Abstract
The majority of T cell responses are restricted to peptide antigens bound by polymorphic major histocompatibility complex (MHC) molecules. However, peptide antigens can be presented to T cells by murine non-MHC–encoded CD1d (mCD1) molecules, and human T cell lines specific for nonpeptide antigens presented on CD1 isoforms have been identified. It is shown here that antigen-specific, mCD1-restricted lymphocytes can be generated in vivo by immunizing mice with a combination of plasmids encoding chicken ovalbumin, murine CD1d, and costimulatory molecules. Splenocytes from immunized mice have CD1d-restricted, MHC- unrestricted, ovalbumin-specific cytolytic activity that can be inhibited by anti-CD1 antibodies as well as a competing CD1-binding peptide. These results suggest a physiologic role for murine CD1d to present exogenous protein antigens.
Classically, MHC class I or class II molecules are responsible for presenting peptide antigens to T cells. However, in both humans and mice, the non-MHC– encoded CD1 family of cell surface proteins has been implicated to also have an antigen-presenting function (1, 2). Although MHC class I molecules mediate recognition of nonself or infected tissues by the immune system, the function of CD1 molecules is still unclear. Unlike the MHC proteins, CD1 molecules are nonpolymorphic and have five isoforms: CD1a, -b, -c, -d, and -e (3). The isoforms are conserved in several mammalian species (4) and have been divided into two groups based on the sequences of their external domains (5). CD1a, -b, -c, and -e comprise group 1, while group 2 contains CD1d. Although all five isoforms are found in humans, only the group 2 isoforms are conserved from rodents to humans.
CD1 molecules share some characteristics with both MHC class I and MHC class II ligands. CD1 proteins bear some resemblance to the classical MHC class I proteins both in overall sequence homology, especially in the α3 domain, and by their usual association with β2-microglobulin (β2m; references 5 and 6). However, unlike MHC class I molecules, CD1 proteins have been reported to be expressed without β2m (7) and do not require the transporter proteins associated with antigen processing (TAP) for stable expression (8–10). The mechanism for antigen processing for CD1 is more similar to that of MHC class II than class I (11–13). Like MHC class II, human CD1b is localized to endocytic compartments, including the specialized endosomes where MHC class II proteins are believed to bind endocytosed antigens (14–17). The non-MHC– encoded CD1 family of nonpolymorphic glycoproteins is, therefore, similar to, yet distinct from, other antigen-presenting molecules in its similarity to MHC class I by sequence, structural homology, and association with β2m, as well as its similarity to MHC class II by its cellular localization and dependence on the endosomal compartment for presentation of exogenous antigens.
Unlike classical MHC, CD1 can present nonpeptide ligands such as mycolic acid (18), lipoarabinomannan (19), and mycobacterial lipid antigens (20) to αβ T cell receptor–bearing lymphocytes. The presentation of foreign nonpeptide antigens by CD1 has been demonstrated for the human CD1b and CD1c isoforms from which human CD1d and its related murine isoforms are divergent (5). Castaño et al. (2) have reported that murine non-MHC– encoded CD1d (mCD1) can bind long peptides with hydrophobic and bulky amino acids. Immunization of mice with CD1-transfected cells preincubated with peptide generated, CD1-restricted, peptide-specific CTL. These data suggest that mCD1 may have a antigen-presenting function by binding peptides with hydrophobic residues (2).
Murine autoreactive, CD1-restricted T cells have been identified in unimmunized mice (21, 22). To test the biological significance of mCD1 presentation of foreign protein antigens, we generated an antigen-specific, CD1- restricted response by plasmid DNA immunization. This immunization protocol raised a CD1-restricted, ovalbumin-specific CTL response, demonstrating that protein antigen is recognized in the context of mCD1 and elicits a cellular immune response in vivo. Lysis by these cytotoxic lymphocytes are antigen and CD1 dependent, can be partially abrogated by anti-CD1 antibodies, and are competitively inhibited by an established CD1-binding peptide. Furthermore, these CTLs lyse allogeneic targets in an antigen-specific manner.
Materials and Methods
Mice.
C57BL/6 mice were purchased from The Jackson Laboratories (Bar Harbor, ME) and maintained under standard conditions in the University of California, San Diego Animal Facility accredited by the American Association of Laboratory Care. Mice of either sex were used at 2–4 mo of age.
Preparation of Plasmid DNA.
