Abstract
The immunoglobulin superfamily member CD147 plays an important role in fetal, neuronal, lymphocyte and extracellular matrix development. Here we review the current understanding of CD147 expression and protein interactions with regard to CD147 function and its role in pathologic conditions including heart disease, Alzheimer’s disease, stroke and cancer. A model linking hypoxic conditions found within the tumor microenvironment to up-regulation of CD147 expression and tumor progression is introduced.
Keywords: CD147, EMMPRIN, MMP, MCT, immunoglobulin superfamily, integrin, tumor, metastasis
1. Introduction
CD147, a member of the immunoglobulin superfamily, was discovered twenty five years ago (Biswas 1982), however, very little is known about its regulation and function in vivo. Originally identified as a tumor surface protein capable of inducing matrix metalloproteinase (MMP) expression in fibroblasts, the protein was initially termed tumor cell-derived collagenase stimulatory factor (TCSF) (Biswas et al. 1987; Ellis et al. 1989). Following its cloning, TCSF was determined to be a transmembrane glycoprotein that was highly homologous with proteins of the immunoglobulin (Ig) superfamily (Fossum et al. 1991; Miyauchi et al. 1991; Kasinrerk et al. 1992; Biswas et al. 1995) and was able to induce the production of several MMPs (Kataoka et al. 1993; Biswas et al. 1995; Guo et al. 1997). These findings resulted in the renaming of the protein as CD147/EMMPRIN (extracellular matrix metalloproteinase inducer) (Biswas et al. 1995; Guo et al. 1997). While increased expression of CD147 occurs in many tumors, it’s expression is not limited to tumor cells (Ellis et al. 1989; Muraoka et al. 1993; Polette et al. 1997; van den Oord et al. 1997; Bordador et al. 2000; Sameshima et al. 2000; Jiang et al. 2001). CD147 is a pleiotropic molecule that is critical in fetal development and retinal function and has been shown to play a role in thymic T cell development, as well as many neurological processes ranging from the development of the nervous system to involvement in spatial learning and plaque formation within Alzheimer’s stricken brains (Naruhashi et al. 1997; Muramatsu et al. 2003; Zhou et al. 2005). Because of the ubiquitous expression and pleiotropic nature of this molecule, understanding the protein-protein interactions and regulation of CD147 expression is essential for therapeutic intervention strategies. Here we will review the functional consequences of CD147 expression under physiologic and pathologic conditions as well as the body of literature on CD147 structure and associated proteins.
2. Structural Organization
CD147 is a transmembrane glycoprotein that was categorized as a member of the immunoglobulin superfamily (IgSF) of receptors (Fossum et al. 1991; Miyauchi et al. 1991; Kasinrerk et al. 1992; Biswas et al. 1995). CD147 was identified independently in various species and are referenced throughout the literature as EMMPRIN (Extracellular Matrix Metalloproteinase-Inducer), M6 (Human) (Kasinrerk et al. 1992), Neurothelin, 5A11 and HT7 (chicken) (Seulberger et al. 1992; Fadool et al. 1993; Schlosshauer et al. 1995), OX47 and CE9 (rat) (Fossum et al. 1991; Nehme et al. 1995), and Basigin, gp42 (human and mouse) (Altruda et al. 1989; Miyauchi et al. 1991; Miyauchi et al. 1995). The CD147 gene, designated BSG for basigin, is located on chromosome 19p13.3 and encodes a 29 kD protein, though migration on SDS-PAGE usually occurs between 35–65 kD, depending on the degree of glycosylation (Kasinrerk et al. 1992; Kirsch et al. 1997; Kasinrerk et al. 1999). Treatment of the protein with endoglycosidase F demonstrated that almost half of CD147’s molecular weight was due to the addition of N-linked carbohydrates (Kasinrerk et al. 1992). The protein is composed of an extracellular domain of 187 residues, a 24 residue transmembrane domain and a 40 amino acid cytoplasmic region (Biswas et al. 1995). CD147 is classified as a member of a small family of related proteins, including the teratocarcinoma antigen GP70 and the synaptic glycoprotein SDR1, that share the features of two Ig-like extracellular domains, a short cytoplasmic tail, and a single transmembrane domain (Fossum et al. 1991; Shirozu et al. 1996). Three glycosylation sites have been identified within the CD147 Ig like domains: two within the membrane proximal Ig domain and one within the distal Ig domain (Hakomori 1996; Sun et al. 2001; Tang et al. 2004). N-linked glycosylation is critical to CD147 and is postulated to serve as a regulatory mechanism of CD147 function.
Recently, a distinct CD147 transcript that encodes for three Ig domains was identified in mouse retina (Hanna et al. 2003; Ochrietor et al. 2003). While this variant has not been fully characterized as of yet, it is believed to aid in the alignment of CD147 associated lactate transporters to sites of cell-cell contact via homophilic CD147 interactions.
3. CD147-Associated Proteins
A single charged glutamic acid residue resides within the hydrophobic transmembrane domain of CD147 (Fossum et al. 1991). This feature is atypical of transmembrane domains and was first described for the neu oncoprotein as a way to promote oligomerization (Weiner et al. 1989) or to potentiate protein-protein interactions. Sequence comparison of the various species-specific CD147 isoforms have demonstrated a high degree of conservation within both the cytoplasmic and transmembrane domains, implying a high degree of functional importance associated with these regions (Miyauchi et al. 1991). Studies performed with a CD2-CD147 chimera implicate the transmembrane and cytoplasmic domains of CD147 as being critical for protein-protein interactions within the plasma membrane (Kirk et al. 2000). Two members of the monocarboxylate transporter (MCT) family of proteins, MCT1 and MCT4, were found to require CD147 as a chaperone for their expression at the plasma membrane (Kirk et al. 2000). Additionally, experimental evidence exists for CD147’s association with integrin (Berditchevski et al. 1997; Curtin et al. 2005) caveolin-1 (Cav-1) (Tang et al. 2004), and cyclophilins (CyP) (Yurchenko et al. 2001; Pushkarsky et al. 2005; Yurchenko et al. 2005). These protein interactions are critical to CD147’s function and explain the complexity of CD147 behavior.
MCT
Monocarboxylate transporter proteins catalyze the proton-linked transport of monocarboxylates, including lactate and pyruvate: elements essential for the maintenance of metabolic homeostasis within most cells (Halestrap et al. 1999). All MCT proteins share the same basic structure, having 12 transmembrane domains and cytosolically-exposed C and N termini, as well as a large loop region between transmembrane domains 6 and 7 (Halestrap et al. 1999). Despite the structural similarities between the fourteen members that comprise this family, experimental evidence of proton-linked transport exists only for the first four isoforms (MCT1-MCT4).
CD147 has been shown to interact and co-immunoprecipitate with two members of the proton-linked MCT family, MCT1 and MCT4 (Kirk et al. 2000). In all tissues expressing MCT1 or MCT4, CD147 expression was consistently found co-localized in the same regions (Kirk et al. 2000). MCT1 and MCT4 require direct association with CD147 within the cell for their transport to the plasma membrane (Kirk et al. 2000). CD147 was initially thought to serve solely as a chaperone in this instance, yet their association persists once the proteins localize within the plasma membrane, intimating another function to this complex. While the significance of this continued association is not yet clear, recent evidence suggests that CD147 association with MCT1 or MCT4 within the plasma membrane is essential for MCT mediated lactate transport (Wilson et al. 2005). Fluorescence resonance energy transfer analyses suggest that the minimal complex consists of a dimer of CD147 interacting with two MCT1 molecules (Wilson et al. 2002). Aggressive tumors were found to express higher than usual levels of CD147, which corresponded with up-regulated levels of MCT1 (Froberg et al. 2001; Jin et al. 2006; Jin et al. 2006; Nabeshima et al. 2006; Zhang et al. 2006); an event potentially necessary to meet the high glycolytic needs of transformed cells. Recently, expression of MCT4 was shown to control plasma membrane localization of CD147 in a metastatic breast cancer cell line (Gallagher et al. 2007). The increased expression of MCTs observed in many forms of cancer is important to tumor viability. Tumor cells thrive under hypoxic conditions, yet their high glycolytic nature results in the production of excess lactic acid, which, in turn, has to be regulated by MCT for tumor survival. Increased expression of MCTs could serve as an indicator of the transition from normalcy to malignancy. Indeed, recently, MCT inhibitors have been proposed as anti-cancer agents (Wahl et al. 2002; Coss et al. 2003; Mathupala et al. 2004; Fang et al. 2006).
Integrin
Integrins constitute a superfamily of membrane proteins that allow individual cells to communicate with their environment. Through their interactions with the extracellular matrix (ECM), external stimuli are transmitted internally through the activation of various signaling cascades, thus allowing cells to adapt to environmental changes. Two types of integrin signaling exist: direct, whereby an extracellualr ligand binds to integrin and induces intracellular signaling and indirect, whereby changes in integrin-mediated cell anchorage result from altered mitogen signaling (Keely et al. 1998; Springer et al. 2004). Most integrins are non-covalently linked heterodimers composed of both an alpha and beta subunit with functional specificity defined by the pairing of these subunits.
Originally thought to function independently, a growing body of evidence has demonstrated that broader functions for integrins exist through their ability to interact with other membrane-bound proteins in cis (Porter et al. 1998); CD147 is one such protein. Immunoprecipitation and immunofluorescent co-localization experiments identified two integrin isoforms, α3β1 and α6β1, that associate with CD147 at points of cell-cell contact (Berditchevski et al. 1997). These integrins, which were shown to preferentially bind to laminins, non-collagenous glycoproteins that serve as the major constituent of the basement membrane (Delwel et al. 1994; Kikkawa et al. 1998), have been implicated in several processes critical to metastasis, including the promotion of cell adhesion (Malinda et al. 1996; Engbring et al. 2003), angiogenesis (Malinda et al. 1999; Engbring et al. 2003), signaling, which includes MMP induction (Reich et al. 1995; Morini et al. 2000) and chemotaxis and proliferation (Engbring et al. 2003). Through the association of CD147 with integrin, it is possible that CD147 may regulate the integrin/laminin association, and in so doing, regulate such diverse processes as basement membrane formation (Malinda et al. 1996), fetal development (Ryan et al. 1996; Ekblom et al. 2003), the promotion of cell adhesion (Malinda et al. 1996; Engbring et al. 2003), angiogenesis (Malinda et al. 1999; Engbring et al. 2003), signaling, MMP induction, chemotaxis and proliferation.
The interaction of CD147 with integrin is conserved in drosophila and is proposed to play a role in dorsal closure and extraembryonic membrane apposition (Reed et al. 2004), as well as in maintenance of cellular architecture through cytoskeletal rearrangement (Curtin et al. 2005). Within cultured insect cells, CD147/integrin interactions are essential for lamellipodia formation and within retinal cells disruption of the CD147-integrin interaction results in aberrant distribution of organelles, including mitochondria, nuclei, and rough endoplasmic reticulum (Curtin et al. 2005).
Cyclophilins
Cyclophilins (CyP) are members of the immunophilin family of peptidyl-prolyl cis-trans isomerases having proposed functions as chaperones and mediators of protein folding (Galat 1993). The notion of cyclophilin A functioning as a chaperone comes from several lines of evidence, including its association with protein transport complexes as well as its ability to regulate the surface expression of the asialoglycoprotein receptor in human cells (Huang et al. 2002) and the rhodopsin photoreceptor in Drosophila (Colley et al. 1991). Recently, several reports implicated cyclophilins as regulators of CD147 surface expression following the observation that cyclosporine A (CsA), which binds CyP, adversely affected CD147 surface expression (Yurchenko et al. 2001; Pushkarsky et al. 2005; Yurchenko et al. 2005). Peptide binding studies identified proline 211 (P211) within the CD147 transmembrane domain as being essential for CyP associated transport of CD147 to the plasma membrane (Yurchenko et al. 2005). In a subsequent study by this group, the lesser-characterized CyP60 was identified as the chaperone of CD147 (Pushkarsky et al. 2005).
