Abstract
Memory B cells isolated from human tonsils are characterized by an activated cell surface phenotype, localization to mucosal epithelium, expression of somatically mutated immunoglobulin (Ig) variable (V) region genes, and a preferential differentiation into plasma cells in vitro. In spleens of both humans and rodents, a subset of memory B cells is believed to reside in the marginal zone of the white pulp. Similar to tonsil-derived memory B cells, splenic marginal zone B cells can be distinguished from naive follicular B cells by a distinct cell surface phenotype and by the presence of somatic mutations in their Ig V region genes. Although differences exist between human naive and memory B cells, no cell surface molecules have been identified that positively identify all memory B cells. In this study, we have examined the expression of the receptor-type protein tyrosine phosphatase CD148 on human B cells. CD148+ B cells present in human spleen exhibited characteristics typical of memory B cells. These included an activated phenotype, localization to the marginal zone, the expression of somatically mutated Ig V region genes, and the preferential differentiation into plasma cells. In contrast, CD148− B cells appeared to be naive B cells due to localization to the mantle zone, the expression of surface antigens typical of unstimulated B cells, and the expression of unmutated Ig V region genes. Interestingly, CD148+ B cells also coexpressed CD27, whereas CD148− B cells were CD27−. These results identify CD148 and CD27 as markers which positively identify memory B cells present in human spleen. Thus, assessing expression of these molecules may be a convenient way to monitor the development of memory B cell responses in immunocompromised individuals or in vaccine trials.
Keywords: human memory B cells, marginal zone B cells, somatic mutation, CD148, CD27
The differentiation of naive B cells into memory B cells is a complex procedure involving antigen (Ag), Ag-specific T cells, cytokines, and accessory cells such as dendritic cells and follicular dendritic cells (1–6). Ag-specific differentiation of naive to memory B cells occurs within germinal centers (GC),1 which are highly specialized areas of secondary lymphoid areas such as lymph nodes, spleen, and tonsil (1, 7–10). Within GC, activated naive B cells undergo vigorous proliferation, somatic mutation of Ig V region genes, Ig isotype switching, and selection after interaction with specific Ag (4, 7, 8, 11–14). Numerous B cell subsets have been identified in tonsil, spleen, and peripheral blood that are believed to represent different stages of development of a naive B cell into a memory B cell. In human tonsil, at least five distinct subpopulations of mature human B cells (Bm1–Bm5) have been identified (6, 10, 14). Bm1 B cells are small resting B cells that express high levels of surface (s)IgD. Bm2 B cells represent activated Bm1 cells that express sIgD and the early activation Ag CD23. Both Bm1 and Bm2 cells express unmutated Ig V region genes (15). Bm3 cells have downregulated expression of sIgD and exhibit a high proliferative index. Somatic hypermutation is believed to occur at the Bm3 stage of B cell development (12, 13, 15). Bm4 cells represent differentiated Bm3 cells that, depending on the affinity of their Ig receptor after somatic hypermutation, can undergo either positive selection or apoptosis. The cells exhibiting the greatest affinity for Ag then develop into the Bm5 subset of B cells, which express the downstream Ig isotypes IgG or IgA as well as activation Ags such as CD80, CD86, and CD95 (10, 14, 16). According to this criterion, naive B cells belong to the Bm1 and Bm2 subsets, whereas the Bm5 subset corresponds to differentiated memory B cells. Naive and memory B cells present in human tonsil also differ from one another with respect to their anatomical distribution. Thus, memory B cells colonize the mucosal epithelium, whereas naive B cells can be found within the follicular area of the lymphoid tissue (16). In peripheral blood and bone marrow, memory B cells have been identified that express either class-switched Ig isotypes, IgM and IgD, or IgM only (17–20). In spleen, there exist two main B cell populations that not only are phenotypically distinct but, similar to tonsillar B cells, also localize to separate areas of the lymphoid tissue, namely the follicular mantle zone and marginal zone. Mantle zone B cells primarily express an sIgD++sIgM+CD21+CD23+ phenotype, whereas marginal zone B cells are sIgD+sIgM++CD21++CD23± (21–24). Additionally, the Ig V region genes of mantle zone B cells are unmutated, whereas those of marginal zone B cells exhibit somatic mutations (25). Finally, in situ analysis of responses to T cell–dependent Ag demonstrated that marginal zone B cells were Ag-specific B cells (26). Based on these differences, mantle zone B cells are believed to be naive B cells, while marginal zone B cells are memory B cells.
Although memory B cells can be identified by the lack of expression of sIgD and CD38 or by the expression of switched Ig isotypes IgG and IgA (2, 3, 6, 16), this criterion excludes non–isotype-switched memory B cells (17–20, 27). To date, no cell surface molecules have been described that are expressed by all memory B cells. The identification of cell surface molecules expressed exclusively by either naive or memory B cells would assist in the further biological characterization of B cell subsets, as well as in the assessment of memory responses in vivo. CD148 is a receptor-type protein tyrosine phosphatase expressed on all human leukocytes (28). We have now examined the expression of CD148 on human B cells and have identified CD148, as well as CD27, as markers for memory B cells present in human spleen.
Materials and Methods
Antibodies/Cytokines.
The anti-CD148 mAb (28) was labeled with FITC by established protocols. PE-labeled anti-CD148 was prepared by PharMingen (San Diego, CA). The following mAbs were used in this study: unconjugated, FITC-, PE-, or PerCp- labeled anti-CD2, CD3, CD4, CD8, CD10, CD14, CD16, CD20, CD23, CD38, CD56, CD80, and HLA-DR (Becton Dickinson, San Jose, CA); PE-labeled anti–human IgD, IgG, IgM, CD21, CD27, and CD86 (PharMingen); F(ab′)2 fragments of affinity-purified goat anti–human IgD antiserum, alkaline phosphatase–conjugated affinity-purified swine anti–goat Ig antiserum (BioSource International, Camarillo, CA); FITC-labeled anti–human IgA (Sigma Chemical Co., St. Louis, MO); anti-CD40 (mAb 89; provided by J. Banchereau, Schering-Plough Laboratory for Immunological Research, Dardilly, France [29]); anti–signaling lymphocytic activation molecule (SLAM; A12 [30]), CD39 (A1 [30]), and CD95 (DX2 [31]). IL-2, IL-4, and IL-10 were provided by S. Menon (DNAX Research Institute of Molecular and Cellular Biology). Staphylococcus aureus Cowan (SAC) was purchased from Calbiochem Corp. (La Jolla, CA).
Immunofluorescent Staining.
Splenic mononuclear cells (MNC) were incubated with FITC- or PE-labeled anti-CD148 mAb, PerCp-labeled anti-CD20 mAb, and either FITC- or PE-labeled isotype control mAb, anti-IgM, IgD, IgG, IgA, CD10, CD21, CD23, CD27, CD38, CD39, CD40, CD80, CD86, HLA-DR, SLAM, or CD95 mAb on ice for 30 min. The cells were then washed twice, fixed in 1% paraformaldehyde, and analyzed on a FACScan® flow cytometer (Becton Dickinson). The expression of the different Ags was assessed by collecting data for CD20+ cells and analyzing CD148+ and CD148− populations of cells. Surface staining was measured on a logarithmic scale. 1–2 × 104 events were collected per sample, and the data were analyzed using CellQuest software (Becton Dickinson).
Purification of Splenic B Cells.
Splenic MNC were depleted of non-B cells by negative selection by incubation on ice for 30 min with saturating amounts of anti-CD2, CD3, CD4, CD8, CD14, CD16, and CD56 mAb. The cells were then washed twice with PBS, mixed with an equal volume of sheep anti–mouse Ig conjugated to magnetic beads (Dynal A.S., Oslo, Norway), and incubated on a rotating platform for 30 min at 4°C. Unbound cells were collected after the removal of positive cells by a magnetic field. Splenic B cells were further purified by cell sorting using a FACSVantage® or a FACStarPlus (Becton Dickinson) by incubating the recovered cell population with saturating amounts of FITC–anti-CD148 and PE–anti-CD20 mAbs. Gates were set to collect CD148+ and CD148−CD20+ B cells. Dead cells were excluded by the inclusion of propidium iodide (2 μg/ml). On reanalysis, the sorted B cell subsets were always >98% CD148+CD20+ and CD148−CD20+ (data not shown).
Giemsa Staining.
