001 | ||
| Nonsmokers | 0.045 | 22.7 | 62.8 | 40.1 (35.3 to ∞) | <.001 | ||
| Counseling (A2–5) | Main outcomee | (Either or both of below.) | 0.063 | 53.4 | 61.9 | 8.6 (0.8 to ∞) | .04 |
| Advice (A2) | Secondary outcome | “If you smoke, did your doctor advise you today to stop smoking?” | 0.064 | 51.5 | 59.9 | 8.4 (0.7 to ∞) | .04 |
| Discussion (A3–5) | Secondary outcome | “If you smoke, did your doctor talk with you today about ideas or plans to help you quit smoking?” | 0.061 | 29.3 | 32.5 | 3.3 (–2.9 to ∞) | .18 |
5As = ask (A1), advise (A2), assess (A3), assist (A4), and arrange (A5); ICC = intracluster correlation coefficient; CI = confidence interval.
a Estimate of the intracluster correlation.
b Nine practices with denominators of n = 3,848 patients for A1 and n = 588 smokers for A2–5.
c Nine practices with denominators of n = 2,881 patients for A1 and n = 561 smokers for A2–5.
d The 95% lower confidence interval.
e Defined as an affirmative response to the question addressing A2, the question addressing A3–5, or both.
Primary Outcome: Smoking Cessation Counseling of Any Intensity (A2–5)
Patients at intervention practices were more likely to report they had received some form of smoking cessation counseling (A2–5) during their visit, whether it was simple advice, a more extensive discussion, or both. The adjusted proportion of patients reporting such counseling was higher in the intervention group than in the control group (61.9% vs 53.4%, P = .04), with a difference of 8.6% (Table 3 ▶).
Secondary Outcomes: Counseling Subcomponents (A2 vs A3–5)
The effect of the vital sign intervention on counseling was largely restricted to the delivery of simple advice (A2). Analyzed by cluster, the adjusted proportion of patients receiving simple advice was significantly greater in the intervention group than in the control group, by 8.4% (59.9% vs 51.5%, P = .04); in contrast, the proportion of patients reporting more extensive discussion (A3–5) did not differ significantly (32.5% vs 29.3%, P = .18) (Table 3 ▶).
DISCUSSION
We found that the vital sign intervention improves the frequency of smoking cessation counseling in primary care practices. We observed an 8.6% proportional increase in counseling that consisted mainly of simple advice to quit (A2), with little additional discussion of how to do so (A3–5). Six months after the study ended, 4 of the 9 intervention sites reported continuing the vital sign assessment.
A key finding of our study—that most of the observed increase in counseling involved the delivery of simple advice—has important relevance to public health. A Cochrane meta-analysis concluded that 1 of 40 patients who receive simple advice will quit.30 On the basis of this effect size, the smoking rates and magnitude of increased cessation advice observed in our study, and an estimated 5,000 visits per clinician per year, we estimate that a practice of 4 clinicians could expect to gain an additional exsmoker every 6 weeks by adopting the vital sign intervention.
Seven prior studies of the vital sign intervention11–17 yielded inconsistent findings with regard to the frequency and intensity of subsequent cessation counseling (Table 4 ▶). The only other randomized trial of the vital sign intervention reported no change or a decrease in cessation advice and assistance.14 Our study may have greater external validity (generalizability) than prior research because we included more practices (18 vs 1–7) and excluded residencies. The internal validity of our study is enhanced by its randomized design and its rigorous approach to data collection and analysis.
Table 4.