Aside from their functioning as protein chaperones, cyclophilins also mediate chemotaxis of neutrophils, eosinophils and T cells when secreted from cells in response to inflammatory stimuli (Sherry et al. 1992; Xu et al. 1992; Allain et al. 2002; Yurchenko et al. 2002). Cell surface heparans are the primary receptors for secreted CyP (Denys et al. 1998; Saphire et al. 1999). Recently, however, CD147 has been identified as being essential for cyclophilin-mediated signaling (Yurchenko et al. 2002; Arora et al. 2005). Binding of extracellular CyPA to CD147 via heparans resulted in Erk activation and chemotaxis. Two amino acids, proline 180 and glycine 181, were identified within the extracellular domain of CD147 as being crucial for CD147 mediated signaling and chemotaxis. Additionally, through the use of CyPA mutants, the rotamase activity of CyPA was found to be important for this CD147 mediated signaling (Yurchenko et al. 2002). Interestingly, the association of CyPA and CD147 is further supported by findings that increased levels of CyPA in synovial fluid and increased CD147 levels on reactive neutrophils in rheumatoid arthritis patients correlated with the severity of disease (Tegeder et al. 1997; Konttinen et al. 2000). Furthermore, CyPA is incorporated into HIV-1 virions and has been shown to play a role in early viral infection (Braaten et al. 1996; Sherry et al. 1998). CD147 has been shown to facilitate HIV-1 infection through its interaction with viral associated CyPA (Pushkarsky et al. 2001).
In a mouse model of induced asthma where elevated airway levels of cyclophilins have been observed, targeting the cyclophilin-CD147 interaction using anti-CD147 mAb decreased recruitment of activated Th2 effector cells into inflamed lung tissues, resulting in decreased cytokine production and eosinophil recruitment and therefore greatly decreased lung inflammation and pathology (Gwinn et al. 2006). Clearly, cyclophilins are capable of modulating the immune response through their specific association with cellular proteins including CD147.
Caveolin-1
Caveolin proteins serve as the major constituents of caveolae, cholesterol- and glycosphingolipid-rich moeities present within the plasma membranes of most cells that mediate the transcytosis of macromolecules in a clathrin-independent manner (Williams et al. 2005). Since their initial characterization, many other functions have been ascribed to these structures, including cholesterol homeostasis, the organization and retention of cellular receptors and signal transducing elements reviewed in (Williams et al. 2004) and, most recently, as a regulator of transformation and tumorigenesis (Williams et al. 2004; Williams et al. 2005; Williams et al. 2005). While the caveolin family of proteins is comprised of three isoforms (caveolins 1, 2 and 3), only caveolin-1 has been shown to directly associate with CD147. This association apparently begins within the Golgi apparatus, with caveolin-1 specifically binding to and escorting the lower glycosylated forms of CD147 to the plasma membrane; thus preventing the formation of the self-aggregating, highly glycosylated species of CD147 which is responsible for MMP production (Tang et al. 2004). Caveolin-1 is proposed to serve as a negative regulator of CD147; the direct association of cav-1 with the second Ig domain of CD147 was found to decrease CD147 clustering (Tang et al. 2004). This hypothesis was supported by the generation of a CD147 mutant lacking the second Ig domain, which was incapable of associating with caveolin-1, as well as following the RNAi knockdown of caveolin-1, both of which resulted in impaired MMP production (Tang et al. 2004).
Recently, studies using murine hepatocarcinoma cell lines demonstrated an opposite effect of caveolin-1, namely that increased caveolin -1 expression results in an increased proportion of highly glycosylated CD147 relative to the lower glycosylated form and increased production of MMP-11 and higher invasiveness. In these studies down regulation of caveolin -1 resulted in a decrease in highly glycosylated CD147 (Jia et al. 2006). Regardless of the outcome of these studies, it is clear that the expression of CD147 glycosylation forms is functionally linked with caveolin -1 expression.
4. CD147 Function: clues from CD147 null mice
Just as CD147 has been found in association with several proteins and has been shown to be expressed on many cell types, the functions ascribed to CD147 are varied and are evidenced in the phenotype observed in the CD147 null mouse. One of the first observations regarding the CD147 null mouse was that implantation of the null fetus was severely impaired (Igakura et al. 1996; Naruhashi et al. 1997; Igakura et al. 1998). Although CD147 null embryos developed normally during the early stages of preimplantation, fewer than 8% of null embryos successfully implanted in the uterus of CD147 heterozygous females mated to heterozygous males (CD147 +/−) (Igakura et al. 1998). Increased expression of CD147 within the uterus of wildtype mice was observed at the stage of blastocyst implantation in the endometrium (Igakura et al. 1998; Kuno et al. 1998). Additionally, during the time of implantation increased levels of CD147 expression was detected in the trophectoderm of the wildtype embryo indicating the possibility that CD147 function, with respect to fetal development, is incumbent upon the induction of MMP production (Igakura et al. 1998); evidence in support of this notion was found in both mice and humans. Within human placentas, it is believed that CD147, through the induction of MMP production, may induce fetal membrane rupture and detachment from the uterus at the time of parturition (Li et al. 2004). Interestingly, CD147 null females were capable of supporting gestation of implanted CD147 wildtype embryos although with severely reduced efficiency (Igakura et al. 1998). Male CD147 null mice are sterile due to arrest of spermatogenesis at the metaphase of the first meiosis (Igakura et al. 1998). Together these observations indicate that CD147 expression within both the uterus and the embryo are required for successful implantation and suggest a functional interaction of CD147 with cell membrane proteins.
CD147 mediated transport of MCT1 and MCT4 to the plasma membrane is crucial to many processes. CD147 null mice were found to be deficient for MCT1, MCT3 and MCT4 within the retinal pigment epithelia, leading to blindness as a result of abnormal photoreceptor function (Hori et al. 2000; Ochrietor et al. 2002; Philp et al. 2003). Retinal development requires the coordinated shuttling of lactate between photoreceptor cells and Muller cells. Muller cells metabolize glucose as their main source of energy and, in the process, produce lactate as a major end product, while this lactate can be used to fuel oxidative phosphorylation within neighboring photoreceptor cells. MCT1, whose expression within the plasma membrane is incumbent upon its association with CD147, transports the lactate between these cells; therefore, the metabolic potential of both cell types are coupled to one another in an MCT1 dependent manner. The importance of this coupling is most evident in CD147 null mice. Absence of CD147 results in impaired surface expression of MCT1, which in turn results in the death of photoreceptor cells and ultimately blindness due to the lack of lactate transport (Philp et al. 2003). Retinal specific expression of the three Ig domain isoform of CD147, (Ochrietor et al. 2003) suggests that the homophillic interaction of this CD147 isoform may direct MCT1 specifically to areas of cell-cell contact (Hanna et al. 2003).
CD147 Function: an inducer of matrix metalloproteinases
CD147 was initially identified following the observation that interstitial collagenase (MMP-1) production was induced during the co-culturing of tumor cells and fibroblasts (Biswas 1982; Biswas 1984). Further studies revealed that CD147 is capable of inducing the expression of several MMPs other than MMP-1, including MMP-2, MMP-3, MMP-9 and MMP-11 and as such was renamed EMMPRIN/CD147 (Kataoka et al. 1993; Biswas et al. 1995; Guo et al. 1997). It was determined that either CD147 expressing tumor cells or conditioned media from the same cells were equally capable of inducing MMP production in co-cultured fibroblast cells (Biswas 1982; Biswas 1984). Increased MMP expression was obtained following the exogenous treatment of fibroblasts with recombinant CD147 protein as well as the adenoviral transduction of fibroblasts with CD147 in the absence of tumor cells (Li et al. 2001) These studies indicated that membrane bound and soluble CD147 are equally capable of inducing MMP production from fibroblast cells. The soluble form of CD147 has been detected in conditioned media as full length protein (Taylor et al. 2002) or as part of shed microvesicles (Sidhu et al. 2004), as well as in forms lacking the transmembrane and cytoplasmic domain derived from MMP mediated cleavage of CD147 from the cell surface (Haug et al. 2004; Tang et al. 2004; Egawa et al. 2006; Maatta et al. 2006).
Recent studies have addressed whether CD147 expressed on tumor cells induces production of MMP from surrounding peritumoral fibroblasts and whether CD147 could induce MMP production from tumor cells themselves. Other features that have been resolved include the fact that CD147, like other Ig containing molecules, interact homotypically. Sun and Hemler produced recombinant soluble CD147 fusion proteins and, using CD147 transfectants, determined that CD147 can interact homotypically via the D1 Ig domain. Additionally, soluble CD147 was shown to induce MMP production from fibroblasts expressing CD147 as well as from tumor cells themselves (Sun et al. 2001). These experiments do not rule out the possibility of a distinct receptor for CD147, however. Transfection of human breast cancer cells with CD147 cDNA followed by injection into mammary tissue of nude mice resulted in an enhancement of tumor growth and increase in metastatic incidences, both of which were directly correlated with high levels of tumor-derived MMP-2 and MMP-9 (Zucker et al. 2001). The induction of MMP-2 transcription was found to be directly linked to the glycosylation status of CD147 (Sun et al. 2001). Treatment of CD147 expressing MDA-435 breast carcinoma cells with either anti-CD147 antibody or purified, degylcosylated CD147 did not lead to the induction of MMP-1 and MMP-2 expression and treatment with deglycosylated CD147 antagonized the MMP induction capabilities of purified, native CD147, as well (Sun et al. 2001). Clearly there is a requirement for the N-linked glycosylation of CD147 in MMP induction.
Further evaluation of CD147 glycosylation demonstrated a link between the complexity of the attached sugars, CD147 function and cancer; whereby the CD147 expressed on cancer cells were found to contain the more complex β 1–6 branched polylactosamine sugars as opposed to the lower glycosylated species which typically contain mannose (Tang et al. 2004). The degree of glycosylation, the complexity of the associated sugars and their link to cancer is thought to occur at the level of cellular association with the extracellular matrix (ECM). It has been postulated that CD147 glycosylation allows for greater association with the ECM; this, coupled with the fact that glycosylation is required for the induction of MMP expression, suggest this process may serve as a regulatory mechanism of CD147 function as well as a possible therapeutic strategy (Hakomori 1996; Sun et al. 2001; Tang et al. 2004).
Role in lymphocyte migration and activation
Thymocyte maturation occurs through a series of distinct stages and is thought to require two distinct signals: one that regulates survival / selection of the population and one that initiates expansion of the population. While the pre-TCR signaling complex, comprised of TCRβ chain, pTα and a functional CD3 complex, was found to regulate selection / survival, evidence suggests that CD147 ligation can serve as the second signal necessary for expansion (Renno et al. 2002). Within the thymus, CD147 expression is highest on late stage, immature thymocytes, which correlates with transition to the next maturational level. The requirement of CD147 in T cell maturation was illustrated following the treatment of immature fetal lymphocytes with an anti-CD147 monoclonal antibody, which arrested any further development (Renno et al. 2002).
Within circulating immune cell populations, CD147 is highly expressed on activated T and B lymphocytes, as well as dendritic cells, monocytes and macrophage (Koch et al. 1999). CD147 was identified as an activation associated antigen on PHA activated T cells (Kasinrerk et al. 1992). Analysis of mRNA levels in both naïve and activated T cells revealed a 3.75-fold increase in CD147 transcription in the activated population (Zhang et al. 2002). Recently, increased expression of CD147 was observed on CD3+ T cells from systemic lupus erythematosus patients compared to healthy donor T cells, likely indicating their increased activation (Pistol et al. 2007). Engagement of CD147 with the MEM-M6/6 anti-CD147 monoclonal antibody was found to inhibit anti-CD3 induced T cell activation (Koch et al. 1999), presumably by inhibiting the reorganization and clustering of GPI-anchored co-receptors within lipid rafts which in turn alters down-stream signaling events (Staffler et al. 2003). For T cells, the level of CD147 expression also correlates with the state of differentiation, whereby immature thymocytes express higher CD147 levels than do mature resting T cells (Kirsch et al. 1997).