Sort-purified CD148+ and CD148− splenic B cells (4 × 104) were cytocentrifuged for 5 min at 500 rpm onto microscope slides, air dried, and then fixed in methanol for 10 min at room temperature. The slides were incubated with Giemsa stain (BDH Chemicals Ltd., Essex, UK) diluted 1:5 with distilled H2O for 15 min at room temperature, then washed with distilled water.
Immunohistology.
Serial sections of spleen tissue were incubated with a control mAb, or with mAb specific for IgM or CD27. Bound mouse mAb was visualized using a mouse Ig–specific Vectastain kit (Vector Laboratories, Inc., Burlingame, CA), according to the manufacturer's instructions. IgD was subsequently detected by the addition of goat anti-IgD antiserum and visualized with alkaline phosphatase–conjugated affinity-purified swine anti–goat Ig antiserum and phosphatase-specific substrate.
Sequence Analysis of VH Genes.
Total RNA was isolated from sort-purified CD148+ and CD148− splenic B cells by guanidine isothiocyanate extraction using the GlassMax RNA Isolation kit (GIBCO BRL, Gaithersburg, MD). 5 μl of total RNA was reverse transcribed into cDNA using oligo-dT (Boehringer Mannheim Corp., Indianapolis, IN) as primer and Superscript II RNase H− Reverse Transcriptase (GIBCO BRL) in a total volume of 25 μl containing 50 mM Tris-HCl, pH 8.3, 75 mM KCl, 3 mM MgCl2, 10 mM dithiothreitol, and 1 mM of each dNTP. The reverse transcription reaction was performed at 42°C for 90 min. The VH5 and VH6 genes were amplified from 1 μl of reverse-transcribed cDNA by PCR in a total volume of 50 μl containing 10 mM Tris-HCl, pH 8.3, 50 mM KCl, 1.5 mM MgCl2, 250 μM of each dNTP, 1.25 U AmpliTaq DNA polymerase (Perkin-Elmer Corp., Foster City, CA), and 50 pmol of primers corresponding to the 5′ region of the VH5 or VH6 leader sequences (VH5 ATG GGG TCA ACC GCC ATC CT; VH6 ATG TCT GTC TCC TTC CTC AT) and to the 3′ Cμ constant region (GTC CTG TGC GAG GCA GCC AA; all primer sequences were from reference 32). The PCR consisted of 35 cycles of 1 min of denaturation at 94°C, 1 min of annealing at 55°C, and 1 min of extension at 72°C in a thermocycler (Gene Amp 9600; Perkin-Elmer Corp.). Amplified PCR products were purified from agarose gels, ligated into the pCR2.1 vector (Invitrogen, Carlsbad, CA), and used to transform TOP10F′ bacteria (Invitrogen). Individual clones were selected, expanded overnight, and then plasmid DNA was recovered. DNA sequencing was performed in both directions using the dideoxy termination technique with a Sequenase 2.0 kit (United States Biochemical Corp., Cleveland, OH) and by using an automated nucleic acid sequencer (Applied Biosystems, Inc., Foster City, CA). Forward (ATT ACG CCA AGC TTG GTA CCG) and reverse (ATT GGG CCC TCT AGA TGC ATG) primers that flanked the multicloning site of the pCR2.1 vector were used for the sequencing reactions. Nucleotide sequences were analyzed using the Sequencher program, and comparisons were performed using the GenBank database.
B Cell Cultures.
For Ig secretion, 1–2.5 × 104 sort-purified B cells were cultured in 200 μl in the wells of a 96-well round-bottomed culture plate with either anti-CD40 mAb (15 μg/ml) plus IL-4 (400 U/ml), anti-CD40 mAb plus IL-10 (100 U/ml), and IL-2 (100 U/ml), or SAC (0.1%) plus IL-2 with or without IL-10 or anti-CD40 mAb. B cells were cultured in four to eight replicates, depending on the number of cells recovered after cell sorting. The secretion of IgA, IgG, IgG4, IgE, and IgM were determined after 12–14 d by Ig H chain isotype-specific ELISAs (33, 34). For induction of differentiation of B cells into plasma cells, sort-purified CD148+ and CD148− splenic B cells were cultured with anti-CD40 mAb (15 μg/ml), IL-2 (100 U/ml), and IL-10 (100 U/ml) for 3 d. The cells were then washed and recultured for 3 d in medium containing IL-2 and IL-10 only (35). All cultures were performed using Yssel's medium (36) supplemented with 10% FCS, 40 μg/ml transferrin (Pierce Chemical Co., Rockford, IL), 0.5% BSA, and 5 μg/ml insulin, and were carried out at 37°C in 5% CO2.
Results
Expression of CD148 Defines Phenotypically and Morphologically Distinct Subpopulations of Human B Cells
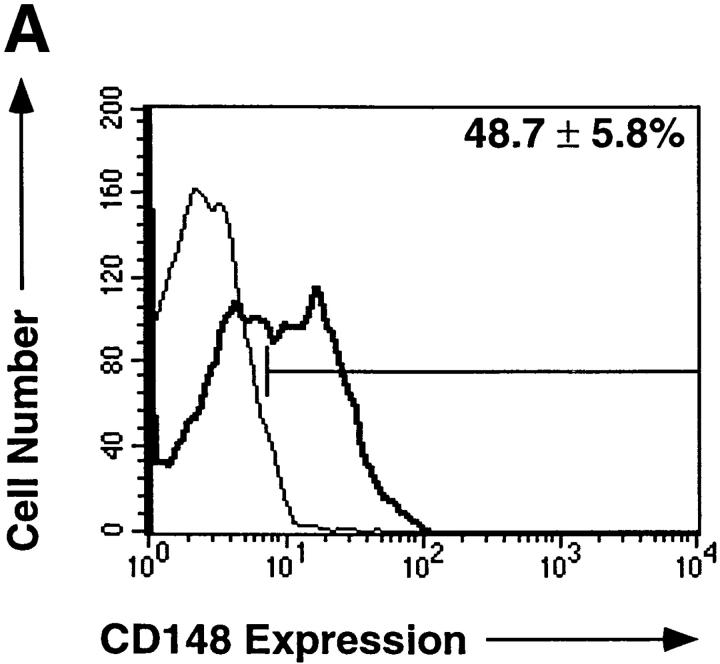
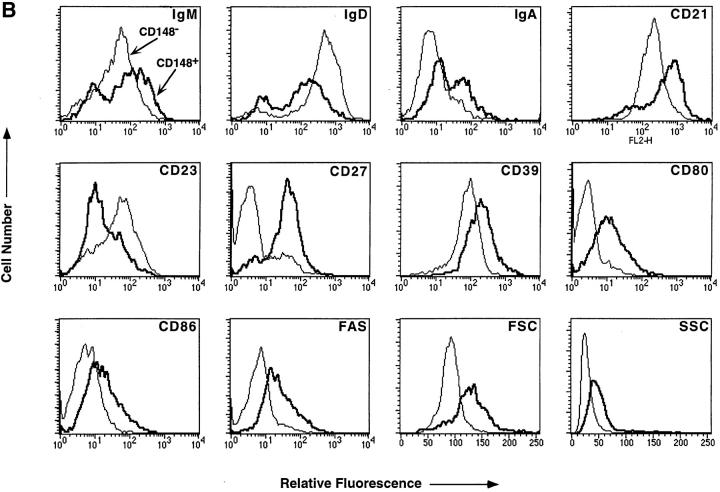
The A3 mAb recognizes CD148, a receptor-type protein tyrosine phosphatase expressed on all human leukocytes (28). In human spleen, CD148 was found to be expressed on ∼50% of B cells (Fig. 1 A). Three-color immunofluorescence revealed that the CD148+ and CD148− splenic B cells were phenotypically distinct. The expression of different sIg isotypes and other B cell molecules by CD148+ and CD148− B cells differed with respect to both the proportion of positive cells and the level of expression of these molecules (Fig. 1 B, and Table 1). Thus, while ∼90% of CD148− B cells expressed a high level of sIgD, fewer CD148+ B cells expressed sIgD, and the level of expression on CD148+ B cells was less than on CD148− B cells. In contrast, a greater proportion of CD148+ B cells expressed sIgM and sIgA than CD148− B cells. Furthermore, the expression level of these Ig isotypes on CD148+ B cells was 2.5-fold greater than CD148− B cells. The majority of CD148− B cells expressed CD23; however, CD27+ B cells resided in the CD148+ B cell subset. CD21 and CD39 were expressed by all B cells, yet their expression was 2–5-fold greater on CD148+ B cells than on CD148− B cells. Similarly, although CD80, CD86, CD95, and SLAM were present at low levels on some CD148− B cells, they were expressed at a level that was also 2–5-fold greater on an increased number of CD148+ B cells (Fig. 1 B, and Table 1). Overall, the phenotype of splenic CD148+ B cells was
Figure 1.