The plasmid pACB-CD1 was constructed by subcloning the BamHI–XhoI fragment from the pBluescript vector encoding murine CD1D1 (reference 6; provided by S. Balk, Beth Israel Hospital, Boston, MA) into the BamHI–SalI site of the pACB vector (23). The nCMVOVA, nCMVB7-1, and nCMVB7-2 plasmids have been previously described (24). DNA was prepared using maxiprep kits (Qiagen Inc., Chatsworth, CA), with the modification of adding 0.1 vol 10% Triton X-114 (Sigma Chemical Co., St. Louis, MO) to the clarified bacterial lysate before applying it to the column in the kit. Before injection, the residual endotoxin level was quantified using a limulus extract clot assay (Associates of Cape Cod, Woods Hole, MA). Plasmid DNA with a level of <5 ng endotoxin/mg DNA was resuspended in sterile isotonic saline solution before injection.
Immunizations.
Mice were immunized once a week for 2 wk. Plasmids were injected intradermally in the tail base with 50 μg of each plasmid indicated in 50 μl normal saline using a 25-gauge needle.
Cytotoxic T Lymphocyte Assay.
Mice were killed and their spleens were removed and teased apart in RPMI 1640 media (BioWhittaker Inc., Walkersville, MD) supplemented with 2% fetal bovine serum (FBS). In 24-well plates (Costar Corp., Cambridge, MA) 7 × 106 responders were incubated with 1 × 106 mitomycin C–treated stimulator cells in the presence of 50 IU/ml recombinant human IL-2. The culture media was RPMI 1640 supplemented with 10% heat-inactivated FBS, 2 mM glutamine, 50 μM 2β-mercaptoethanol, and 1% penicillin and streptomycin. The stimulator cells, EL4-CD1 or RMAS-CD1, were stably transfected with a pCDNA3 vector expressing murine CD1d1 (pCDNA3 CD1D1). These transfectants were incubated with 40–200 μg/ml ovalbumin protein (Sigma Chemical Co.) for 1 h at 37°C and 5% CO2 and then treated with mitomycin C (Sigma Chemical Co.) 50 μg/ml shaking for 30 min at 37°C. After 5 d, the restimulated cells were harvested and separated from dead cells on a lympholyte M gradient (Accurate Chemical and Scientific Corp., Westbury, NY). In 96-well, round-bottom plates, target cells were incubated in 200-μl volumes with restimulated T cells at titrated E/T ratios for 4 h. The assay medium used was phenol red–free RPMI 1640 supplemented with 2% BSA, 2 mM glutamine, and 1% penicillin and streptomycin. The target cells used were peptide transporter–deficient RMAS cells or RMAS cells stably transfected with pCDNA3 CD1D1. Targets were pulsed with 40–200 μg/ml ovalbumin protein at 37°C and 5% CO2 for 1 h and then washed three times. 50 μl per well of the supernatant was then transferred to 96-well plates and lysis was assessed by measuring lactate dehydrogenase release using the Cytotox 96 assay kit (Promega Corp., Madison, WI). Controls were included on each plate for spontaneous LDH release from target and effector cells. Percent lysis was calculated according to manufacturer's instructions using the formula: [(test release − spontaneous release)/(maximum release − spontaneous release)] × 100%.
Peptide and Antibodies.
The p99 peptide, YEHDFHHIREWGNHWKNFLAVM, that has been reported to bind murine CD1d1 (2) was purchased from PeptidoGenic Research and Company (Livermore, CA) HPLC purified. Antibodies used were anti-Kb and Db, 20-8-4S (American Type Culture Collection [ATCC], Rockville, MD), anti-CD1, 1B1 (PharMingen, San Diego, CA) or 1H1 (PharMingen) or 3C11 (generously provided by S. Balk), anti-CD8, 3.155 (ATCC), anti-CD4, YTS177, and anti-Fas ligand, K-10 (PharMingen). All antibodies were used at 10 μg/ml with the exception of 3C11 ascites used at a dilution of 1:50, 3.155 ascites used at a dilution of 1:300 and YTS177 ascites used at a dilution of 1:20. The antibodies 20-8-4S and 3.155 were capable of blocking class I–restricted CD8+ T cell responses at these concentrations in pilot experiments.
Generation of T Cell Lines.
C57BL/6 mice were immunized with plasmids encoding OVA, mCD1d1, and B7.1 twice. Splenocytes were harvested 2 wk after the first injection and restimulated in vitro for 6 d with CD1-transfected RMAS cells pulsed with ovalbumin protein in IMDM (BioWhittaker) supplemented with 10% Con A supernatant, 10% heat-inactivated FBS, 2 mM glutamine, 50 μM 2β-mercaptoethanol, and 1% penicillin and streptomycin. The cultured splenocytes were restimulated weekly in 24-well plates with 105 each of mitomycin C–treated, thioglycollate-activated syngeneic peritoneal macrophages and CD1-transfected RMAS cells pulsed with ovalbumin protein in Con A–supplemented complete IMDM.
Results and Discussion
Generation of Ovalbumin-specific, CD1-restricted CTL.
