Abstract
Treatment of WEHI 231 immature B lymphoma cells with an antibody against their surface immunoglobulin M (anti-IgM) induces apoptosis and has been studied extensively as a model of self-induced B cell tolerance. Since the tumor suppressor protein p53 has been implicated in apoptosis in a large number of cell types and has been found to be mutated in a variety of B cell tumors, here we sought to determine whether p53 and the p53 target gene cyclin-dependent kinase inhibitor p21WAF1/CIP1 were involved in anti-IgM–induced cell death. Anti-IgM treatment of WEHI 231 cells increased expression of p53 and p21 protein levels. Ectopic expression of wild-type p53 in WEHI 231 cells induced both p21 expression and apoptosis. Ectopic expression of p21 similarly induced apoptosis. Rescue of WEHI 231 cells from apoptosis by costimulation with CD40 ligand ablated the increase in p21 expression. Lastly, a significant decrease in anti-IgM–mediated apoptosis was seen upon downregulation of endogenous p53 activity by expression of a dominant-negative p53 protein or upon microinjection of an antisense p21 expression vector or antibody. Taken together, the above data demonstrate important roles for p53 and p21 proteins in receptor-mediated apoptosis of WEHI 231 B cells.
Keywords: p53 tumor suppressor, p21WAF1/CIP1, apoptosis, WEHI 231 B-lymphocytes, CD40 ligand
The WEHI 231 lymphoma has been used as a model of self-induced B cell tolerance (1). These cells express surface IgM (1–3), and cross-linking of these surface immunoglobulin receptors with an anti-IgM antibody inhibits DNA synthesis (1, 4). Anti-IgM treatment is followed by cell cycle arrest in the late G1 phase followed by apoptosis (5–8). Because of these features, WEHI 231 cells have been used extensively as a model of clonal deletion of B cells, facilitating dissection of molecular pathways leading to cell death. For example, we recently demonstrated that anti-IgM treatment of these cells causes a drop in activity of the nuclear factor (NF)-κB/Rel transcription factor family, resulting in decreased c-myc expression and induction of cell death (9–12).
A growing number of gene products have been revealed as components of the machinery leading to cell death. Among these, p53 is of particular interest. The p53 protein, originally identified as a cellular nuclear phosphoprotein bound to the large transforming antigen of the SV40 DNA virus (13, 14), has been shown to play important roles in control of progression through G1 into S phase, DNA repair, differentiation, tumor formation, and apoptosis (15– 17). Induction of p53 is often associated with activation of cell death, and ectopic expression of p53 can induce apoptosis (18). Thymocytes and hematopoietic cells from mice lacking a p53 gene show resistance to radiation and drug-induced apoptosis (19, 20), and fibroblasts from these mice show resistance to apoptosis (21). Interestingly, anti-IgM– induced cell death of immature B cells from mice null for the p53 gene was significantly reduced (22). The mechanism by which p53 exerts these effects is not clear, but seems to depend on the ability of p53 protein to act as a transcription factor.
One of the important p53 transcriptional target genes is the cyclin-dependent kinase (CDK)1 inhibitor p21WAF1/CIP1 (23–26). The p21 protein can convert active CDK to inactive species, controlling and coordinating cell cycle progression (27). The increase in p21 levels elicited by p53 protein upon cellular damage caused by irradiation or other toxic agents leads to CDK inhibition and cell cycle arrest (28, 29). Moreover, p21 activity has been implicated in apoptosis. Ectopic p21 expression induces cell death in MCF-7 breast carcinoma cells, and p21 levels increase during apoptosis of the RT4 human bladder tumor cell line (30–32). These findings suggest that at least some of the ability of p53 to promote apoptosis is mediated through its effects on p21 expression. Here we have investigated the involvement of p53 and its putative target gene p21 in apoptosis of WEHI 231 cells induced by anti-IgM treatment. Our results indicate p53 and p21 play important roles as intermediates in receptor-mediated apoptosis of these immature B lymphoma cells.
Materials and Methods
Cell Culture and Treatment Conditions.
WEHI 231 cells were maintained at 37°C in DMEM supplemented with 10% fetal bovine serum (FBS), 0.35% glucose, 4 mM glutamine, nonessential amino acids, 100 U/ml penicillin, 100 μg/ml streptomycin, and 50 μM 2-ME as previously described (9). Before treatment, cells were diluted to a density of 4 × 105 cells/ml with fresh warm media and allowed to incubate for a minimum of 4–5 h. Cells were treated with 1:1,000 dilution anti-μ heavy chain antibody (anti-IgM, GIBCO BRL, Gaithersburg, MD) for the indicated periods of time. For cotreatment with CD40 ligand (CD40L) and anti-IgM, WEHI 231 cells were incubated with CD40L fusion protein (1:10), consisting of the extracellular domains of CD40L and CD8α, and anti-CD8 (1:100) plus anti-IgM, as previously described (33). Stable temperature-sensitive p53-transfected WEHI 231 cells, prepared as described below, were cultured at 38.5°C. Where indicated, untreated cultures were shifted to 32.5°C to induce wild-type p53 activity.
Cell Death Assays.
For analysis of DNA laddering, ∼106 cells were used per sample according to the procedure of Smith et al. (34) as we have previously described (10, 11). For trypan blue analysis, cells were incubated with 0.2% trypan blue for 10–20 min and the percentage of cells excluding dye (viable cells) determined by examination under phase-contrast microscopy at ×100.
Construction of Transfected Cell Lines.
Electroporation of WEHI 231 cells was performed essentially as described (9, 10). WEHI 231 cells in exponential growth were washed once with medium and resuspended at a concentration of 2 × 107 cells/ml in DMEM supplemented with 20% FBS. Cells (250 μl) were preincubated on ice for 10 min with DNA (20–40 μg). Cells were transfected by electroporation at 240V and 960 μF using a Gene Pulser (Bio-Rad, Hercules, CA). After incubation on ice for 5 min, the cell suspension was mixed with 1.75 ml of complete medium, and incubated for 10 min at room temperature. Cell cultures were incubated for 48 h at 37° C before selection.
Stably transfected WEHI 231 cell lines expressing a temperature-sensitive p53 dominant negative protein were prepared using the expression vector pLTRp53cGVal135 (35–37). This vector expresses a p53 mutant in which alanine at position 135 is replaced with valine (35). WEHI 231 cells were electroporated with 40 μg DNA plus 2 μg of pSV2neo vector as a selectable marker. After 24–48 h, 1,000 μg/ml G418 (GIBCO BRL) was added to the cells, and cultures were maintained under antibiotic selection for ∼2 wk. Individual clones were obtained via limiting dilution. WEHI 231 cells transfected with pSV2neo DNA alone were used as controls.
For preparation of p21 inducible expression WEHI 231 cell lines, a modification of the Lac-Switch inducible mammalian expression vector (Stratagene, La Jolla, CA) was used. This system consists of p3′SS, a eukaryotic vector carrying the lacI gene expressing lac-repressor, and a eukaryotic lac operator–containing vector pOPRSVICAT driven by the RSV-LTR. To construct an inducible p21 expression vector, the HindIII and NotI cDNA fragment, which encodes full-length p21 protein, was excised from a human cDNA vector (pBS-p21A, gift of Dr. Y. Xiong, University of North Carolina, Chapel Hill, NC), and used to replace the chloramphenicol acetyl transferase (CAT) reporter gene in the pOPRSVICAT vector, generating a clone termed pOPRSVI-p21. Cells were electroporated with 30 μg pOPRSVI-p21 and 10 μg p3′SS, and selected for stable transfectants under 350 μg/ml hygromycin B (Boehringer Mannheim GmbH, Mannheim, Germany) and 1,000 μg/ml G418 to isolate mixed populations of p21 expressing cells, which were used in the experiments. As a control, cells were transfected with p3′SS only and selected with hygromycin.
Immunoblot Analysis.