CD148+ and CD148− human B cells represent phenotypically distinct subpopulations of B cells. (A) MNC from human spleen were incubated with PE-labeled anti-CD20 mAb and either FITC-labeled anti-CD148 mAb (bold histogram) or FITC-labeled isotype control IgG1 mAb (thin histogram). The FITC fluorescence of CD20+ B cells was assessed by flow cytometric analysis. The values represent the mean ± SD of CD148+ B cells from nine different donor spleens. (B) MNC from human spleen were incubated with FITC- or PE-labeled anti-CD148, PerCp-labeled anti-CD20, and one of the following PE- or FITC-labeled mAbs: anti-IgM, anti-IgD, anti-IgA, anti-CD21, anti-CD23, anti-CD27, anti-CD39, anti-CD80, anti-CD86, or anti-CD95. CD148+CD20+ (bold histogram) and CD148−CD20+ (thin histogram) cells were gated, and the fluorescence and the light scattering characteristics of each population were assessed by flow cytometric analysis. The fluorescence of CD148+CD20+ and CD148−CD20+ cells incubated with an isotype control mAb was within the first log of the histogram plot. These results are representative of six different spleen samples.
Table 1.
Cell Surface Phenotypes of CD148+ and CD148− B Cells
| CD148− B cells | CD148+ B cells | |||||||
|---|---|---|---|---|---|---|---|---|
| % positive | MFI | % positive | MFI | |||||
| Surface Ag | ||||||||
| sIgM | 67.9 ± 17.0 | 55.4 ± 24.0 | 80.3 ± 11.4 | 131.1 ± 34.3 | ||||
| sIgD | 84.5 ± 8.0 | 508.3 ± 112 | 76.8 ± 8.3 | 261.7 ± 98.2 | ||||
| sIgG | 14.5 ± 7.5 | 335.4 ± 42 | 15.5 ± 3.0 | 223.3 ± 93.3 | ||||
| sIgA | 18.5 ± 7.3 | 75.0 ± 34.7 | 41.3 ± 8.0 | 170 ± 94.8 | ||||
| CD21 | 98.0 ± 1.2 | 251.8 ± 55.5 | 92.9 ± 7.0 | 602.9 ± 213.5 | ||||
| CD23 | 78.3 ± 6.4 | 63.1 ± 17.0 | 30.8 ± 6.3 | 72.7 ± 12.5 | ||||
| CD27 | 17.4 ± 4.0 | 46.9 ± 21.8 | 90.3 ± 3.2 | 58.0 ± 30 | ||||
| CD39 | 96.7 ± 1.6 | 115.6 ± 27.9 | 99.4 ± 0.6 | 591.9 ± 443 | ||||
| CD40 | 100 | 355.1 ± 54 | 99.6 ± 0.7 | 545.5 ± 92.8 | ||||
| CD80 | 17.4 ± 9.6 | 21.3 ± 6.4 | 46.3 ± 8.6 | 37.4 ± 18.9 | ||||
| CD86 | 40.5 ± 16.6 | 20.3 ± 4.3 | 55.8 ± 5.7 | 85.8 ± 41.8 | ||||
| CD95 | 59.1 ± 13.6 | 21.5 ± 5.6 | 86.5 ± 11.1 | 76.3 ± 23.8 | ||||
| HLA-DR | 99.6 ± 0.7 | 618 ± 193 | 99.6 ± 1.0 | 696.5 ± 233 | ||||
| SLAM | 26.4 ± 18.9 | 18.8 ± 4.5 | 36.6 ± 19.3 | 70.5 ± 22.3 | ||||
| FSC | NA | 94.1 ± 8.2 | NA | 138.6 ± 14.8 | ||||
| SSC | NA | 32.0 ± 5.6 | NA | 61.0 ± 5.8 | ||||
Splenic B cells were stained with FITC- or PE-labeled anti-CD148, PerCp–anti-CD20 and FITC- or PE-labeled mAbs specific for the indicated Ag. The percentage of CD148− and CD148+ B cells' positive cells, as well as the mean fluorescence intensity (MFI) of the positive cell population, of the different surface molecules was assessed by gating on CD148−CD20+ or CD148+CD20+ cells. Each value represents the mean ± SD of values obtained from flow cytometric analysis of six different donor spleens. NA, Not applicable.
IgM++IgD+IgA+/++CD21++CD23±CD27+CD39++ CD80+CD86+CD95+,
while that of splenic CD148− B cells was
IgM+IgD++IgA−/±CD21+CD23+CD27−CD39+ CD80±CD86−CD95±.
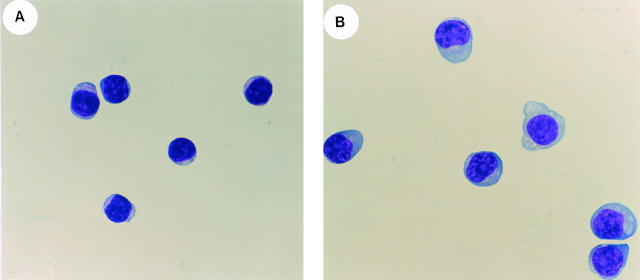
In contrast to these molecules, expression of sIgG, CD10, CD38, CD40, HLA-DR, or bcl-2 did not differ between CD148− and CD148+ B cells (Table 1, and data not shown). CD148+ and CD148− splenic B cells also differed morphologically. The forward and 90° angle light-scatter (FSC and SSC), indicative of cell size and internal complexity, respectively, of CD148+ B cells was up to twofold greater than that of CD148− B cells (Fig. 1 B, and Table 1). These morphological differences were further highlighted after Giemsa staining of the sort-purified B cell populations. CD148− B cells were homogeneously small cells with a large nucleus, scant cytoplasm, and very few cytoplasmic granules (Fig. 2 A). In contrast, CD148+ B cells comprised predominantly larger cells with large cytoplasms, eccentric nuclei, and cytoplasmic granules (Fig. 2 B).
Figure 2.
Morphological differences between sort-purified human CD148+ and CD148− splenic B cells. Human splenic B cells were incubated with FITC-labeled anti-CD148 and PE-labeled anti-CD20 and sorted into CD20+ B cell populations that were either CD148− or CD148+. After sort-purification, the (A) CD148− B cells and (B) CD148+ B cells were cytocentrifuged onto glass slides and analyzed by Giemsa staining.
CD148+CD27+ B cells Are Located in the Marginal Zone of Human Spleen
Splenic white pulp contains phenotypically distinct B cell subsets which reside in the marginal or follicular mantle zones (21–24, 37). The phenotype of CD148− and CD148+ B cells strongly suggested that these cells were mantle and marginal zone B cells, respectively. To confirm that the B cell subsets resided in distinct areas of human spleen, tissue sections were stained with anti-IgD–specific antiserum alone or in conjunction with either anti-CD27 mAb (to identify sIgD++CD27−CD148− and sIgD±/+CD27+CD148+ B cells) or anti-IgM mAb (to identify sIgM+IgD++CD148− and sIgM++IgD+CD148+ B cells). This was done because these molecules defined CD148+ and CD148− B cells (Fig. 1 B, and Table 1) and also because the A3 mAb did not react well with tissue sections (data not shown). Consistent with previous data (21, 24, 37), the mantle and marginal zones could be distinguished by the presence of sIgDhigh and sIgDdim B cells, respectively (Fig. 3, A and D). Within the mantle zone is a GC, containing sIgD− cells (Fig. 3, A and D). The marginal zone is interrupted by the (sIgD−) T cell zone (Fig. 3 A). CD27+ cells were present in the T cell zone, the marginal zone, and the GC, but not the mantle zone (Fig. 3, B and E). Within the marginal zone, all sIgDdim B cells were CD27+, whereas the T cell zone comprised single-stained CD27+ cells (Fig. 3, B and E). The CD27+ cells in the GC may represent T cells (38), GC B cells (39), or both. B cells expressing the brightest level of sIgM were present in the marginal zone, whereas mantle zone B cells expressed a lower level of sIgM (Fig. 3 C). Similar to the lack of expression of CD27, few sIgMhigh cells were detected in the follicular mantle zone (Fig. 3 C). Thus, CD27+ B cells are sIgDdimsIgMhigh and reside in the marginal zone, whereas CD27− B cells are sIgDhighsIgMdim and occupy the follicular mantle zone. Extrapolating these differences to those observed for cell surface phenotypes (Fig. 1 B, and Table 1) indicates that CD148+ and CD148− B cells are marginal and mantle zone B cells, respectively.