Intradermal administration of plasmid DNA induces antigen-specific immune responses (25) and allows for the ectopic expression of membrane-bound molecules such as CD1 and costimulators, as well as the expression of the antigen, chicken ovalbumin. Therefore, we chose this approach to test the biological significance of CD1 presentation of peptide from whole protein antigens. CD1 expression has been detected on gastrointestinal epithelium, thymus, liver, and spleen (6, 26). As murine CD1 is not prevalent in the dermis, a coinjection strategy was used to ectopically express the antigen-presenting molecule (CD1d1) as well as the antigen. Additionally, plasmids encoding costimulatory molecules B7.1 or B7.2 were included to provide the responsive T cell with a nonspecific second signal in the same milieu as the cognate signal (27–29). Chicken ovalbumin was selected as the test antigen as it contains a peptide with the reported mCD1-binding motif (2). In these experiments, antigen loading of target cells was accomplished by incubating them with whole protein, because as recent reports suggest, CD1 antigen loading occurs through a TAP-independent endocytic mechanism (11–13). The TAP independence of CD1 antigen processing made TAP-deficient, CD1-transfected RMAS cells the preferred target cells (9, 10).
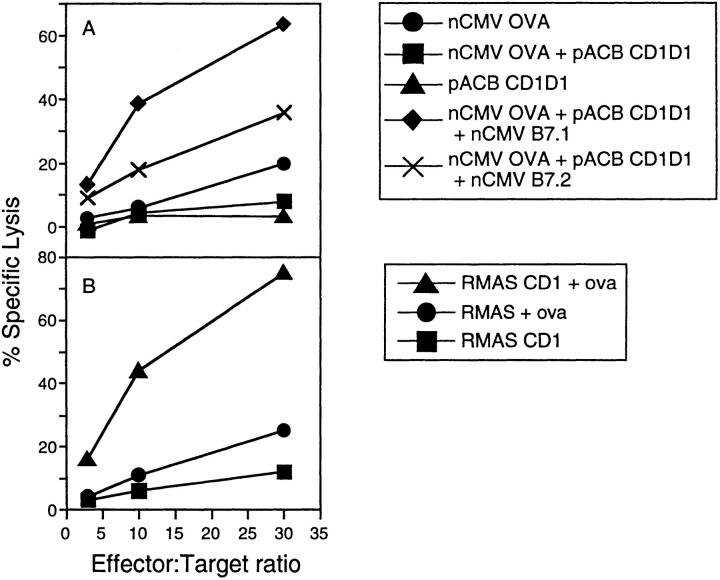
Hence, C57BL/6 mice were immunized with different combinations of plasmid DNA encoding mCD1D1 (pACB-CD1), chicken ovalbumin (nCMV-OVA), and costimulator molecules (nCMV-B7.1 or nCMV-B7.2) at weeks zero and one. Mice were killed at week 2, and after in vitro restimulation, their splenocytes were assayed for their ability to lyse H-2b CD1-transfected RMAS cells. Mice immunized with a combination including B7.1-expressing plasmid repeatedly gave a higher specific lysis than mice immunized with a B7.2-encoding plasmid (Fig. 1 A). The in vivo requirement for adding a CD28 ligand is in contrast to in vitro studies with CD1b- or CD1c-restricted human T cell hybridomas that use a pathway of costimulation outside the interaction between CD28 and its counterreceptors (30).
Figure 1.
Recognition of ovalbumin by CD1d1-restricted CTL. Three mice per group were immunized with 50 μg of each plasmid at days 0 and 7. 2 wk after the first injection, mice were killed and restimulated in vitro with CD1d1-transfected EL4 cells pulsed with ovalbumin protein for 5 d in the presence of 50 IU rIL-2. The specificity of the resulting pooled CTL was determined by measuring the lysis of CD1d1-transfected RMAS cells incubated with ovalbumin in triplicate. (A) Immunizing mice with a combination of plasmids enhances the CD1-restricted CTL response. Mice were immunized with plasmids encoding ovalbumin, mCD1d1, and B7.1 (diamonds); ovalbumin, mCD1d1, and B7.2 (crosses); ovalbumin (circles); ovalbumin and mCD1d1 (squares); or mCD1d1 (triangles). The values plotted are average protein-specific lysis − background lysis. (B) CTL are specific for CD1d1 and ovalbumin. The splenocytes from the three mice injected with plasmids encoding ovalbumin, mCD1d1, and B7.1 were tested for their ability to lyse CD1-transfected RMAS cells pulsed with ovalbumin (triangles), CD1-transfected RMAS (squares), or RMAS cells pulsed with ovalbumin (circles) in triplicate. The values plotted represent the average lysis for three mice. These experiments were repeated twice.
CTL Activity Is Antigen and CD1 Dependent.