For nuclear protein extraction, cells were washed twice in cold PBS and resuspended in cold TKM 10-10-5 (10 mM Tris-HCl, pH 7.6, 10 mM KCl, and 5 mM MgCl2) with 0.2 mM PMSF and 10 μg/ml leupeptin. After incubation on ice for 10 min, cells were lysed by addition of NP-40 to 0.5% (final concentration). Nuclei were removed by centrifugation and nuclear protein were extracted with RIPA buffer (10 mM Tris-HCl pH 7.5, 150 mM NaCl, 1% NP-40, 0.1% SDS, 1% sodium sarcosyl, 0.2 mM PMSF, 10 μg/ml leupeptin, and 1 mM dithiothreitol) as previously described (11). For total cellular protein isolation, the PBS-washed cell pellets were extracted with RIPA as above. Protein samples (20 μg for nuclear protein and 50 μg for total protein) were fractionated on a 10 or 12% polyacrylamide/SDS gel and transferred to polyvinylidene fluoride membrane. Immunoblotting was performed as previously described (11). The antibodies used were: p53, Pab 421 (Oncogene Science, Cambridge, MA); p21, sc-397 (Santa Cruz Biotechnology, Santa Cruz, CA); c-Myc, 50-23, an affinity-purified murine c-Myc specific antibody (provided by S. Hann, Vanderbilt University, Nashville, TN) (38).
Northern Blot Analysis.
Cytoplasmic RNA was isolated from 2 × 106 cells per sample. Cells were washed twice in ice cold PBS, resuspended in 400 μl of cold TKM, and swelled on ice for 10 min. After lysis upon addition of 20 μl 10% NP-40, the nuclei were removed by centrifugation. The resulting cytoplasmic supernatant was extracted with phenol/chloroform/isoamyl alcohol (10:10:1). RNA was precipitated from the aqueous supernatant with 0.1 vol of 3 M NaAc (pH 5.2) and 2.5 vol of absolute ethanol, and dissolved in diethyl pyrocarbonate–treated water. The p53 probe pLTRp53cGVal135 (35) was labeled by the random primer method of Feinberg and Vogelstein (39). Hybridization was performed as previously described (40).
Microinjection Analysis.
For microinjection analysis, a pOPRSVI-p21 construct with reverse orientation of the p21 cDNA insert was prepared. This vector expresses a noncoding p21 transcript (antisense) and was termed pOPRSVI-anti-p21. Similarly, a human p27 cDNA insert was cut from vector pGlu-p27H (provided by Dr. Y. Xiong) with HindIII and BamHI, and used to replace the CAT gene of pOPRSVICAT in an orientation yielding expression of antisense p27 RNA. This expression vector was termed pOPRSVI-anti-p27. Alternatively, affinity-purified antibody preparations against p21 or p27 (sc-397 and sc-528, respectively; Santa Cruz Biotechnology) were used.
WEHI 231 cells were allowed to attach to tissue culture plastic in the presence of culture medium containing 0.4% FBS, and were supplemented with 20 mM Hepes, pH 7.3. After 30 min of incubation at 37°C, duplicate areas (containing ∼200–300 cells/ sample) were encircled. For microinjection of expression plasmids, DNA samples were adjusted to 1 μg/μl in 130 mM KCl, and 10 mM sodium phosphate buffer, pH 7.3. Microinjections were performed using a Narishige micromanipulator (Narishige Co., Ltd., Tokyo, Japan) and borosilicate glass capillaries (tip diameter: 0.2 μm) under conditions of constant nitrogen flow at 0.4 pounds per square inch. After microinjection, cloning rings were placed over the microinjected areas and the medium was replaced with 10% FBS/DMEM. After 30 min of incubation at 37°C, cells were removed by gentle trituration, transferred to multiwell plates, and incubated at 37°C in the presence of anti-IgM. After 45 h, 0.1 vol trypan blue solution (0.04% final) was added to the wells and the wells were incubated for another 15 min, then the percentage of trypan blue positive was determined by phase-contrast microscopy. Alternatively, cells were microinjected with 4 μg of affinity-purified antibody in the absence or presence of cognate peptide. After microinjection, cells were treated with anti-IgM and processed after 18 h, as above.
Results
Anti-IgM Treatment of WEHI 231 Cells Induces p53 Protein Expression.
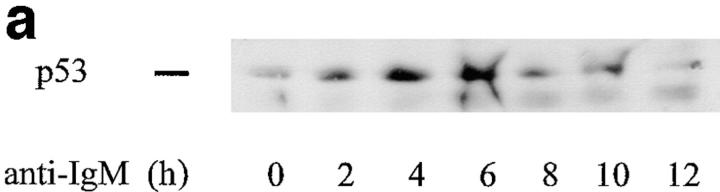
As an initial assessment of the contribution of p53 to receptor-mediated apoptosis of WEHI 231 cells, the expression of p53 protein after anti-IgM treatment was analyzed. Nuclear proteins were isolated from cells treated with anti-IgM for 0, 2, 4, 6, 8, 10, or 12 h, and were subjected to immunoblotting using a mouse monoclonal anti-p53 antibody. As seen in Fig. 1 a, untreated cells (0 h) expressed a low but detectable level of p53 protein. By 2 h of stimulation, p53 levels had increased ∼2.7-fold, and levels peaked between 4 and 6 h after treatment. Compared with untreated control cells, p53 levels had increased 6.2- and 6.7-fold, respectively, by 4 and 6 h. Between 10 and 12 h, p53 protein levels gradually returned to baseline values (2.1- to 1.1-fold). Thus, apoptosis initiated by anti-IgM treatment leads to a relatively rapid increase in p53 protein expression; the rise in p53 levels was similar to that seen in other cell types induced to undergo cell death (18).
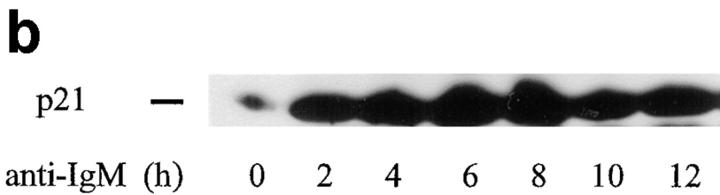
Figure 1.
Anti-IgM treatment induces p53 and p21 proteins. (a) p53 analysis. Nuclear proteins were isolated from exponentially growing WEHI 231 cells treated with anti-IgM for 0, 2, 4, 6, 8, 10, or 12 h, and samples (20 μg) were subjected to immunoblotting for p53 expression with a mouse monoclonal antibody. Detection was achieved using a horseradish peroxidase–conjugated secondary antibody and chemiluminescence as previously described (10). (b) p21 analysis. Total cellular proteins were prepared from WEHI 231 cells treated with anti-IgM for 0, 2, 4, 6, 8, 10, or 12 h, and samples (50 μg) were subjected to immunoblotting for expression of the CDK inhibitor p21 using an affinity-purified rabbit antibody (sc-397; Santa Cruz Biotechnology).
To assess whether the p53 protein that was induced after anti-IgM treatment was functional, we next determined whether expression of the CDK inhibitor p21, previously shown to be transcriptionally activated by p53 (23–26), followed a kinetic profile consistent with p53-mediated activation. Total cellular proteins were extracted from WEHI 231 cells treated with anti-IgM for the same time course as above, and subjected to immunoblot analysis for p21 expression (Fig.1 b). The low levels of p21 protein found in untreated cells were increased at 2 h after stimulation; the levels peaked at 6–8 h, and remained elevated throughout the 12-h time course. The kinetics of the observed changes in p21 expression paralleled, but were delayed with respect to, p53 induction. Thus, p53 expression increases after anti-IgM treatment, and p21 protein levels are elevated in a manner consistent with functionally active p53.
Ectopic Expression of p53 Induces Apoptosis of WEHI 231 Cells.
To determine whether ectopic expression of p53 protein can induce the death of WEHI 231 cells, stably transfected WEHI 231 cell lines expressing a temperature-sensitive murine p53 protein were prepared by using the expression vector pLTRp53cGVal135 (35). At the nonpermissive temperature of 32.5°C, these transfectants express a p53 that is wild type; at 38.5°C, they express a dominant-negative p53 that abrogates the endogenous p53 function (35–37).