Summary of Previously Published Studies of the Vital Sign Intervention
| Study | |||||||||
| Feature | Robinson et al12 (1995) | Fiore et al11 (1995) | Ahluwalia et al13 (1999) | Piper et al14 (2003) | Boyle and Solberg15 (2004) | Milch et al16 (2004) | Maizlish et al17 (2006) | ||
| Setting | 1 family practice residency training site | 1 academic internal medicine clinic | 1 inner-city residency walk-in clinic | 5 clinics | 2 clinics | 5 teams in 1 hospital-based primary care practice | 7 community health centers | ||
| Design | Before and after | Before and after | Intervention and control alternating every2 weeks | Randomization of practices to intervention or control | Phone survey before and after institution of vital sign measure | Nonrandom allocation of 2 teams to routine vital signs or enhanced vital signs; 3 teams as controls | Before and after | ||
| Number of participants (smokers) | 637 (179) | 1,864 (254) | 2,811 (883) | 9,439 (1,611) | 4,667 (332) | 3,063 (644) | 1,571 (≈267)a | ||
| Ask (A1) | |||||||||
| Measure | – | Patients’ report of physician asking if they smoke | Patients’ report of physician asking if they smoked cigarettes | Patients’ report of physician asking if they smoke | Visits with chart evidence of tobacco use documentation | Medical record documentation of smoking status | Medical record documentation of smoking status | ||
| Changeb | – | 25.5% vs 52.6%c | 45.6% vs 78.4%c | 24.0% vs 41.2%c | 38.0% vs 78.4%c | 49% vs 86%c,d | 57% vs 85%c | ||
| Any counseling (A2–5) | |||||||||
| Measure | Visits in which physician discussed smoking | – | – | – | Visits with chart evidence of tobacco use documentation | Chart evidence of advising, quitting, setting quit date, referral or pharmacotherapy | – | ||
| Changeb | 47% vs 86%c | – | – | – | 33.5% vs 18.8%c | 30% vs 38%d | – | ||
| Advise (A2) | |||||||||
| Measure | Visits in which physician advised quitting | Smokers’ report of physician advice to quit | Smokers’ report of physician telling them to quit | Smokers’ report of physician advice to quit | Smokers’ report of advice to quit | – | Medical record documentation of advice to quit | ||
| Changeb | 50% vs 80%c | 48.8% vs 69.8%c | 26.9% vs 39.9%c | 60.0% vs 37.1%c | 66.3% vs 66.5% | – | 26% vs 26% | ||
| Further discussion (A3–5) | |||||||||
| Measure | Smokers’ report of physician advice on how to quit | – | – | – | – | – | – | ||
| Changeb | 23.8% vs 42.6%c | – | – | – | – | – | – | ||
| Assist (A4) | |||||||||
| Measure | – | – | Smokers’ report of physician helping to set quit date | Smokers’ report of physician assistance with how to quit | Smokers’ report of physician helping to set quit date | Smokers’ report of NRT prescription | – | – | – |
| Changeb | – | – | <1% vs <1% | 2.8% vs 4.8%c | 4.4% vs 1.5%c | 8.5% vs 1.9%c | – | – | – |
| Arrange (A5) | |||||||||
| Measure | – | – | Smokers’ report that physician arranged follow-up | – | – | – | – | ||
| Changeb | – | – | 6.2% vs 12.3%c | – | – | – | – | ||
NRT = nicotine replacement therapy.
Note: For cells containing a dash (–), there were no equivalent measures or data.
a Exact number of smokers in baseline sample was not reported.
b Control vs intervention.
c Change was statistically significant.
d Control vs routine vital sign. Change was larger for control vs enhanced vital sign arm of the study.
Our trial has several limitations. First, as in most prior studies, our outcome measure was counseling rather than smoking cessation, although extensive evidence establishes the close linkage between the two.9 Second, as in most prior work, we relied on patient report of counseling rather than direct observation (eg, audiotaping), because the latter was too expensive and intrusive. Third, a Hawthorne effect was possible if clinicians knew RAs were at the practice, although such knowledge was less likely given that the visits were unannounced. Fourth, we measured effects for only 6 months and cannot predict sustainability over longer periods. Fifth, office managers at intervention practices received feedback on the rooming staff’s performance of the study intervention; although this feedback is unlikely to have affected the clinicians’ counseling behavior, it might limit generalizability to practices without such reinforcement. Intervention practices also received $1,000 more than did control practices, but this amount is unlikely to explain sustained performance of the intervention for 6 months beyond the study period. Finally, although almost 80% of smokers reported being asked if they smoke, better performance of this intervention, perhaps through different training than the 1-hour session we used, may have resulted in higher counseling rates.
We conclude that inquiring about smoking status as a routine vital sign in primary care practices should modestly enhance cessation when implemented as a stand-alone intervention. Reminder systems, by themselves, are easy for practices to implement. Our trial confirms data reported by others,13,14 however, that this reminder system alone does not translate into more frequent intensive counseling within the practice. To offer this level of assistance, practices and the health plans within which they operate must establish additional systems to overcome barriers to the delivery of smoking cessation counseling and to ensure that patients receive the intensive interventions needed to effect tobacco cessation. They must augment the identification of smokers—the focus of this study—by redesigning management systems, modifying reimbursement algorithms, and forging alliances with community resources to overcome these barriers.
