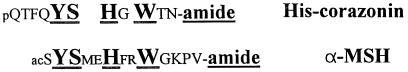Abstract
In response to crowding, locusts develop characteristic black patterns that are well discernible in the gregarious phase at outbreaks. We report here a dark-color-inducing neuropeptide (dark-pigmentotropin) from the corpora cardiaca of two plague locusts, Schistocerca gregaria and Locusta migratoria. The chromatographic isolation of this neuropeptide was monitored by using a bioassay with an albino mutant of L. migratoria. The neurohormone, consisting of 11 amino acids, is identical to [His7] corazonin, previously isolated from corpora cardiaca of another acridid without known function. The present results show that even in isolated (solitary) nymphs, [His7] corazonin induces gregarious black patterns. Its primary structure shows some similarity with the vertebrate melanophore stimulating hormone.
Body-color polymorphism is widespread among animals. Two locust species, Schistocerca gregaria and Locusta migratoria, display conspicuous differences in body color, particularly during the nymphal stage. A major extrinsic factor influencing locust body color is phase polymorphism, a term used to describe continuous polymorphism in response to population density: locusts at a low density (solitary phase) are often green or brown, whereas those at outbreaks (gregarious phase) develop black patterns (1–4). Although the role of juvenile hormone in the induction of the green color is well established (2–4), little information is available about the hormonal factor that induces dark color in locusts. It has long been known that some factor present in the brain and the corp cardiaca (CC) promotes darkening in locusts (2, 3), but progress in identifying its chemical nature has been hampered by the lack of a convenient bioassay.
Recently, we discovered an albino mutant, originating from a laboratory colony of an Okinawa (Japan) strain of L. migratoria (5). Albinism in this mutant is controlled by a single recessive Mendelian unite (5), as described also for other albino mutants of this species (6, 7), as well as of S. gregaria (8) and the grasshopper Melanoplus sanguinipes (under the name Melanoplus bilituratus) (9). The albinism in the Okinawa strain of L. migratoria is caused by the deficiency of a peptide(s) present in the central nervous system and the CC. Implantation of a brain or CC taken from normal (pigmented) individuals or injection of their methanolic extract induces dark color in albino locusts (10–12), but injection of such methanolic extract made from albino individuals has no dark-color-inducing effect in albino locusts (11). Of interest, implantation of brains or CC taken from other taxa, including S. gregaria and other acridids, cockroaches, katydids, crickets, and moths also are effective in inducing dark color in albino L. migratoria (10, 12, 13). This result indicates that similar substances inducing dark color in L. migratoria may exist in diverse groups of insects. Because whitish albino locusts can be obtained easily by mass rearing, they provide an excellent bioassay system for the characterization of this dark-color inducing peptide. Its role in body color polymorphism and phase polymorphism in locusts can then be determined by means of the synthetic analog.
MATERIALS AND METHODS
Insects and Tissue Extraction.
The colony of the desert locust S. gregaria was maintained according to Ashby’s method (14) and that of the migratory locust L. migratoria migratorioides as described (15). We collected 2,950 pairs of CC from adult S. gregaria and 4,700 pairs from adult L. migratoria migratorioides and extracted the glands of each species with a methanol/water/acetic acid (vol/vol/vol 90:9:1) solution.
HPLC Purification.
Methanol was evaporated, and the remaining aqueous residue was reextracted with ethyl acetate and hexane to remove the lipids. The organic layers were discarded, and the aqueous layer was dried and redissolved in 0.1% trifluoroacetic acid (TFA). Subsequently, each sample was loaded onto a Megabond Elute cartridge (Varian), which was eluted in 24 ml each of, respectively, 30, 60, and 90% CH3CN/0.1% TFA. The elutes were dried and tested for activity. The fraction containing the dark-color-inducing activity was further purified on a Gilson HPLC system with variable detector (set at 214 nm). Column conditions were (i) for the Bondapak C18 PrepLC Cartridge (25 × 100 mm; 30 nm, 15 μm, Waters), solvent A, 0.1% TFA in water; solvent B, 50% CH3CN in 0.1% aqueous TFA; 100% solvent A for 30 min followed by a linear gradient to 100% solvent B in 150 min; flow rate, 6 ml/min; detector range, 0.5 absorption units full scale; fractions were collected every 2 min; (ii) for a Spherisorb C1 column (4.6 × 250 mm, 5 μm, Phase Separations B5m, Waddinxveen, The Netherlands), solvent A and B, the same as above; a linear gradient from 0% to 30% B in 60 min; flow rate, 1.5 ml/min; detector range, 0.5 absorption units full scale; 1-min fractions were collected; (iii) for a Supelcosil C8-DB column (4.6 × 250 mm, 5 μm, Supelco); solvents A and B were the same as above; linear gradient to 50% solvent B over 80 min; flow rate, 1.5 ml/min; 0.05 absorption units full scale. Peaks were collected manually. After each run, a volume containing approximately 10 CC equivalents was dried and assayed.