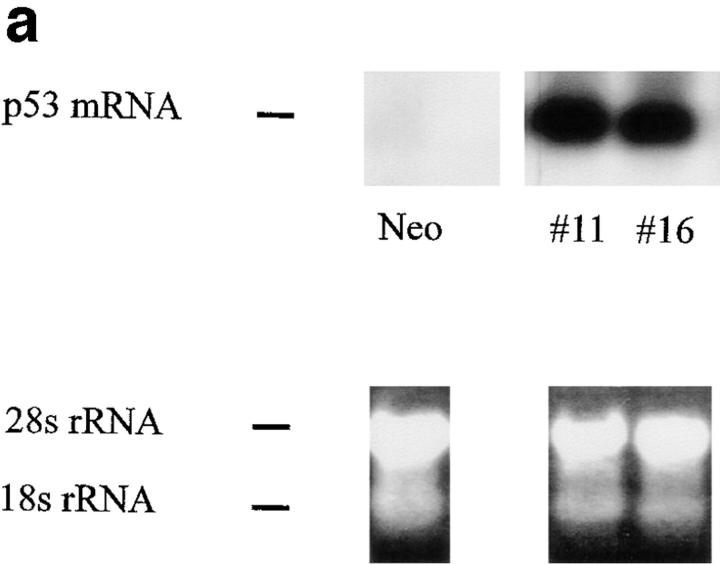
Northern and immunoblot analyses were used to verify the ectopic expression of p53 in the pLTRp53cGVal135 transfected lines maintained at 38.5°C. Cytoplasmic RNA was isolated from clones p53#11 and p53#16 and pSV2Neo cells, and samples subjected to Northern blotting (Fig. 2 a). At the nonpermissive temperature, clones p53#11 and p53#16 expressed high levels of p53 mRNA, whereas barely detectable levels of p53 were noted in the pSV2Neo clone. Ethidium bromide staining of the 28S and 18S rRNA in the gel indicated equal loading and integrity of the RNA samples. Furthermore, ectopic expression of p53 protein was confirmed using immunoblotting of total protein extracts from the above cell lines (Fig. 2 b). Lastly, to verify that these clones expressed functional p53 in a temperature-dependent fashion, the effects of a temperature shift on endogenous p21 expression were monitored (Fig. 2 c). 24 h after shifting to the permissive temperature, p21 protein levels were dramatically induced. These results confirm that the transfectants express a functional p53 protein.
Figure 2.
Expression of p53 and p21 in the temperature-sensitive p53 cell lines. WEHI 231 cell lines were electroporated with 40 μg DNA of pLTRp53cGVal135 ts p53 expression vector (35) plus 2 μg of pSV2Neo vector as a selective marker, and p53#11, p53#13, and p53#16 stable lines obtained by limiting dilution. Similarly, WEHI 231 cells were transfected with 2 μg of pSV2Neo alone to establish a population of control cells (Neo). Cell lines were maintained at 38.5°C where p53 is in a dominant negative mutant form. (a) p53 RNA analysis. Cytoplasmic RNA was isolated from clones p53#11 and p53#16 lines and pSV2Neo cells, and samples (20 μg) subjected to Northern blotting using the murine p53 DNA pLTRp53cGVal135 as probe (35). Bottom panel shows ethidium bromide staining of the 28S and 18S rRNA on the Northern blot, which indicates equal loading and RNA integrity. (b) p53 protein analysis. Total cellular proteins were extracted with RIPA buffer from p53#11, p53#16 and control Neo cells, and samples (50 μg) subjected to immunoblotting using the p53 antibody Pab421. (c) p21 protein analysis. Cultures of p53#11 and p53#16 cell lines were kept at 38.5°C or switched to 32.5°C, and total cellular proteins isolated after 24 h. Samples (50 μg) were analyzed for p21 protein by immunoblotting using an affinity purified rabbit antibody against p21 protein (sc-397, Santa Cruz Biotechnology).
Ectopic Expression of p53 Induces Apoptosis of WEHI 231 Cells.
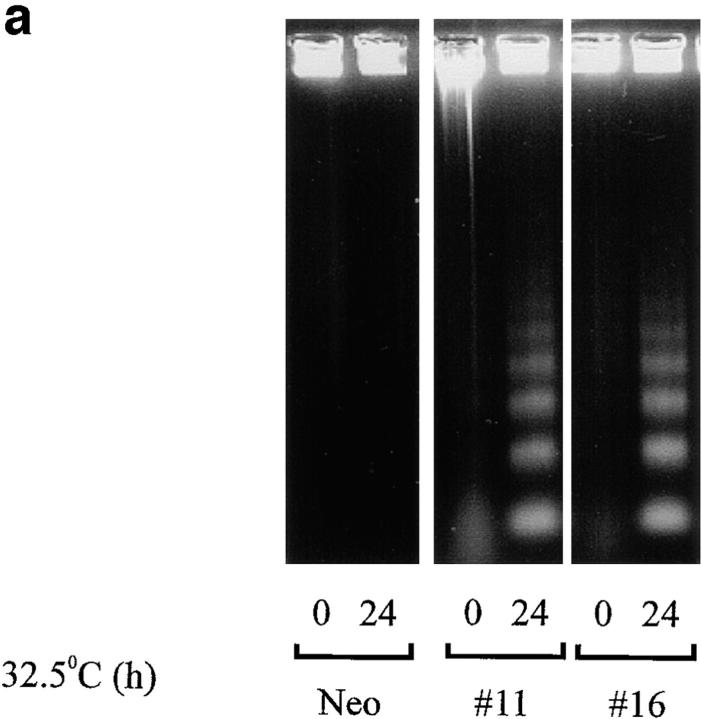
To determine whether induction of p53 expression activates apoptosis, DNA fragmentation assays were performed. The p53 clones p53#11, p53#16, and Neo cells were analyzed 24 h after a temperature switch to 32.5°C. Significant DNA laddering was observed after induction of wild-type p53 expression in the p53#11 and p53#16 cell lines (Fig. 3 a). As expected, only a low basal level of DNA laddering was detectable in the Neo cells after 24 (Fig. 3 a) or 48 (data not shown) h. Thus, ectopic expression of wild-type p53 is sufficient to induce apoptosis of WEHI 231 cells.
Figure 3.
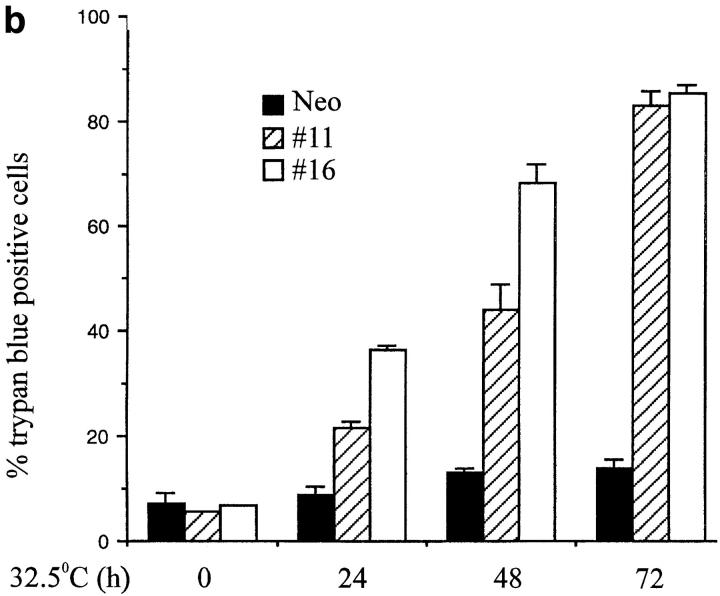
Induction of apoptosis by ectopic p53 expression. (a) DNA laddering. The p53 expressing clones p53#11, p53#16, and control Neo cells were cultured at 32.5°C for 0 or 24 h, and subjected to DNA ladder assays (34), as a measure of induction of apoptosis. (b) Trypan blue staining. The p53 expressing clones p53#11 (hatched bars), p53#16 (open bars) and control Neo (filled bars) cells were cultured in duplicate at 32.5°C for 0, 24, 48, or 72 h in 96-well dishes, and induction of cell death determined by trypan blue staining. Numbers of cells stained trypan are plotted as a function of time in hours. The statistical significance was obtained using the Student's t test, and the vertical bars represent the standard deviation.