Blocking CD147 expressed on antigen presenting cells inhibits T cell activation. Studies on antibody pre-pulsed monoblastoid cells or peripheral blood dendritic cells co-cultured with T cells revealed inhibition of anti-CD3 induced T cell proliferation as well as inhibition of alloantigen induced T cell responses, suggesting that CD147-dependent T cell activation occurs at the level of the antigen presenting cell (APC) rather than the T cell (Stonehouse et al. 1999; Woodhead et al. 2000). This may be attributable to CD147’s function as an adhesion molecule. Studies performed with anti-CD147 treated human monoblastoid U937 cells demonstrated the importance of CD147-induced cellular adhesion and highlighted the involvement of the LFA-1/ICAM-1 pathway in this process (Kasinrerk et al. 1999). These findings, coupled with the fact that CD147 is expressed on both APCs and T cells, suggests a more general function for CD147 as a mediator of cell-cell interaction and regulator of antigen presentation (Woodhead et al. 2000).
The specific targeting of CD147 has proven efficacious in the treament of steroid-refractory acute graft-versus-host disease (GVHD) (Deeg et al. 2001). GVHD is a T cell mediated disorder whereby donor lymphocytes recognize host tissues as foreign and initiate immune responses against them. As a result, acute GVHD is a major complication following hematopoietic stem cell transplantations. Because CD147 is primarily expressed on activated T cell populations, those cells that would mediate GVHD can be selectively targeted without disrupting/depleting the entire complement of T cells. Administration of the anti-CD147 antibody ABX-CBL, an immunoglobulin M murine monoclonal antibody, extended the median survival time from 20 days post-transplantation to 117 days with little to no adverse side effects reported (Deeg et al. 2001).
5. CD147 in Disease: Cancer
The metastatic potential of cancerous cells is dependent upon their ability to disengage themselves from environmental cues that serve to regulate and constrain them. Typically, successful metastasis requires alterations in cytoskeletal architecture, expression of surface adhesion molecules and penetration of the basement membrane; phenomena that have been allied with known CD147 function. While the sequence of events leading to unregulated growth has yet to be elucidated, the induction of MMP activity has been shown to mediate the degradation of the ECM, thus alleviating the physical constraint to cell movement in vivo. The fact that CD147 is found over-expressed in a number of cancerous specimens, coupled with its ability to induce MMP expression, suggests that it serves as a key regulator of oncogenesis and that it is linked with one or more signaling pathways.
Evaluation of several signaling pathways has demonstrated a link between CD147 and the MAPK cascade (Lim et al. 1998). In metastatic ovarian carcinoma, the activities of the extracellular signal-regulated kinase (ERK), p38 and c-Jun N-terminal kinase (JNK) were found to be elevated concomitantly with increased MMP-2 production (Davidson et al. 2003). Further evaluation of these signaling pathways demonstrated that a distinct pattern existed between these signaling proteins and the protein expression patterns they subsequently induced. MMP-2 expression was found to correlate with activation of both ERK and p38 kinases, MMP-9 correlated positively with p38 activation and inversely with ERK expression and activation, while CD147 expression strongly correlated only with activated ERK (Davidson et al. 2003). While fibroblasts normally constitutively express MMPs at low levels, dysregulated MMP expression and activation aids in cancer cell migration. It has been widely documented that tumorigenic cells expressing CD147 induce MMP expression by neighboring fibroblasts (Biswas 1982; Kataoka et al. 1993; Biswas et al. 1995; Guo et al. 1997). Analyses of both tumor and peritumoral fibroblasts in breast, colon, lung, skin and head/neck tumors by in situ hybridization revealed that CD147 expression is primarily tumor associated, while MMP expression is fibroblast associated (Pyke et al. 1992; Majmudar et al. 1994; Noel et al. 1994; Heppner et al. 1996; Polette et al. 1997). Of the MMPs induced by CD147, malignant progression has been most closely correlated with the expression of MMP-2 in several forms of cancer (Sameshima et al. 2000; Ishikawa et al. 2004; Kawamura et al. 2004; Pasieka et al. 2004; Morgia et al. 2005; Redondo et al. 2005) and these increased levels are typically indicative of poor prognostic outcome (Vaisanen et al. 1996; Ishibashi et al. 2004; Morgia et al. 2005). Antibodies to CD147 have potential anti-cancer therapeutic utility and have been shown to decrease MMP expression leading to an inhibition of tumor cell invasion (Bordador et al. 2000; Sun et al. 2001; Kanekura et al. 2002).
Aside from stimulating MMP production in neighboring stromal cells, recent evidence suggests CD147 stimulates its own expression through a positive feedback mechanism and induces the production of vascular endothelial growth factor (VEGF) (Tang et al. 2005) as well as a soluble form of CD147 (Tang et al. 2004). The result of proteolytic cleavage of the carboxy terminus, the generation of a soluble form of CD147 increases the potential for stimulation of MMP expression from adjacent stromal cells to distal ones and is thought to aid metastasis to distant sites (Tang et al. 2004). Furthering the metastatic potential of CD147 expressing tumor cells is their ability to regulate the angiogenic process through the induction of VEGF (Tang et al. 2005). VEGF serves as a major regulator of the angiogenic process in a wide variety of circumstances, including tumor formation. In vitro and in vivo analyses demonstrate the inextricable link between CD147 expression and the induction of VEGF. In human breast cancer and glioblastoma cell lines, suppression of CD147 by antisense RNA not only reduced the production of MMP-2 and MMP-9 but also downregulated VEGF secretion (Liang et al. 2005; Tang et al. 2005). In vivo, tumor growth and angiogenesis were stimulated by the over-expression of CD147 and this phenomenon was inhibited following the antisense suppression of CD147 (Tang et al. 2005).
CD147 is associated with the α3β1 and α6β1 integrins which interact with laminins (Berditchevski et al. 1997). Integrins serve as the major receptors for cellular attachment to the ECM, therefore, it is not surprising that their interaction with laminin, the major component of the basement membrane, serves as a key event in the metastatic process (Engbring et al. 2003). The elevated expression levels of both α3β1 and α6β1 integrins has been witnessed in most metastatic and primary cancers (Bartolazzi et al. 1994; Demetriou et al. 2004). Comparison of normal prostate cells to cancerous cells showed a preferential pairing of α6 with β4 that transitioned to α6β1 in cancer cells (Demetriou et al. 2004). The significance of this transition, while still speculated, is most likely related to alterations in downstream signaling events.
Recent evidence suggests that stimulation of α3β1 activates signaling mechanisms that specifically promote tumor cell growth and progression (Choma et al. 2004; Manohar et al. 2004; Wang et al. 2004). The aberrant expression of α3β1 and α6β1 integrin species in many forms of cancer coupled with the fact that they are capable of associating with CD147 suggest these proteins work in concert to mediate ECM breakdown for tumor cell invasion to occur.
Tissue Repair/Remodeling
A delicate balance exists between MMP-induced stromal remodeling/restoration of homeostasis and stromal destruction; intuitively, MMP production must be a tightly regulated process. CD147 has been postulated to mediate this balance directly via a feed-back mechanism that links the affected epithelial cells with neighboring fibroblasts (Gabison et al. 2005). Within the eye, corneal ulcerations predominantly occur as a result of the breakdown of the protective epithelial barrier of the eye, leaving it susceptible to infection by bacteria, viruses and fungi and, if left untreated, they can result in blindness. CD147 has been identified in both healthy and ulcerated corneas, but was found at much greater levels within diseased corneas and its expression was found localized to areas of greater MMP expression (Gabison et al. 2005).
Within the cardiovascular system, this delicate balance is even more evident. MMP expression is critical for the prevention of hypertension and, at the same time, implicated in the progression of congestive heart failure (CHF) (Spinale et al. 2000; Ergul et al. 2004). Remodeling of the human vasculature for the maintenance of pliable vessels occurs through the constant degradation and regeneration of the ECM. Analyses of hypertensive patients identified a deficit in MMP production concomitant with decreased CD147 expression and increased production of collagen that could contribute to target organ damage, stroke, left ventricular hypertrophy and coronary artery disease (Ergul et al. 2004). Interestingly, greatly decreased CD147 expression and MMP levels were found in diabetic arteries implicating deregulation of CD147 in the diabetic vasculature where pathologic vascular remodeling is characterized by increased collagen deposition (Portik-Dobos et al. 2002).
End-stage CHF is typified by increased wall stress that leads to dysfunctional pumping of the heart resulting from tissue remodeling of the left ventricle. Evaluation of diseased and normal myocardium demonstrated CD147 expression in both populations; however, the diseased samples showed significantly higher levels as well as increased MMP expression (Spinale et al. 2000). Therefore, it is believed that the left ventricular problems associated with CHF are the direct result of dysregulated CD147 expression and MMP activity.
Ischemic disease
Low oxygen conditions exist in the brain during stroke and in the heart during myocardial infarction. During these conditions of hypoxia and ischemia cells within the tissues become dependent upon glycolysis for their energy metabolism. Transport of lactic acid that is produced as a byproduct of glycolysis is required for continued cell viability and is accomplished by the CD147 associated lactate transporters MCT-1 and MCT-4 (Halestrap et al. 1999; Kirk et al. 2000). Upregulation of MCT and CD147 expression is reported under ischemic conditions in neuronal and cardiac cells (Zhang et al. 2005; Han et al. 2006). The up-regulation of MCTs serves to protect cells from lactic acid buildup due to low oxygen levels, however, the coordinated up-regulation of CD147 expression has other effects on the cells and tissues as a result of CD147 interactions with other proteins.
CD147, in conjunction with CyPA, has been reported to protect neurons from oxidative stress and ischemia (Boulos et al. 2007). CD147 has been shown to serve as a receptor for CyPA (Yurchenko et al. 2002). The addition of CyPA to neuronal cultures resulted in activation of ERK1/2 (Boulos et al. 2007), similar to the downstream signaling events observed upon CD147/CyPA association (Yurchenko et al. 2002). CyPA levels are higher in the brain than they are in any other organ, predominantly localizing to neurons. Interestingly, CyPA was found to be released from cardiac myocytes upon hypoxia / reoxygenation in vitro or ischemia / reperfusion in vivo and was accompanied by upregulation of CD147 (Seko et al. 2004). The addition of rhCyPA to cardiac myocyte cultures led to the activation of ERK1/2, p38MAPK, SAPK and Akt. Cardiac mycoytes may increase the secretion of CyPA and expression of CD147 in response to hypoxia/reoxygenation as a means to initiate pro-survival signaling in an autocrine fashion thus limiting the amount of cell death (Seko et al. 2004).
Although the CD147 / CyPA association and related signaling may provide protection to neurons and cardiac myocytes from ischemic damage, the induction of matrix metalloproteinases through increased CD147 expression may promote ischemic related pathology by increasing tissue destruction. Increased levels of CD147 within the basal ganglia and cortexes of ischemic brains correlated with increased MMP-2 and MMP-9 levels potentially resulting in destruction of the brain microvasculature following an ischemic event (Burggraf et al. 2005). In cardiac disease CD147, MT-MMP-1, MMP-2 and MMP-9 levels are increased in patients suffering from acute myocardial infarction (MI) but not those with stable angina (Schmidt et al. 2006). MIs are induced by the rupturing of atherosclerotic plaques; a process that is mediated by MMPs, all of which have been detected within vulnerable plaques.