Amino Acid Sequence Determination and Mass Analyses.
Two activity units of pyroglutamine aminopeptidase (Sigma) dissolved in 40 μl of distilled water were added to the dried natural peptide. After incubation (37°C for 30 min), the sample was purified on the Supelcosil-C8-DB column. Conditions: solvent A, 0.1% TFA; solvent B, 50% CH3CN/0.1% TFA; linear gradient from 20% to 60% solvent B; flow rate 1.0 ml/min; detector range, 0.1 absorption units full scale. After deblocking and purification on C8-DB, 1/10 of the final volume (1.5 ml) was loaded onto a precycled Biobren Plus-coated glass filter. N-terminal amino acid sequence analysis was carried out on a Perkin–Elmer/Applied Biosystems Procise 492 microsequencer running in the pulsed liquid mode. Mass analysis of the natural and deblocked peptide was performed on a Micromass Tofspec matrix-assisted laser desorption ionization–time-of-flight mass spectrometer as described (16). An aliquot (15 μl) of the final volume was mixed with an equal volume of matrix solution (50 mM α-cyano 4-hydroxycinnamic acid in 30% CH3CN containing 0.1% TFA). One microliter of this mixture was deposited on the metal target, allowed to air-dry, and introduced into the matrix-assisted laser desorption ionization–time-of-flight mass spectrometer source.
The amino acid sequence, so determined, was synthesized at Research Genetics (Huntsville, AL).
Bioassays.
Albino locusts were reared either in groups of 100 individuals per cage or in isolation at 50–60% relative humidity and 30°C. They were whitish, and no color difference was noticed between crowded and isolated locusts before the experiment. Bioassays were conducted with newly ecdysed third- or fourth-instar crowded albino nymphs of L. migratoria at 30°C. Dried samples were mixed with peanut oil or olive oil and injected into the hemocoel (4 μl per animal). No appreciable difference was noted in biological activity when extracts were mixed with different oils. The results were evaluated 4 days later by the visible colors, and locusts were categorized into three categories: unpigmented, slightly pigmented, and more strongly pigmented.
Hatchlings of S. gregaria obtained from a line maintained in isolation for two generations (except for a short mating period) were reared individually on fresh leaves of the orchard grass at 70–85% relative humidity and 30°C. Individually isolated green locusts with no black spot on the head and thorax were injected (in oil as above) with the synthetic hormone. To compare the effect of this hormone injection with that of crowding, five isolated green locusts were put together with crowded individuals of the same age in a small wood-framed cage (15 × 28 × 28 cm) covered with nylon mesh. One antenna was removed from each of the latter to avoid confusion with the test individuals. All locusts were derived from the same egg pod. The test was repeated twice.
The dark-color-inducing effect of synthetic [Arg7] corazonin (Sigma) or [His7] corazonin (Research Genetics) injected to newly ecdysed albino locusts was categorized into five color grades: 0, unpigmented; 1–3, increasingly darker coloration; 4, practically black (13). In another experiment, [His 7] corazonin was injected to mid-instar (3 days after prior molt) albino locusts, and the color induced in the next instar was compared with that of normally pigmented conspecific crowded (gregarious) locusts.