To obtain more quantitative assessment of this killing, analysis of cell viability was performed using trypan blue exclusion at various times after temperature shift. The p53#11, p53#16, and control Neo cells were cultured in duplicate at 32.5°C for 0, 24, 48, or 72 h, and trypan blue– positive cells were measured. Fig. 3 b shows that both clone p53#11 and p53#16 exhibited a steady increase in cell death during the time course; the percentage of dead cells increased from 7% (in both) on day 0 to 82 and 84% on day 3 in the p53#11 and p53#16 cultures, respectively. In contrast, only a modest increase in cell death was observed with the Neo clone (from 7 to 12%). These results demonstrate that overexpression of p53 protein is sufficient to induce apoptosis of WEHI 231 cells.
Downregulation of Endogenous p53 Activity Inhibits Anti-IgM–induced Apoptosis.
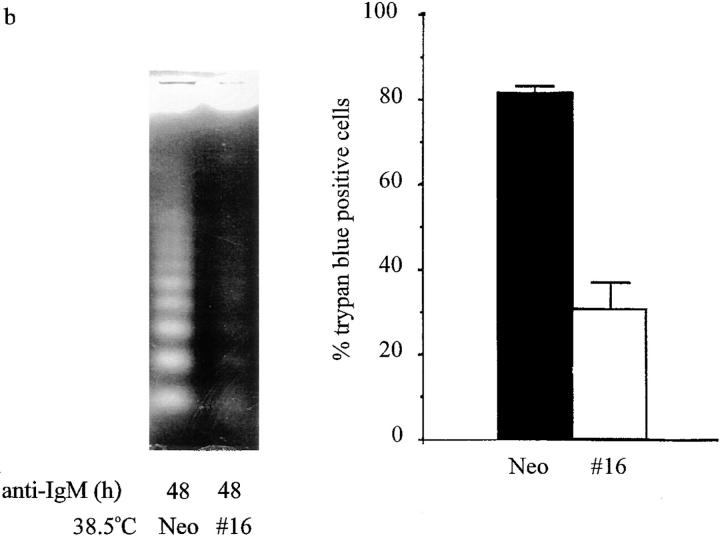
To determine whether p53 expression is necessary for anti-IgM–induced apoptosis, p53 clones p53#11, p53#16, and Neo cells were cultured at 38.5°C and treated with anti-IgM for 20, 32, or 48 h, as indicated. DNA fragmentation was extensive in the Neo cells treated with anti-IgM for 20 or 32 h (Fig. 4 a), or 48 h (Fig. 4 b), as expected. In contrast, inhibition of endogenous p53 in clone p53#11 (Fig. 4 a) and clone p53#16 (Fig. 4 b) greatly reduced DNA fragmentation in comparison with the Neo clone. Quantitative analysis of cell death in these clones in response to 48 h of anti-IgM treatment was obtained using trypan blue exclusion. Clones p53#11 and p53#16 demonstrated significant protection against anti-IgM–induced apoptosis compared with the Neo control line (Fig. 4, a and b). The percentage of cell death was reduced from 80% in the Neo line to 25 and 35% in clones p53#11 and p53#16, respectively. Similarly, in the mixed population of p53 transfectants, the percentage of cell death was reduced from 76.5 to 47.3% after treatment with anti-IgM for 48 h at 38.5°C (data not shown). Thus, absence of functional p53 activity allows for significant rescue of WEHI 231 cells from receptor-mediated apoptosis.
Figure 4.
Downregulation of endogenous p53 activity inhibits anti-IgM–induced apoptosis. (a) The p53 expressing clone p53#11 and Neo control cells were grown at 38.5°C and treated with anti-IgM for 0, 20, or 32 h, and analyzed for apoptosis by DNA ladder assays (left panel). Alternatively, p53#11 and Neo control cells were cultured in duplicate at 38.5°C and treated with anti-IgM for 48 h, and cell death was analyzed by trypan blue staining (right panel). Data are plotted as the mean percentage of cells stained positive for trypan blue. The statistical significance was obtained using the Student's t test, and the vertical bars represent the standard deviation. (b) The p53-expressing clone p53#16 and Neo control cells were cultured at 38.5°C and treated with anti-IgM for 48 h, and analyzed as above. Alternatively, p53#16 and Neo control cells were cultured in duplicate at 38.5°C and treated with anti-IgM for 0 or 48 h, and analyzed by trypan blue staining for cell death. Data are plotted as above.
Ectopic Expression of p21 Induces Apoptosis of WEHI 231 Cells.
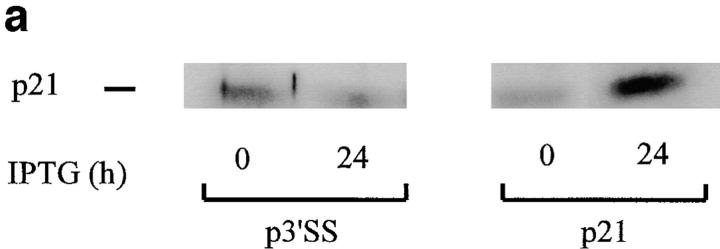
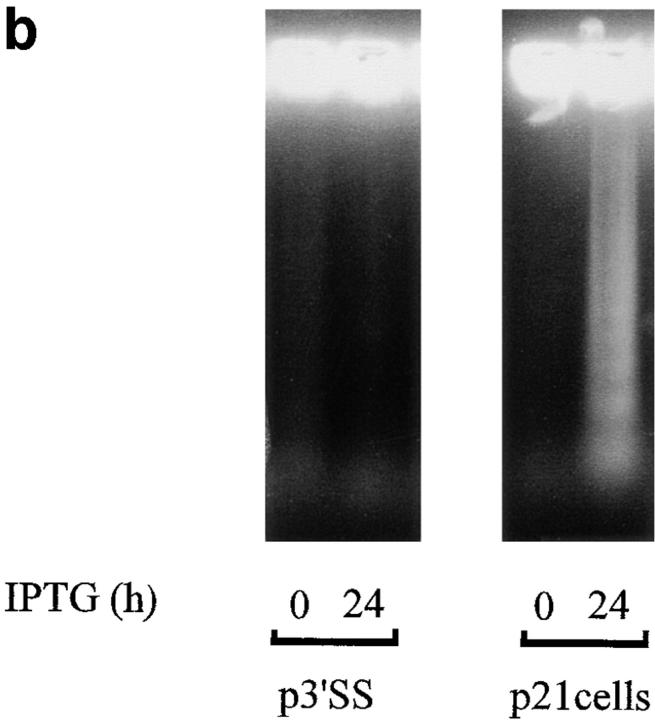
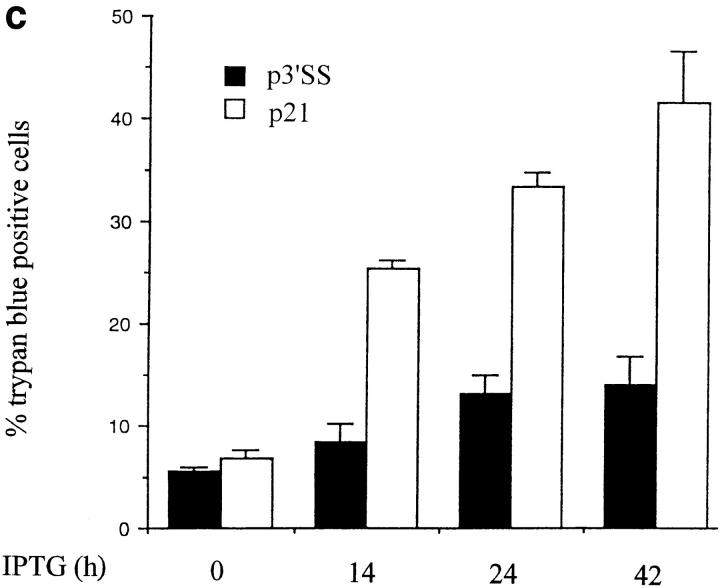
The above results indicate that p53 induction is sufficient to induce apoptosis, and that p53 induces p21 expression. To determine whether p21 induction alone is sufficient to produce apoptosis, stable WEHI 231 cells containing an inducible p21 expression vector were prepared using a modification of the isopropyl-β-d-thiogalacto-pyranoside (IPTG)-inducible LacSwitch expression vector system. This modified version consists of the p3′SS vector carrying the lacI gene, and a lac operator–containing eukaryotic expression vector pOPRSVICAT, in which the CAT reporter gene was replaced by a full length cDNA insert encoding p21; this new vector was termed pOPRSVI-p21. Stably transfected WEHI 231 cells were prepared with a combination of pOPRSVI-p21 and p3′SS vector, or with p3′SS vector alone, as control. A pool of stable transfectants were treated with 20 mM IPTG for 24 h to induce p21 protein expression; this dose of IPTG was found sufficient to inactivate the repressor protein (data not shown). Total cellular proteins were prepared and subjected to immunoblotting analysis to verify induction of p21 expression. IPTG treatment induced p21 expression in the population of pOPRSVI-p21 transfectants, but not in the control p3′SS cells, as expected (Fig. 5 a). To determine whether ectopic expression of p21 induced cell death, cultures were treated with 20 mM IPTG for 24 h and subjected to DNA fragmentation analysis. IPTG treatment of the population of pOPRSVI-p21 transfectants resulted in significant DNA fragmentation, whereas similar treatment of control p3′SS cells showed essentially no DNA fragmentation (Fig. 5 b). To more quantitatively measure cell death induced by p21 expression, cultures were treated in duplicate with 20 mM IPTG for 0, 14, 24, or 42 h, and cell viability was assessed via trypan blue staining (Fig. 5 c). A time-dependent increase in cell death was noted with the pOPRSVI-p21 transfectant population. By 42 h after treatment, ∼45% of the cells were dead. In contrast, only a slight 8% change in cell death was observed with the control p3′SS cells. Similarly, 31.0 ± 2.8% of WEHI 231 cells microinjected with the p21 expression vector were trypan blue positive after 18 h compared with 5.2 ± 2.0% of cells not microinjected or 6.5 ± 0.8% of cells microinjected with pBluescript vector DNA (data not shown). Taken together, these results demonstrate that ectopic p21 expression leads to apoptosis of WEHI 231 cells.