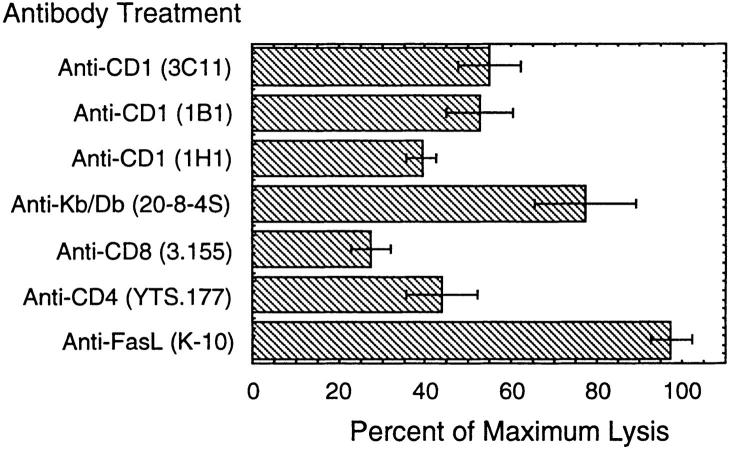
Splenocytes from mice immunized with the combination of plasmids encoding ovalbumin, mCD1d1, and B7.1 were able to lyse MHC class I–deficient, CD1+ RMAS transfectants pulsed with ovalbumin more effectively than untransfected RMAS cells pulsed with ovalbumin (Fig. 1 B). Although RMAS cells have very low levels of detectable CD1 expression on their surface by cytofluorometric analysis, augmenting the level of surface CD1 by transfection resulted in a threefold increase in cytotoxic lysis in the 4-h period of the assays. To verify that the incubation with ovalbumin did not spuriously result in MHC class I surface loading of peptide that was resulting in CTL recognition, antigen presentation was inhibited with antibodies. An antibody directed against MHC class I (H-2K b/D b) partially decreased lysis by 23%, whereas the anti-CD1 antibodies inhibited lysis by ∼45–60% (Fig. 2). None of the anti-CD1 antibodies were able to completely inhibit lysis at the concentrations used. In fact, the extent of inhibition varied between experiments, with the 1H1 antibody being the most effective. This variation between the different anti-CD1 antibodies may reflect differences in their affinities for antigen or may represent different epitopes seen by mixed populations of T cells in each experiment.
Figure 2.
Antibody inhibition of CD1-restricted CTL lysis. Four C57BL/6 mice were immunized with plasmids encoding OVA, mCD1d1, and B7.1 as in Fig. 1. Spleens were harvested after 2 wk and restimulated in vitro for 5 d with CD1d1 transfected RMAS cells pulsed with ovalbumin protein for 5 d with rIL-2. The splenocytes were pooled and assayed for CTL activity in duplicate. The percentage of maximum lysis relative to the value in the presence of targets and antigen alone ± SEM is shown. This experiment was repeated five times.
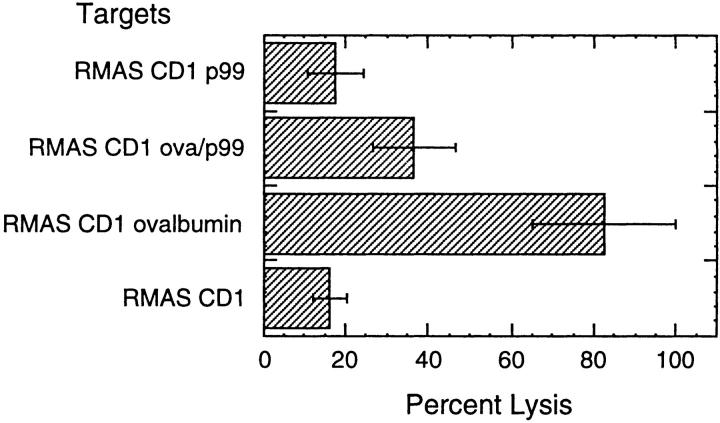
To confirm that the CTL response was ovalbumin specific and the effector cells were not simply recognizing a conformation of CD1d1 stabilized by binding a peptide, we tested the CTL for lysis activity of CD1+ RMAS cells pulsed with the p99 peptide previously described to bind murine CD1d1 (reference 2; Fig 3). The lysis of targets pulsed with p99 (16%) was not different from unpulsed targets (18%). Furthermore, p99 competitively inhibited lysis of CD1+ RMAS cells pulsed with ovalbumin protein, decreasing lysis from 82 to 37%.
Figure 3.
Peptide inhibition of ovalbumin-specific, CD1-restricted CTL lysis. T cell lines from mice injected with plasmids encoding OVA, mCD1d1, and B7.1 were assayed for cytolytic activity. CD1+ RMAS targets incubated with effector cells in triplicate wells were either unpulsed, pulsed for 2 h with 4 μg/ml of p99, pulsed with 200 μg/ml of ovalbumin protein for 1 h, washed, and then pulsed with 12 μg/ml of p99 for 1 h, or pulsed with ovalbumin protein only. The percentage of lysis ± SEM is shown at an E/T ratio of 30:1. This experiment was repeated twice.