RESULTS AND DISCUSSION
Among the three fractions obtained with the Megabond Elute cartridge, the dark-color-inducing activity eluted in the 30% CH3CN/0.1% TFA fraction in S. gregaria. The 60% and 90% fractions were devoid of this activity. The initial HPLC fractionation of the 30% fraction on the Bondapak preparative C18 column yielded only two subsequent 2-min fractions containing dark-color-inducing activity. These two fractions, at 76–80 min, were further purified on the C1 column, and the activity eluted in the 26 min fraction. The final purification on the C8-DB column yielded three high-absorbance peaks, the highest of which (57.5 min) contained the active material. The extract of the L. migratoria migratorioides CC was processed in exactly the same way, and the dark-color-inducing fraction eluted from the respective columns at exactly the same retention times as the active fraction for S. gregaria.
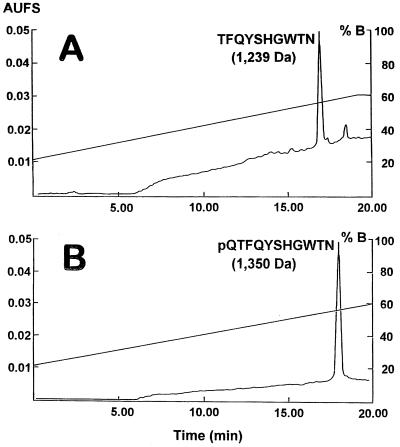
For both S. gregaria and L. migratoria purified factors, matrix-assisted laser desorption ionization–time-of-flight mass spectrometry revealed a molecular mass of 1,350 Da ([M + H]+ at 1,351.1). Edman degradation-based amino acid sequencing of the samples yielded no sequence, indicating that the peptide is N-terminally blocked. This result was confirmed by the comparison of the molecular mass and the HPLC retention of the natural and the deblocked peptide. Deblocking with pyroglutamate aminopeptidase of half of the peptide sample followed by HPLC fractionation on Supelcosil C8-DB resulted in the isolation of a fragment eluting at 17 min (Fig. 1A), which is 1 min earlier than the elution time of the blocked peptide (half of the sample was run on the same column in the same conditions; see Fig. 1B). The molecular mass of the deblocked peptide of both locusts was established as 1,239 Da ([M + H]+ at 1, 240), which corresponds to the mass of the blocked peptide minus that of pGlu (111.1) (Fig. 1). The deblocked peptide of S. gregaria yielded the following primary structure: Thr-Phe-Gln-Tyr-Ser-His-Gly-Trp-Thr-Asn. The same sequence was obtained for L. migratoria migratorioides. Based on the difference of 1 Da between the calculated (1,351.6) and the experimental (1,350.1) molecular mass, we concluded that the carboxyl terminus was most probably amidated and that the primary structure of the dark-color-inducing factor was pGlu-Thr-Phe-Gln-Tyr-Ser-His-Gly-Trp-Thr-Asn-NH2. This was confirmed by coelution of the synthetic and the natural analog in the respective reverse-phase HPLC systems.
Figure 1.
HPLC fractionation on the Supelcosil C8-DB column of the dark-color-inducing neuropeptide digested with pyroglutamate aminopeptidase (A) and of the natural peptide (B) (for column conditions, see text). The hardly noticeable peak of the parent peptide at 18 min in A indicated that nearly all of the parent peptide had been digested by the pyroglutamine aminopeptidase. AUFS, absorption units full scale.
The primary structure of this peptide is identical to [His7] corazonin, previously isolated from the CC of Schistocerca americana (17). This is one of the many peptides identified as myotropic factors from locusts and other insects during the past decade (18, 19). Corazonin (with an Arg7 instead of His7) was first isolated from CC of a cockroach Periplaneta americana as the most potent cardiostimulatory peptide (20). However, neither [Arg7] corazonin nor [His7] corazonin affects the rate of heartbeat in the locust S. americana, from which the latter was first isolated (17). Thus, the physiological function of [His7] corazonin was unknown.