Alzheimer’s disease
Alzheimer’s Disease is a neurodegenerative disease characterized by progressive cognitive deficiency that is manifested by impaired spatial reasoning, motor commands and language skills. It is the errant production of amyloid β peptides that is believed to lead to the formation of the characteristic amyloid plaques and neurofibrillary tangles within the neocortical and subcortical regions of the brain resulting in disease pathology. The γ secretase complex regulates production of these amyloid β peptides reviewed in (Caltagarone et al. 2006; Zhou et al. 2007). Recently CD147 was observed to be a regulatory component of the γ secretase complex (Zhou et al. 2005). Depletion of CD147 from the γ secretase complex through RNA interference in cell lines resulted in an increase in amyloid β peptide production but had no effect on the expression levels of other complex components or complex substrates suggesting that CD147 may regulate γ secretase activity (Zhou et al. 2005). Interestingly, neurological abnormalities observed in CD147 null mice including spatial learning and memory deficits (Naruhashi et al. 1997) are similar to those observed in mouse models of Alzheimer’s disease (Higgins et al. 2003; Richardson et al. 2003). Although the mechanism by which CD147 attenuates the production of amyloid β peptides is unknown, further study of CD147 function within the γ secretase complex may aid Alzheimer’s disease therapy. Interestingly, CD147 associated α6β1 integrins bind amyloid β peptides initiating a signal transduction cascade in microglial cells (Bamberger et al. 2003) indicating the potential for another level of regulation influenced by CD147 expression in Alzheimer’s disease.
6. Hypoxic induction of MCT and CD147 expression promotes tumor progression
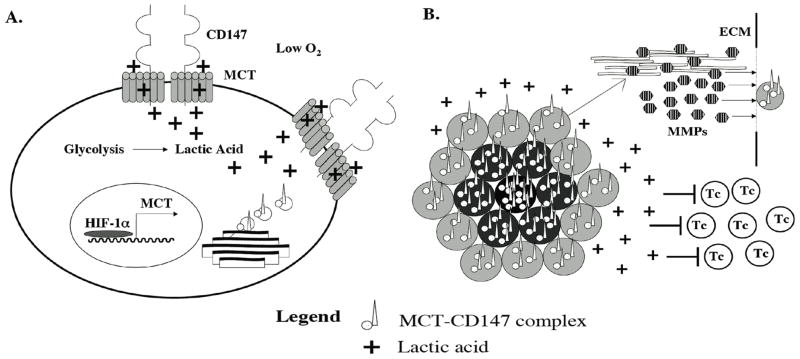
As tumor size increases hypoxic conditions arise within the tumor microenvironment resulting in the induction of hypoxia inducible factor, HIF-1α, a transcription factor that has been shown to induce MCT-4 gene expression in cells (Moeller et al. 2005; Moeller et al. 2006). Upregulation of MCTs in tumor cells is advantageous to tumor survival. Rapid tumor proliferation under hypoxic conditions is mediated by glycolysis which produces increased levels of intracellular lactic acid requiring export via MCTs (Figure 1A). Due to the action of upregulated MCTs the concentration of lactic acid increases in the extracellular tumor microenvironment as well. This excess lactic acid has been determined to inhibit peritumoral cytotoxic T cell function, thus permitting continued unchecked growth of the tumor (Fischer et al. 2007). Since MCT surface expression is linked to CD147 due to the requirement for interaction between the two proteins for successful cell surface expression (Kirk et al. 2000), a coordinate upregulation of CD147 is observed on tumor cells. Indeed, screening of numerous clinical tumor specimens consistently reveals a link between the most aggressive tumors and the highest expression levels of CD147 (Davidson et al. 2003; Rosenthal et al. 2003; Ishibashi et al. 2004; Reimers et al. 2004; Li et al. 2005; Cheng et al. 2006; Jin et al. 2006; Jin et al. 2006; Nabeshima et al. 2006; Riethdorf et al. 2006; Vigneswaran et al. 2006; Zhang et al. 2006; Zheng et al. 2006). Increased expression of CD147 results in induction of increased MMP production by peritumoral fibrobasts resulting in degradation of the extracellular matrix and a favorable environment for tumor metastasis (Figure 1B). Inhibition of CD147, MCT, or HIF-1α within tumor cells would coordinately decrease metastatic potential and tumor progression.
Figure 1. The CD147-MCT complex promotes tumor progression.
A.) Under hypoxic conditions within the tumor microenvironment rapid tumor proliferation is achieved by anaerobic glycolysis, the byproduct of which is lactic acid. Coordinate upregulation of hypoxia inducible factor, HIF-1α, results in increased transcription of MCT (Moeller et al. 2005; Moeller et al. 2006) and increased cell surface expression of the CD147-MCT complex after their assembly in the Golgi apparatus. Tumor viability is maintained by the MCT facilitated transport of lactic acid into the peritumoral space thus regulating intracellular pH. B) Peritumoral cytotoxic T cell (Tc) function is inhibited by increased lactic acid in the tumor microenvironment (Fischer et al. 2007) enabling unchecked tumor growth. Increased CD147 expression induces MMP production by neighboring fibroblasts (Kataoka et al. 1993; Biswas et al. 1995; Guo et al. 1997; Sun et al. 2001) resulting in breakdown of the extracellular matrix and metastasis of CD147 expressing tumor cells.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Allain F, Vanpouille C, Carpentier M, Slomianny MC, Durieux S, Spik G. Interaction with glycosaminoglycans is required for cyclophilin B to trigger integrin-mediated adhesion of peripheral blood T lymphocytes to extracellular matrix. Proc Natl Acad Sci U S A. 2002;99:2714–2719. doi: 10.1073/pnas.052284899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Altruda F, Cervella P, Gaeta ML, Daniele A, Giancotti F, Tarone G, Stefanuto G, Silengo L. Cloning of cDNA for a novel mouse membrane glycoprotein (gp42): shared identity to histocompatibility antigens, immunoglobulins and neural-cell adhesion molecules. Gene. 1989;85:445–451. doi: 10.1016/0378-1119(89)90438-1. [DOI] [PubMed] [Google Scholar]
- Arora K, Gwinn WM, Bower MA, Watson A, Okwumabua I, MacDonald HR, Bukrinsky MI, Constant SL. Extracellular cyclophilins contribute to the regulation of inflammatory responses. J Immunol. 2005;175:517–522. doi: 10.4049/jimmunol.175.1.517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bamberger ME, Harris ME, McDonald DR, Husemann J, Landreth GE. A cell surface receptor complex for fibrillar beta-amyloid mediates microglial activation. J Neurosci. 2003;23:2665–2674. doi: 10.1523/JNEUROSCI.23-07-02665.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bartolazzi A, Cerboni C, Nicotra MR, Mottolese M, Bigotti A, Natali PG. Transformation and tumor progression are frequently associated with expression of the alpha 3/beta 1 heterodimer in solid tumors. Int J Cancer. 1994;58:488–491. doi: 10.1002/ijc.2910580405. [DOI] [PubMed] [Google Scholar]
- Berditchevski F, Chang S, Bodorova J, Hemler ME. Generation of monoclonal antibodies to integrin-associated proteins. Evidence that alpha3beta1 complexes with EMMPRIN/basigin/OX47/M6. J Biol Chem. 1997;272:29174–29180. doi: 10.1074/jbc.272.46.29174. [DOI] [PubMed] [Google Scholar]
- Biswas C. Tumor cell stimulation of collagenase production by fibroblasts. Biochem Biophys Res Commun. 1982;109:1026–1034. doi: 10.1016/0006-291x(82)92042-3. [DOI] [PubMed] [Google Scholar]
- Biswas C. Collagenase stimulation in cocultures of human fibroblasts and human tumor cells. Cancer Lett. 1984;24:201–207. doi: 10.1016/0304-3835(84)90137-x. [DOI] [PubMed] [Google Scholar]
- Biswas C, Nugent MA. Membrane association of collagenase stimulatory factor(s) from B-16 melanoma cells. J Cell Biochem. 1987;35:247–258. doi: 10.1002/jcb.240350307. [DOI] [PubMed] [Google Scholar]
- Biswas C, Zhang Y, DeCastro R, Guo H, Nakamura T, Kataoka H, Nabeshima K. The human tumor cell-derived collagenase stimulatory factor (renamed EMMPRIN) is a member of the immunoglobulin superfamily. Cancer Res. 1995;55:434–439. [PubMed] [Google Scholar]
- Bordador LC, Li X, Toole B, Chen B, Regezi J, Zardi L, Hu Y, Ramos DM. Expression of emmprin by oral squamous cell carcinoma. Int J Cancer. 2000;85:347–352. [PubMed] [Google Scholar]
- Boulos S, Meloni BP, Arthur PG, Majda B, Bojarski C, Knuckey NW. Evidence that intracellular cyclophilin A and cyclophilin A/CD147 receptor-mediated ERK1/2 signalling can protect neurons against in vitro oxidative and ischemic injury. Neurobiol Dis. 2007;25:54–64. doi: 10.1016/j.nbd.2006.08.012. [DOI] [PubMed] [Google Scholar]
- Braaten D, Franke EK, Luban J. Cyclophilin A is required for the replication of group M human immunodeficiency virus type 1 (HIV-1) and simian immunodeficiency virus SIV(CPZ)GAB but not group O HIV-1 or other primate immunodeficiency viruses. J Virol. 1996;70:4220–4227. doi: 10.1128/jvi.70.7.4220-4227.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burggraf D, Liebetrau M, Martens HK, Wunderlich N, Jager G, Dichgans M, Hamann GF. Matrix metalloproteinase induction by EMMPRIN in experimental focal cerebral ischemia. Eur J Neurosci. 2005;22:273–277. doi: 10.1111/j.1460-9568.2005.04187.x. [DOI] [PubMed] [Google Scholar]
- Caltagarone J, Jing Z, Bowser R. Focal adhesions regulate Abeta signaling and cell death in Alzheimer’s disease. Biochim Biophys Acta. 2006 doi: 10.1016/j.bbadis.2006.11.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng MF, Tzao C, Tsai WC, Lee WH, Chen A, Chiang H, Sheu LF, Jin JS. Expression of EMMPRIN and matriptase in esophageal squamous cell carcinoma: correlation with clinicopathological parameters. Dis Esophagus. 2006;19:482–486. doi: 10.1111/j.1442-2050.2006.00613.x. [DOI] [PubMed] [Google Scholar]
- Choma DP, Pumiglia K, DiPersio CM. Integrin alpha3beta1 directs the stabilization of a polarized lamellipodium in epithelial cells through activation of Rac1. J Cell Sci. 2004;117:3947–3959. doi: 10.1242/jcs.01251. [DOI] [PubMed] [Google Scholar]
- Colley NJ, Baker EK, Stamnes MA, Zuker CS. The cyclophilin homolog ninaA is required in the secretory pathway. Cell. 1991;67:255–263. doi: 10.1016/0092-8674(91)90177-z. [DOI] [PubMed] [Google Scholar]
- Coss RA, Storck CW, Daskalakis C, Berd D, Wahl ML. Intracellular acidification abrogates the heat shock response and compromises survival of human melanoma cells. Mol Cancer Ther. 2003;2:383–388. [PubMed] [Google Scholar]