Acknowledgments
Dr Rothemich, whose work on this topic was made possible by the Robert Wood Johnson Foundation Generalist Physician Faculty Scholars Program, thanks the National Program Office and is especially grateful for the support and guidance he received from C. Tracy Orleans, PhD, and Cynthia D. Mulrow, MD, MSc. The authors also thank the Virginia Ambulatory Care Outcomes Research Network (ACORN); the research assistants who administered the exit questionnaires (Ann Luck, Carolyn Mitchell, and Frances Stewart); and the clinicians, staff, managers, and patients of the participating practices: Ashland Medical Center, Ashland; Aylett Medical Center, Aylett; Cold Harbor Family Medicine, Mechanicsville; Family Physicians of Chester, Chester; Harbour Point Family Practice, Midlothian; Huguenot Primary Care, Midlothian; Internal Medicine Associates of Virginia, Richmond; Ironbridge Family Practice, Chester; King and Queen Family Practice, St Stephens Church; Laburnum Medical Center, Richmond; Memorial Medical Center, Mechanicsville; Midlothian Family Practice, Powhatan; Midlothian Family Practice, Watercove; Midlothian Primary Care, Midlothian; Drs Titus, Hendrix, Turner, Pahle, and Christensen, Richmond; Tuckahoe Family Practice Center, Richmond; Village Green Family Medicine, Midlothian; and Village Medical Associates, Midlothian. The authors are also grateful for editorial assistance and review rendered by Michelle Kienholz.
Conflicts of interest: Dr. Johnson has been paid by Altria Group, Inc, for statistical consulting services unrelated to this study. All of the other authors report no potential conflicts of interest.
Funding support: This study was funded through 2 grants to Dr Rothemich from the Robert Wood Johnson Foundation, including a Generalist Physician Faculty Scholars Program award (036798) and a supplemental grant (043145).
Disclaimer: The Foundation had no role in the design or conduct of the study; the collection, management, analysis, or interpretation of the data; or the preparation, review, or approval of the manuscript. Drs Rothemich and Johnson had full access to all data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Trial registration: ClinicalTrials.gov: NCT00245323.
REFERENCES
- 1.Orleans CT, Woolf SH, Rothemich SF, Marks JS, Isham GJ. The top priority: building a better system for tobacco-cessation counseling. Am J Prev Med. 2006;31(1):103–106. [DOI] [PubMed] [Google Scholar]
- 2.Centers for Disease Control and Prevention. Annual smoking-attributable mortality, years of potential life lost, and economic costs—United States, 1995–1999. MMWR Morb Mortal Wkly Rep. 2002;51(14):300–303. [PubMed] [Google Scholar]
- 3.40th Anniversary of the First Surgeon General’s Report on Smoking and Health. http://www.cdc.gov/mmwr/preview/mmwrhtml/mm5303a1.htm. Accessed May 13, 2005.
- 4.Ockene JK. Smoking intervention: the expanding role of the physician. Am J Public Health. 1987;77(7):782–783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Owen N, Davies MJ. Smokers’ preference for assistance with cessation. Prev Med. 1990;19(4):424–431. [DOI] [PubMed] [Google Scholar]
- 6.Russell MA, Wilson C, Taylor C, et al. Effect of general practitioners’ advice against smoking. BMJ. 1979;2(6184):231–235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Pine D, Sullivan S, Conn SA, David C. Promoting tobacco cessation in primary care practice. Prim Care. 1999;26(3):591–610. [DOI] [PubMed] [Google Scholar]
- 8.The Agency for Health Care Policy and Research Smoking Cessation Clinical Practice Guideline. JAMA. 1996;275(16):1270–1280. [PubMed] [Google Scholar]
- 9.Fiore MC, Bailey WC, Cohen SJ, et al. Treating Tobacco Use and Dependence. Rockville, MD: US Department of Health and Human Services; 2000.