In arthropods, the only pigment-affecting hormones identified so far are the red pigment-concentrating hormone, the pigment-dispersing hormones, and the melanization and reddish-coloration hormone (19). The function of the insect peptides such as the adipokinetic hormones and the pigment dispersing factors, related to the crustacean red pigment-concentrating hormone and pigment-dispersing hormones, is not, as in crustaceans, to regulate pigment dispersal. [His7] corazonin is not related to melanization and reddish-coloration hormone or the pigment-dispersing hormones. However, the sequences of adipokinetic hormones/red pigment-concentrating hormone peptides display some structural similarities with corazonin, as do the predicted structures of their respective peptide precursors (21). From this comparative viewpoint, the structural resemblance between [His7] corazonin and mammalian melanophore-stimulating hormone (α-MSH) (Fig. 2) is also intriguing. Preliminary experiments showed that [His7] corazonin (10 μg) does not mimic the effect of MSH with respect to darkening of the frog skin by either in vivo or in vitro tests, but injection of α-, β,- or γ-MSH (1 nmol) into albino L. migratoria causes some darkening in the latter.
Figure 2.
Comparison of the primary structures of mammalian α-MSH and locust [His7] corazonin. Identical components are marked with underlined and in boldface.
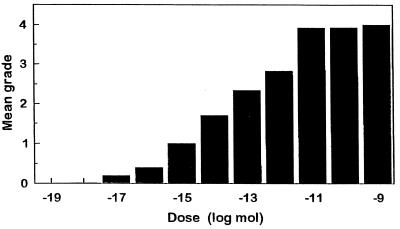
To examine dose–response relations, various concentrations of the synthetic [His7] corazonin in peanut oil were injected into albino locusts, and a dose-dependent response was seen (Fig. 3). All nymphs tested were darkened when injected at or above a dose of 10 fmol. Injections of 10 pmol or higher concentrations caused most albino nymphs to turn black completely (grade 4). We also found that [Arg7] corazonin had a similar effect in locusts. Injection of 100 or 10 pmol of [Arg7] corazonin in peanut oil into newly ecdysed fourth-instar albino nymphs of L. migratoria caused them to turn black after ecdysis to the following instar, with a mean color grade of 4.0 and 3.6, respectively (n = 10 each).
Figure 3.
Effect of graded doses of [His7] corazonin injected, in peanut oil, on the pigmentation of albino locusts in L. migratoria. Albino locusts were injected at the beginning of the fourth instar, and their body color was checked 4 days later in the same (fourth) instar according to the color grades previously published (13); 0, unpigmented; 1–3, increasingly darker coloration; 4, practically black. Columns indicate means (n = 9–15 for each column).
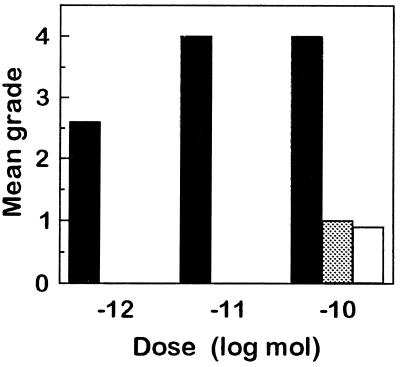
In contrast to the strong dark-color-inducing effect of [His7] corazonin injected in oil (see above), no dark color was induced when aqueous solutions of the peptide were injected into albino nymphs except for the highest dose (100 pmol), which induced slightly dark color after injection (Fig. 4). In our previous tests, biological activity was detected only when crude extracts of CC were injected in oil instead of aqueous solutions (11, 12). It is possible that [His7] corazonin, which may otherwise be degraded or excreted rapidly, is released from the oil droplets slowly into the hemolymph, and its long-lasting presence is necessary to elicit the response.
Figure 4.
Effect of [His7] corazonin in peanut oil (filled bars), 10% aqueous ethanol (stippled bar), and water (open bar) on the pigmentation of albino locusts in L. migratoria. Albino locusts were injected at the beginning of the fourth instar, and their body color was checked 4 days later, as in Fig. 3. Columns indicate means (n = 10 each).