CTL Are Antigen Specific and MHC Unrestricted.
Given that CD1 is nonpolymorphic, the same immunization protocol that was effective in generating CD1-restricted CTL in H-2b mice should also generate CTL that recognize CD1 on cells of another MHC haplotype. Splenocytes from C57BL/6 (H-2b) mice immunized with plasmids encoding mCD1D1 and B7.1 or mCD1D1, B7.1, and ovalbumin were tested for CTL activity to CD1D1-transfected P815 targets pulsed in the presence or absence of ovalbumin protein. The background lysis of CD1D1-transfected P815 in the absence of ovalbumin was ∼45% at the highest E/T ratio, suggesting a partial allogeneic response, which was subtracted to give the percent antigen-specific lysis. However, H-2b mice immunized with the three plasmids were repeatedly able to mount a significant CTL response against both CD1D1-transfected, ovalbumin-pulsed H-2b RMAS cells and H-2d P815 cells (Figs. 4, A and B), when compared to the response in the negative control group.
Figure 4.
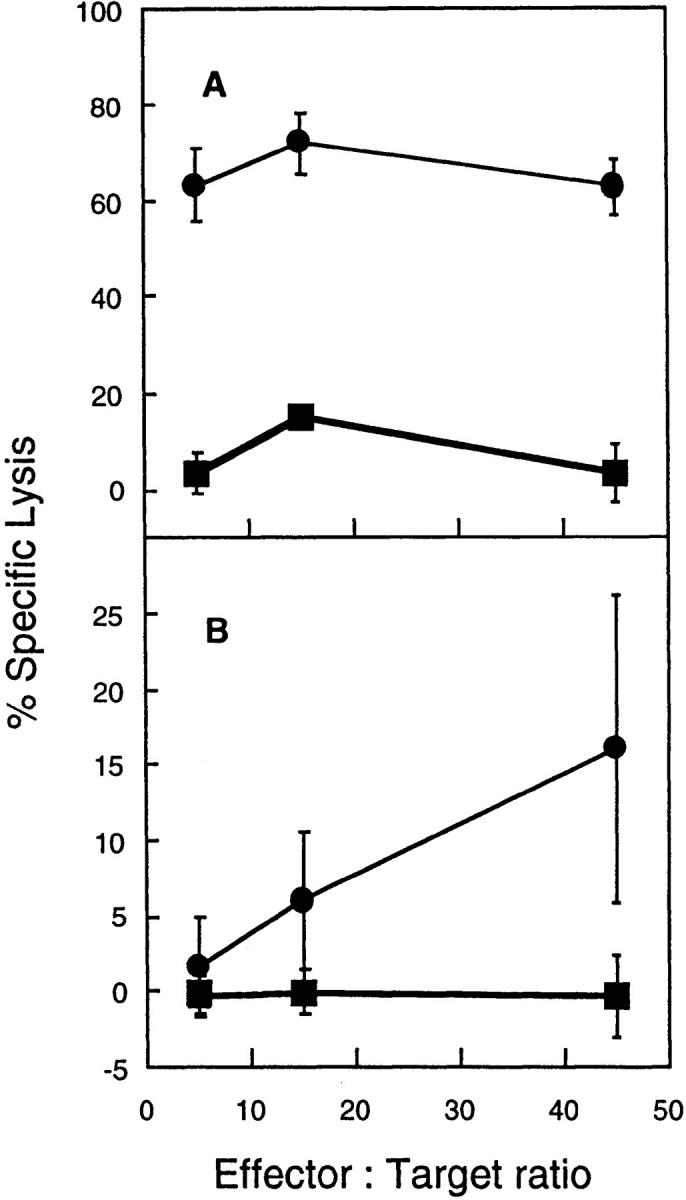
Ovalbumin-specific, CD1-restricted CTL lysis of allogeneic targets. 10 C57BL/6 mice (H-2b) were immunized with plasmids encoding OVA, mCD1d1, and B7.1 (circles), and four mice were immunized with mCD1d1 and B7.1 (squares) on days 0 and 10. 2 wk after the first injection, mice were killed and restimulated in vitro with CD1d1-transfected RMAS cells pulsed with ovalbumin protein for 5 d in the presence of 50 IU/ ml rIL-2. CD1+ RMAS or CD1+ P815 targets were incubated in the presence or absence of ovalbumin protein (100 μg/ ml) for 1 h and washed three times before being plated with effector cells in triplicate wells. The specific lysis was determined by measuring the lysis of CD1+ RMAS cells pulsed with ovalbumin minus unpulsed transfectants (A) or CD1+ P815 cells pulsed with ovalbumin minus unpulsed transfectants (B). The values plotted represent the average lysis ± SEM. This experiment was repeated twice.