- 10.Manley MW, Griffin T, Foldes SS, Link CC, Sechrist RA. The role of health plans in tobacco control. Annu Rev Public Health. 2003;24:247–266. [DOI] [PubMed] [Google Scholar]
- 11.Fiore MC, Jorenby DE, Schensky AE, Smith SS, Bauer RR, Baker TB. Smoking status as the new vital sign: effect on assessment and intervention in patients who smoke. Mayo Clin Proc. 1995;70(3):209–213. [DOI] [PubMed] [Google Scholar]
- 12.Robinson MD, Laurent SL, Little JM Jr. Including smoking status as a new vital sign: it works! J Fam Pract. 1995;40(6):556–561 [PubMed] [Google Scholar]
- 13.Ahluwalia JS, Gibson C, Kenney E, Wallace D, Resnicow K. Smoking status as a vital sign. J Gen Intern Med. 1999;14(7):402–408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Piper ME, Fiore MC, Smith SS, et al. Use of the vital sign stamp as a systematic screening tool to promote smoking cessation. Mayo Clin Proc. 2003;78(6):716–722. [DOI] [PubMed] [Google Scholar]
- 15.Boyle R, Solberg LI. Is making smoking status a vital sign sufficient to increase cessation support actions in clinical practice? Ann Fam Med. 2004;2(1):22–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Milch CE, Edmunson JM, Beshansky JR, Griffith JL, Selker HP. Smoking cessation in primary care: a clinical effectiveness trial of two simple interventions. Prev Med. 2004;38(3):284–294. [DOI] [PubMed] [Google Scholar]
- 17.Maizlish NA, Ruland J, Rosinski ME, Hendry KA. A systems-based intervention to promote smoking as a vital sign in patients served by community health centers. Am J Med Qual. 2006;21(3):169–177. [DOI] [PubMed] [Google Scholar]
- 18.Hopkins DP, Briss PA, Ricard CJ, et al. The Task Force on Community Preventive Services. Reviews of evidence regarding interventions to reduce tobacco use and exposure to environmental tobacco smoke. Am J Prev Med. 2001;20(2 Suppl):16–66.11173215 [Google Scholar]
- 19.Lancaster T, Stead L, Silagy C, Sowden A. Effectiveness of interventions to help people stop smoking: findings from the Cochrane Library. BMJ. 2000;321(7257):355–358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Silagy C. Physician advice for smoking cessation. Cochrane Database Syst Rev. 2000;(2):CD000165. Review. Update in Cochrane Database Syst Rev. 2001;(2):CD000165.
- 21.Lancaster T, Stead LF. Individual behavioural counselling for smoking cessation. Cochrane Database Syst Rev. 2002;(3):CD001292. [DOI] [PubMed]
- 22.Nutting PA, Beasley JW, Werner JJ. Practice-based research networks answer primary care questions. JAMA. 1999;281(8):686–688. [DOI] [PubMed] [Google Scholar]
- 23.Current estimates from the National Health Interview Survey, 1993. Vital Health Stat 10. 1994;(190):1–221. [PubMed]
- 24.Pbert L, Adams A, Quirk M, Hebert JR, Ockene JK, Luippold RS. The patient exit interview as an assessment of physician-delivered smoking intervention: a validation study. Health Psychol. 1999;18(2):183–188. [DOI] [PubMed] [Google Scholar]
- 25.Johnson RA, Wichern DW. Applied Multivariate Statistical Analysis. 4th ed. Upper Saddle River, NJ: Prentice Hall; 1998.
- 26.Cornuz J, Zellweger JP, Mounoud C, Decrey H, Pecoud A, Burnand B. Smoking cessation counseling by residents in an outpatient clinic. Prev Med. 1997;26(3):292–296. [DOI] [PubMed] [Google Scholar]
- 27.Goldstein MG, Niaura R, Willey-Lessne C, et al. Physicians counseling smokers. A population-based survey of patients’ perceptions of health care provider-delivered smoking cessation interventions. Arch Intern Med. 1997;157(12):1313–1319. [DOI] [PubMed] [Google Scholar]
- 28.Sesney JW, Kreher NE, Hickner JM, Webb S. Smoking cessation interventions in rural family practices: an UPRNet study. J Fam Pract. 1997;44(6):578–585. [PubMed] [Google Scholar]
- 29.Agresti A. Categorical Data Analysis. 2nd ed. Hoboken, NJ: John Wiley & Sons, Inc; 2002.
- 30.Lancaster T, Stead L. Physician advice for smoking cessation. Cochrane Database Syst Rev. 2004;(4):CD000165. [DOI] [PubMed]