Figure 3.
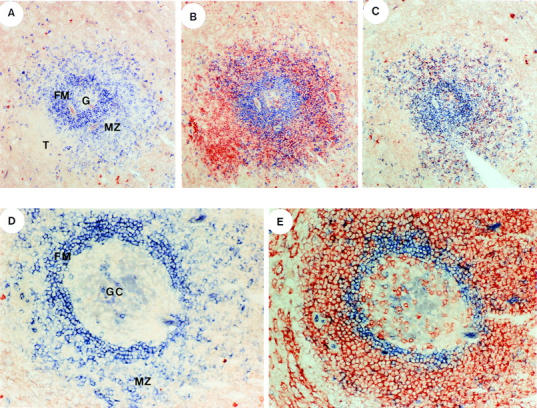
Localization of CD148+CD27+ and CD148−CD27− B cells in follicles of human spleen. Serial tissue sections of human spleen were incubated with anti-IgD antiserum alone (blue; A and D), anti-IgD antiserum (blue) and anti-CD27 mAb (red; B and E), or anti-IgD antiserum (blue) and anti-IgM mAb (red; C). The anti-IgD polyclonal antibody was visualized after the addition of alkaline phosphatase–conjugated anti–goat Ig and phosphatase-specific substrate. The anti-CD27 or anti-IgM mAb was visualized by an anti–mouse Ig-specific Vectastain kit. Original magnification of A, B, and C: ×40; of D and E: ×100. The follicular mantle zone (FM), marginal zone (MZ), germinal center (G or GC), and T cell zones (T) are indicated.
CD148+CD27+ B Cells Express Somatically Mutated Ig V Genes whereas Ig V Genes from CD148− CD27− B Cells Are in Germ-line Configuration
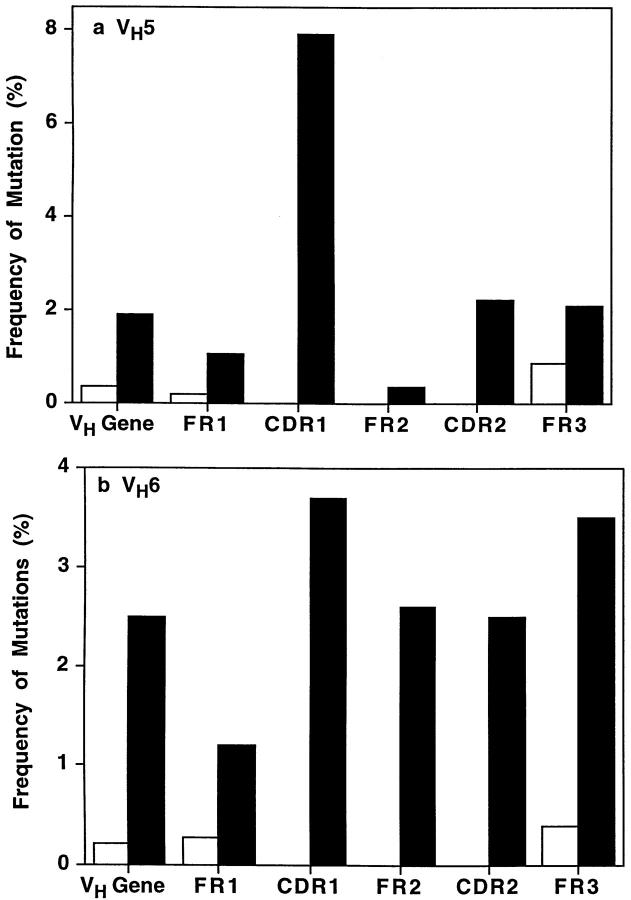
To investigate somatic mutation, Ig V region genes belonging to the small VH5 family and nonpolymorphic VH6 family were amplified by PCR using 5′ family-specific leader sequence primers and a 3′ Cμ constant region primer. Of 12 VH5 genes amplified from CD148− B cells, 6 were 100% identical to 1 of the 2 published VH5 germline genes (VH251 or VH32; Fig. 4 [40]). Similarly, five out of eight VH6 gene sequences obtained from CD148− B cells were unmutated, and therefore identical to the single VH6 germ-line gene here identified (VHVI; Fig. 5 [40]). No mutations were detected in the CDR1, CDR2, or framework region (FR)2 of the remaining sequences analyzed. However, a small number of mutations were present in FR1 and FR3 (Figs. 4 and 5). The overall frequency of mutations (no. of nucleotide changes/no. of base pairs sequenced) in the different VH5 and VH6 genes was 0.34 and 0.21%, respectively (Fig. 6, A and B); the average number of mutations detected per sequence was 1.0 ± 1.35 and 0.625 ± 1.06 (mean ± SD), respectively. These rates of mutation do not differ greatly from that reported for the endogenous error rate of Taq polymerase (1/500–1,000 bp; 0.1–0.2% [17]). This indicates that, similar to naive and mantle zone B cells (15, 25), the Ig V genes expressed by CD148− B cells are unmutated and in germ-line conformation.
Figure 4.
Nucleotide sequence of Ig VH5 genes isolated from CD148− and CD148+ splenic B cells. Nucleotide sequences of VH5 Ig genes cloned from sort-purified CD148− and CD148+ human splenic B cells are shown. The germline VH5 (VH251) sequence was derived from Tomlinson et al. (reference 40). For clarity, only sequences homologous to VH251 are shown. Each dash represents identity with the germ-line sequence; nucleotide differences are indicated. The FR and CDRs are indicated.
Figure 5.
Nucleotide sequence of Ig VH6 genes isolated from CD148− and CD148+ splenic B cells. Nucleotide sequences of VH6 Ig V genes cloned from sort-purified CD148− and CD148+ human splenic B cells are shown. The germline VH6 (VHVI) sequence was derived from Tomlinson et al. (reference 40). Each dash represents identity with the germ-line sequence; nucleotide differences are indicated. The FR and CDRs are indicated.
Figure 6.
Frequency of mutations in Ig V genes isolated from CD148− and CD148+ splenic B cells. The frequency of somatic mutations in (a) VH5 or (b) VH6 genes isolated from CD148− (white bars) and CD148+ B cells (black bars) was calculated as the number of nucleotide changes detected per base pairs sequenced. Each value represents the mean rate of mutation for either the entire VH5 or VH6 gene (VH), or for the different regions (FR, CDR) of the Ig V gene for all of the VH5 and VH6 clones analyzed.
For analysis of Ig V genes from CD148+ B cells, 14 different VH5 and 9 different VH6 genes were obtained by PCR amplification (Figs. 4 and 5). Unlike CD148− B cells, nucleotide changes were detected in Ig V genes expressed by CD148+ B cells (Figs. 4 and 5). Analysis of the Ig VH genes isolated from CD148+ B cells revealed that 14/14 VH5 and 8/9 VH6 genes had accumulated between 1 and 16 mutations, with >60% of sequences acquiring 5 or more mutations (Figs. 4 and 5). Somatic mutations were present in all three FR as well as both CDRs, with the frequency of mutation of the different regions of the Ig gene as high as ∼8% (Fig. 6). The overall frequency of mutations in the different VH5 and VH6 genes was 1.9 and 2.5% (Fig. 6), and the average number of mutations detected per sequence was 5.64 ± 4.0 and 7.67 ± 5.07, respectively. There were a total of 79 and 69 nucleotide differences detected in the different VH5 and VH6 genes, respectively, and >60% yielded amino acid replacements (data not shown). Thus, CD148+ B cells have undergone somatic mutation, a characteristic of memory and marginal zone B cells (4, 15, 20, 25).