CTL Effectors Are Predominantly CD8+ and Lysis Is Fas Independent.
To further characterize the phenotype of the CD1-restricted CTL, blocking antibodies to CD4 and CD8 were used (Fig. 2). Anti-CD8 repeatedly blocked lysis, whereas anti-CD4 blocked lysis less consistently. The variation of inhibition between experiments by anti-CD4 implies a mixed effector population from the whole spleen preparations. Recently, Stenger et al. (31) have identified two mechanisms of CD1-restricted, cell-mediated cytotoxicity to contribute to the outcome of infection with intracellular pathogens. CD1-restricted T cells that are CD4−CD8− lysed targets through the Fas–Fas ligand pathway while having no effect on intracellular bacterial survival, whereas CD8+ effector cells used perforin and granzymes to kill the bacteria as well as their host cell. In our studies, antibodies against Fas ligand had no effect on the lysis of CD1-transfected cells pulsed with ovalbumin (Fig. 2).
In summary, these studies demonstrate that normal mice are able to generate ovalbumin-specific CTL restricted to CD1 in vivo. The highly conserved nature of CD1 proteins suggests that they may have evolved a specialized function to present nonpeptidic ligands to T cells (1). The crystal structure of CD1d1 reveals many differences from classical MHC molecules (32), including a deep and highly hydrophobic groove that would be structurally difficult to bind peptides in the manner of MHC molecules (33). However, there remains the possibility that CD1d may also present hydrophobic peptides that bind in a different configuration as suggested by the CD1d peptide–binding motif and synthetic peptide binding studies (2). The data presented here suggest that in addition to lipids and other nonpeptidic molecules, proteins can be recognized as antigens in the context of CD1 in vivo to prime CTL that are antigen specific.
CD1-reactive cells have been identified in mice, however, the physiological role of CD1 antigen presentation is unknown. One population in particular, the CD4+ NK1+ T cells, has been shown to make IL-4 (21, 34) in response to CD1. Although MHC class II° mice are relatively deficient in CD4+ cells, they have been described to contain greater numbers of CD4+ NK1+ T cells compared to wild-type mice (22). In these mice, with the absence of MHC II–restricted CD4+ T cells, CD4+ NK1+ cells appear to be important in providing help by serving as an early source of IL-2 in the priming of CD8+ CTL in the induction of resistance to infection by the intracellular parasite, Toxoplasma gondii (35). The physiologic relevance of CD1 presentation of antigens remains unclear since T cells that recognize these molecules have been described as having similar effector functions as MHC class I– and class II–restricted T cells. Whether CD1-restricted T cells fulfill an ancillary, regulatory, or completely independent role in the immune system warrants further investigation.
Acknowledgments
We gratefully acknowledge Dr. S. Balk for gifts of the CD1d1 cDNA and 3C11 ascites. We would like to thank P. Charos, N. Noon, and J. Uhle for their assistance. We are grateful to Dawne Page for her critical reading of the manuscript.
This work was supported in part by grant A96192 from the American Federation for Research on Aging, and grants AR07567 and AR25443 from the National Institutes of Health. M. Corr is an investigator of the Arthritis Foundation. D.J. Lee is supported in part by grants from the Markey Charitable Trust and the National Institute of General Medical Sciences GM07198.