Figure 5.
Ectopic expression of p21 induces WEHI 231 cell death. WEHI 231 cells were electroporated with 30 μg pOPRSVI-p21 and 10 μg p3′SS vector encoding the Lac I repressor and a mixed population of p21 transfectants were selected using 350 μg/ml hygromycin B and 1,000 μg/ml G418. Alternatively, WEHI 231 cells electroporated with control p3′SS were selected with hygromycin alone. (a) p21 protein analysis. Total proteins were prepared from pOPRSVI-p21 and control p3′SS transfected cells after treatment with 20 mM IPTG for 24 h, and samples (50 μg) were subjected to immunoblot analyses for expression of p21. (b) DNA ladder assay. Cultures of the control pOPRSVI-p21 and p3′SS transfected cells were treated with 20 mM IPTG for 24 h and analyzed for apoptosis by a DNA ladder assay. (c)Trypan blue staining. Cultures of the pOPRSVI-p21 and p3′SS transfected cells were treated in duplicate with 20 mM IPTG for 24 h, and extent of cell death was assessed by trypan blue staining. Data are plotted as the mean percentage of cells stained positive for trypan blue. The statistical significance was obtained using the Student's t test, and the vertical bars represent the standard deviation.
CD40L Rescue of Anti-IgM–induced Apoptosis Correlates with Decreased Expression of p21.
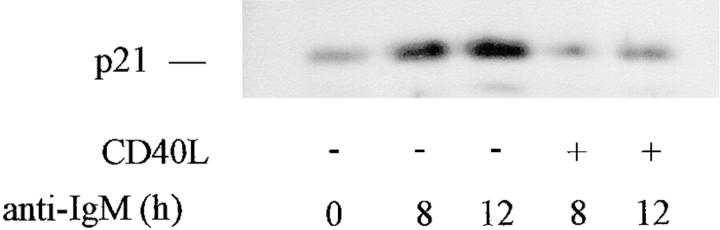
CD40L rescue of WEHI 231 cells from anti-IgM–induced apoptosis is accompanied by maintenance of NF-κB/Rel and c-myc expression (33), as well as of cellular proliferation (41). To determine whether CD40L affected p21 expression in response to anti-IgM treatment, exponentially growing WEHI 231 cells were treated with anti-IgM in the presence or absence of CD40L for 0, 8, or 12 h. Total cellular proteins were extracted, and samples were subjected to immunoblotting for p21 protein expression (Fig. 6). Treatment with anti-IgM alone led to the expected induction in p21 protein levels. However, induction of p21 was significantly ablated by cotreatment with CD40L plus anti-IgM (Fig. 6). Thus, CD40L-induced rescue of WEHI 231 cells from receptor-mediated apoptosis is accompanied by a block in the induction of the CDK inhibitor p21.
Figure 6.
CD40L rescue of anti-IgM–induced apoptosis is accompanied by a block in induction of p21 expression. WEHI 231 cultures were treated with anti-IgM in the absence (−) or presence (+) of CD40L for the indicated times, and total cellular proteins were prepared and samples (20 μg) were subjected to immunoblot analysis for p21 expression.
Reduction of CDK Inhibitor Expression Ablates Anti-IgM– induced Apoptosis.
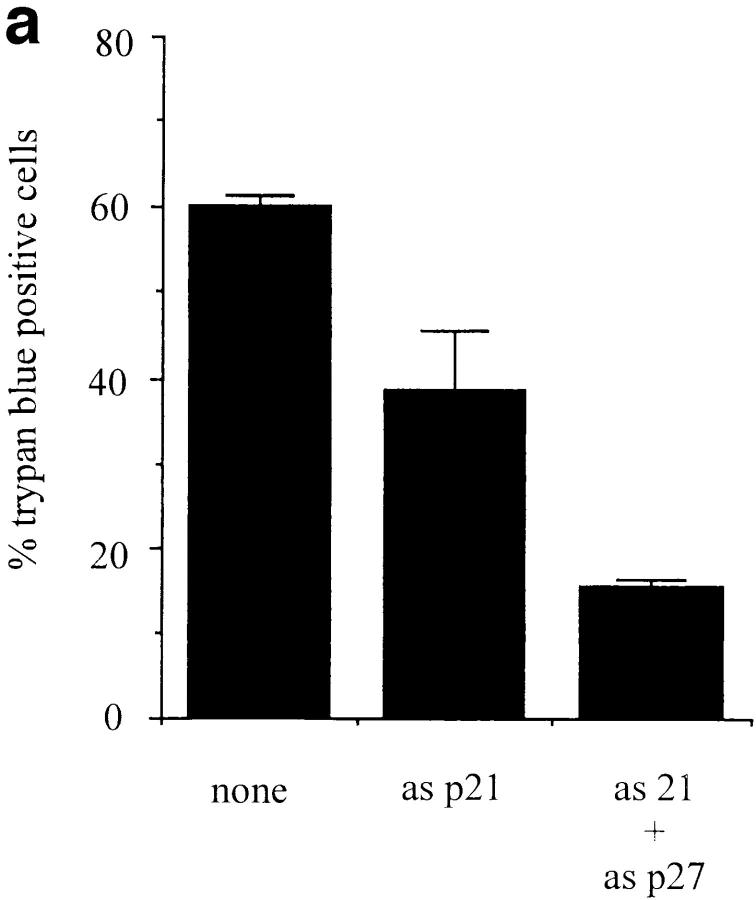
Since induction of p21 was sufficient to induce apoptosis of WEHI 231 cells, and CD40L rescue prevented the normal increase seen upon anti-IgM treatment, we next sought to determine whether the receptor-mediated increase in p21 expression was a necessary part of the signal transduction pathway(s) leading to apoptosis. WEHI 231 cells were microinjected with pOPRSVI-anti-p21, a plasmid yielding inducible expression of p21 transcript in an antisense orientation, and then treated immediately with anti-IgM. After incubation at 37°C for 45 h, cultures were analyzed for cell viability (Fig. 7). Treatment of nonmicroinjected cells with anti-IgM induced death in 60 ± 1.4% of the cells after 45 h, as previously seen (11). In contrast, only 38.7 ± 6.8% of pOPRSVI-anti-p21 microinjected cells were dead (P <0.05, Student's t test). Thus, antisense p21 is capable of inducing a significant amount of protection against anti-IgM–induced apoptosis. Recently, anti-IgM treatment has also been shown to induce expression of another CDK inhibitor, p27Kip1 (42). Thus, the effects of antisense expression of p27 in combination with antisense p21 on anti-IgM–mediated apoptosis were similarly assessed. The combination of antisense p21 and antisense p27 provided very significant protection, superior to that with the p21 vector alone. Cells microinjected with the combination of vectors exhibited 15.4 ± 1.1% cell death at 45 h after treatment with anti-IgM.