CD148+ B Cells Preferentially Differentiate to Plasma Cells In Vitro
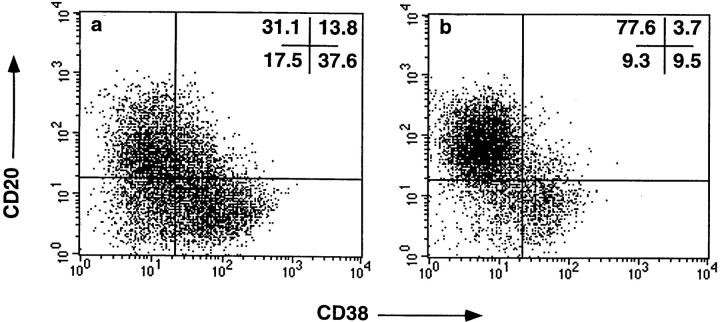
Memory B cells obtained from human tonsil are biased to differentiating into plasma B cells after in vitro activation (35, 41). To determine whether CD148− or CD148+ B cells preferentially differentiated to plasma cells, sort-purified B cells were cultured under conditions to induce plasma cell differentiation (35). After the culture, ∼40% of CD148+ B cells were CD38+CD20−, and thus resembled plasma cells (35, 41). In contrast, only ∼10% of CD148− B cells appeared as plasma cells, while the majority (>75%) continued to express high levels of CD20 (Fig. 7). Thus, similar to tonsil-derived memory B cells, splenic CD148+ B cells differentiate into plasma cells at a greater rate than CD148− B cells.
Figure 7.
CD148+ B cells differentiate into plasma cells in vitro. Sort-purified (a) CD148+ and (b) CD148− B cells were cultured for 3 d with anti-CD40 mAb (15 μg/ml), IL-2 (100 U/ml), and IL-10 (100 U/ml), and then recultured for an additional 3 d with IL-2 and IL-10. Plasma cell differentiation was then assessed by flow cytometric analysis after immunofluorescent staining with FITC–anti-CD38 mAb (x axis) and PE–anti-CD20 mAb (y axis). The values represent the percentage of cells in each quadrant of the dot plot. Similar results were obtained in a second experiment.
CD148+ and CD148− B Cell Subsets Secrete Distinct Ig Isotypes
CD148− B Cells Undergo Isotype Switching to Secrete IgE and IgG4.
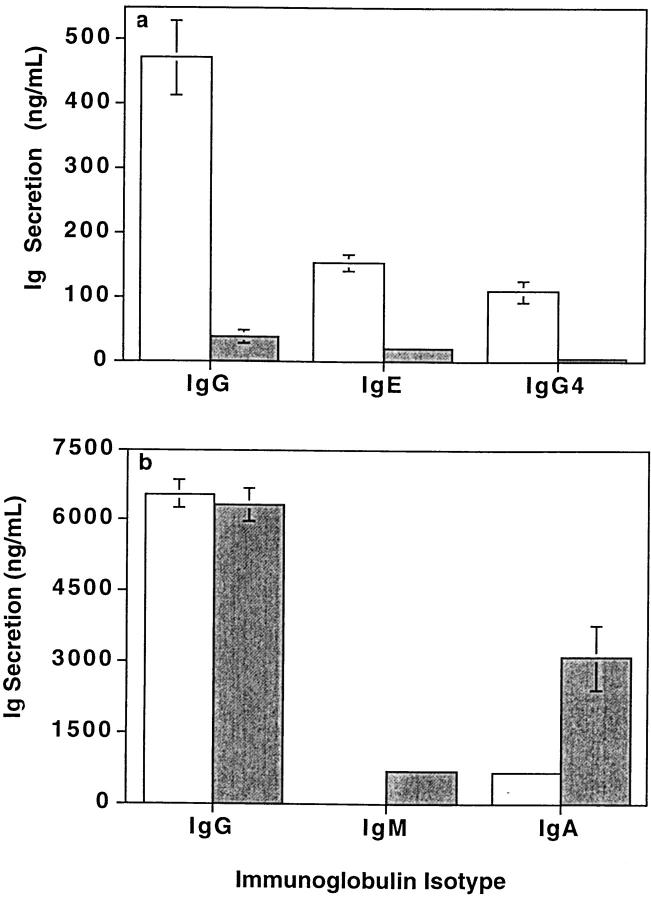
Activation of B cells with anti-CD40 mAb and IL-4 induces isotype switching to secrete IgG4 and IgE (2, 42). Under these conditions, CD148− B cells secreted 87.6 ± 10.1 and 89.9 ± 6.3% (mean ± SD, n = 9), respectively, of the IgG4 and IgE produced by total splenic B cells (Fig. 8 a). CD148− B cells secreted 16.6 ± 5.1 and 19.0 ± 10.3 (mean ± SEM, n = 9)-fold more IgE and IgG4 than CD148+ B cells. A similar dichotomy was observed for total IgG secretion in response to anti-CD40 mAb plus IL-4, where it was found that CD148− B cells secreted 6.6 ± 1.2–fold more (n = 11) IgG than CD148+ B cells (Fig. 8 a). Thus, only CD148− B cells are induced to undergo isotype switching to IgE and IgG4, and secrete the majority of total IgG, after activation with anti-CD40 mAb plus IL-4.
Figure 8.
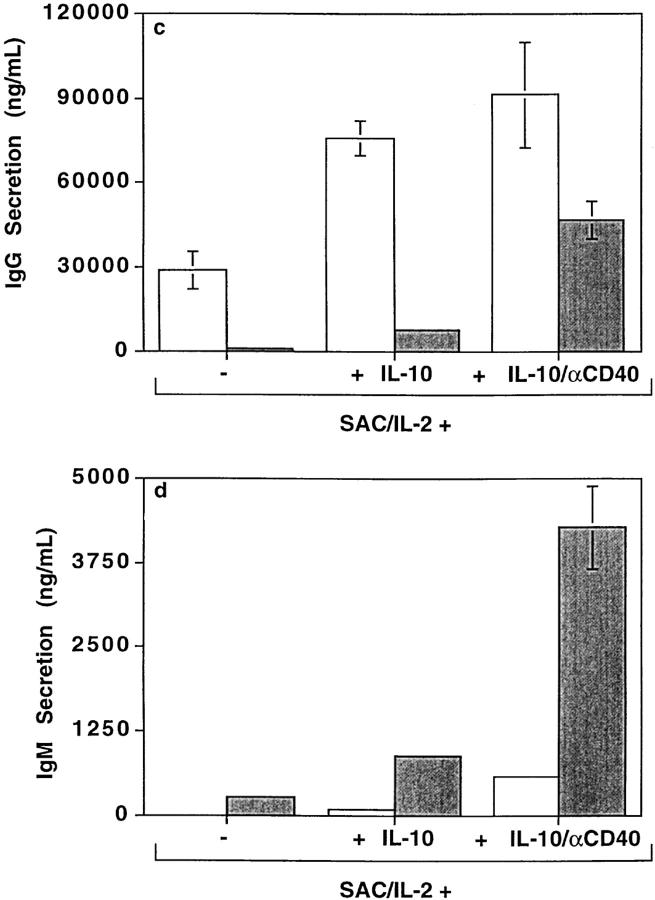
CD148− and CD148+ B cells differ in their ability to secrete Igs. Sort-purified CD148+ (gray bars) and CD148− B cells (white bars) were cultured with (a) anti-CD40 mAb (15 μg/ml) and IL-4 (400 U/ml), (b) anti-CD40 mAb and IL-2 (100 U/ ml) and IL-10 (100 U/ml), or (c and d) SAC (0.01%) plus IL-2 with or without IL-10 or anti-CD40 mAb. The amounts of IgG, IgE, IgG4, IgA, and IgM secreted were determined by isotype-specific immunoassays after 12 d. Each value represents the mean ± SEM of four to six replicates. The results shown are representative of data obtained from between 5 and 11 independent experiments.
Differential Secretion of IgA, IgG, and IgM.
Activation with anti-CD40 mAb plus IL-2 and IL-10 resulted in secretion of comparable levels of total IgG by CD148− and CD148+ B cells (Fig. 8 b). However, activated CD148+ B cells were the main producers of IgA and IgM, secreting 4.6 ± 1.3– fold more IgA (mean ± SEM; n = 8) and 20.6 ± 4.9–fold more IgM (n = 11) than CD148− B cells (Fig. 8 b). This indicates that CD148+ B cells can secrete high levels of particular Ig isotypes after the appropriate in vitro activation. Activation with SAC plus IL-2 revealed that CD148− B cells secreted 18.0 ± 7.5–fold more IgG than CD148+ B cells (n = 5; Fig. 8 c), whereas CD148+ B cells secreted 36.0 ± 16.0–fold more IgM than CD148− B cells (n = 5; Fig. 8 d). The secretion of IgG by activated CD148+ B cells and of IgM by activated CD148− B cells could be increased in the presence of IL-10, or IL-10 plus anti-CD40 mAb (Fig. 8, c and d). However, under these conditions, secretion of IgG by CD148− B cells and IgM by CD148+ B cells was also markedly increased, such that the levels of IgG and IgM secreted by CD148− and CD148+ B cells, respectively, continued to exceed those secreted by CD148+ and CD148− B cells by 6–13.0-fold (Fig. 8, c and d). This demonstrates that under these in vitro culture conditions, CD148− B cells are the primary producers of IgG, whereas CD148+ B cells secrete the majority of IgM.