- Curtin KD, Meinertzhagen IA, Wyman RJ. Basigin (EMMPRIN/CD147) interacts with integrin to affect cellular architecture. J Cell Sci. 2005;118:2649–2660. doi: 10.1242/jcs.02408. [DOI] [PubMed] [Google Scholar]
- Davidson B, Givant-Horwitz V, Lazarovici P, Risberg B, Nesland JM, Trope CG, Schaefer E, Reich R. Matrix metalloproteinases (MMP), EMMPRIN (extracellular matrix metalloproteinase inducer) and mitogen-activated protein kinases (MAPK): co-expression in metastatic serous ovarian carcinoma. Clin Exp Metastasis. 2003;20:621–631. doi: 10.1023/a:1027347932543. [DOI] [PubMed] [Google Scholar]
- Davidson B, Goldberg I, Berner A, Kristensen GB, Reich R. EMMPRIN (extracellular matrix metalloproteinase inducer) is a novel marker of poor outcome in serous ovarian carcinoma. Clin Exp Metastasis. 2003;20:161–169. doi: 10.1023/a:1022696012668. [DOI] [PubMed] [Google Scholar]
- Deeg HJ, Blazar BR, Bolwell BJ, Long GD, Schuening F, Cunningham J, Rifkin RM, Abhyankar S, Briggs AD, Burt R, Lipani J, Roskos LK, White JM, Havrilla N, Schwab G, Heslop HE. Treatment of steroid-refractory acute graft-versus-host disease with anti-CD147 monoclonal antibody ABX-CBL. Blood. 2001;98:2052–2058. doi: 10.1182/blood.v98.7.2052. [DOI] [PubMed] [Google Scholar]
- Delwel GO, de Melker AA, Hogervorst F, Jaspars LH, Fles DL, Kuikman I, Lindblom A, Paulsson M, Timpl R, Sonnenberg A. Distinct and overlapping ligand specificities of the alpha 3A beta 1 and alpha 6A beta 1 integrins: recognition of laminin isoforms. Mol Biol Cell. 1994;5:203–215. doi: 10.1091/mbc.5.2.203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Demetriou MC, Cress AE. Integrin clipping: a novel adhesion switch? J Cell Biochem. 2004;91:26–35. doi: 10.1002/jcb.10675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Denys A, Allain F, Carpentier M, Spik G. Involvement of two classes of binding sites in the interactions of cyclophilin B with peripheral blood T-lymphocytes. Biochem J. 1998;336(Pt 3):689–697. doi: 10.1042/bj3360689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Egawa N, Koshikawa N, Tomari T, Nabeshima K, Isobe T, Seiki M. Membrane type 1 matrix metalloproteinase (MT1-MMP/MMP-14) cleaves and releases a 22-kDa extracellular matrix metalloproteinase inducer (EMMPRIN) fragment from tumor cells. J Biol Chem. 2006;281:37576–37585. doi: 10.1074/jbc.M606993200. [DOI] [PubMed] [Google Scholar]
- Ekblom P, Lonai P, Talts JF. Expression and biological role of laminin-1. Matrix Biol. 2003;22:35–47. doi: 10.1016/s0945-053x(03)00015-5. [DOI] [PubMed] [Google Scholar]
- Ellis SM, Nabeshima K, Biswas C. Monoclonal antibody preparation and purification of a tumor cell collagenase-stimulatory factor. Cancer Res. 1989;49:3385–3391. [PubMed] [Google Scholar]
- Engbring JA, Kleinman HK. The basement membrane matrix in malignancy. J Pathol. 2003;200:465–470. doi: 10.1002/path.1396. [DOI] [PubMed] [Google Scholar]
- Ergul A, Portik-Dobos V, Hutchinson J, Franco J, Anstadt MP. Downregulation of vascular matrix metalloproteinase inducer and activator proteins in hypertensive patients. Am J Hypertens. 2004;17:775–782. doi: 10.1016/j.amjhyper.2004.06.025. [DOI] [PubMed] [Google Scholar]
- Fadool JM, Linser PJ. Differential glycosylation of the 5A11/HT7 antigen by neural retina and epithelial tissues in the chicken. J Neurochem. 1993;60:1354–1364. doi: 10.1111/j.1471-4159.1993.tb03296.x. [DOI] [PubMed] [Google Scholar]
- Fang J, Quinones QJ, Holman TL, Morowitz MJ, Wang Q, Zhao H, Sivo F, Maris JM, Wahl ML. The H+-linked monocarboxylate transporter (MCT1/SLC16A1): a potential therapeutic target for high-risk neuroblastoma. Mol Pharmacol. 2006;70:2108–2115. doi: 10.1124/mol.106.026245. [DOI] [PubMed] [Google Scholar]
- Fischer K, Hoffmann P, Voelkl S, Meidenbauer N, Ammer J, Edinger M, Gottfried E, Schwarz S, Rothe G, Hoves S, Renner K, Timischl B, Mackensen A, Kunz-Schughart L, Andreesen R, Krause SW, Kreutz M. Inhibitory effect of tumor cell derived lactic acid on human T cells. Blood. 2007 doi: 10.1182/blood-2006-07-035972. [DOI] [PubMed] [Google Scholar]
- Fossum S, Mallett S, Barclay AN. The MRC OX-47 antigen is a member of the immunoglobulin superfamily with an unusual transmembrane sequence. Eur J Immunol. 1991;21:671–679. doi: 10.1002/eji.1830210320. [DOI] [PubMed] [Google Scholar]
- Froberg MK, Gerhart DZ, Enerson BE, Manivel C, Guzman-Paz M, Seacotte N, Drewes LR. Expression of monocarboxylate transporter MCT1 in normal and neoplastic human CNS tissues. Neuroreport. 2001;12:761–765. doi: 10.1097/00001756-200103260-00030. [DOI] [PubMed] [Google Scholar]
- Gabison EE, Hoang-Xuan T, Mauviel A, Menashi S. EMMPRIN/CD147, an MMP modulator in cancer, development and tissue repair. Biochimie. 2005;87:361–368. doi: 10.1016/j.biochi.2004.09.023. [DOI] [PubMed] [Google Scholar]
- Gabison EE, Mourah S, Steinfels E, Yan L, Hoang-Xuan T, Watsky MA, De Wever B, Calvo F, Mauviel A, Menashi S. Differential expression of extracellular matrix metalloproteinase inducer (CD147) in normal and ulcerated corneas: role in epithelio-stromal interactions and matrix metalloproteinase induction. Am J Pathol. 2005;166:209–219. doi: 10.1016/S0002-9440(10)62245-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galat A. Peptidylproline cis-trans-isomerases: immunophilins. Eur J Biochem. 1993;216:689–707. doi: 10.1111/j.1432-1033.1993.tb18189.x. [DOI] [PubMed] [Google Scholar]
- Gallagher SM, Castorino JJ, Wang D, Philp NJ. Monocarboxylate transporter 4 regulates maturation and trafficking of CD147 to the plasma membrane in the metastatic breast cancer cell line MDA-MB-231. Cancer Res. 2007;67:4182–4189. doi: 10.1158/0008-5472.CAN-06-3184. [DOI] [PubMed] [Google Scholar]
- Guo H, Zucker S, Gordon MK, Toole BP, Biswas C. Stimulation of matrix metalloproteinase production by recombinant extracellular matrix metalloproteinase inducer from transfected Chinese hamster ovary cells. J Biol Chem. 1997;272:24–27. [PubMed] [Google Scholar]
- Gwinn WM, Damsker JM, Falahati R, Okwumabua I, Kelly-Welch A, Keegan AD, Vanpouille C, Lee JJ, Dent LA, Leitenberg D, Bukrinsky MI, Constant SL. Novel approach to inhibit asthma-mediated lung inflammation using anti-CD147 intervention. J Immunol. 2006;177:4870–4879. doi: 10.4049/jimmunol.177.7.4870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hakomori S. Tumor malignancy defined by aberrant glycosylation and sphingo(glyco)lipid metabolism. Cancer Res. 1996;56:5309–5318. [PubMed] [Google Scholar]
- Halestrap AP, Price NT. The proton-linked monocarboxylate transporter (MCT) family: structure, function and regulation. Biochem J. 1999;343(Pt 2):281–299. [PMC free article] [PubMed] [Google Scholar]
- Han M, Trotta P, Coleman C, Linask KK. MCT-4, A511/Basigin and EF5 expression patterns during early chick cardiomyogenesis indicate cardiac cell differentiation occurs in a hypoxic environment. Dev Dyn. 2006;235:124–131. doi: 10.1002/dvdy.20531. [DOI] [PubMed] [Google Scholar]
- Hanna SM, Kirk P, Holt OJ, Puklavec MJ, Brown MH, Barclay AN. A novel form of the membrane protein CD147 that contains an extra Ig-like domain and interacts homophilically. BMC Biochem. 2003;4:17. doi: 10.1186/1471-2091-4-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haug C, Lenz C, Diaz F, Bachem MG. Oxidized low-density lipoproteins stimulate extracellular matrix metalloproteinase Inducer (EMMPRIN) release by coronary smooth muscle cells. Arterioscler Thromb Vasc Biol. 2004;24:1823–1829. doi: 10.1161/01.ATV.0000142806.59283.11. [DOI] [PubMed] [Google Scholar]
- Heppner KJ, Matrisian LM, Jensen RA, Rodgers WH. Expression of most matrix metalloproteinase family members in breast cancer represents a tumor-induced host response. Am J Pathol. 1996;149:273–282. [PMC free article] [PubMed] [Google Scholar]
- Higgins GA, Jacobsen H. Transgenic mouse models of Alzheimer’s disease: phenotype and application. Behav Pharmacol. 2003;14:419–438. doi: 10.1097/01.fbp.0000088420.18414.ff. [DOI] [PubMed] [Google Scholar]
- Hori K, Katayama N, Kachi S, Kondo M, Kadomatsu K, Usukura J, Muramatsu T, Mori S, Miyake Y. Retinal dysfunction in basigin deficiency. Invest Ophthalmol Vis Sci. 2000;41:3128–3133. [PubMed] [Google Scholar]
- Huang T, Deng H, Wolkoff AW, Stockert RJ. Phosphorylation-dependent interaction of the asialoglycoprotein receptor with molecular chaperones. J Biol Chem. 2002;277:37798–37803. doi: 10.1074/jbc.M204786200. [DOI] [PubMed] [Google Scholar]
- Igakura T, Kadomatsu K, Kaname T, Muramatsu H, Fan QW, Miyauchi T, Toyama Y, Kuno N, Yuasa S, Takahashi M, Senda T, Taguchi O, Yamamura K, Arimura K, Muramatsu T. A null mutation in basigin, an immunoglobulin superfamily member, indicates its important roles in peri-implantation development and spermatogenesis. Dev Biol. 1998;194:152–165. doi: 10.1006/dbio.1997.8819. [DOI] [PubMed] [Google Scholar]
- Igakura T, Kadomatsu K, Taguchi O, Muramatsu H, Kaname T, Miyauchi T, Yamamura K, Arimura K, Muramatsu T. Roles of basigin, a member of the immunoglobulin superfamily, in behavior as to an irritating odor, lymphocyte response, and blood-brain barrier. Biochem Biophys Res Commun. 1996;224:33–36. doi: 10.1006/bbrc.1996.0980. [DOI] [PubMed] [Google Scholar]
- Ishibashi Y, Matsumoto T, Niwa M, Suzuki Y, Omura N, Hanyu N, Nakada K, Yanaga K, Yamada K, Ohkawa K, Kawakami M, Urashima M. CD147 and matrix metalloproteinase-2 protein expression as significant prognostic factors in esophageal squamous cell carcinoma. Cancer. 2004;101:1994–2000. doi: 10.1002/cncr.20593. [DOI] [PubMed] [Google Scholar]
- Ishikawa S, Takenaka K, Yanagihara K, Miyahara R, Kawano Y, Otake Y, Hasegawa S, Wada H, Tanaka F. Matrix metalloproteinase-2 status in stromal fibroblasts, not in tumor cells, is a significant prognostic factor in non-small-cell lung cancer. Clin Cancer Res. 2004;10:6579–6585. doi: 10.1158/1078-0432.CCR-04-0272. [DOI] [PubMed] [Google Scholar]