Figure 7.
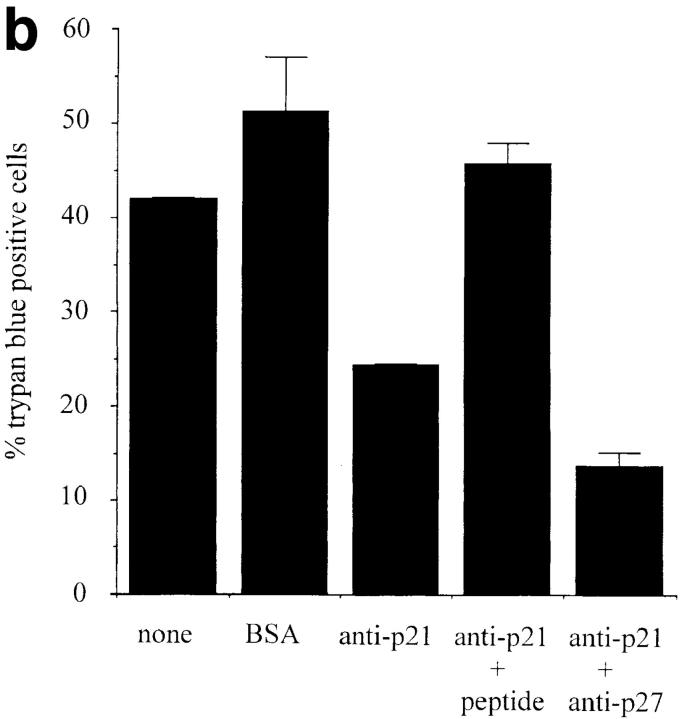
Antisense p21 and antisense p27 expression ablates anti-IgM– induced apoptosis. (a) WEHI 231 cells were microinjected with the indicated plasmids at a concentration of 0.5 μg/μl, and with enough pBluescript plasmid to bring the final DNA concentration to 1.0 μg/μl. Immediately after microinjection, cells were treated with anti-IgM and cultured for 45 h, and cell viability was assessed by trypan blue staining. Data are presented as the mean of duplicates, with standard deviation indicated. This data represents one experiment that is typical of three independent experiments with essentially identical results. The total number of cells analyzed per treatment ranged from 163 (asp21 + asp27) to 231 (none). (b) WEHI 231 cells either were not microinjected (none) or were microinjected with 4 μg/μl. BSA or affinity-purified antibody against p21 protein in the absence or presence of 4 μg/μl cognate peptide or 4 μg/μl affinity-purified antibody against p27 protein. Immediately after microinjection, cells were treated with anti-IgM and cultured for 18 h, and cell viability was assessed by trypan blue staining. Data are presented as the mean of duplicates, with standard deviation indicated. This data represents one experiment that is typical of two independent experiments with essentially identical results. The total number of cells analyzed per treatment ranged from 135 (BSA) to 231 (anti-p21).
To inhibit p21 activity more directly, cells were also microinjected with affinity purified antibodies to either p21 or p27 protein, and the effects on anti-IgM–mediated killing assessed. After 18 h, in the absence of microinjection, 41.9 ± 0.1% of cells were trypan blue positive, and this value was essentially unaffected by microinjection of BSA protein. In contrast, microinjection of an antibody against p21 protein significantly reduced the level of cell death (24.4 ± 0.1%), and this was specific as judged by reversal of protection upon incubation with the cognate peptide. Microinjection of the peptide alone did not affect cell viability (data not shown). Again, inhibition of both p21 and p27 protected to even a greater extent (only 13.7 ± 1.5% cell death). The specificity of the effect of the p27 antibody was also confirmed with its cognate peptide (data not shown). Thus, these results indicate that a portion of the signals leading to apoptosis is dependent on p21; many of the remaining signals appear mediated via a p27 pathway.
Discussion
The results presented here indicate that induction of p53 and p21 proteins after anti-IgM treatment is part of a critical pathway in receptor-mediated apoptosis of WEHI 231 B cells. Treatment of WEHI 231 cells with anti-IgM led to early increases in p53 protein expression and subsequently to increases in p21 protein levels. Ectopic expression of wild-type p53 induced both p21 expression and apoptosis. Consistent with a direct role for induction of p21 in cell death, ectopic expression of p21 alone also induced apoptosis of WEHI 231 cells. Furthermore, downregulation of endogenous p53 activity by expression of a dominant-negative p53 protein or by p21 antibody or antisense expression significantly ablated receptor-mediated apoptosis. Taken together, the above data demonstrate a pivotal involvement of p53 in anti-IgM–induced apoptosis of WEHI 231 B cells, which is probably mediated, at least in part, by the product of its target gene p21.
Mounting evidence, both in vitro and in vivo, supports a role for p53 function in control of B cell fate. Mutations of the p53 gene have been reported in 41% of Burkitt's lymphomas, 60% of B cell chronic lymphocytic leukemia, and 30% of high-grade non-Hodgkin's lymphomas (for reviews see references 43, 44). In addition, a recent report found p53 abnormalities in 75% of B cell prolymphocytic leukemia (45). Analysis of B cell tumors from p53-null mice revealed significantly decreased levels of immunoglobulin deposition when compared with tumors from wild-type mice (22). Furthermore, the p53-null mice contained more immature B cells in the bone marrow compared with wild-type mice. Importantly, these effects appear to relate to loss of normal receptor-mediated killing of these immature B cells (46). Treatment of the immature B cell population from p53-null mice with anti-IgM resulted in hyperproliferation (46) rather than the typical cell death response seen with immature B cells from normal mice (22), suggesting a role of p53 in control of B cell death. Furthermore, wild-type p53 has been implicated as a control protein in the differentiation pathway of B cells. Ectopic expression of wild-type p53 protein promotes differentiation of the Abelson murine leukemia virus–transformed pre-B cell line L12 (47). Differentiation of the pre-B cell line 70Z/3 in response to lipopolysaccharide is accompanied by increases in p53 expression (48). Lastly, ectopic expression of a dominant-negative p53 mutant is capable of blocking LPS-mediated differentiation (48).
Our work extends the list of genes whose protein products are involved in receptor-induced apoptotic pathways of WEHI 231 cells. Recent work from our lab has implicated the NF-κB/Rel transcription factor family and the c-myc oncogene as important intermediates in the signal transduction pathway leading to apoptosis (10, 11). Ectopic expression of either c-myc or c-rel prevents anti-IgM mediated apoptosis; direct inhibition of either c-Myc or Rel activity induces death of WEHI 231 cells (10, 11, 49). One potential model for relating the findings of this work to our previous studies is to postulate that c-myc is upstream of p53. Anti-IgM treatment leads to a transient increase by 1 h followed by eventual downmodulation of NF-κB/Rel activity; these modulations precede concomitant changes in transcription of the c-myc gene, which is strongly regulated by NF-κB/Rel in these cells (9). Interestingly the p53 promoter has been found to be activated by c-Myc in fibroblasts (50,51). Furthermore, p53 has been found to repress c-myc transcription in a human glial cell line (52), and we have observed that induction of wild-type p53, in our temperature-sensitive p53 lines, led to downregulation of c-myc expression (data not shown). Thus, one can propose that the initial increase in c-Myc expression leads to induction of p53, which then participates in promoting the subsequent downregulation of c-myc expression. The potential interrelationships between c-Myc and p53 expression after anti-IgM treatment, which are probably complex, are currently under investigation.