Discussion
Expression of CD148 allowed the identification of two phenotypically distinct populations of splenic B cells. CD148+ B cells were larger and expressed lower levels of sIgD and CD23, but increased levels of sIgM, sIgA, CD21, CD39, and the activation/costimulatory Ags CD80, CD86, CD95, and SLAM. In contrast, CD148− B cells are smaller cells and express a high level of sIgD and CD23 and a lower level of activation Ags. Coincidentally, CD27 was expressed on all CD148+ but not CD148− B cells. In vitro stimulation of B cells by ligating sIg or CD40 induces B cell activation as evidenced by an increase in cell size (29, 43) and upregulation of expression of CD80, CD86 (16, 44, 45), CD95 (46), and SLAM (47). Splenic CD148+ B cells appear to have undergone Ag-specific T cell–mediated activation in vivo, and therefore represent a population of activated B cells.
Splenic white pulp comprises T cell zones, B cell follicles, and marginal zones, and these areas are populated by phenotypically distinct B cell subsets (21–24, 37). Based on phenotype, CD148+ and CD148− human B cells corresponded to marginal and mantle zone B cells, respectively. This was confirmed by demonstrating that CD27+IgD+ IgM++(CD148+) B cells colonized the marginal zone whereas CD27−IgD++IgM+(CD148−) B cells resided in the mantle zone. It has been suggested that mantle zone B cells are naive B cells while the marginal zone of spleen is a depot for memory B cells (25, 26). Interestingly, by virtue of IgD, CD23, CD80, CD86, and CD95 expression, CD148− and CD148+ splenic B cells phenotypically resembled naive and memory B cells, respectively, isolated from peripheral blood and tonsil (2, 16, 20). Taken together, it would appear that CD148 is expressed on human splenic memory B cells that reside in the marginal zone, whereas naive B cells present in the follicular mantle are CD148−.
In addition to cell surface phenotype, a key difference between naive and memory B cells is the presence of Ag-induced somatic mutations within Ig V region genes of memory B cells (3, 4, 15, 20). Analysis of the mutational status of Ig V region genes expressed by CD148− and CD148+ B cells revealed that CD148− B cells expressed unmutated Ig genes. In contrast, mutations were present in all regions of Ig V genes isolated from CD148+ B cells. The majority of these mutations gave rise to amino acid replacements, and exhibited characteristics typical of point mutations within somatically mutated Ig V genes, i.e., a frequent exchange of G to A, A to G, and C to T (4, 20). It is important to emphasize that the mutation status of Ig V genes rearranged to μ H chain, and consequently of sIgM-expressing B cells, was analyzed. Thus, consistent with previous studies (13, 20), somatic mutation can occur before Ig H chain isotype switching. The frequency of mutations and the average number of mutations detected per gene sequence within the VH5 and VH6 genes derived from CD148+ B cells were similar to that previously reported for Ig V genes isolated from sIgM+ memory B cells derived from human peripheral blood, tonsil, spleen, and bone marrow (∼2.0%, and 5.0–8.5 mutations/gene sequence; 15, 17–20, 25). The presence of somatic mutations in Ig V genes in CD148+ B cells confirms that these B cells are indeed memory B cells, whereas the expression of unmutated Ig V genes by CD148− B cells demonstrates that these cells are unprimed naive B cells.
CD27 is a TNF superfamily member expressed on resting T cells (38) and a subset of peripheral blood and tonsil B cells (48, 49). Based on phenotype, CD148+ splenic B cells and CD27+ peripheral blood and tonsillar B cells shared characteristics in addition to the coexpression of CD27. CD27+ peripheral blood B cells were also larger and expressed lower levels of sIgD but higher levels of sIgA than CD27− B cells (48, 49). Because CD148+CD27+ splenic B cells expressed mutated Ig V region genes, it would be predicted that this would also be true for peripheral blood and tonsil CD27+ B cells. Indeed, it has recently been reported that IgM+IgD+CD27+ peripheral blood B cells exhibited a similar frequency (∼2.0% [20]) of mutations in Ig V region genes as we found for CD148+CD27+ splenic B cells. Interestingly, CD27+ peripheral blood B cells, similar to human tonsil memory B cells (35, 41), differentiate into plasma cells after in vitro activation (50). In vitro activation of sort-purified splenic B cells induced plasmacytoid differentiation in a greater proportion of CD148+ B cells than CD148− B cells. These results, as well as those of others, provided additional evidence that splenic B cells coexpressing CD148 and CD27, as well as CD27+ peripheral blood B cells, are memory B cells.
CD148− and CD148+ B cells also differed in their ability to undergo isotype switching and to secrete Ig. CD148− B cells secreted >80% of total IgG when cultured with SAC plus IL-2. More striking was the observation that CD148− cells secreted all of the IgE, IgG4, and the majority of total IgG, when cultured with anti-CD40 mAb plus IL-4. It has been previously reported that only CD23+ B cells undergo switching to IgE (51, 52). Therefore, it was important to note that CD148− IgE-secreting B cells expressed CD23. Consistent with our proposal that CD148− B cells are naive, these cells underwent isotype switching to downstream isotypes as described for IgDbright naive B cells obtained from human tonsil and spleen (2, 42, 53). The inability of CD148+ B cells to secrete IgE and IgG4 did not result from a complete inability to secrete any Ig. In fact, after culture with anti-CD40 mAb plus IL-10 and IL-2, CD148+ and CD148− B cells secreted comparable levels of IgG. Importantly, CD148+ B cells secreted all of the IgM and the majority of IgA induced by this culture condition. Additionally, CD148+ cells secreted the majority of IgM in response to SAC plus IL-2. Because a significant proportion of CD148+ B cells expressed sIgA, it is unlikely that elevated secretion of IgA in the presence of anti-CD40 mAb plus IL-10 and IL-2 by CD148+ B cells is due to isotype switching, but rather results from activation of B cells that have already undergone isotype switching to IgA in vivo. Thus, the Ig isotype profile of activated CD148− and CD148+ splenic B cells is distinct. This may reflect specific roles that naive and memory B cells play in the humoral immune response to different pathogens.
In conclusion, by virtue of their phenotype, localization within the marginal zone, the presence of mutations in Ig V region genes, and their enhanced ability to differentiate to plasma cells, CD148+ splenic B cells are memory B cells. In contrast, CD148− B cells characteristically resembled naive B cells. Thus, expression of CD148 clearly identifies memory B cells. This feature was also found for the expression of CD27. Expression of CD27 on memory B cells is important for differentiation because ligation of CD27 enhanced the rate of plasma cell development (50, 54). The physiological significance of the expression of CD148 on memory B cells is presently unknown. It is tempting to speculate that it too may have a role in the subsequent differentiation of memory B cells. The finding that CD148 and CD27 are cell surface molecules capable of defining naive and memory splenic B cells will allow further characterization of these B cell subsets, and may reveal the exact role of CD148 in B cell activation and differentiation. Furthermore, assessment of expression of CD148 and CD27 represents a useful means to quantitate the development of a memory B cell response in vivo.
Acknowledgments
We thank Dr. Jim Cupp, Eleni Callas, Melinda Cook, Erin Murphy, and Dixie Pollakoff for cell sorting; Bruce Bennett for expert assistance with the Ig ELISAs; Svetlana Antonenko for expert assistance with histology; Natalie Davidson-Bennett for assistance with photography; Debbie Liggett for synthesis of oligonucleotides; Dan Gorman and the DNAX Sequencing Facility for DNA sequencing; and Dr. Lewis Lanier for critical review of this manuscript.
Abbreviations used in this paper
- FR
framework region(s)
- FSC
forward angle light scatter
- GC
germinal center(s)
- MFI
mean fluorescence intensity
- MNC
mononuclear cell(s)
- SSC
side scatter
- SAC
Staphylococcus aureus Cowan
- SLAM
signaling lymphocytic activation molecule
Footnotes
This paper is dedicated to Bruce F. Bennett, a dear friend and colleague of everyone at DNAX.