References
- 1.Porcelli SA. The CD1 family: a third lineage of antigen-presenting molecules. Adv Immunol. 1995;59:1–98. doi: 10.1016/s0065-2776(08)60629-x. [DOI] [PubMed] [Google Scholar]
- 2.Castano AR, Tangri S, Miller JE, Holcombe HR, Jackson MR, Huse WD, Kronenberg M, Peterson PA. Peptide binding and presentation by mouse CD1. Science. 1995;269:223–226. doi: 10.1126/science.7542403. [DOI] [PubMed] [Google Scholar]
- 3.Martin LH, Calabi F, Milstein C. Isolation of CD1 genes: a family of major histocompatibility complex- related differentiation antigens. Proc Natl Acad Sci USA. 1986;83:9154–9158. doi: 10.1073/pnas.83.23.9154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Calabi F, Belt KT, Yu CY, Bradbury A, Mandy WJ, Milstein C. The rabbit CD1 and the evolutionary conservation of the CD1 gene family. Immunogenetics. 1989;30:370–377. doi: 10.1007/BF02425277. [DOI] [PubMed] [Google Scholar]
- 5.Calabi F, Jarvis JM, Martin L, Milstein C. Two classes of CD1 genes. Eur J Immunol. 1989;19:285–292. doi: 10.1002/eji.1830190211. [DOI] [PubMed] [Google Scholar]
- 6.Balk SP, Bleicher PA, Terhorst C. Isolation and expression of cDNA encoding the murine homologues of CD1. J Immunol. 1991;146:768–774. [PubMed] [Google Scholar]
- 7.Balk SP, Burke S, Polischuk JE, Frantz ME, Yang L, Porcelli S, Colgan SP, Blumberg RS. Beta 2-microglobulin-independent MHC class Ib molecule expressed by human intestinal epithelium. Science. 1994;265:259–262. doi: 10.1126/science.7517575. [DOI] [PubMed] [Google Scholar]
- 8.Hanau D, Fricker D, Bieber T, Esposito-Farese ME, Bausinger H, Cazenave JP, Donato L, Tongio MM, de la Salle H. CD1 expression is not affected by human peptide transporter deficiency. Hum Immunol. 1994;41:61–68. doi: 10.1016/0198-8859(94)90086-8. [DOI] [PubMed] [Google Scholar]
- 9.Brutkiewicz RR, Bennink JR, Yewdell JW, Bendelac A. TAP-independent, β2-microglobulin-dependent surface expression of functional mouse CD1.1. J Exp Med. 1995;182:1913–1919. doi: 10.1084/jem.182.6.1913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Teitell M, Holcombe HR, Brossay L, Hagenbaugh A, Jackson MJ, Pond L, Balk SP, Terhorst C, Peterson PA, Kronenberg M. Nonclassical behavior of the mouse CD1 class I-like molecule. J Immunol. 1997;158:2143–2149. [PubMed] [Google Scholar]
- 11.Sugita M, Jackman RM, van Donselaar E, Behar SM, Rogers RA, Peters PJ, Brenner MB, Porcelli SA. Cytoplasmic tail-dependent localization of CD1b antigen-presenting molecules to MIICs. Science. 1996;273:349–352. doi: 10.1126/science.273.5273.349. [DOI] [PubMed] [Google Scholar]
- 12.Porcelli S, Morita CT, Brenner MB. CD1b restricts the response of human CD4-8-T lymphocytes to a microbial antigen. Nature. 1992;360:593–597. doi: 10.1038/360593a0. [DOI] [PubMed] [Google Scholar]
- 13.Prigozy TI, Sieling PA, Clemens D, Stewart PL, Behar SM, Porcelli SA, Brenner MB, Modlin RL, Kronenberg M. The mannose receptor delivers lipoglycan antigens to endosomes for presentation to T cells by CD1b molecules. Immunity. 1997;6:187–197. doi: 10.1016/s1074-7613(00)80425-2. [DOI] [PubMed] [Google Scholar]
- 14.Peters PJ, Raposo G, Neefjes JJ, Oorschot V, Leijendekker RL, Geuze HJ, Ploegh HL. Major histocompatibility complex class II compartments in human B lymphoblastoid cells are distinct from early endosomes. J Exp Med. 1995;182:325–334. doi: 10.1084/jem.182.2.325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Peters PJ, Neefjes JJ, Oorschot V, Ploegh HL, Geuze HJ. Segregation of MHC class II molecules from MHC class I molecules in the Golgi complex for transport to lysosomal compartments. Nature. 1991;349:669–676. doi: 10.1038/349669a0. [DOI] [PubMed] [Google Scholar]
- 16.Tulp A, Verwoerd D, Dobberstein B, Ploegh HL, Pieters J. Isolation and characterization of the intracellular MHC class II compartment [see comments] Nature. 1994;369:120–126. doi: 10.1038/369120a0. [DOI] [PubMed] [Google Scholar]
- 17.Sanderson F, Kleijmeer MJ, Kelly A, Verwoerd D, Tulp A, Neefjes JJ, Geuze HJ, Trowsdale J. Accumulation of HLA-DM, a regulator of antigen presentation, in MHC class II compartments. Science. 1994;266:1566–1569. doi: 10.1126/science.7985027. [DOI] [PubMed] [Google Scholar]
- 18.Beckman EM, Porcelli SA, Morita CT, Behar SM, Furlong ST, Brenner MB. Recognition of a lipid antigen by CD1-restricted alpha beta+T cells. Nature. 1994;372:691–694. doi: 10.1038/372691a0. [DOI] [PubMed] [Google Scholar]