Anti-IgM has been previously shown to induce expression of p27 protein (42), although the mechanism is unknown. The evidence presented here suggests that p21 and p27 cooperate in mediating anti-IgM–induced apoptosis, as judged by the antisense experiments. Thus, these observations implicate p27 in receptor-mediated apoptosis in WEHI 231 cells. The fact that neither antisense reagent alone gives full protection suggests a divergence in pathways leading to apoptosis of these B cells: portions of the apoptotic signals initiated after cross-linking of surface IgM channel through p21 and others through p27. Thus, these findings suggest the existence of parallel signal transduction pathways diverging from the initiating events mediated by receptor–ligand interaction and working together to promote apoptosis.
Acknowledgments
We thank Drs. Y. Xiong, S. Hann, and T. Rothstein for generously providing cDNA clones, c-Myc antibody, and CD40 ligand, respectively.
This work was supported by National Institutes of Health grant CA-36355.
Abbreviations used in this paper
- CAT
chloramphenicol acetyl transferase
- CD40L
CD40 ligand
- CDK
cyclin-dependent kinase
- FBS
fetal bovine serum
- IPTG
isopropyl-β-d-thiogalacto-pyranoside
- NF
nuclear factor
- RSV-LTR
Rous Sarcoma Virus–long terminal repeat
Footnotes
Min Wu and Robert E. Bellas contributed equally to this work.
References
- 1.Boyd AW, Schrader JW. The regulation of growth and differentiation of a murine B-cell lymphoma. II. The inhibition of WEHI 231 by anti-immunoglobulin antibodies. J Immunol. 1981;126:2466–2469. [PubMed] [Google Scholar]
- 2.Monroe JG, Seyfert VL. Studies of surface immunoglobulin-dependent B cell activation. Immunol Res. 1988;7:136–151. doi: 10.1007/BF02918097. [DOI] [PubMed] [Google Scholar]
- 3.Ralph P. Functional subsets of murine and human B lymphocyte cell lines. Immunol Rev. 1979;48:107–121. doi: 10.1111/j.1600-065x.1979.tb00300.x. [DOI] [PubMed] [Google Scholar]
- 4.McCormack JE, Pepe VH, Kent RB, Dean M, Marshak-Rothstein A, Sonenshein GE. Specific regulation of c-myconcogene expression in a murine B-cell lymphoma. Proc Natl Acad Sci USA. 1984;811:5546–5550. doi: 10.1073/pnas.81.17.5546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Benhamou LE, Cazenave P-A, Sarthou P. Anti-immunoglobulins induce death by apoptosis in WEHI 231 B lymphoma cells. Eur J Immunol. 1990;20:1405–1407. doi: 10.1002/eji.1830200630. [DOI] [PubMed] [Google Scholar]
- 6.Hasbold J, Klaus GG. Anti-immunoglobulin antibodies induce apoptosis in immature B cell lymphomas. Eur J Immunol. 1990;20:1685–1690. doi: 10.1002/eji.1830200810. [DOI] [PubMed] [Google Scholar]
- 7.Warner GL, Ludlow JW, Nelson DO, Gaur A, Scott D. Anti-immunoglobulin treatment of B cell lymphomas induces active transforming growth factor beta but pRB hypophosphorylation is transforming growth factor beta independent. Cell Growth Differ. 1992;3:175–181. [PubMed] [Google Scholar]
- 8.Maheswaran S, McCormack JE, Sonenshein GE. Changes in phosphorylation of myc oncogene and RBantioncogene protein products during growth arrest of the murine lymphoma WEHI 231 cell line. Oncogene. 1993;6:1965–1971. [PubMed] [Google Scholar]
- 9.Lee H, Arsura M, Wu M, Duyao M, Buckler AJ, Sonenshein GE. Role of Rel-related factors in control of c-mycgene transcription in receptor-mediated apoptosis of the murine B cells WEHI 231 line. J Exp Med. 1995;181:1169–1177. doi: 10.1084/jem.181.3.1169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wu M, Lee H, Bellas RE, Schauer LS, Arsura M, Katz D, FitzGerald MJ, Rothstein TL, Sherr DH, Sonenshein GE. Inhibition of NF-κB/Rel induces apoptosis of murine B cells. EMBO (Eur Mol Biol Organ) J. 1996;15:4682–4690. [PMC free article] [PubMed] [Google Scholar]
- 11.Wu M, Arsura M, Bellas RE, FitzGerald MJ, Lee H, Schauer SL, Sherr DH, Sonenshein GE. Inhibition of c-mycexpression induces apoptosis of WEHI 231 murine B cells. Mol Cell Biol. 1996;16:5015–5025. doi: 10.1128/mcb.16.9.5015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sonenshein GE. Down-modulation of c-Myc expression induces apoptosis of B lymphocyte models of tolerance via clonal deletion. J Immunol. 1997;178:1994–1997. [PubMed] [Google Scholar]
- 13.Lane DP, Crawford LV. T antigen is bound to a host protein in SV40 transformed cells. Nature. 1979;278:261–263. doi: 10.1038/278261a0. [DOI] [PubMed] [Google Scholar]
- 14.Linzer DP, Levine AJ. Characterization of a 54K dalton cellular SV40 tumor antigen present in SV40-transformed cells and uninfected embryonal carcinoma cells. Cell. 1979;17:43–52. doi: 10.1016/0092-8674(79)90293-9. [DOI] [PubMed] [Google Scholar]
- 15.Vogelstein B, Kinzler KW. p53 function and dysfunction. Cell. 1992;70:523–526. doi: 10.1016/0092-8674(92)90421-8. [DOI] [PubMed] [Google Scholar]
- 16.Harris CC, Hollstein M. Medical progress: clinical implications of the p53 tumor-suppressor gene. N Engl J Med. 1993;329:1318–1327. doi: 10.1056/NEJM199310283291807. [DOI] [PubMed] [Google Scholar]
- 17.Carson DA, Lois A. Cancer progression and p53. Lancet. 1995;346:1009–1011. doi: 10.1016/s0140-6736(95)91693-8. [DOI] [PubMed] [Google Scholar]
- 18.Meikrantz W, Schlegel R. Apoptosis and cell cycle. J Cell Biochem. 1995;58:160–174. doi: 10.1002/jcb.240580205. [DOI] [PubMed] [Google Scholar]
- 19.Clarke AR, Purdie CA, Harrison DJ, Morris RG, Bird CC, Hooper ML, Wyllie AH. Thymocyte apoptosis induced by p53-dependent and independent pathways. Nature. 1993;362:849–852. doi: 10.1038/362849a0. [DOI] [PubMed] [Google Scholar]
- 20.Clarke AR, Gledhill S, Hooper ML, Bird CC, Wyllie AH. p53 dependence of early apoptotic and proliferative responses within the mouse intestinal epithelium following gamma-irradiation. Oncogene. 1994;9:1767–1773. [PubMed] [Google Scholar]
- 21.Hermeking H, Eick D. Mediation of c-Myc induced apoptosis of by p53. Science. 1994;265:2091–2093. doi: 10.1126/science.8091232. [DOI] [PubMed] [Google Scholar]
- 22.Shick L, Carman JA, Choi JK, Somasundaram K, Burrell M, Hill DE, Zeng YX, Wang Y, Wiman KG, Salhany K, et al. Decreased immunoglobulin deposition in tumor and increased immature B cells in p53-null mice. Cell Growth Differ. 1997;8:121–131. [PubMed] [Google Scholar]
- 23.Harper JW, Adami GR, Wei N, Keyomarsi K, Elledge SJ. The p21 cdk-interacting protein Cip1 is a potent inhibitor of G1 cyclin-dependent kinase. Cell. 1993;75:805–816. doi: 10.1016/0092-8674(93)90499-g. [DOI] [PubMed] [Google Scholar]
- 24.Xiong Y, Zhang H, Beach D. Subunit rearrangement of the cyclin-dependent kinases is associated with cellular transformation. Genes Dev. 1993;7:1572–1583. doi: 10.1101/gad.7.8.1572. [DOI] [PubMed] [Google Scholar]