DNAX Research Institute is supported by the Schering-Plough Corporation.
G. Aversa and J. de Vries's current address is Novartis Research Institute, Brunner Strasse 59, A-1235 Vienna, Austria.
References
- 1.MacLennan IC, Liu Y-J, Johnson GD. Maturation and dispersal of B-cell clones during T cell-dependent antibody responses. Immunol Rev. 1992;126:143–161. doi: 10.1111/j.1600-065x.1992.tb00635.x. [DOI] [PubMed] [Google Scholar]
- 2.Banchereau J, Rousset F. Human B lymphocytes: phenotype, proliferation, and differentiation. Adv Immunol. 1992;52:125–262. doi: 10.1016/s0065-2776(08)60876-7. [DOI] [PubMed] [Google Scholar]
- 3.Gray D. Immunological memory. Annu Rev Immunol. 1993;11:49–77. doi: 10.1146/annurev.iy.11.040193.000405. [DOI] [PubMed] [Google Scholar]
- 4.Rajewsky K. Clonal selection and learning in the antibody system. Nature. 1996;381:751–758. doi: 10.1038/381751a0. [DOI] [PubMed] [Google Scholar]
- 5.Liu Y-J, Grouard G, de Bouteiller O, Banchereau J. Follicular dendritic cells and germinal centers. Int Rev Cytol. 1996;166:139–179. doi: 10.1016/s0074-7696(08)62508-5. [DOI] [PubMed] [Google Scholar]
- 6.Liu Y-J, Banchereau J. The paths and molecular controls of peripheral B-cell development. The Immunologist. 1996;4:55–66. [Google Scholar]
- 7.MacLennan IC. Germinal centers. Annu Rev Immunol. 1994;12:117–139. doi: 10.1146/annurev.iy.12.040194.001001. [DOI] [PubMed] [Google Scholar]
- 8.Thorbecke GJ, Amin AR, Tsiagbe VK. Biology of germinal centers in lymphoid tissue. FASEB J. 1994;8:832–840. doi: 10.1096/fasebj.8.11.8070632. [DOI] [PubMed] [Google Scholar]
- 9.Han S, Zheng B, Takahashi Y, Kelsoe G. Distinctive characteristics of germinal center B cells. Semin Immunol. 1997;9:255–260. doi: 10.1006/smim.1997.0081. [DOI] [PubMed] [Google Scholar]
- 10.Liu Y-J, Arpin C. Germinal center development. Immunol Rev. 1997;156:111–126. doi: 10.1111/j.1600-065x.1997.tb00963.x. [DOI] [PubMed] [Google Scholar]
- 11.Liu Y-J, Joshua DE, Williams GT, Smith CA, Gordon J, MacLennan IC. Mechanisms of antigen-driven selection in germinal centres. Nature. 1989;342:929–931. doi: 10.1038/342929a0. [DOI] [PubMed] [Google Scholar]
- 12.Küppers R, Zhao M, Hansmann ML, Rajewsky K. Tracing B cell development in human germinal centres by molecular analysis of single cells picked from histological sections. EMBO J. 1993;12:4955–4967. doi: 10.1002/j.1460-2075.1993.tb06189.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Liu Y-J, Malisan F, de Bouteiller O, Guret C, Lebecque S, Banchereau J, Mills FC, Max EE, Martinez-Valdez H. Within germinal centers, isotype switching of immunoglobulin genes occurs after the onset of somatic mutation. Immunity. 1996;4:241–250. doi: 10.1016/s1074-7613(00)80432-x. [DOI] [PubMed] [Google Scholar]
- 14.Liu Y-J, Arpin C, de Bouteiller O, Guret C, Banchereau J, Martinez-Valdez H, Lebecque S. Sequential triggering of apoptosis, somatic mutation and isotype switch during germinal center development. Semin Immunol. 1996;8:169–177. doi: 10.1006/smim.1996.0021. [DOI] [PubMed] [Google Scholar]
- 15.Pascual V, Liu Y-J, Magalski A, de Bouteiller O, Banchereau J, Capra JD. Analysis of somatic mutation in five B cell subsets of human tonsil. J Exp Med. 1994;180:329–339. doi: 10.1084/jem.180.1.329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Liu Y-J, Barthelemy C, de Bouteiller O, Arpin C, Durand I, Banchereau J. Memory B cells from human tonsils colonize mucosal epithelium and directly present antigen to T cells by rapid up-regulation of B7-1 and B7-2. Immunity. 1995;2:239–248. doi: 10.1016/1074-7613(95)90048-9. [DOI] [PubMed] [Google Scholar]
- 17.Klein U, Küppers R, Rajewsky K. Human IgM+IgD+B cells, the major B cell subset in the peripheral blood, express Vκ genes with no or little somatic mutation throughout life. Eur J Immunol. 1993;23:3272–3277. doi: 10.1002/eji.1830231232. [DOI] [PubMed] [Google Scholar]
- 18.Paramithiotis E, Cooper MD. Memory B lymphocytes migrate to bone marrow in humans. Proc Natl Acad Sci USA. 1997;94:208–212. doi: 10.1073/pnas.94.1.208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Klein U, Küppers R, Rajewsky K. Evidence for a large compartment of IgM-expressing memory B cells in humans. Blood. 1997;89:1288–1298. [PubMed] [Google Scholar]
- 20.Klein U, Goossens T, Fischer M, Kanzler H, Braeuninger A, Rajewsky K, Küppers R. Somatic hypermutation in normal and transformed human B cells. Immunol Rev. 1998;162:261–280. doi: 10.1111/j.1600-065x.1998.tb01447.x. [DOI] [PubMed] [Google Scholar]
- 21.Timens WM, Poppema S. Lymphocyte compartments in human spleen. An immunohistologic study in normal spleens and noninvolved spleens in Hodgkin's disease. Am J Pathol. 1985;120:443–454. [PMC free article] [PubMed] [Google Scholar]
- 22.Timens W, Boes A, Poppema S. Human marginal zone B cells are not an activated B cell subset: strong expression of CD21 as a putative mediator for rapid B cell activation. Immunology. 1989;19:2163–2166. doi: 10.1002/eji.1830191129. [DOI] [PubMed] [Google Scholar]
- 23.Smith-Ravin J, Spencer J, Beverley PCL, Isaacson PG. Characterization of two monoclonal antibodies (UCL4D12 and UCL3D3) that discriminate between human mantle zone and marginal zone B cells. Clin Exp Immunol. 1990;82:181–187. doi: 10.1111/j.1365-2249.1990.tb05424.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kraal G. Cells in the marginal zone of the spleen. Int Rev Cytol. 1992;132:31–74. doi: 10.1016/s0074-7696(08)62453-5. [DOI] [PubMed] [Google Scholar]
- 25.Dunn-Walters DK, Isaacson PG, Spencer J. Analysis of mutations in immunoglobulin heavy chain variable region genes of microdissected marginal zone (MGZ) B cells suggests that the MGZ of human spleen is a reservoir of memory B cells. J Exp Med. 1995;182:559–566. doi: 10.1084/jem.182.2.559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Liu Y-J, Oldfield S, MacLennan IC. Memory B cells in T cell-dependent antibody responses colonize the splenic marginal zones. Eur J Immunol. 1988;18:355–362. doi: 10.1002/eji.1830180306. [DOI] [PubMed] [Google Scholar]
- 27.Liu Y-J, de Bouteiller O, Arpin C, Briere F, Galibert L, Ho S, Martinez-Valdez H, Banchereau J, Lebecque S. Normal human IgD+IgM−germinal center B cells can express up to 80 mutations in the variable region of their IgD transcripts. Immunity. 1996;4:603–613. doi: 10.1016/s1074-7613(00)80486-0. [DOI] [PubMed] [Google Scholar]
- 28.Tangye SG, Phillips JH, Lanier LL, de Vries JE, Aversa G. CD148: a receptor-type protein tyrosine phosphatase involved in the regulation of human T cell activation. J Immunol. 1998;161:3249–3255. [PubMed] [Google Scholar]
- 29.Valle A, Zuber CE, Defrance T, Djossou O, de Rie M, Banchereau J. Activation of human B lymphocytes through CD40 and interleukin-4. Eur J Immunol. 1989;19:1463–1467. doi: 10.1002/eji.1830190818. [DOI] [PubMed] [Google Scholar]