- Jia L, Wang S, Zhou H, Cao J, Hu Y, Zhang J. Caveolin-1 up-regulates CD147 glycosylation and the invasive capability of murine hepatocarcinoma cell lines. Int J Biochem Cell Biol. 2006;38:1584–1593. doi: 10.1016/j.biocel.2006.03.019. [DOI] [PubMed] [Google Scholar]
- Jiang JL, Zhou Q, Yu MK, Ho LS, Chen ZN, Chan HC. The involvement of HAb18G/CD147 in regulation of store-operated calcium entry and metastasis of human hepatoma cells. J Biol Chem. 2001;276:46870–46877. doi: 10.1074/jbc.M108291200. [DOI] [PubMed] [Google Scholar]
- Jin JS, Hsieh DS, Lin YF, Wang JY, Sheu LF, Lee WH. Increasing expression of extracellular matrix metalloprotease inducer in renal cell carcinoma: tissue microarray analysis of immunostaining score with clinicopathological parameters. Int J Urol. 2006;13:573–580. doi: 10.1111/j.1442-2042.2006.01353.x. [DOI] [PubMed] [Google Scholar]
- Jin JS, Yao CW, Loh SH, Cheng MF, Hsieh DS, Bai CY. Increasing expression of extracellular matrix metalloprotease inducer in ovary tumors: tissue microarray analysis of immunostaining score with clinicopathological parameters. Int J Gynecol Pathol. 2006;25:140–146. doi: 10.1097/01.pgp.0000189244.57145.84. [DOI] [PubMed] [Google Scholar]
- Kanekura T, Chen X, Kanzaki T. Basigin (CD147) is expressed on melanoma cells and induces tumor cell invasion by stimulating production of matrix metalloproteinases by fibroblasts. Int J Cancer. 2002;99:520–528. doi: 10.1002/ijc.10390. [DOI] [PubMed] [Google Scholar]
- Kasinrerk W, Fiebiger E, Stefanova I, Baumruker T, Knapp W, Stockinger H. Human leukocyte activation antigen M6, a member of the Ig superfamily, is the species homologue of rat OX-47, mouse basigin, and chicken HT7 molecule. J Immunol. 1992;149:847–854. [PubMed] [Google Scholar]
- Kasinrerk W, Tokrasinwit N, Phunpae P. CD147 monoclonal antibodies induce homotypic cell aggregation of monocytic cell line U937 via LFA-1/ICAM-1 pathway. Immunology. 1999;96:184–192. doi: 10.1046/j.1365-2567.1999.00653.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kataoka H, DeCastro R, Zucker S, Biswas C. Tumor cell-derived collagenase-stimulatory factor increases expression of interstitial collagenase, stromelysin, and 72-kDa gelatinase. Cancer Res. 1993;53:3154–3158. [PubMed] [Google Scholar]
- Kawamura K, Kamiya N, Suyama T, Shimbo M, Oosumi N, Suzuki H, Ueda T, Tobe T, Igarashi T, Ito H, Ishikura H. In situ gelatinolytic activity correlates with tumor progression and prognosis in patients with bladder cancer. J Urol. 2004;172:1480–1484. doi: 10.1097/01.ju.0000137749.99255.3e. [DOI] [PubMed] [Google Scholar]
- Keely P, Parise L, Juliano R. Integrins and GTPases in tumour cell growth, motility and invasion. Trends Cell Biol. 1998;8:101–106. doi: 10.1016/s0962-8924(97)01219-1. [DOI] [PubMed] [Google Scholar]
- Kikkawa Y, Sanzen N, Sekiguchi K. Isolation and characterization of laminin-10/11 secreted by human lung carcinoma cells. laminin-10/11 mediates cell adhesion through integrin alpha3 beta1. J Biol Chem. 1998;273:15854–15859. doi: 10.1074/jbc.273.25.15854. [DOI] [PubMed] [Google Scholar]
- Kirk P, Wilson MC, Heddle C, Brown MH, Barclay AN, Halestrap AP. CD147 is tightly associated with lactate transporters MCT1 and MCT4 and facilitates their cell surface expression. Embo J. 2000;19:3896–3904. doi: 10.1093/emboj/19.15.3896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kirsch AH, Diaz LA, Jr, Bonish B, Antony PA, Fox DA. The pattern of expression of CD147/neurothelin during human T-cell ontogeny as defined by the monoclonal antibody 8D6. Tissue Antigens. 1997;50:147–152. doi: 10.1111/j.1399-0039.1997.tb02853.x. [DOI] [PubMed] [Google Scholar]
- Koch C, Staffler G, Huttinger R, Hilgert I, Prager E, Cerny J, Steinlein P, Majdic O, Horejsi V, Stockinger H. T cell activation-associated epitopes of CD147 in regulation of the T cell response, and their definition by antibody affinity and antigen density. Int Immunol. 1999;11:777–786. doi: 10.1093/intimm/11.5.777. [DOI] [PubMed] [Google Scholar]
- Konttinen YT, Li TF, Mandelin J, Liljestrom M, Sorsa T, Santavirta S, Virtanen I. Increased expression of extracellular matrix metalloproteinase inducer in rheumatoid synovium. Arthritis Rheum. 2000;43:275–280. doi: 10.1002/1529-0131(200002)43:2<275::AID-ANR6>3.0.CO;2-#. [DOI] [PubMed] [Google Scholar]
- Kuno N, Kadomatsu K, Fan QW, Hagihara M, Senda T, Mizutani S, Muramatsu T. Female sterility in mice lacking the basigin gene, which encodes a transmembrane glycoprotein belonging to the immunoglobulin superfamily. FEBS Lett. 1998;425:191–194. doi: 10.1016/s0014-5793(98)00213-0. [DOI] [PubMed] [Google Scholar]
- Li HG, Xie DR, Shen XM, Li HH, Zeng H, Zeng YJ. Clinicopathological significance of expression of paxillin, syndecan-1 and EMMPRIN in hepatocellular carcinoma. World J Gastroenterol. 2005;11:1445–1451. doi: 10.3748/wjg.v11.i10.1445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li R, Huang L, Guo H, Toole BP. Basigin (murine EMMPRIN) stimulates matrix metalloproteinase production by fibroblasts. J Cell Physiol. 2001;186:371–379. doi: 10.1002/1097-4652(2000)9999:999<000::AID-JCP1042>3.0.CO;2-8. [DOI] [PubMed] [Google Scholar]
- Li W, Alfaidy N, Challis JR. Expression of extracellular matrix metalloproteinase inducer in human placenta and fetal membranes at term labor. J Clin Endocrinol Metab. 2004;89:2897–2904. doi: 10.1210/jc.2003-032048. [DOI] [PubMed] [Google Scholar]
- Liang Q, Xiong H, Gao G, Xiong K, Wang X, Zhao Z, Zhang H, Li Y. Inhibition of Basigin Expression in Glioblastoma Cell Line via Antisense RNA Reduces Tumor Cell Invasion and Angiogenesis. Cancer Biol Ther. 2005;4:759–762. doi: 10.4161/cbt.4.7.1828. [DOI] [PubMed] [Google Scholar]
- Lim M, Martinez T, Jablons D, Cameron R, Guo H, Toole B, Li JD, Basbaum C. Tumor-derived EMMPRIN (extracellular matrix metalloproteinase inducer) stimulates collagenase transcription through MAPK p38. FEBS Lett. 1998;441:88–92. doi: 10.1016/s0014-5793(98)01474-4. [DOI] [PubMed] [Google Scholar]
- Maatta M, Tervahartiala T, Kaarniranta K, Tang Y, Yan L, Tuukkanen J, Sorsa T. Immunolocalization of EMMPRIN (CD147) in the human eye and detection of soluble form of EMMPRIN in ocular fluids. Curr Eye Res. 2006;31:917–924. doi: 10.1080/02713680600932290. [DOI] [PubMed] [Google Scholar]
- Majmudar G, Nelson BR, Jensen TC, Johnson TM. Increased expression of matrix metalloproteinase-3 (stromelysin-1) in cultured fibroblasts and basal cell carcinomas of nevoid basal cell carcinoma syndrome. Mol Carcinog. 1994;11:29–33. doi: 10.1002/mc.2940110106. [DOI] [PubMed] [Google Scholar]
- Malinda KM, Kleinman HK. The laminins. Int J Biochem Cell Biol. 1996;28:957–959. doi: 10.1016/1357-2725(96)00042-8. [DOI] [PubMed] [Google Scholar]
- Malinda KM, Nomizu M, Chung M, Delgado M, Kuratomi Y, Yamada Y, Kleinman HK, Ponce ML. Identification of laminin alpha1 and beta1 chain peptides active for endothelial cell adhesion, tube formation, and aortic sprouting. Faseb J. 1999;13:53–62. [PubMed] [Google Scholar]
- Manohar A, Shome SG, Lamar J, Stirling L, Iyer V, Pumiglia K, DiPersio CM. Alpha 3 beta 1 integrin promotes keratinocyte cell survival through activation of a MEK/ERK signaling pathway. J Cell Sci. 2004;117:4043–4054. doi: 10.1242/jcs.01277. [DOI] [PubMed] [Google Scholar]
- Mathupala SP, Parajuli P, Sloan AE. Silencing of monocarboxylate transporters via small interfering ribonucleic acid inhibits glycolysis and induces cell death in malignant glioma: an in vitro study. Neurosurgery. 2004;55:1410–1419. doi: 10.1227/01.neu.0000143034.62913.59. discussion 1419. [DOI] [PubMed] [Google Scholar]
- Miyauchi T, Jimma F, Igakura T, Yu S, Ozawa M, Muramatsu T. Structure of the mouse basigin gene, a unique member of the immunoglobulin superfamily. J Biochem (Tokyo) 1995;118:717–724. doi: 10.1093/oxfordjournals.jbchem.a124971. [DOI] [PubMed] [Google Scholar]
- Miyauchi T, Masuzawa Y, Muramatsu T. The basigin group of the immunoglobulin superfamily: complete conservation of a segment in and around transmembrane domains of human and mouse basigin and chicken HT7 antigen. J Biochem (Tokyo) 1991;110:770–774. doi: 10.1093/oxfordjournals.jbchem.a123657. [DOI] [PubMed] [Google Scholar]
- Moeller LC, Cao X, Dumitrescu AM, Seo H, Refetoff S. Thyroid hormone mediated changes in gene expression can be initiated by cytosolic action of the thyroid hormone receptor beta through the phosphatidylinositol 3-kinase pathway. Nucl Recept Signal. 2006;4:e020. doi: 10.1621/nrs.04020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moeller LC, Dumitrescu AM, Refetoff S. Cytosolic action of thyroid hormone leads to induction of hypoxia-inducible factor-1alpha and glycolytic genes. Mol Endocrinol. 2005;19:2955–2963. doi: 10.1210/me.2004-0542. [DOI] [PubMed] [Google Scholar]
- Morgia G, Falsaperla M, Malaponte G, Madonia M, Indelicato M, Travali S, Mazzarino MC. Matrix metalloproteinases as diagnostic (MMP-13) and prognostic (MMP-2, MMP-9) markers of prostate cancer. Urol Res. 2005;33:44–50. doi: 10.1007/s00240-004-0440-8. [DOI] [PubMed] [Google Scholar]
- Morini M, Mottolese M, Ferrari N, Ghiorzo F, Buglioni S, Mortarini R, Noonan DM, Natali PG, Albini A. The alpha 3 beta 1 integrin is associated with mammary carcinoma cell metastasis, invasion, and gelatinase B (MMP-9) activity. Int J Cancer. 2000;87:336–342. [PubMed] [Google Scholar]
- Muramatsu T, Miyauchi T. Basigin (CD147): a multifunctional transmembrane protein involved in reproduction, neural function, inflammation and tumor invasion. Histol Histopathol. 2003;18:981–987. doi: 10.14670/HH-18.981. [DOI] [PubMed] [Google Scholar]
- Muraoka K, Nabeshima K, Murayama T, Biswas C, Koono M. Enhanced expression of a tumor-cell-derived collagenase-stimulatory factor in urothelial carcinoma: its usefulness as a tumor marker for bladder cancers. Int J Cancer. 1993;55:19–26. doi: 10.1002/ijc.2910550105. [DOI] [PubMed] [Google Scholar]