Studies with crude extracts of CC indicate that injection of the extracts at the middle of a nymphal instar causes albino locusts to develop gregarious black patterns in L. migratoria (11). To determine whether [His7] corazonin induces such black patterns, 0.1 nmol of the peptide was injected into albino nymphs at the middle of the third instar (day 3), and their body color was examined 2 days after the subsequent ecdysis; i.e., 4 or 5 days after injection. Here we tested both crowded and isolated albino locusts; i.e., locusts reared under crowded and isolated conditions. After injection, all crowded albino locusts (n = 10) developed black patterns with an orange background color (Fig. 5C) similar to that of normal crowded locusts (Fig. 5B; n = 10), whereas oil-injected controls remained unpigmented (Fig. 5A). This result demonstrates that [His7] corazonin induces a body coloration like that typically observed in gregarious individuals. Of interest, a similar coloration was obtained in isolated albino locusts after injection of the hormone (Fig. 5D), indicating that crowding is not necessary for the induction of black patterns and orange color.
Figure 5.
Photographs showing the effect of [His7] corazonin injected in peanut oil on the body color in L. migratoria (A–D) and S. gregaria (E–H). Locusts were injected with the hormone in 4 μl of oil or with 4 μl of oil alone at day 3 of the third instar and photographed 2 days after the subsequent ecdysis. Hormone (0.1 nmol and 1 nmol) was injected into L. migratoria and S. gregaria, respectively. (A) Oil-injected crowded albino nymph. (B) Untreated crowded normal nymph. (C) Hormone-injected crowded albino nymph. (D) Hormone-injected isolated albino nymph. (E) Oil-injected isolated nymph. (F) Untreated crowded nymph. (G) Nymph transferred from isolation to crowding at the third instar. (H) Hormone-injected isolated nymph.
Because no albino strain was available in S. gregaria, we used green nymphs obtained from isolated rearing to test the above effect in this species. Green third-instar nymphs remained green after ecdysis to the fourth instar (Fig. 5E; n = 10), whereas those continuously reared under crowded conditions (10 individuals per cage) had dark patterns typical of gregarious individuals (Fig. 5F; n = 10). Crowding influences locust body color greatly (1–3). When 5 green nymphs from individual rearing were put into a cage together with 10 crowded nymphs at the beginning of the third instar, they all showed dark patterns by day 2 of the fourth instar; i.e., 7 or 8 days after transfer (n = 10,) and looked similar to those continuously kept crowded (Fig. 5G; n = 10,). Injection of [His7] corazonin mimicked this effect; green nymphs injected with 1 nmol of the peptide on day 1 of the third instar developed black patterns after ecdysis to the fourth instar (n = 10), even though they were continuously kept in isolation (Fig. 5H). They also developed reddish spots at the rear portion of the head and on the thorax, like crowded individuals. These results clearly indicate that [His7] corazonin is responsible for the induction of darkening that can be observed in crowded individuals in the laboratory or at outbreaks in the field.
The discovery of the biological function of [His7] corazonin raises an important question as to its functional relationship to the induction of other body colors and their patterns observed in the field. It seems that the timing and intrinsic titer of this hormone and its interaction with other hormones, such as the juvenile hormone, exert a major effect on the color polymorphism of locusts and possibly of some other insects. Many acridids show homochromy, i.e., adaptation of the body color to that of the background (1–2), and this hormone may induce dark homochromy. Another important question is related to the possible involvement of this hormone in the control of other phase-related morphological, behavioral, and physiological differences in locusts. The discovery of the structural similarity between [His7] corazonin and vertebrate MSHs, together with the presence of dark-color-inducing substances in diverse groups of insects (10, 12, 13), will stimulate further research to understand the function, diversity, and evolution of neuropeptides.
We name this hormone “dark-color-inducing neurohormone” (Lom-DCIN and Scg-DCIN), or “dark-pigmentotropin.” It presumably induces both melanin and ommochrome pigments because cuticular malanins are overlying dark areas of epidermal ommochromes in locusts (22). On the other hand, the DCIN seems to control primarily dark colors; its absence in the albino locusts does not prevent changes in some lighter colors, such as green coloration, or yellowing of the adult males with sexual maturation, also as induced by juvenile hormone analogs (5).