- 30.Aversa G, Chang CC, Carballido JM, Cocks BG, de Vries JE. Engagement of the signaling lymphocytic activation molecule (SLAM) on activated T cells results in IL-2-independent, cyclosporin A-sensitive T cell proliferation and IFN-gamma production. J Immunol. 1997;158:4036–4044. [PubMed] [Google Scholar]
- 31.Cifone MG, De Maria R, Roncaioli P, Rippo MR, Azuma M, Lanier LL, Santoni A, Testi R. Apoptotic signaling through CD95 (Fas/Apo-1) activates an acidic sphingomyelinase. J Exp Med. 1994;180:1547–1552. doi: 10.1084/jem.180.4.1547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Campbell MJ, Zelenetz AD, Levy S, Levy R. Use of family specific leader region primers for PCR amplification of the human heavy chain variable region gene repertoire. Mol Immunol. 1992;29:193–203. doi: 10.1016/0161-5890(92)90100-c. [DOI] [PubMed] [Google Scholar]
- 33.Gascan H, Aversa GG, Gauchat J-F, van Vlasselaer P, Roncarlo M-G, Yssel H, Kehry M, Spits H, de Vries JE. Membranes from activated CD4+T cells expressing T cell receptor (TcR) αβ or TcR γδ induce IgE synthesis by human B cells in the presence of interleukin-4. Eur J Immunol. 1992;22:1133–1141. doi: 10.1002/eji.1830220505. [DOI] [PubMed] [Google Scholar]
- 34.Punnonen J, Aversa GG, Vandekerckhove B, Roncarolo MG, de Vries JE. Induction of isotype switching and Ig production by CD5+ and CD10+human fetal B cells. J Immunol. 1992;148:3398–3404. [PubMed] [Google Scholar]
- 35.Arpin C, Dechanet J, Van Kooten C, Merville P, Grouard G, Briere F, Banchereau J, Liu Y-J. Generation of memory B cells and plasma cells in vitro. Science. 1995;268:720–722. doi: 10.1126/science.7537388. [DOI] [PubMed] [Google Scholar]
- 36.Yssel H, de Vries JE, Koken M, van Blitterswijk W, Spits H. Serum-free medium for the generation and propagation of functional human cytotoxic and helper T cell clones. J Immunol Methods. 1984;72:219–227. doi: 10.1016/0022-1759(84)90450-2. [DOI] [PubMed] [Google Scholar]
- 37.MacLennan, I.C.M., P.D. Nathan, G.D. Johnson, M. Khan, L. Walker, and N.R. Ling. 1986. Analysis of B and L workshop antibodies on sections of normal and neoplastic lymphoid tissue and cell lines. In Leukocyte Typing II, Vol. 2: Human B Lymphocytes. E.L. Reinherz, B.F. Haynes, L.M. Nadler, and I.D. Bernstein, editors. Springer-Verlag, New York. 257–275.
- 38.Hintzen RQ, de Jong R, Lens SM, van Lier RA. CD27: marker and mediator of T-cell activation? . Immunol Today. 1994;15:307–311. doi: 10.1016/0167-5699(94)90077-9. [DOI] [PubMed] [Google Scholar]
- 39.van Oers MH, Pals ST, Evers LM, van der Schoot CE, Koopman G, Bonfrer JM, Hintzen RQ, von dem Borne AE, van Lier RA. Expression and release of CD27 in human B-cell malignancies. Blood. 1993;82:3430–3436. [PubMed] [Google Scholar]
- 40.Tomlinson IM, Walter G, Marks JD, Llewelyn MB, Winter G. The repertoire of human germline VH sequences reveals about fifty groups of VH segments with different hypervariable loops. J Mol Biol. 1992;227:776–798. doi: 10.1016/0022-2836(92)90223-7. [DOI] [PubMed] [Google Scholar]
- 41.Arpin C, Banchereau J, Liu Y-J. Memory B cells are biased towards terminal differentiation: a strategy that may prevent repertoire freezing. J Exp Med. 1997;186:931–940. doi: 10.1084/jem.186.6.931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Rousset F, Garcia E, Banchereau J. Cytokine-induced proliferation and immunoglobulin production of human B lymphocytes triggered through their CD40 antigen. J Exp Med. 1991;173:705–710. doi: 10.1084/jem.173.3.705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Hodgkin PD, Go NF, Cupp JE, Howard M. Interleukin-4 enhances anti-IgM stimulation of B cells by improving cell viability and by increasing the sensitivity of B cells to the anti-IgM signal. Cell Immunol. 1991;134:14–30. doi: 10.1016/0008-8749(91)90327-8. [DOI] [PubMed] [Google Scholar]
- 44.Azuma M, Ito D, Yagita H, Okumura K, Phillips JH, Lanier LL, Somoza C. B70 antigen is a second ligand for CTLA-4 and CD28. Nature. 1993;366:76–79. doi: 10.1038/366076a0. [DOI] [PubMed] [Google Scholar]
- 45.Lenschow DJ, Sperling AI, Cooke MP, Freeman G, Rhee L, Decker DC, Gray G, Nadler LM, Goodnow CC, Bluestone JA. Differential up-regulation of the B7-1 and B7-2 costimulatory molecules after Ig receptor engagement by antigen. J Immunol. 1994;153:1990–1997. [PubMed] [Google Scholar]
- 46.Garrone P, Neidhardt EM, Garcia E, Galibert L, van Kooten C, Banchereau J. Fas ligation induces apoptosis of CD40-activated human B lymphocytes. J Exp Med. 1995;182:1265–1273. doi: 10.1084/jem.182.5.1265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Punnonen J, Cocks BG, Carballido JM, Bennett B, Peterson D, Aversa G, de Vries JE. Soluble and membrane-bound forms of signaling lymphocytic activation molecule (SLAM) induce proliferation and Ig synthesis by activated human B lymphocytes. J Exp Med. 1997;185:993–1004. doi: 10.1084/jem.185.6.993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Maurer D, Holter W, Majdic O, Fischer GF, Knapp W. CD27 expression by a distinct subpopulation of human B lymphocytes. Eur J Immunol. 1990;20:2679–2684. doi: 10.1002/eji.1830201223. [DOI] [PubMed] [Google Scholar]
- 49.Agematsu K, Nagumo H, Yang FC, Nakazawa T, Fukushima K, Ito S, Sugita K, Mori T, Kobata T, Morimoto C, Komiyama A. B cell subpopulations separated by CD27 and crucial collaboration of CD27+B cells and helper T cells in immunoglobulin production. Eur J Immunol. 1997;27:2073–2079. doi: 10.1002/eji.1830270835. [DOI] [PubMed] [Google Scholar]
- 50.Agematsu K, Nagumo H, Oguchi Y, Nakazawa T, Fukushima K, Yasui K, Ito S, Kobata T, Morimoto C, Komiyama A. Generation of plasma cells from peripheral blood memory B cells: synergistic effect of interleukin-10 and CD27/CD70 interaction. Blood. 1998;91:173–180. [PubMed] [Google Scholar]
- 51.Pene J, Rousset F, Briere F, Chreitien I, Wideman J, Bonnefoy JY, de Vries JE. Interleukin 5 enhances interleukin 4-induced IgE production by normal human B cells: the role of soluble CD23 antigen. Eur J Immunol. 1988;18:929–935. doi: 10.1002/eji.1830180615. [DOI] [PubMed] [Google Scholar]
- 52.Bonnefoy JY, Gauchat JF, Life P, Graber P, Mazzei G, Aubry JP. Pairs of surface molecules involved in human IgE regulation: CD23-CD21 and CD40-CD40L. Eur Respir J. 1996;22:63S–66S. [PubMed] [Google Scholar]
- 53.Cocks BG, de Waal R, Malefyt, Galizzi J-P, de Vries JE, Aversa G. IL-13 induces proliferation and differentiation of human B cells activated by the CD40 ligand. Int Immunol. 1993;5:657–663. doi: 10.1093/intimm/5.6.657. [DOI] [PubMed] [Google Scholar]
- 54.Jacquot S, Kobata T, Iwata S, Morimoto C, Schlossman SF. CD154/CD40 and CD70/CD27 interactions have different and sequential functions in T cell-dependent B cell responses: enhancement of plasma cell differentiation by CD27 signaling. J Immunol. 1997;159:2652–2657. [PubMed] [Google Scholar]