- Nabeshima K, Iwasaki H, Nishio J, Koga K, Shishime M, Kikuchi M. Expression of emmprin and matrix metalloproteinases (MMPs) in peripheral nerve sheath tumors: emmprin and membrane-type (MT)1-MMP expressions are associated with malignant potential. Anticancer Res. 2006;26:1359–1367. [PubMed] [Google Scholar]
- Naruhashi K, Kadomatsu K, Igakura T, Fan QW, Kuno N, Muramatsu H, Miyauchi T, Hasegawa T, Itoh A, Muramatsu T, Nabeshima T. Abnormalities of sensory and memory functions in mice lacking Bsg gene. Biochem Biophys Res Commun. 1997;236:733–737. doi: 10.1006/bbrc.1997.6993. [DOI] [PubMed] [Google Scholar]
- Nehme CL, Fayos BE, Bartles JR. Distribution of the integral plasma membrane glycoprotein CE9 (MRC OX-47) among rat tissues and its induction by diverse stimuli of metabolic activation. Biochem J. 1995;310(Pt 2):693–698. doi: 10.1042/bj3100693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Noel AC, Polette M, Lewalle JM, Munaut C, Emonard HP, Birembaut P, Foidart JM. Coordinate enhancement of gelatinase A mRNA and activity levels in human fibroblasts in response to breast-adenocarcinoma cells. Int J Cancer. 1994;56:331–336. doi: 10.1002/ijc.2910560306. [DOI] [PubMed] [Google Scholar]
- Ochrietor JD, Moroz TP, Clamp MF, Timmers AM, Muramatsu T, Linser PJ. Inactivation of the Basigin gene impairs normal retinal development and maturation. Vision Res. 2002;42:447–453. doi: 10.1016/s0042-6989(01)00236-x. [DOI] [PubMed] [Google Scholar]
- Ochrietor JD, Moroz TP, van Ekeris L, Clamp MF, Jefferson SC, deCarvalho AC, Fadool JM, Wistow G, Muramatsu T, Linser PJ. Retina-specific expression of 5A11/Basigin-2, a member of the immunoglobulin gene superfamily. Invest Ophthalmol Vis Sci. 2003;44:4086–4096. doi: 10.1167/iovs.02-0995. [DOI] [PubMed] [Google Scholar]
- Pasieka Z, Stepien H, Czyz W, Pomorski L, Kuzdak K. Concentration of metalloproteinase-2 and tissue inhibitor of metalloproteinase-2 in the serum of patients with benign and malignant thyroid tumours treated surgically. Endocr Regul. 2004;38:57–63. [PubMed] [Google Scholar]
- Philp NJ, Ochrietor JD, Rudoy C, Muramatsu T, Linser PJ. Loss of MCT1, MCT3, and MCT4 expression in the retinal pigment epithelium and neural retina of the 5A11/basigin-null mouse. Invest Ophthalmol Vis Sci. 2003;44:1305–1311. doi: 10.1167/iovs.02-0552. [DOI] [PubMed] [Google Scholar]
- Pistol G, Matache C, Calugaru A, Stavaru C, Tanaseanu S, Ionescu R, Dumitrache S, Stefanescu M. Roles of CD147 on T lymphocytes activation and MMP-9 secretion in Systemic Lupus Erythematosus. J Cell Mol Med. 2007;11:339–348. doi: 10.1111/j.1582-4934.2007.00022.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Polette M, Gilles C, Marchand V, Lorenzato M, Toole B, Tournier JM, Zucker S, Birembaut P. Tumor collagenase stimulatory factor (TCSF) expression and localization in human lung and breast cancers. J Histochem Cytochem. 1997;45:703–709. doi: 10.1177/002215549704500508. [DOI] [PubMed] [Google Scholar]
- Porter JC, Hogg N. Integrins take partners: cross-talk between integrins and other membrane receptors. Trends Cell Biol. 1998;8:390–396. doi: 10.1016/s0962-8924(98)01344-0. [DOI] [PubMed] [Google Scholar]
- Portik-Dobos V, Anstadt MP, Hutchinson J, Bannan M, Ergul A. Evidence for a matrix metalloproteinase induction/activation system in arterial vasculature and decreased synthesis and activity in diabetes. Diabetes. 2002;51:3063–3068. doi: 10.2337/diabetes.51.10.3063. [DOI] [PubMed] [Google Scholar]
- Pushkarsky T, Yurchenko V, Vanpouille C, Brichacek B, Vaisman I, Hatakeyama S, Nakayama KI, Sherry B, Bukrinsky MI. Cell Surface Expression of CD147/EMMPRIN Is Regulated by Cyclophilin 60. J Biol Chem. 2005;280:27866–27871. doi: 10.1074/jbc.M503770200. [DOI] [PubMed] [Google Scholar]
- Pushkarsky T, Zybarth G, Dubrovsky L, Yurchenko V, Tang H, Guo H, Toole B, Sherry B, Bukrinsky M. CD147 facilitates HIV-1 infection by interacting with virus-associated cyclophilin A. Proc Natl Acad Sci U S A. 2001;98:6360–6365. doi: 10.1073/pnas.111583198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pyke C, Ralfkiaer E, Huhtala P, Hurskainen T, Dano K, Tryggvason K. Localization of messenger RNA for Mr 72,000 and 92,000 type IV collagenases in human skin cancers by in situ hybridization. Cancer Res. 1992;52:1336–1341. [PubMed] [Google Scholar]
- Redondo P, Lloret P, Idoate M, Inoges S. Expression and serum levels of MMP-2 and MMP-9 during human melanoma progression. Clin Exp Dermatol. 2005;30:541–545. doi: 10.1111/j.1365-2230.2005.01849.x. [DOI] [PubMed] [Google Scholar]
- Reed BH, Wilk R, Schock F, Lipshitz HD. Integrin-dependent apposition of Drosophila extraembryonic membranes promotes morphogenesis and prevents anoikis. Curr Biol. 2004;14:372–380. doi: 10.1016/j.cub.2004.02.029. [DOI] [PubMed] [Google Scholar]
- Reich R, Blumenthal M, Liscovitch M. Role of phospholipase D in laminin-induced production of gelatinase A (MMP-2) in metastatic cells. Clin Exp Metastasis. 1995;13:134–140. doi: 10.1007/BF00133618. [DOI] [PubMed] [Google Scholar]
- Reimers N, Zafrakas K, Assmann V, Egen C, Riethdorf L, Riethdorf S, Berger J, Ebel S, Janicke F, Sauter G, Pantel K. Expression of extracellular matrix metalloproteases inducer on micrometastatic and primary mammary carcinoma cells. Clin Cancer Res. 2004;10:3422–3428. doi: 10.1158/1078-0432.CCR-03-0610. [DOI] [PubMed] [Google Scholar]
- Renno T, Wilson A, Dunkel C, Coste I, Maisnier-Patin K, Benoit de Coignac A, Aubry JP, Lees RK, Bonnefoy JY, MacDonald HR, Gauchat JF. A role for CD147 in thymic development. J Immunol. 2002;168:4946–4950. doi: 10.4049/jimmunol.168.10.4946. [DOI] [PubMed] [Google Scholar]
- Richardson JC, Kendal CE, Anderson R, Priest F, Gower E, Soden P, Gray R, Topps S, Howlett DR, Lavender D, Clarke NJ, Barnes JC, Haworth R, Stewart MG, Rupniak HT. Ultrastructural and behavioural changes precede amyloid deposition in a transgenic model of Alzheimer’s disease. Neuroscience. 2003;122:213–228. doi: 10.1016/s0306-4522(03)00389-0. [DOI] [PubMed] [Google Scholar]
- Riethdorf S, Reimers N, Assmann V, Kornfeld JW, Terracciano L, Sauter G, Pantel K. High incidence of EMMPRIN expression in human tumors. Int J Cancer. 2006;119:1800–1810. doi: 10.1002/ijc.22062. [DOI] [PubMed] [Google Scholar]
- Rosenthal EL, Shreenivas S, Peters GE, Grizzle WE, Desmond R, Gladson CL. Expression of extracellular matrix metalloprotease inducer in laryngeal squamous cell carcinoma. Laryngoscope. 2003;113:1406–1410. doi: 10.1097/00005537-200308000-00027. [DOI] [PubMed] [Google Scholar]
- Ryan MC, Christiano AM, Engvall E, Wewer UM, Miner JH, Sanes JR, Burgeson RE. The functions of laminins: lessons from in vivo studies. Matrix Biol. 1996;15:369–381. doi: 10.1016/s0945-053x(96)90157-2. [DOI] [PubMed] [Google Scholar]
- Sameshima T, Nabeshima K, Toole BP, Yokogami K, Okada Y, Goya T, Koono M, Wakisaka S. Expression of emmprin (CD147), a cell surface inducer of matrix metalloproteinases, in normal human brain and gliomas. Int J Cancer. 2000;88:21–27. doi: 10.1002/1097-0215(20001001)88:1<21::aid-ijc4>3.0.co;2-s. [DOI] [PubMed] [Google Scholar]
- Sameshima T, Nabeshima K, Toole BP, Yokogami K, Okada Y, Goya T, Koono M, Wakisaka S. Glioma cell extracellular matrix metalloproteinase inducer (EMMPRIN) (CD147) stimulates production of membrane-type matrix metalloproteinases and activated gelatinase A in co-cultures with brain-derived fibroblasts. Cancer Lett. 2000;157:177–184. doi: 10.1016/s0304-3835(00)00485-7. [DOI] [PubMed] [Google Scholar]
- Saphire AC, Bobardt MD, Gallay PA. Host cyclophilin A mediates HIV-1 attachment to target cells via heparans. Embo J. 1999;18:6771–6785. doi: 10.1093/emboj/18.23.6771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlosshauer B, Bauch H, Frank R. Neurothelin: amino acid sequence, cell surface dynamics and actin colocalization. Eur J Cell Biol. 1995;68:159–166. [PubMed] [Google Scholar]
- Schmidt R, Bultmann A, Ungerer M, Joghetaei N, Bulbul O, Thieme S, Chavakis T, Toole BP, Gawaz M, Schomig A, May AE. Extracellular matrix metalloproteinase inducer regulates matrix metalloproteinase activity in cardiovascular cells: implications in acute myocardial infarction. Circulation. 2006;113:834–841. doi: 10.1161/CIRCULATIONAHA.105.568162. [DOI] [PubMed] [Google Scholar]
- Seko Y, Fujimura T, Taka H, Mineki R, Murayama K, Nagai R. Hypoxia followed by reoxygenation induces secretion of cyclophilin A from cultured rat cardiac myocytes. Biochem Biophys Res Commun. 2004;317:162–168. doi: 10.1016/j.bbrc.2004.03.021. [DOI] [PubMed] [Google Scholar]
- Seulberger H, Unger CM, Risau W. HT7, Neurothelin, Basigin, gp42 and OX-47--many names for one developmentally regulated immuno-globulin-like surface glycoprotein on blood-brain barrier endothelium, epithelial tissue barriers and neurons. Neurosci Lett. 1992;140:93–97. doi: 10.1016/0304-3940(92)90690-9. [DOI] [PubMed] [Google Scholar]
- Sherry B, Yarlett N, Strupp A, Cerami A. Identification of cyclophilin as a proinflammatory secretory product of lipopolysaccharide-activated macrophages. Proc Natl Acad Sci U S A. 1992;89:3511–3515. doi: 10.1073/pnas.89.8.3511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sherry B, Zybarth G, Alfano M, Dubrovsky L, Mitchell R, Rich D, Ulrich P, Bucala R, Cerami A, Bukrinsky M. Role of cyclophilin A in the uptake of HIV-1 by macrophages and T lymphocytes. Proc Natl Acad Sci U S A. 1998;95:1758–1763. doi: 10.1073/pnas.95.4.1758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shirozu M, Tada H, Tashiro K, Nakamura T, Lopez ND, Nazarea M, Hamada T, Sato T, Nakano T, Honjo T. Characterization of novel secreted and membrane proteins isolated by the signal sequence trap method. Genomics. 1996;37:273–280. doi: 10.1006/geno.1996.0560. [DOI] [PubMed] [Google Scholar]