- 25.Zhang H, Xiong Y, Beach D. Proliferating cell nuclear antigen and p21 are components of multiple cell cycle kinase complex. Mol Cell Biol. 1993;4:897–906. doi: 10.1091/mbc.4.9.897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Waga S, Hannon GJ, Beach D, Stillman B. The p21 inhibitor of cyclin-dependent kinases controls DNA replication by interaction with PCNA. Nature. 1994;369:574–578. doi: 10.1038/369574a0. [DOI] [PubMed] [Google Scholar]
- 27.Sherr CJ, Roberts JM. Inhibitors of mammalian G1cyclin-dependent kinases. Genes Dev. 1995;9:1149–1163. doi: 10.1101/gad.9.10.1149. [DOI] [PubMed] [Google Scholar]
- 28.El-Deiry WS, Tokino T, Velculesu VE, Levy DB, Parsons R, Trent JM, Lin D, Mercer WE, Kinzler KW, Vogelstein B. WAF1, a potential mediator of p53 tumor suppression. Cell. 1993;75:817–825. doi: 10.1016/0092-8674(93)90500-p. [DOI] [PubMed] [Google Scholar]
- 29.Dulic V, Kaufmann WK, Wilson SJ, Tlsty TD, Lees E, Harper JW, Elledge SJ, Reed SI. p53-dependent inhibition of cyclin-dependent kinase in human fibroblasts during radiation-induced G1arrest. Cell. 1994;76:1013–1023. doi: 10.1016/0092-8674(94)90379-4. [DOI] [PubMed] [Google Scholar]
- 30.Sheikh MS, Rochefort H, Garcia M. Overexpression of p21WAF1/CIP1induces growth arrest, giant cell formation and apoptosis in human breast carcinoma cell lines. Oncogene. 1995;11:1899–1905. [PubMed] [Google Scholar]
- 31.Sheikh MS, Garcia M, Zhan Q, Liu Y, Fornace AJ., Jr Cell cycle–independent regulation of p21Waf1/Cip1and retinoblastoma protein during okadaic acid–induced apoptosis is coupled with induction of Bax protein in human breast carcinoma cells. Cell Growth Differ. 1996;7:1599–1607. [PubMed] [Google Scholar]
- 32.Chresta CM, Masters JR, Hickman JA. Hypersensitivity of human testicular tumors to etoposide-induced apoptosis is associated with functional p53 and a high Bax: Bcl-2 ratio. Cancer Res. 1996;56:1834–1841. [PubMed] [Google Scholar]
- 33.Schauer SL, Wang Z, Sonenshein GE, Rothstein TL. Maintenance of NF-κB/Rel and c-mycexpression during CD4 ligand rescue of WEHI 231 cells from receptor-mediated apoptosis through modulation of IκB proteins. J Immunol. 1996;157:81–86. [PubMed] [Google Scholar]
- 34.Smith CA, Williams GT, Kingston R, Jenkinson EJ, Owen JJ. Antibodies to CD3/T-cell receptor complex induce death by apoptosis in immature T cells in thymic cultures. Nature. 1989;337:181–184. doi: 10.1038/337181a0. [DOI] [PubMed] [Google Scholar]
- 35.Finlay CA, Hinds PW, Tan TH, Eliyahu D, Oren M, Levine AJ. Activating mutations for transformation by p53 produce a gene product that forms an hsc70-p53 complex with an altered half-life. Mol Cell Biol. 1988;8:531–539. doi: 10.1128/mcb.8.2.531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Michalovitz D, Halevy O, Oren M. Conditional inhibition of transformation and of cell proliferation by a temperature-sensitive mutant of p53. Cell. 1990;62:671–680. doi: 10.1016/0092-8674(90)90113-s. [DOI] [PubMed] [Google Scholar]
- 37.Martinez J, Georgoff I, Martinez J, Levine AJ. Cellular localization and cell cycle regulation by a temperature-sensitive p53 protein. Genes Dev. 1991;5:151–159. doi: 10.1101/gad.5.2.151. [DOI] [PubMed] [Google Scholar]
- 38.Hann SR, Dixit M, Sears RC, Sealy L. The alternatively initiated c-Myc proteins differentially regulate transcription through a non-canonical DNA-binding site. Genes Dev. 1994;8:2441–2452. doi: 10.1101/gad.8.20.2441. [DOI] [PubMed] [Google Scholar]
- 39.Feinberg AP, Vogelstein B. A technique for radiolabeling DNA restriction endonuclease fragments to high specific activity. Anal Biochem. 1983;132:6–13. doi: 10.1016/0003-2697(83)90418-9. [DOI] [PubMed] [Google Scholar]
- 40.Arsura M, Wu M, Sonenshein GE. TGF-β1 inhibits NF-κB/Rel activity inducing apoptosis of B cells: transcriptional activation of IκBα. Immunity. 1996;5:31–40. doi: 10.1016/s1074-7613(00)80307-6. [DOI] [PubMed] [Google Scholar]
- 41.Tsubata T, Wu J, Honjo T. B-cell apoptosis induced by antigen receptor crosslinking is blocked by a T-cell signal through CD40. Nature. 1993;364:645–648. doi: 10.1038/364645a0. [DOI] [PubMed] [Google Scholar]
- 42.Ezhevsky SA, Toyoshima H, Hunter T, Scott DW. Role of cyclin A and p27 in anti-IgM–induced G1growth arrest of murine B cell lymphomas. Mol Biol Cell. 1996;7:553–564. doi: 10.1091/mbc.7.4.553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Newcomb EW. p53 gene mutations in lymphoid diseases and their possible relevance to drug resistance. Leuk Lymphoma. 1995;17:211–221. doi: 10.3109/10428199509056825. [DOI] [PubMed] [Google Scholar]
- 44.Ichikawa A, Hotta T, Saito H. Mutations of the p53 gene in B-cell lymphoma. Leuk Lymphoma. 1993;11:21–25. doi: 10.3109/10428199309054727. [DOI] [PubMed] [Google Scholar]
- 45.Lens D, De Schouwer PJ, Hamoudi RA, Abdul-Rauf M, Farahat N, Matutes E, Crook T, Dyer MJ, Catovsky D. p53 abnormalities in B-cell prolymphocytic leukemia. Blood. 1997;89:2015–2023. [PubMed] [Google Scholar]
- 46.Norvell A, Mandik L, Monroe JG. Engagement of the antigen receptor on immature murine B lymphocytes results in death by apoptosis. J Immunol. 1995;154:4403–4413. [PubMed] [Google Scholar]
- 47.Shaulsky G, Goldfinger A, Peled A, Rotter V. Involvement of wild type p53 in pre-B-cells differentiation in vitro. . Proc Natl Acad Sci USA. 1991;88:8982–8986. doi: 10.1073/pnas.88.20.8982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Aloni-Grinstein R, Zan-Bar I, Alboum I, Goldfinger N, Rotter V. Wild-type p53 functions as a control protein in the differentiation pathway of the B cell-lineage. Oncogene. 1993;8:3297–3305. [PubMed] [Google Scholar]
- 49.Wu M, Yang W, Bellas RE, Arsura M, Schauer LS, Katz D, FitzGerald MJ, Lee HY, Sonenshein GE. c-Myc promotes survival of WEHI 231 B lymphoma cells from apoptosis. Curr Top Microbiol Immunol. 1997;224:91–101. doi: 10.1007/978-3-642-60801-8_9. [DOI] [PubMed] [Google Scholar]
- 50.Reisman D, Elkind NB, Roy B, Beamon J, Rotter V. c-myctransactivates the p53 promoter through a required downstream CACGTG motif. Cell Growth Differ. 1993;4:57–65. [PubMed] [Google Scholar]
- 51.Roy B, Beamon J, Balint E, Reisman D. Transactivation of the human p53 tumor suppressor gene by c-Myc/ Max contributes to elevated mutant p53 expression in some tumors. Mol Cell Biol. 1994;14:7805–7815. doi: 10.1128/mcb.14.12.7805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Moberg KH, Tyndall WA, Hall DJ. Wild-type murine p53 represses transcription from the murine c-mycpromoter in a human glial cell line. J Cell Biochem. 1992;49:208–215. doi: 10.1002/jcb.240490213. [DOI] [PubMed] [Google Scholar]