Acknowledgments
Insect rearing was assisted by N. Kenmochi, S. Ogawa and H. Ikeda in Tsukuba, by H. Abu-Hilal in Jerusalem and by R. Jonckers in Leuven. S. Tanaka thanks Emer. Prof. Sinzo Masaki for introducing S.T. to acridology. A. De Loof acknowledges the E.U. FAIR-CT-96-1648 sponsoring. The research was supported by the Ministry of Agriculture, Forestry and Fisheries of Japan and the Science and Technology Agency of Japan. The part of the research carried out in Jerusalem was supported by The Israel Science Foundation founded by The Israel Academy of Sciences and Humanities.
ABBREVIATIONS
- CC
corpora cardiaca
- MSH
vertebrate melanophore stimulating hormone
- TFA
trifluoroacetic acid
Footnotes
This paper was submitted directly (Track II) to the Proceedings Office.
References
- 1.Uvarov B. Grasshoppers and Locusts. Vol. 1. Cambridge, U.K.: Cambridge Univ. Press; 1966. [Google Scholar]
- 2.Fuzeau-Braesch S. In: Comprehensive Insect Physiology, Biochemistry, and Pharmacology. Kerkut G A, Gilbert L I, editors. Vol. 9. Oxford: Pergamon; 1985. pp. 549–589. [Google Scholar]
- 3.Pener M P. Adv Insect Physiol. 1991;23:1–79. [Google Scholar]
- 4.Pener M P, Yerushalmi Y. J Insect Physiol. 1998;44:365–377. doi: 10.1016/s0022-1910(97)00169-8. [DOI] [PubMed] [Google Scholar]
- 5.Hasegawa E, Tanaka S. Jap J Entomol. 1994;62:315–324. [Google Scholar]
- 6.Verdier M. Bull Soc Zool Fr. 1965;90:493–501. [Google Scholar]
- 7.Nolte D J. Chromosoma. 1969;26:287–297. doi: 10.1007/BF00326523. [DOI] [PubMed] [Google Scholar]
- 8.Hunter-Jones P. Nature (London) 1957;180:236–237. [Google Scholar]
- 9.Putnam L G. Nature (London) 1958;182:1529. [Google Scholar]
- 10.Tanaka S. Zool Sci. 1993;10:467–471. [Google Scholar]
- 11.Tanaka S, Pener M P. J Insect Physiol. 1994;40:997–1005. [Google Scholar]
- 12.Tanaka S, Yagi S. Jap J Entomol. 1997;65:447–457. [Google Scholar]
- 13.Tanaka S. J Insect Physiol. 1996;42:287–294. [Google Scholar]
- 14.Ashby G. In: The UFAW Handbook on the Care and Management of Laboratory Animals. Worden A N, Lane-Petter W, editors. London: The Universities Federation for Animal Welfare; 1972. pp. 582–587. [Google Scholar]
- 15.Shalom U, Pener M P. Physiol Entomol. 1986;11:441–452. [Google Scholar]
- 16.Veelaert D, Schoofs L, Verhaert P, De Loof A. Biochem Biophys Res Commun. 1997;241:530–534. doi: 10.1006/bbrc.1997.7843. [DOI] [PubMed] [Google Scholar]
- 17.Veenstra J A. Peptides. 1991;12:1285–1289. doi: 10.1016/0196-9781(91)90208-7. [DOI] [PubMed] [Google Scholar]
- 18.Schoofs L, Veelaert D, Vanden Broeck J, De Loof A. Peptides. 1997;18:145–156. doi: 10.1016/s0196-9781(96)00236-7. [DOI] [PubMed] [Google Scholar]
- 19.Gäde G, Hoffmann K-H, Spring J H. Physiol Rev. 1997;79:963–1032. doi: 10.1152/physrev.1997.77.4.963. [DOI] [PubMed] [Google Scholar]
- 20.Veenstra J A. FEBS Lett. 1989;250:231–234. doi: 10.1016/0014-5793(89)80727-6. [DOI] [PubMed] [Google Scholar]
- 21.Veenstra J A. Biochem Biophys Res Commun. 1994;204:292–296. doi: 10.1006/bbrc.1994.2458. [DOI] [PubMed] [Google Scholar]
- 22.Fuzeau-Braesch S. J Insect Physiol. 1966;12:1363–1368. [Google Scholar]