- Sidhu SS, Mengistab AT, Tauscher AN, LaVail J, Basbaum C. The microvesicle as a vehicle for EMMPRIN in tumor-stromal interactions. Oncogene. 2004;23:956–963. doi: 10.1038/sj.onc.1207070. [DOI] [PubMed] [Google Scholar]
- Spinale FG, Coker ML, Heung LJ, Bond BR, Gunasinghe HR, Etoh T, Goldberg AT, Zellner JL, Crumbley AJ. A matrix metalloproteinase induction/activation system exists in the human left ventricular myocardium and is upregulated in heart failure. Circulation. 2000;102:1944–1949. doi: 10.1161/01.cir.102.16.1944. [DOI] [PubMed] [Google Scholar]
- Springer TA, Wang JH. The three-dimensional structure of integrins and their ligands, and conformational regulation of cell adhesion. Adv Protein Chem. 2004;68:29–63. doi: 10.1016/S0065-3233(04)68002-8. [DOI] [PubMed] [Google Scholar]
- Staffler G, Szekeres A, Schutz GJ, Saemann MD, Prager E, Zeyda M, Drbal K, Zlabinger GJ, Stulnig TM, Stockinger H. Selective inhibition of T cell activation via CD147 through novel modulation of lipid rafts. J Immunol. 2003;171:1707–1714. doi: 10.4049/jimmunol.171.4.1707. [DOI] [PubMed] [Google Scholar]
- Stonehouse TJ, Woodhead VE, Herridge PS, Ashrafian H, George M, Chain BM, Katz DR. Molecular characterization of U937-dependent T-cell co-stimulation. Immunology. 1999;96:35–47. doi: 10.1046/j.1365-2567.1999.00670.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun J, Hemler ME. Regulation of MMP-1 and MMP-2 production through CD147/extracellular matrix metalloproteinase inducer interactions. Cancer Res. 2001;61:2276–2281. [PubMed] [Google Scholar]
- Tang W, Chang SB, Hemler ME. Links between CD147 function, glycosylation, and caveolin-1. Mol Biol Cell. 2004;15:4043–4050. doi: 10.1091/mbc.E04-05-0402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tang W, Hemler ME. Caveolin-1 regulates matrix metalloproteinases-1 induction and CD147/EMMPRIN cell surface clustering. J Biol Chem. 2004;279:11112–11118. doi: 10.1074/jbc.M312947200. [DOI] [PubMed] [Google Scholar]
- Tang Y, Kesavan P, Nakada MT, Yan L. Tumor-stroma interaction: positive feedback regulation of extracellular matrix metalloproteinase inducer (EMMPRIN) expression and matrix metalloproteinase-dependent generation of soluble EMMPRIN. Mol Cancer Res. 2004;2:73–80. [PubMed] [Google Scholar]
- Tang Y, Nakada MT, Kesavan P, McCabe F, Millar H, Rafferty P, Bugelski P, Yan L. Extracellular matrix metalloproteinase inducer stimulates tumor angiogenesis by elevating vascular endothelial cell growth factor and matrix metalloproteinases. Cancer Res. 2005;65:3193–3199. doi: 10.1158/0008-5472.CAN-04-3605. [DOI] [PubMed] [Google Scholar]
- Taylor PM, Woodfield RJ, Hodgkin MN, Pettitt TR, Martin A, Kerr DJ, Wakelam MJ. Breast cancer cell-derived EMMPRIN stimulates fibroblast MMP2 release through a phospholipase A(2) and 5-lipoxygenase catalyzed pathway. Oncogene. 2002;21:5765–5772. doi: 10.1038/sj.onc.1205702. [DOI] [PubMed] [Google Scholar]
- Tegeder I, Schumacher A, John S, Geiger H, Geisslinger G, Bang H, Brune K. Elevated serum cyclophilin levels in patients with severe sepsis. J Clin Immunol. 1997;17:380–386. doi: 10.1023/a:1027364207544. [DOI] [PubMed] [Google Scholar]
- Vaisanen A, Tuominen H, Kallioinen M, Turpeenniemi-Hujanen T. Matrix metalloproteinase-2 (72 kD type IV collagenase) expression occurs in the early stage of human melanocytic tumour progression and may have prognostic value. J Pathol. 1996;180:283–289. doi: 10.1002/(SICI)1096-9896(199611)180:3<283::AID-PATH662>3.0.CO;2-3. [DOI] [PubMed] [Google Scholar]
- van den Oord JJ, Paemen L, Opdenakker G, de Wolf-Peeters C. Expression of gelatinase B and the extracellular matrix metalloproteinase inducer EMMPRIN in benign and malignant pigment cell lesions of the skin. Am J Pathol. 1997;151:665–670. [PMC free article] [PubMed] [Google Scholar]
- Vigneswaran N, Beckers S, Waigel S, Mensah J, Wu J, Mo J, Fleisher KE, Bouquot J, Sacks PG, Zacharias W. Increased EMMPRIN (CD 147) expression during oral carcinogenesis. Exp Mol Pathol. 2006;80:147–159. doi: 10.1016/j.yexmp.2005.09.011. [DOI] [PubMed] [Google Scholar]
- Wahl ML, Owen JA, Burd R, Herlands RA, Nogami SS, Rodeck U, Berd D, Leeper DB, Owen CS. Regulation of intracellular pH in human melanoma: potential therapeutic implications. Mol Cancer Ther. 2002;1:617–628. [PubMed] [Google Scholar]
- Wang H, Fu W, Im JH, Zhou Z, Santoro SA, Iyer V, DiPersio CM, Yu QC, Quaranta V, Al-Mehdi A, Muschel RJ. Tumor cell alpha3beta1 integrin and vascular laminin-5 mediate pulmonary arrest and metastasis. J Cell Biol. 2004;164:935–941. doi: 10.1083/jcb.200309112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weiner DB, Kokai Y, Wada T, Cohen JA, Williams WV, Greene MI. Linkage of tyrosine kinase activity with transforming ability of the p185neu oncoprotein. Oncogene. 1989;4:1175–1183. [PubMed] [Google Scholar]
- Williams TM, Hassan GS, Li J, Cohen AW, Medina F, Frank PG, Pestell RG, Di Vizio D, Loda M, Lisanti MP. Caveolin-1 promotes tumor progression in an autochthonous mouse model of prostate cancer: genetic ablation of Cav-1 delays advanced prostate tumor development in tramp mice. J Biol Chem. 2005;280:25134–25145. doi: 10.1074/jbc.M501186200. [DOI] [PubMed] [Google Scholar]
- Williams TM, Lisanti MP. The caveolin proteins. Genome Biol. 2004;5:214. doi: 10.1186/gb-2004-5-3-214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams TM, Lisanti MP. Caveolin-1 in oncogenic transformation, cancer, and metastasis. Am J Physiol Cell Physiol. 2005;288:C494–506. doi: 10.1152/ajpcell.00458.2004. [DOI] [PubMed] [Google Scholar]
- Williams TM, Medina F, Badano I, Hazan RB, Hutchinson J, Muller WJ, Chopra NG, Scherer PE, Pestell RG, Lisanti MP. Caveolin-1 gene disruption promotes mammary tumorigenesis and dramatically enhances lung metastasis in vivo. Role of Cav-1 in cell invasiveness and matrix metalloproteinase (MMP-2/9) secretion. J Biol Chem. 2004;279:51630–51646. doi: 10.1074/jbc.M409214200. [DOI] [PubMed] [Google Scholar]
- Wilson MC, Meredith D, Fox JE, Manoharan C, Davies AJ, Halestrap AP. Basigin (CD147) is the target for organomercurial inhibition of monocarboxylate transporter isoforms 1 and 4: the ancillary protein for the insensitive MCT2 is EMBIGIN (gp70) J Biol Chem. 2005;280:27213–27221. doi: 10.1074/jbc.M411950200. [DOI] [PubMed] [Google Scholar]
- Wilson MC, Meredith D, Halestrap AP. Fluorescence resonance energy transfer studies on the interaction between the lactate transporter MCT1 and CD147 provide information on the topology and stoichiometry of the complex in situ. J Biol Chem. 2002;277:3666–3672. doi: 10.1074/jbc.M109658200. [DOI] [PubMed] [Google Scholar]
- Woodhead VE, Stonehouse TJ, Binks MH, Speidel K, Fox DA, Gaya A, Hardie D, Henniker AJ, Horejsi V, Sagawa K, Skubitz KM, Taskov H, Todd RF, 3rd, van Agthoven A, Katz DR, Chain BM. Novel molecular mechanisms of dendritic cell-induced T cell activation. Int Immunol. 2000;12:1051–1061. doi: 10.1093/intimm/12.7.1051. [DOI] [PubMed] [Google Scholar]
- Xu Q, Leiva MC, Fischkoff SA, Handschumacher RE, Lyttle CR. Leukocyte chemotactic activity of cyclophilin. J Biol Chem. 1992;267:11968–11971. [PubMed] [Google Scholar]
- Yurchenko V, O’Connor M, Dai WW, Guo H, Toole B, Sherry B, Bukrinsky M. CD147 is a signaling receptor for cyclophilin B. Biochem Biophys Res Commun. 2001;288:786–788. doi: 10.1006/bbrc.2001.5847. [DOI] [PubMed] [Google Scholar]
- Yurchenko V, Pushkarsky T, Li JH, Dai WW, Sherry B, Bukrinsky M. Regulation of CD147 cell surface expression: involvement of the proline residue in the CD147 transmembrane domain. J Biol Chem. 2005;280:17013–17019. doi: 10.1074/jbc.M412851200. [DOI] [PubMed] [Google Scholar]
- Yurchenko V, Zybarth G, O’Connor M, Dai WW, Franchin G, Hao T, Guo H, Hung HC, Toole B, Gallay P, Sherry B, Bukrinsky M. Active site residues of cyclophilin A are crucial for its signaling activity via CD147. J Biol Chem. 2002;277:22959–22965. doi: 10.1074/jbc.M201593200. [DOI] [PubMed] [Google Scholar]
- Zhang F, Vannucci SJ, Philp NJ, Simpson IA. Monocarboxylate transporter expression in the spontaneous hypertensive rat: effect of stroke. J Neurosci Res. 2005;79:139–145. doi: 10.1002/jnr.20312. [DOI] [PubMed] [Google Scholar]
- Zhang Q, Chen X, Zhou J, Zhang L, Zhao Q, Chen G, Xu J, Qian F, Chen Z. CD147, MMP-2, MMP-9 and MVD-CD34 are significant predictors of recurrence after liver transplantation in hepatocellular carcinoma patients. Cancer Biol Ther. 2006;5:808–814. doi: 10.4161/cbt.5.7.2754. [DOI] [PubMed] [Google Scholar]
- Zhang X, Chen Z, Huang H, Gordon JR, Xiang J. DNA microarray analysis of the gene expression profiles of naive versus activated tumor-specific T cells. Life Sci. 2002;71:3005–3017. doi: 10.1016/s0024-3205(02)02164-1. [DOI] [PubMed] [Google Scholar]
- Zheng HC, Takahashi H, Murai Y, Cui ZG, Nomoto K, Miwa S, Tsuneyama K, Takano Y. Upregulated EMMPRIN/CD147 might contribute to growth and angiogenesis of gastric carcinoma: a good marker for local invasion and prognosis. Br J Cancer. 2006;95:1371–1378. doi: 10.1038/sj.bjc.6603425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou S, Zhou H, Walian PJ, Jap BK. CD147 is a regulatory subunit of the gamma-secretase complex in Alzheimer’s disease amyloid beta-peptide production. Proc Natl Acad Sci U S A. 2005;102:7499–7504. doi: 10.1073/pnas.0502768102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou S, Zhou H, Walian PJ, Jap BK. Regulation of gamma-Secretase Activity in Alzheimer’s Disease. Biochemistry. 2007;46:2553–2563. doi: 10.1021/bi602509c. [DOI] [PubMed] [Google Scholar]
- Zucker S, Hymowitz M, Rollo EE, Mann R, Conner CE, Cao J, Foda HD, Tompkins DC, Toole BP. Tumorigenic potential of extracellular matrix metalloproteinase inducer. Am J Pathol. 2001;158:1921–1928. doi: 10.1016/S0002-9440(10)64660-3. [DOI] [PMC free article] [PubMed] [Google Scholar]