- 19.Sieling PA, Chatterjee D, Porcelli SA, Prigozy TI, Mazzaccaro RJ, Soriano T, Bloom BR, Brenner MB, Kronenberg M, Brennan PJ, et al. CD1-restricted T cell recognition of microbial lipoglycan antigens. Science. 1995;269:227–230. doi: 10.1126/science.7542404. [DOI] [PubMed] [Google Scholar]
- 20.Beckman EM, Melian A, Behar SM, Sieling PA, Chatterjee D, Furlong ST, Matsumoto R, Rosat JP, Modlin RL, Porcelli SA. CD1c restricts responses of mycobacteria-specific T cells. Evidence for antigen presentation by a second member of the human CD1 family. J Immunol. 1996;157:2795–2803. [PubMed] [Google Scholar]
- 21.Bendelac A, Lantz O, Quimby ME, Yewdell JW, Bennink JR, Brutkiewicz RR. CD1 recognition by mouse NK1+ T lymphocytes. Science. 1995;268:863–865. doi: 10.1126/science.7538697. [DOI] [PubMed] [Google Scholar]
- 22.Cardell S, Tangri S, Chan S, Kronenberg M, Benoist C, Mathis D. CD1-restricted CD4+T cells in major histocompatibility complex class II–deficient mice. J Exp Med. 1995;182:993–1004. doi: 10.1084/jem.182.4.993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sato Y, Roman M, Tighe H, Lee D, Corr M, Nguyen MD, Silverman GJ, Lotz M, Carson DA, Raz E. Immunostimulatory DNA sequences necessary for effective intradermal gene immunization. Science. 1996;273:352–354. doi: 10.1126/science.273.5273.352. [DOI] [PubMed] [Google Scholar]
- 24.Corr, M., H. Tighe, D.J. Lee, J. Dudler, M. Trieu, D.C. Brinson, and D.A. Carson. 1997. Costimulation provided by DNA immunization enhances anti-tumor immunity. J. Immunol. . 159:4999–5004. [PubMed]
- 25.Raz E, Carson DA, Parker SE, Parr TB, Abai AM, Aichinger G, Gromkowski SH, Singh M, Lew D, Yankauckas MA, et al. Intradermal gene immunization: the possible role of DNA uptake in the induction of cellular immunity to viruses. Proc Natl Acad Sci USA. 1994;91:9519–9523. doi: 10.1073/pnas.91.20.9519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Bradbury A, Belt KT, Neri TM, Milstein C, Calabi F. Mouse CD1 is distinct from and co-exists with TL in the same thymus. EMBO (Eur Mol Biol Organ) J. 1988;7:3081–3086. doi: 10.1002/j.1460-2075.1988.tb03173.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Bretscher P, Cohn M. A theory of self-nonself discrimination. Science. 1970;169:1042–1049. doi: 10.1126/science.169.3950.1042. [DOI] [PubMed] [Google Scholar]
- 28.Lafferty KJ, Cunningham AJ. A new analysis of allogeneic interactions. Aust J Exp Biol Med Sci. 1975;53:27–42. doi: 10.1038/icb.1975.3. [DOI] [PubMed] [Google Scholar]
- 29.Schwartz RH. Costimulation of T lymphocytes: the role of CD28, CTLA-4, and B7/BB1 in interleukin-2 production and immunotherapy. Cell. 1992;71:1065–1068. doi: 10.1016/s0092-8674(05)80055-8. [DOI] [PubMed] [Google Scholar]
- 30.Behar SM, Porcelli SA, Beckman EM, Brenner MB. A pathway of costimulation that prevents anergy in CD28- T cells: B7-independent costimulation of CD1- restricted T cells. J Exp Med. 1995;182:2007–2018. doi: 10.1084/jem.182.6.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Stenger S, Mazzaccaro R, Uyemura K, Cho S, Barnes PF, Rosat JP, Sette A, Brenner MB, Porcelli SA, Bloom B, et al. Differential effects of cytolytic T cell subsets on intracellular infection. Science. 1997;276:1684–1687. doi: 10.1126/science.276.5319.1684. [DOI] [PubMed] [Google Scholar]
- 32.Zeng Z-H, Castano AR, Segelke BW, Stura EA, Peterson PA, Wilson IA. Crystal structure of mouse CD1: an MHC-like fold with a large hydrophobic binding groove. Science. 1997;277:339–345. doi: 10.1126/science.277.5324.339. [DOI] [PubMed] [Google Scholar]
- 33.Brenner M, Porcelli S. Antigen presentation: a balanced diet. Science. 1997;277:332. doi: 10.1126/science.277.5324.332. [DOI] [PubMed] [Google Scholar]
- 34.Yoshimoto T, Paul WE. CD4pos, NK1.1pos T cells promptly produce interleukin 4 in response to in vivo challenge with anti-CD3. J Exp Med. 1994;179:1285–1295. doi: 10.1084/jem.179.4.1285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Denkers EY, Scharton-Kersten T, Barbieri S, Caspar P, Sher A. A role for CD4+ NK1.1+T lymphocytes as major histocompatibility complex class II–independent helper cells in the generation of CD8+ effector function against intracellular infection. J Exp Med. 1996;184:131–139. doi: 10.1084/jem.184.1.131. [DOI] [PMC free article] [PubMed] [Google Scholar]